Cooperation between distinct viral variants promotes growth of H3N2 influenza in cell culture
Figures
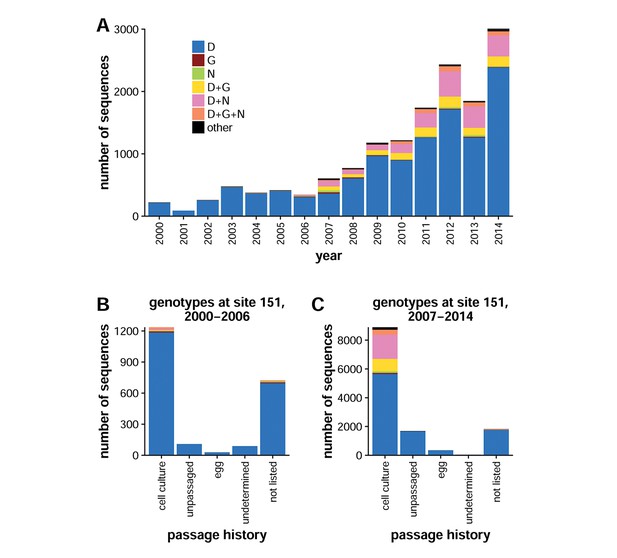
Ambiguous identities are common at NA site 151 after 2007.
(A) Shown are the number of human H3N2 influenza NA sequences in the GISAID EpiFlu database with the given identity at site 151 for each year from 2000 to 2014. Since 2007, ambiguous amino-acid identities have been present at residue 151 in about 20% of sequences. Sequences from (B) 2000 to 2006 and (C) 2007 to 2014 were classified into groups based on their passage history. Ambiguous amino-acid identities were present almost exclusively in isolates that had been passaged in cell culture. Sequences were classified as 'undetermined' if the passage history was difficult to interpret and as 'not listed' if the passage history was absent altogether. Mixed genotypes were inferred on the basis of IUPAC nucleotide ambiguity codes; for instance, the triplet GRT could refer to GAT or GGT, corresponding to amino acids D and G, respectively. Genotypes are indicated if they exceeded a frequency of 0.5% among all analyzed sequences; otherwise, they are categorized as 'other.' The computer code used for analysis is available in Figure 1—source data 1.
-
Figure 1—source data 1
This 7-zip archive contains the source code used for Figure 1 (the analysis of mutation frequencies at site 151 in naturally occurring sequences).
- https://doi.org/10.7554/eLife.13974.005
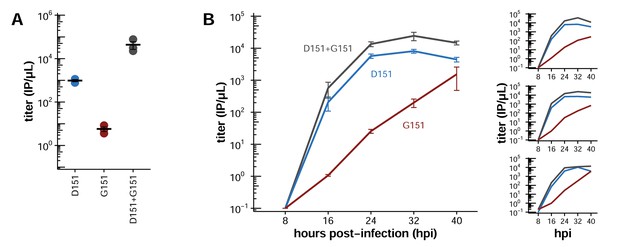
Mixed populations grow to higher titers than either pure population alone.
(A) Pure and mixed populations were generated by reverse genetics. Cells were transfected with a Hanoi/2007 NA plasmid encoding D151, G151, or an equal mix of the two, along with isogenic plasmids for the other genes. The total amount of NA plasmid was the same in all cases; that is, the pure populations were transfected with 250 ng of the indicated variant, and the mixed populations were transfected with 125 ng of each variant. The HA was also derived from Hanoi/2007, and the other genes were derived from the lab-adapted A/WSN/33 strain with GFP packaged in the PB1 segment. The titer was determined after 72 hr using the GFP reporter. Black lines indicate the mean and standard error of the titers for three biological replicates, with titers for each replicate plotted as points. Figure 2—figure supplement 1 shows a comparable effect when the virus does not package GFP. Figure 2—figure supplement 2 shows that growth of the G151 variant is improved by adding oseltamivir. (B) Cells were infected at an MOI of 0.2 with pure D151 virus, pure G151 virus, or an equal mix of the two. The total MOI of infecting virus was the same in all cases. The main plots show titers averaged across three biological replicates, with each replicate plotted individually in the small insets.
A mixed population outgrows either pure population when viruses are generated by reverse genetics with an unmodified PB1 gene.
The data here differ from Figure 2A in that the virus populations were generated by reverse genetics using the unmodified A/WSN/33 PB1 gene rather than the PB1 segment modified to package GFP. Titers were determined at 74 hr post-transfection by staining for nucleoprotein in infected cells. Black lines indicate the mean and standard error of the titers for three biological replicates, with titers for each replicate plotted as points.
Growth of the G151 variant is improved by adding oseltamivir during the generation of viral populations by reverse genetics.
Presumably, this improvement occurs because oseltamivir blocks the binding of G151 NA to receptor, allowing newly formed virions to be released more efficiently. Oseltamivir also slightly increases the growth of the D151 variant. We speculate this is because oseltamivir blocks receptor cleavage by the D151 NA, leaving more receptors that can be bound by HA during secondary viral replication.
Serial passage selects for a stable mix of the two variants.
Shown are the allele frequencies at NA site 151 over five tissue-culture passages of initially pure D151 viruses, pure G151 viruses, or an equal mix of the two. Each passage was seeded at a total MOI of 0.2. Passage 0 refers to the ratio of variants in the viral inoculum for passage 1. Allele frequencies were determined by targeted Illumina deep-sequencing of the NA gene. Based on sequencing of pure plasmid, the error rate was less than 1%. The raw data and computer code are available in Figure 3—source data 1.
-
Figure 3—source data 1
This 7-zip archive contains the data and source code used for Figure 3 (the analysis of mutation frequencies at site 151 after serial passage in the lab).
The code and all the FASTQ files are also available at http://dx.doi.org/10.5061/dryad.s3rs0.
- https://doi.org/10.7554/eLife.13974.010
The N151 variant also cooperates with D151.
Shown are the titers after reverse genetics with the indicated variant of the Hanoi/2007 NA. The experiments here parallel those in Figure 2A. Black lines indicate the mean and standard error of the titers for three biological replicates, with titers for each replicate plotted as points.
Cooperative dynamics depend on multiplicity of infection.
Cells were infected at an MOI of (A) 0.02 or (B) 0.5 with pure D151 virus, pure G151 virus, or an equal mix of the two. The total MOI of infecting virus was the same across the mixed and pure populations for infection at each MOI. The main plots show titers averaged across three biological replicates, with each replicate plotted individually in the small insets. The experiments here parallel those in Figure 2B. Black lines indicate the mean and standard error of the titers for three biological replicates, with titers for each replicate plotted as points.
Changes in HA between 2005 and 2007 potentiated cooperation.
Cooperation occurs between the D151 and G151 NA variants in viruses with HA from the Hanoi/2007 strain, but not in viruses with HA from the Wisconsin/2005 strain. Shown are the titers after reverse genetics with the indicated HA and NA. The experiments here parallel those in Figure 2A. Black lines indicate the mean and standard error of the titers for three biological replicates, with titers for each replicate plotted as points.
Cooperation is obligate when HA lacks receptor-binding activity.
Shown are the titers after reverse genetics with the indicated Hanoi/2007 NAs in combination with an engineered binding-deficient H3 HA with multiple mutations to the receptor-binding pocket (Hooper and Bloom, 2013). The experiments here parallel those in Figure 2A. Black lines indicate the mean and standard error of the titers for three biological replicates, with titers for each replicate plotted as points.
Tables
Prior reports of variation at neuraminidase site 151 when H3N2 clinical specimens are passaged in cell culture.
| Reference | Summary |
|---|---|
| (McKimm-Breschkin et al., 2003) | Sanger sequencing of 38 oseltamivir- and zanamivir-resistant MDCK-passaged clinical isolates found that 7 had G, N, E, or V at site 151. |
| (Lin et al., 2010) | Sanger sequencing of 18 isolates after passage in MDCK or MDCK-SIAT1 cells found 4 isolates as D+G, 3 as D+N, and 2 as D+A. Pyrosequencing detected low frequencies of G151 and N151 in some clinical samples. |
| (Tamura et al., 2013) | Pyrosequencing of 150 isolates after 1-4 passages in MDCK cells found that 85% developed mixed populations at site 151; 29% did so after a single passage. Mixed populations consisted of D+N, D+G, D+G+N, and D+G+A genotypes. T148I/K/P mutations were also observed in 23% of isolates. |
| (Lee et al., 2013) | 77 clinical specimens were Sanger-sequenced before and after a single passage in MDCK cells. 18 acquired a mutation at NA site 151: 10 were D+N, 7 were D+G and one fixed D151N at the limit of detection. No mutations were detected in the unpassaged specimens. |
| (Chambers et al., 2014) | 9 A/Victoria/361/11-like clinical specimens were passaged twice in MDCK cells and Sanger-sequenced before and after expansion. 4 isolates developed NA-dependent cell binding; 3 had D151G, the other D151N. |
| (Mishin et al., 2014) | Pyrosequencing of 150 MDCK-grown isolates found that 42 were D+G, 34 were D+N, and 57 were D+G+N. Pyrosequencing of 50 matched clinical specimens detected no variation at site 151. |
| (Mohr et al., 2015) | 16 pairs of isolates cultured in parallel in MDCK cells and in eggs were sequenced using Ion Torrent. 5 MDCK isolates were D+N, 4 were D+G, and 2 were D+N+G. No egg-passaged isolates had mutations at site 151. T148I/K mutations were observed in 7 MDCK isolates. |
Additional files
-
Supplementary file 1
This text file contains the coding sequences for the HA and NA genes used in this study.
- https://doi.org/10.7554/eLife.13974.015





