Distinct modes of endocytotic presynaptic membrane and protein uptake at the calyx of Held terminal of rats and mice
Figures
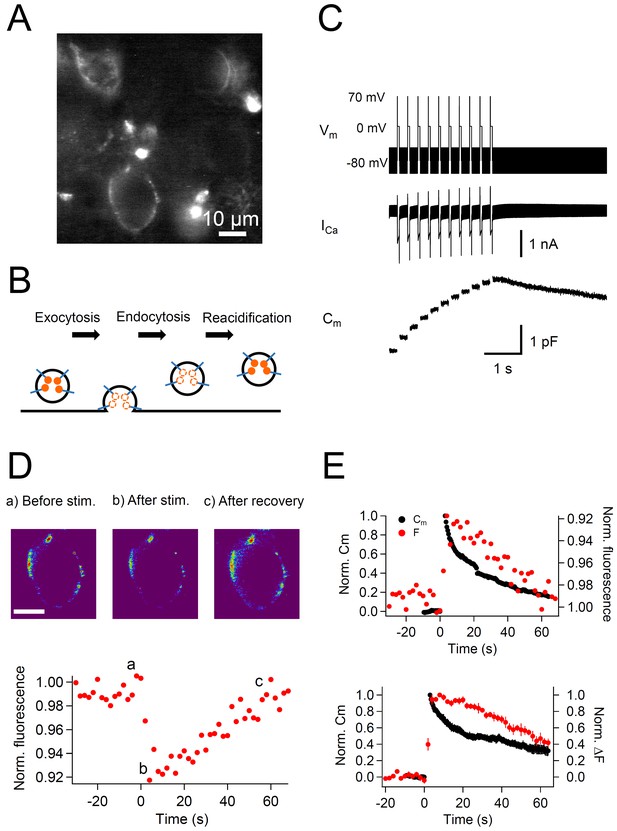
Simultaneous recording of membrane capacitance and anti-Syt2-cypHer fluorescence.
(A) Example fluorescence image of calyx of Held presynaptic terminals labeled with anti-Syt2-cypHer. Intravesicular anti-Syt2-cypHer emits fluorescence upon excitation at 645 nm.(B) Schematic view of fluorescence changes of anti-Syt2-cypHer during exo-endocytosis. The orange dots show cypHer coupled to antibodies against the luminal domain of Syt2. The cypHer fluorescence is maximal at intravesicular pH 5.5 and almost quenched at the extracellular pH 7.4. Upon exocytosis, the fluorescence is quenched because of the exposure to the extracellular pH. During endocytosis and re-acidification, the fluorescence is de-quenched again. (C) A train of depolarizing pulses (0 mV for 50 ms following a prepulse to +70 mV for 2 ms, 10 stimuli, interstimulus interval 200 ms, Vm) was applied to elicit a Ca2+ current (ICa), and membrane capacitance (Cm) was measured during the sweep. The prepulse (+70 mV) was applied to activate Ca2+ channels maximally without causing Ca2+ influx. A sine wave (30 mV in amplitude, 1,000 Hz in frequency) was superimposed on a holding potential of -80 mV to measure membrane capacitance (Cm). (D) The top panel shows example fluorescence images showing the cypHer fluorescence image (a) before stimulation, (b) after stimulation, and (c) after recovery, shown in a pseudo-colored scale. Each image was taken at the time point shown in the bottom trace. Scale bar, 10 μm. The bottom panel shows an example of a normalized fluorescence trace of anti-Syt2-cypHer in response to a train of depolarizing pulses. The fluorescence intensity was normalized to the first point in the plot. (E) The top panel shows example traces of normalized Cm (black circles, left axis) and cypHer fluorescence (red circles, right axis) at a calyx terminal stimulated by a train of depolarizing pulses. The Cm trace was normalized to the amplitude of the capacitance jump, and the fluorecence trace was normalized to the initial intensity. The fluorescence trace was inverted to compare the time courses of Cm and fluorescence traces.The bottom panel shows average traces of normalized Cm (black circles, n = 7) and cypHer fluorescence change (red circles, n = 10) at the calyx terminal evoked by a train of depolarizing pulses (7 data were obtained from simultaneous measurements of capacitance and cypHer). Cm traces were normalized to the peak capacitance change (left axis), and fluorecence traces were normalized to the peak fluorescence change (right axis).
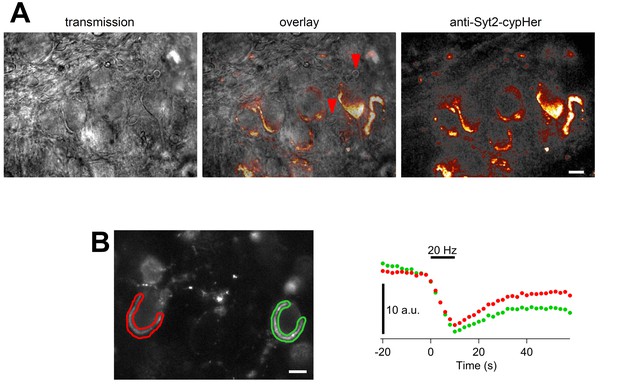
Calyx specific anti-Syt2-cypHer staining and fluorescence change evoked by field stimulation.
(A) Transmission image (left), and anti-Syt2-cypHer fluorescent image (right) in the MNTB region of the rat brainstem slice. The center shows an overlay. (B) A series of field stimulations (20 Hz for 10 s) was applied to the anti-Syt2-cypHer labeled slice (left). The fluorescence intensity at the ROIs is shown on the right. The apparent partial recovery is likely to be due to photobleaching (Hua et al., 2011). Scale bars, 10 μm.
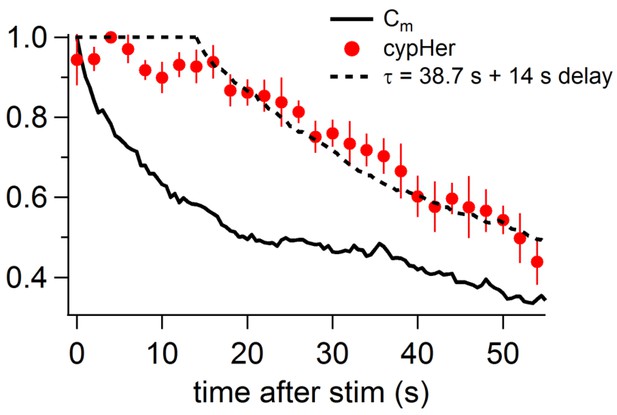
Comparison of the recovery time course of capacitance and cypHer (rat).
By assuming a certain delay and a re-acidification time constant of the endocytosed organelle after membrane retrieval, the recovery time course of cypHer signal (red circles) was fitted.The re-acidification time course of the endocytosed organelle was assumed to be an exponential from pH7.4 to pH5.5. The fitting curve is convolution of the Cm time course (bold line) with best fit delay and re-acidification time constant, calculated by least squares method. During the delay, the value was held to be 1. The cypHer signal was best fitted with a 14 s delay with a 38.7 s re-acidification time constant after membrane retrieval (dotted line).
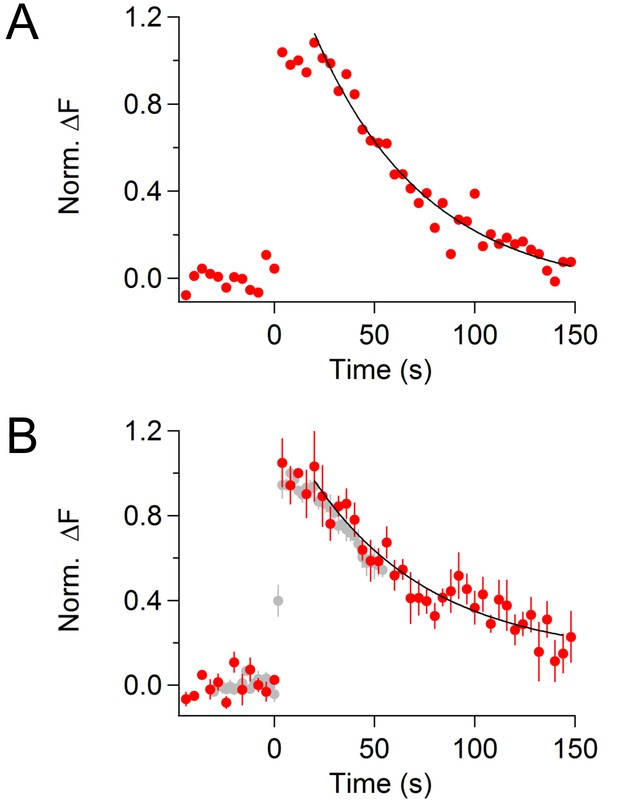
CypHer signal recovery after a train of depolarizing pulses (longer recording).
(A) An example trace of cypHer signal recovery after a train of depolarization (see Figure 1C) is shown for a longer recording period. To avoid bleaching of cypHer, the frequency of image acquisition was reduced to every 4 s. The cypHer signal recovery was best fitted with an exponential (τ = 55.5 s) with a 16 s delay.B. Averaged trace of cypHer signal recovery for a longer recording period (red circles, n = 5). The averaged cypHer signal recovery was best fitted with an exponential (τ = 59.7 s) with a 16 s delay. Gray circles are averaged cypHer signal from shorter recordings, same trace as shown in Figure 1E (bottom).
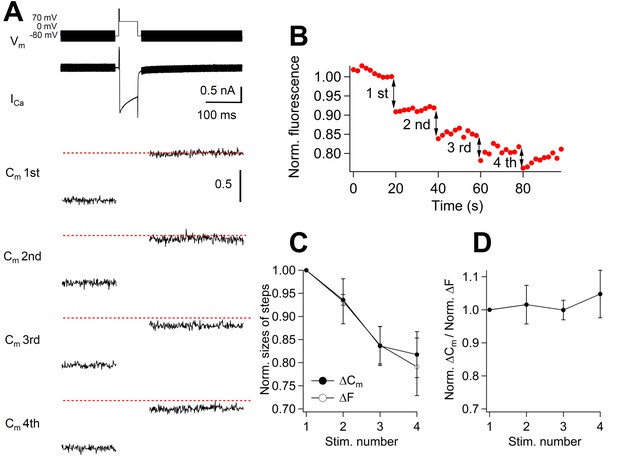
Correlation between the amount of membrane capacitance changes and cypHer signal changes.
(A) A 50 ms depolarizing pulse (0 mV for 50 ms, following a prepulse to +70 mV for 2 ms), as shown on the top, was applied four times with an inter stimulus interval of 20 s. The bottom panels show averaged capacitance traces of four consecutive stimulations (n = 6). Traces from each cell were normalized to the first ΔCm amplitude. The dotted red lines show the first ΔCm amplitudes. (B) Example of cypHer fluorescence changes induced by four consecutive 50 ms depolarizations. (C) The extent of capacitance jumps (filled circles) and cypHer fluorescence changes (open circles) evoked by four consecutive depolarizations. (D) The ratio of normalized ΔCm and normalized ΔF of the cypHer signals plotted against the stimulus number.
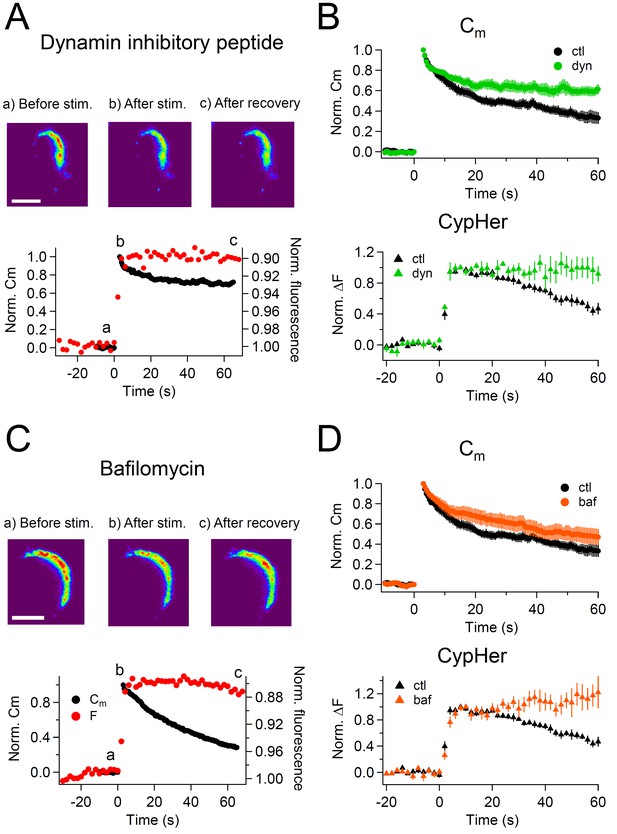
CypHer signal recovery depends on endocytosis and re-acidification.
(A) A train of depolarizing pulses (see Figure 1C) was applied to elicit exocytosis in the presence of 1 mM dynamin inhibitory peptide. Images in the top panel show the cypHer fluorescence at the time point indicated in the plot below (see also Figure 1B). Scale bar, 10 μm. The bottom panel shows example traces of Cm and cypHer fluorescence in the presence of dynamin inhibitory peptide. (B) The top panel shows averaged Cm traces under control conditions (n = 7, black circles) and in the presence of dynamin inhibitory peptide (n = 4, green circles). The bottom panel shows cypHer fluorescence changes under control conditions (n = 10, black triangles) and in the presence of dynamin inhibitory peptide (n = 4, green triangles). (C) Same as in A, with 5 μM bafilomycin applied during the recording. (D) The top panel shows averaged Cm traces under control conditions (n = 7, black circles) and in the presence of bafilomycin (n = 5, orange circles). The bottom panel shows averaged fluorescence traces under control conditions (n = 10, black triangles) and in the presence of bafilomycin (n = 5, orange triangles).
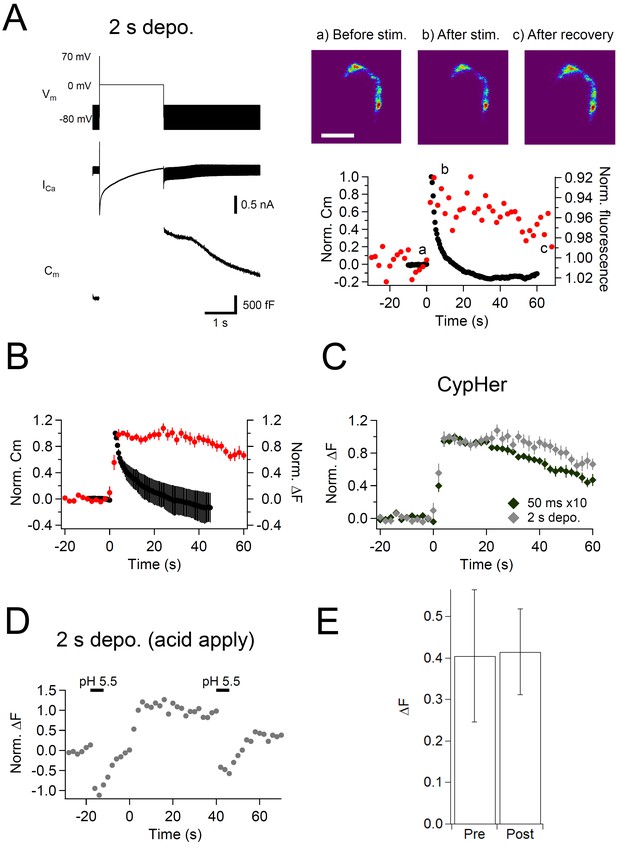
Effect of a prolonged depolarizing pulse on the kinetics of capacitance and cypHer signal recovery.
(A) Similar to Figure 1, but a single prolonged (2 s) depolarization (Vm) was applied, which induced large calcium currents (ICa) and capacitance jumps (Cm). Images (right top) show the cypHer fluorescence at each time point shown in the panel below. Scale bar, 10 μm. The right bottom panel shows example traces of Cm and cypHer fluorescence obtained with the 2 s depolarization paradigm. (B) Average traces of Cm (black circles) and cypHer fluorescence (red circles) at the calyx terminal evoked by a 2 s depolarizing pulse (n=7). (C) Averaged cypHer fluorescence traces obtained by applying a train of depolarizing pulses (the same as Figure 1C, n=10, black diamonds) or by a 2 s depolarizing pulse (n=7, gray diamonds). CypHer fluorescence recovery was slower after a 2 s depolarization (40 s after the stimulus; 50 ms x 10, 0.70 ± 0.04; 2 s, 0.93 ± 0.07, p=0.019). (D) Example trace of cypHer fluorescence change when an acidic extracellular solution (pH 5.5) was puff-applied during the recording. The pH 5.5 solution was puff-applied twice for 5 s, 20 s before and 40 s after stimulation. (E) Comparison of ∆F induced by the puff-application of pH 5.5 solution before (Pre) and after (Post) stimulation. There was no significant difference (Pre, 0.40 ± 0.16; Post, 0.41 ± 0.10, n = 5, p=0.66).
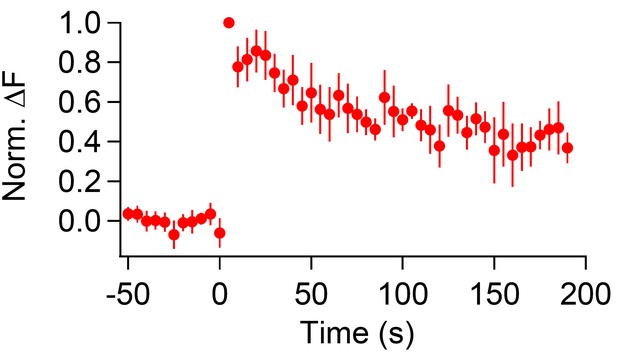
CypHer signal recovery after a prolonged depolarizing pulse (longer recording).
The time course of cypHer signal recovery after a 2 s depolarization (see Figure 4A) is shown for a longer recording period.To avoid bleaching of cypHer, the frequency of image acquisition was reduced to every 5 s. The cypHer signal recovered to 0.36 ± 0.08 after 190 s (n = 4), similar to 60 s after train stimulation with ten 50 ms depolarizations (0.47 ± 0.07, n = 10).
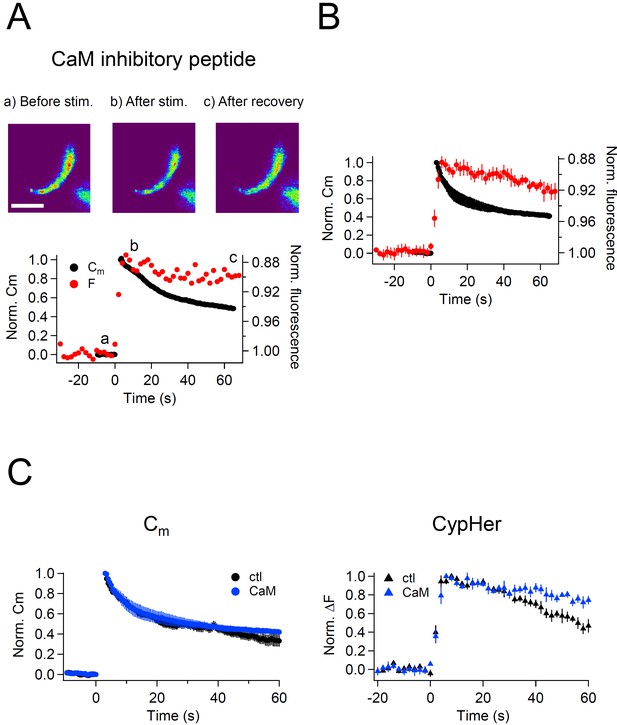
Effect of calmodulin inhibitory peptide on the kinetics of capacitance and cypHer signal recovery.
(A) A train of depolarizing pulses (the same as Figure 1C) was applied to elicit exocytosis in the presence of 20 μM CaM inhibitory peptide. Images (top) show the cypHer fluorescence at each time point depicted in the bottom panel. The bottom panel shows example traces of Cm and cypHer fluorescence in the presence of the CaM inhibitory peptide. Scale bar, 10 μm.(B) Average traces of Cm (black circles) and cypHer fluorescence (red circles) in the presence of CaM inhibitory peptide (n = 5).C. The left panel shows averaged Cm traces under control conditions (n = 7, black circles) and in the presence of CaM inhibitory peptide (n = 5, blue circles). The right panel shows averaged cypHer fluorescence traces under control conditions (n = 10, black triangles) and in the presence of CaM inhibitory peptide (n = 5, blue triangles).
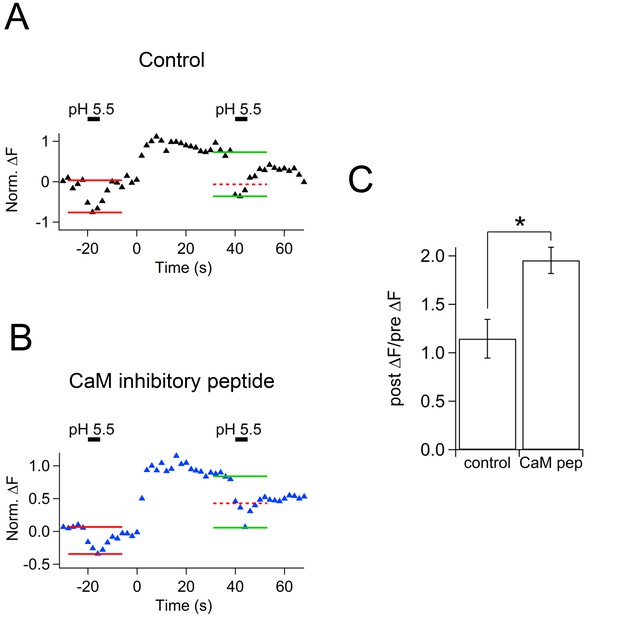
Effect of calmodulin inhibitory peptide on cypHer fluorescence changes induced by acidic solution application.
(A) An example trace of the cypHer fluorescence change when an acidic extracellular solution (pH 5.5) was puff-applied during the recording without CaM inhibitory peptide. The pH 5.5 solution was puff applied for 5 s, once 20 s before and once 40 s after stimulation (50 ms pulse x 10). Red and green lines show the amount of fluorescence change induced by the pH 5.5 solution before and after stimulation, respectively. The dotted red line shows the amplitude of the 'before' signal. (B) The same as A, but in the presence of 20 μM CaM inhibitory peptide. (C) The de-quenching ratio before and after the stimulus (post ΔF/pre ΔF value) was larger in the presence of CaM inhibitory peptide (control, 1.15 ± 0.20, n = 5; CaM inhibitory peptide, 1.95 ± 0.14, n = 5, p=0.012).
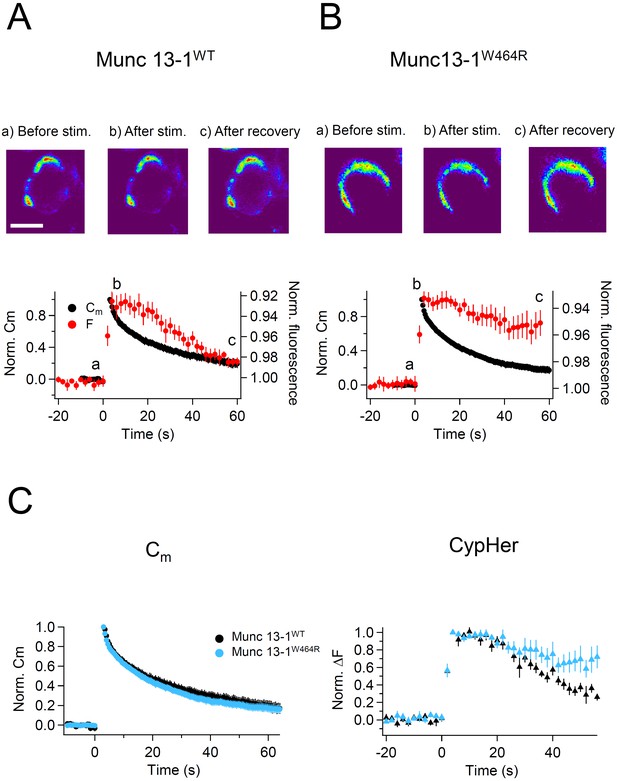
Capacitance and cypHer signal recovery in the calyx of Held of Munc 13-1W464R KI mice.
(A) The same as Figure 1D and E, but recordings from calyx of Held terminals of wild-type (Munc13-1WT) mice. The top panel shows the cypHer images at the time points shown in the bottom panel, and the bottom panel shows averaged Cm (black circles) and cypHer fluorescence (red circles) changes (n = 8). Scale bar, 10 μm. (B) The same as A, but recordings from calyx of Held terminals of Munc 13-1W464R mice (n = 6). Black and red traces show average normalized Cm and fluorescence changes. 3 out of 6 data were obtained from simultaneous measurements of capacitance and cypHer. (C) The left panel shows averaged Cm traces from Munc13-1WT calyces (n = 8, black circles) and Munc13-1W464R calyces (n = 6, turquoise circles). The right panel shows averaged cypHer fluorescence traces from Munc13-1WT calyces (n = 8, black triangles) and Munc13-1W464R calyces (n = 6, turquoise blue triangles).
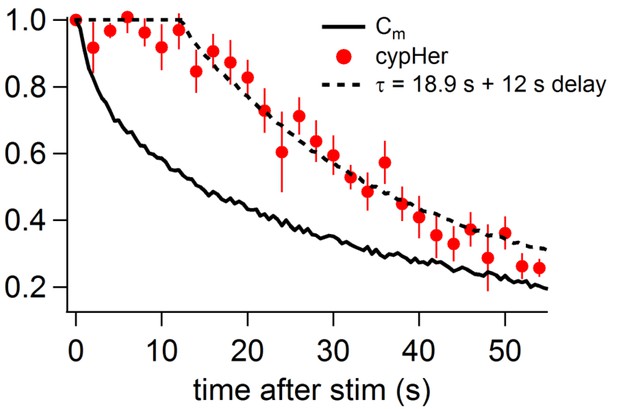
Comparison of the recovery time course of capacitance and cypHer (mouse).
By assuming a certain delay and a re-acidification time constant of the endocytosed organelle after membrane retrieval, the recovery time course of cypHer signal (red circles) was fitted. The re-acidification time course of the endocytosed organelle was assumed to be an exponential from pH7.4 to pH5.5. The fitting curve is convolution of the Cm time course (bold line) with best fit delay and re-acidification time constant, calculated by least squares method. During the delay, the value was held to be 1. The cypHer signal was best fitted with a 12 s delay with a 18.9 s re-acidification time constant after membrane retrieval (dotted line).
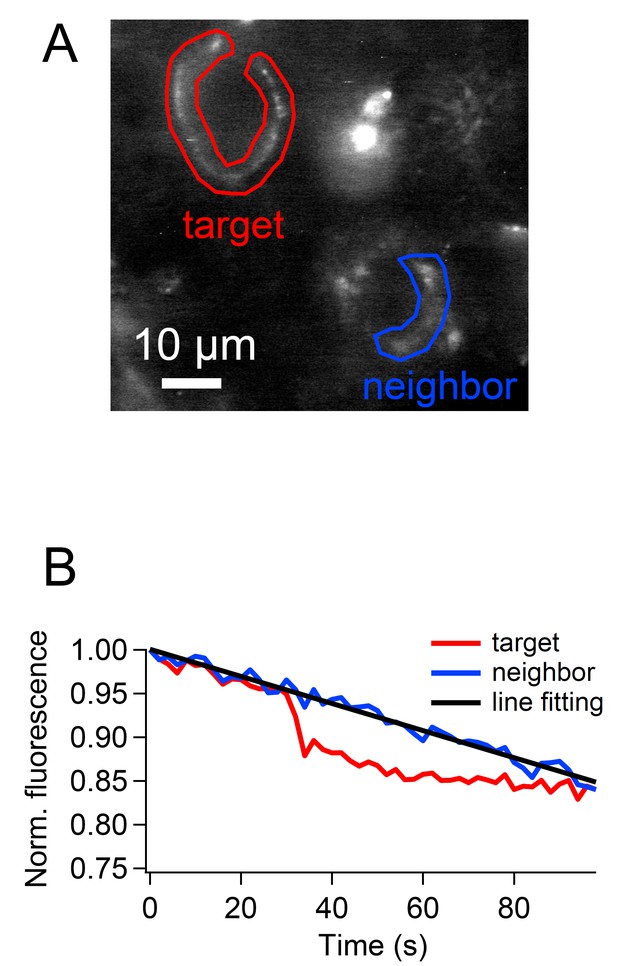
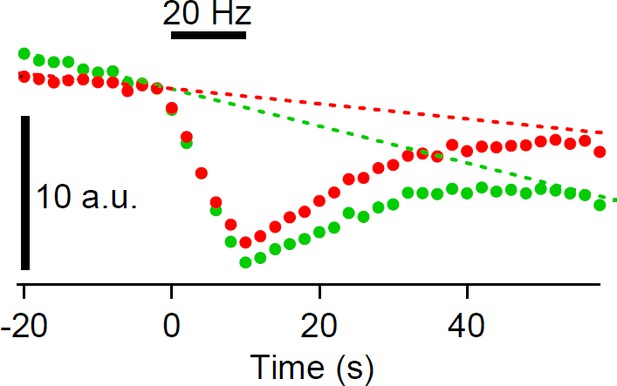
Bleach correction for the cypHer fluorescence.
Bleach was corrected based on the fluorescence of a neighboring unstimulated calyx (A). Fluorescence intensity of the ROI was normalized after background subtraction, and the line (or exponential) was fitted to the neighboring calyx fluorescence. The fitted line (or exponential) was subtracted from the target calyx fluorescence.