Dopamine and opioid systems interact within the nucleus accumbens to maintain monogamous pair bonds
Figures
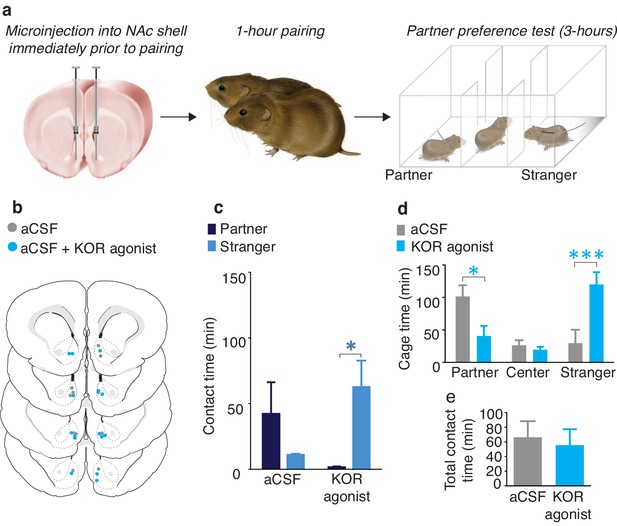
NAc shell KORs encode social aversion.
(a) Experimental design. (b) Histological verification of injection sites. (c) Control males (aCSF) paired with a female partner for 1 hr showed no social preference or aversion (n = 6). In contrast, activation of NAc shell KORs via site-specific administration of a KOR agonist induced a partner aversion (n = 7). (d) Males that received site-specific injections of the KOR agonist also spent significantly less time in the partner’s cage as well as more time in the chamber containing the stranger. (e) There was no difference in total contact time between the two groups. Summary data are presented as mean ± SEM. *p<0.05, **p<0.005.
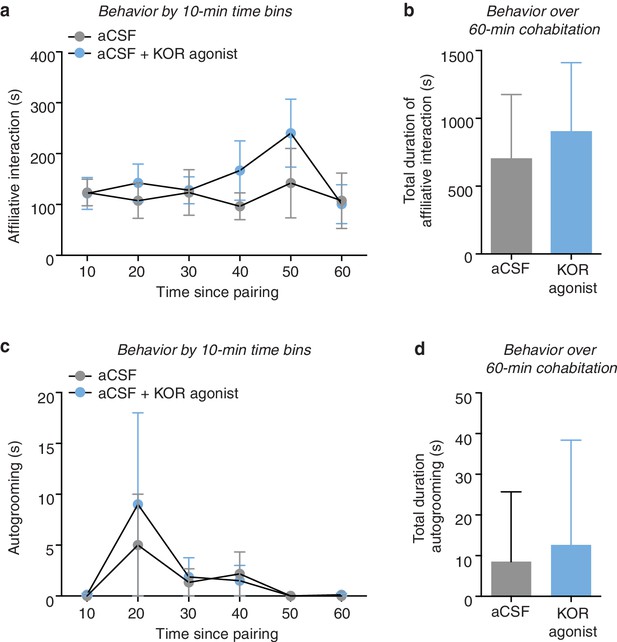
Social and grooming behavior during 1-hr cohabitation.
Prior to partner preference testing, subjects were administered either aCSF (control) or a KOR agonist (1 μg U50,488) into the NAc shell and paired with a female partner for 1 hr. (a, b) Control males (aCSF) and males who were administered a KOR agonist into the NAc shell prior to pairing with the female partner did not differ in the duration of time spent engaging in affiliative behavior at (a) any 10-min time bin (two-way ANOVA, F(1,12) = 0.55, p = 0.47) or (b) over the entire duration of the 60-min pairing (t-test, t(12) = 0.74, p = 0.47). (c, d) Similarly, there was no difference in the duration of time spent autogrooming at (c) any 10-min time bin (two-way ANOVA, F(1,12) = 0.11, p = 0.74) or (d) over the entire duration of the 60-min pairing (t-test, t(12) = 0.34, p = 0.74) (n = 6–8/group).
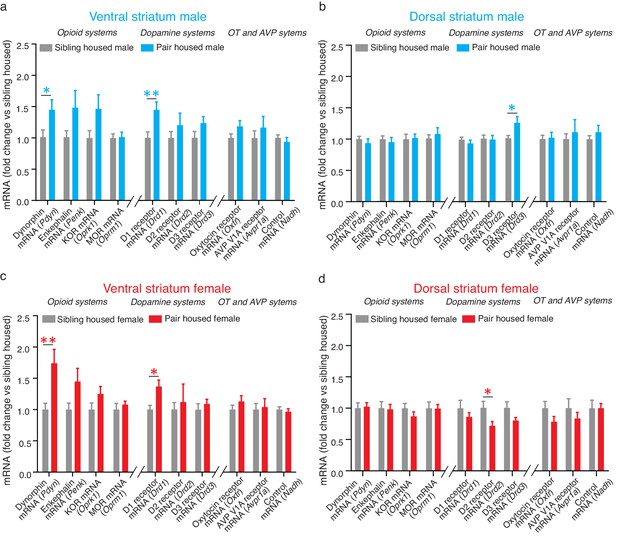
Pair bonding alters mRNA expression within the NAc.
(a) Pair bonding increased the expression of Drd1 and Pdyn mRNA within the VS of males (n = 15/group). (b) Pair bonding decreased Drd3 mRNA expression within the DS of males (n = 15–16/group). (c) Similar to males, pair bonding increased the expression of Drd1 and Pdyn within the VS of females (n = 6–23/group). (d) Pair bonding significantly decreased Drd2 mRNA within the DS of paired females (n = 16/group). *p<0.05, **p<0.005.
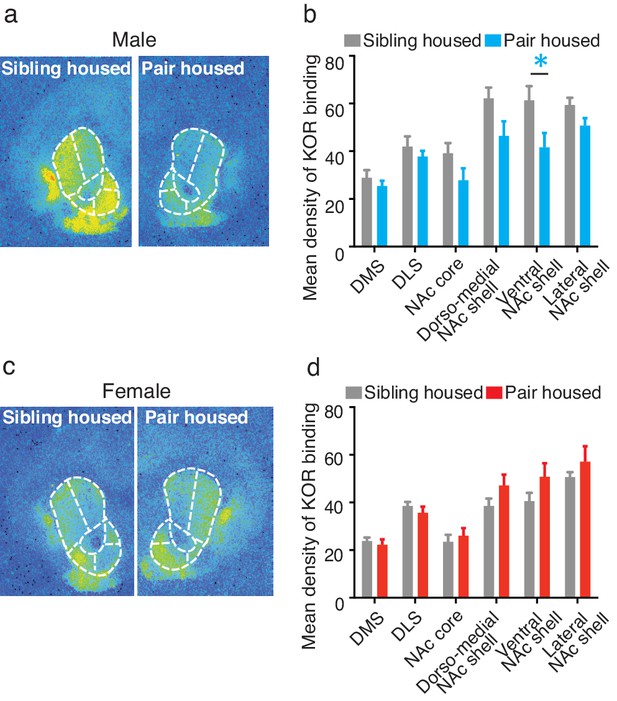
Pair bonding alters KOR binding within the striatum of males.
(a,b) Pair bonding decreased KOR binding in the dorso-medial and ventral NAc shell of males (n = 11/group). (c,d) There was no significant effect on KOR binding density in females (n = 10/group). Summary data are presented as mean ± SEM. *p<0.05.
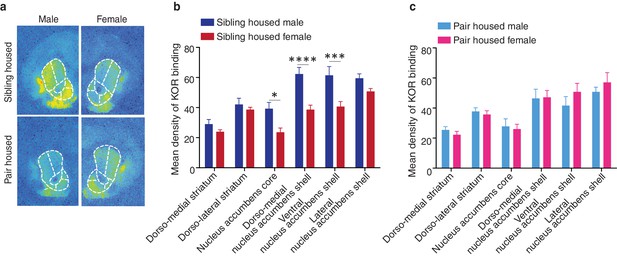
Sex differences in KOR binding density before and after pair bonding.
(a) Representative autoradiographs from sibling (top) and pair housed (bottom) male (left) and female (right) prairie voles. (b) Prior to pair bonding, sibling housed male and female prairie voles show a sex difference in KOR binding density within the striatum (two-way ANOVA, F(1,114) = 38.14, p=0.0001). Specifically, compared to sibling housed females (i.e., non-paired), sibling housed males have higher levels of KOR binding density within ventral regions of the striatum. Bonferroni’s post hoc test revealed that within the striatum, sibling housed males had significantly higher levels of KOR binding density within the NAc core (p=0.02), the dorso-medial region of the NAc shell (p=0.0001), and the ventral NAc shell (p=0.005). (c) Interestingly, following the establishment of a pair bond, these sex differences no longer exist (two-way ANOVA, F(1,114) = 0.36, p=0.55). These results are consistent with findings presented in Figure 3A, which demonstrate that pair bonding decreases KOR binding density within the ventral striatum of males while leaving the KOR binding density in females unaltered. Together, these data suggest that pair bonding in males lowers KOR binding density to the levels of females regardless of the female’s social housing condition. Summary data are presented as mean ± SEM. *p<0.05, **p<0.005, ***p<0.0005.
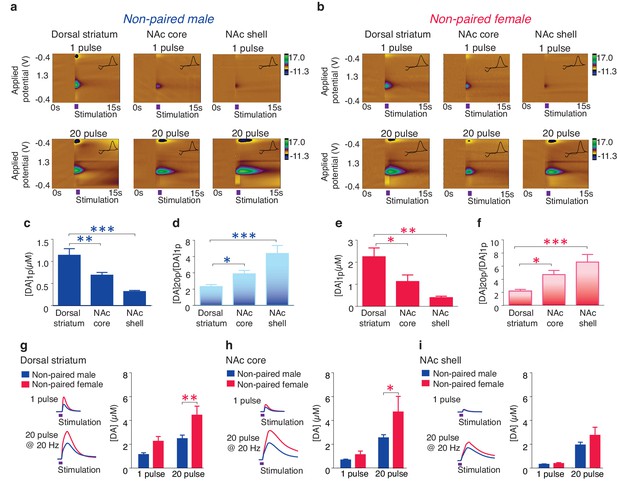
Striatal DA transmission in non-pair bonded prairie voles.
(a,b) Representative color plots of DA transmission throughout the striatum of (a) male and (b) female prairie voles. (c,e) A 1-pulse depolarizing stimulation evokes the greatest magnitude of DA release within the dorsal striatum and the magnitude of this release decreases along a dorsal to ventral gradient within the striatum of (c) males and (e) females. (d,f) An inverse relationship is seen with burst facilitation as the greatest ratio of DA release occurs within the NAc shell, an intermediate ratio occurs within the NAc core, and the lowest ratio occurs within the dorsal striatum of (d) males and (f) females. (g–i) Compared to male prairie voles, a 20-pulse stimulation evokes a greater magnitude of DA release within the (g) dorsal striatum and the (h) NAc core of females. (i) No sex difference in DA transmission occurred within the NAc shell. Summary data are presented as mean ± SEM. *p<0.05, **p<0.005, ***p<0.0005.
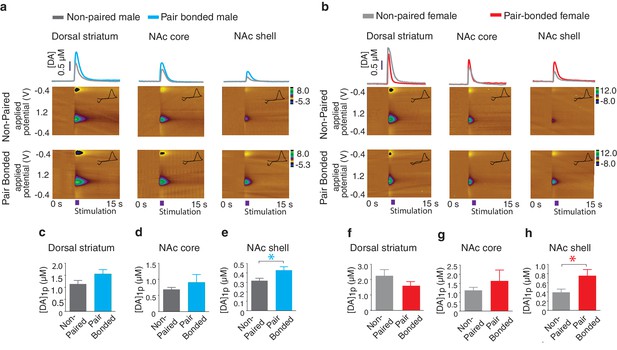
Pair bonding enhances NAc shell DA release.
(a,b) Representative color plots of stimulated DA release following a 1-pulse depolarizing stimulation in (a) male and (b) female subjects. For both sexes, top row shows representative color plots for non-paired subjects and bottom row shows representative color plots for pair bonded subjects. (c,d) Pair bonding had no effect on DA transmission within the (c) dorsal striatum (n = 11–12/group) or (d) the NAc core of males (n = 10/group). (e) Within the NAc shell, a 1-pulse stimulation resulted in significantly greater DA release within the NAc shell of paired males compared to non-paired male controls (n = 9–10/group). (f,g) There was no difference in peak DA release between non-paired and pair bonded females following a 1-pulse stimulation within the (f) dorsal striatum (n = 8–11/group) or (g) the NAc core (n = 8–9/group). (h) Similar to males, a 1-pulse depolarizing stimulation resulted in a greater level of DA release within of the NAc shell of paired females compared to non-paired females (n = 7–8/group). Summary data are presented as mean ± SEM. *p<0.05.
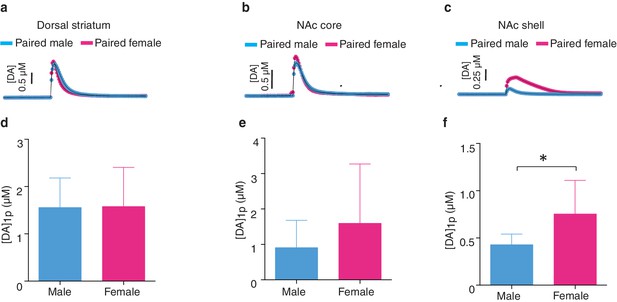
Sex differences in striatal dopamine release following the establishment of a pair bond.
(ac) Representative traces showing changes in DA release following a 1-pulse depolarizing stimulation applied to either the (a) dorsal striatum, (b) the NAc core, or (c) the NAc shell. (d,e) Within both the (d) dorsal striatum (t-test, t(18) = 0.06, p = 0.95; n = 12–8) and the (e) NAc core (t-test, t(16) = 1.16, p = 0.26; n = 10–8), pair bonding did not result in significant differences in peak DA release between paired males and females. (f) However, following the establishment of a pair bond, a 1-pulse depolarizing stimulation resulted in significantly greater levels of peak DA release within the NAc shell of paired females compared to paired males (t-test, t(14) = 2.60, p = 0.02; n = 9–7). Summary data are presented as mean ± SEM. *p<0.05.
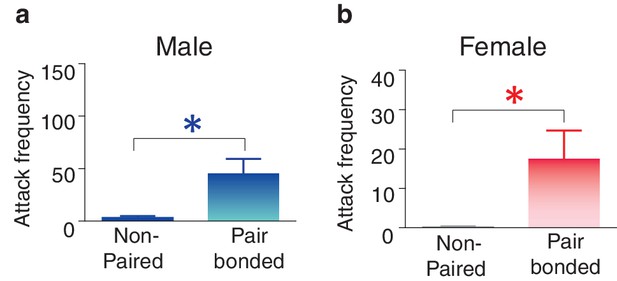
Pair bonding increases selective aggression in both male and female prairie voles.
(a,b) Prior to pair bonding, both males and females show low levels of aggression. However, following the establishment of a pair bond, both (a) males (Mann-Whitney test, U(1,22) = 33, z = −2.26, p = 0.03) (n = 11–13/group) and (b) females increased levels of selective aggression toward novel conspecifics (Mann-Whitney test, U(1, 15) = 11, z = −2.34, p = 0.03) (n = 6–11/group) with males showing qualitatively higher levels of aggression. Summary data are presented as mean ± SEM. *p<0.05.
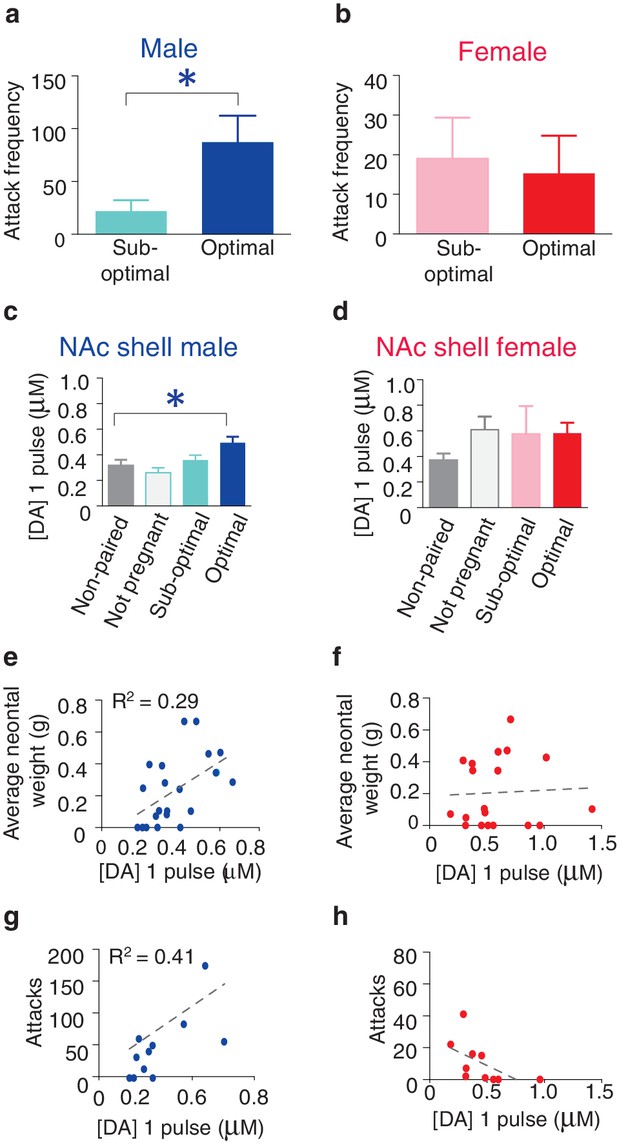
Relationship between striatal DA release and characteristics of pair bonding.
(a) Pair bond induced increases in selective aggression was dependent on fecundity in males as males from optimally pregnant pairs were more aggressive than males from sub-optimally pregnant pairs (n = 5–6/group). (b) Conversely, pregnancy optimality had no effect on attack frequency in females (n = 4–7/group). (c) Within the NAc shell, males whose females were optimally pregnant showed significantly greater levels of DA release (n = 4–18/group). (d) In contrast, for females, there was no difference in peak DA release within the NAc shell between non-paired females and paired females categorized by their reproductive status (n = 5–13/group). (e) Among pair bonded males, neonatal weight (an established indicator of gestational stage) was positively correlated with peak DA release within the NAc shell (n = 23). (f) However, there was no relationship between peak DA release and reproductive status in paired females. (g,h) Finally, in relation to attack frequency, there was a positive correlation between peak DA release and attack frequency within the within the NAc shell of (g) paired males (n = 8), but no such relationship was identified among paired females (n = 10). Summary data are presented as mean ± SEM. *p<0.05.
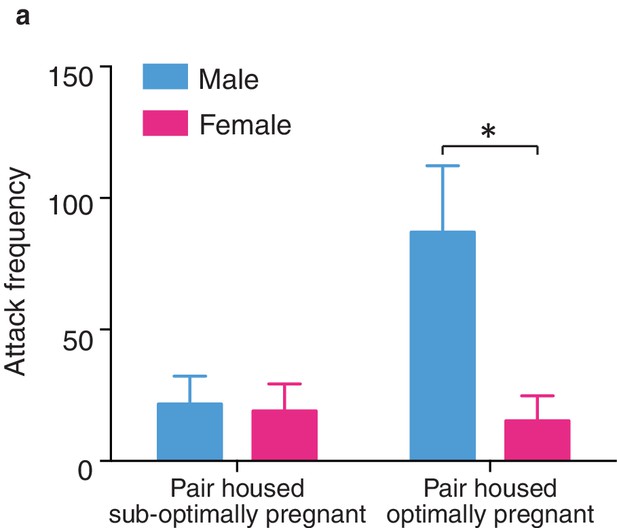
Sex differences in selective aggression by fecundity.
(a) When selective aggression levels were compared between paired males and females based on fecundity (suboptimal versus optimal) sex differences in selective aggression emerged as a function of pregnancy status (two-way ANOVA, F(1,18) = 6.01, p = 0.02) (n = 6–13/group). Specifically, paired males and females categorized as sub-optimally pregnant did not significantly differ in levels of selective aggression (Bonferroni’s post hoc tes, p>0.99). In contrast, males from pairs with optimally pregnant females showed robustly higher levels of selective aggression than paired females categorized as optimally pregnant (p = 0.01) (n = 4–7/group). Summary data are presented as mean ± SEM. *p<0.05.
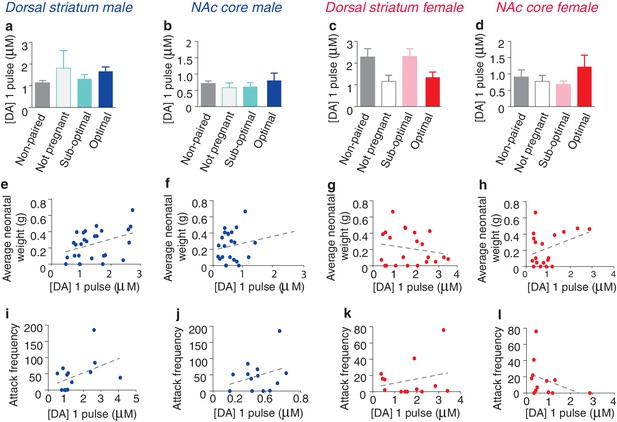
Impact of fecundity on dopamine transmission within the dorsal striatum and NAc core of paired male and female prairie voles.
(a,b) Within the striatum of paired males, males categorized by their partner’s stage of pregnancy did not differ in peak DA release within the (a) dorsal striatum (n = 4–22/group) or (b) the NAc core (n = 3–19/group). (c,d) Similarly, no differences were found within the (c) dorsal striatum (n = 22) or (d) NAc core (n = 19) of paired females. (e) Although categorization of males by their pregnancy status did not result in any significant differences in DA transmission within the dorsal striatum, there was a positive correlation between peak DA transmission and neonatal weight within this region (n = 26). (f) In contrast, there was no relationship between peak DA release and neonatal weight within the NAc core of paired males (n = 24). (g,h) There was no relationship between peak DA release and neonatal weight within the (g) dorsal striatum (n = 22) or (h) NAc core (n = 19) of paired females. (i,j) For paired males, there was no relationship between attack frequency and peak DA release within the (i) dorsal striatum (n = 14) or (j) NAc core (n = 14). (k,l) Similarly, there was no relationship between attack frequency and stimulated DA release within the (k) dorsal striatum (n = 13) or the (l) Nac core (n = 12) of paired females. Summary data are presented as mean ± SEM.
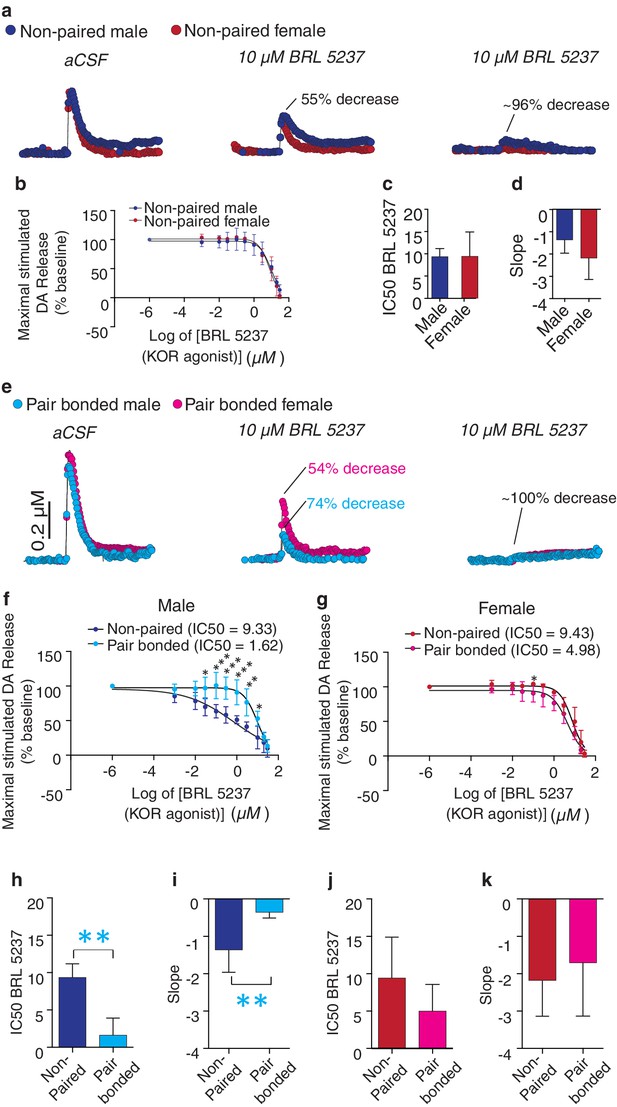
Pair bonding increases KOR modulation of NAc shell DA release in male prairie voles.
(a,b) Similar to other species, bath application of a KOR agonist decreases DA release in the NAc shell of male and female prairie voles. (c,d) Non-paired males and females did not differ in (c) the IC50 of BRL 5237 (a KOR agonist) or (d) in the slope of the dose response curve. (e) Pair bonding induced sex-specific alterations in KOR modulation of DA transmission within the NAc shell (f,g) Following the establishment of a pair bond, KOR mediated decrease of stimulated DA release was enhanced within the NAc shell of (f) males (n = 3–5/group), but not (g) females. (h,i) Compared to non-paired males, pair bonding significantly decreased (h) the IC50 of BRL 5237 as well as (i) the slope of the dose response curve in paired males. (j,k) Pair bonding did not alter KOR mediated DA transmission in females (n = 3–4/group). Summary data are presented as mean ± SEM. **p<0.005.

Comparison of the prairie vole KOR protein sequence to other rodent species and humans.
(a) Cladogram showing that the prairie vole (Michrotus ochrogaster) KOR is distinct from that of guinea pigs (Cavia porcellus), humans (Homo sapiens), rats (Rattus Norvegicus), and mice (Mus musculus). (b) Protein sequence comparison of the prairie vole KOR to the above listed species. * indicates completely conserved regions of the gene between all species shown here, + indicates regions of the gene that are similar between prairie voles, humans, and guinea pigs, but not rats or mice, and the # indicates portions of the KOR sequence that are unique to prairie voles. Consistent with other opioid receptors, the transmembrane regions of the gene (black lines) are highly conserved between species. In contrast, the N-terminus region of the KOR gene contains the most protein sequence differences between prairie voles and the other species shown. Notably, there is one portion of the dynorphin binding site, located in extracellular loop 2, where a threonine has been substituted for a valine, which is unique to the prairie vole KOR.
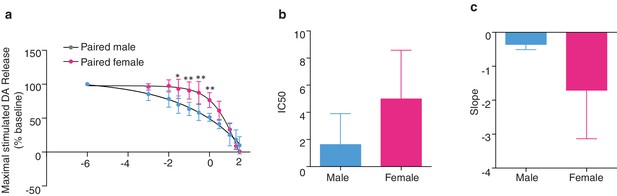
Sex differences KOR modulation of NAc shell DA release following the establishment of a pair bond.
(a) Comparison of dose response curves between pair bonded males and females (n = 4–5/group). (b–c) Although pair bonding significantly altered KOR modulation over DA release in paired males, but not paired females, these groups did not significantly differ in the IC50 of BRL5237 (a KOR agonist) (t(7) = 1.72, p = 0.13) or in the slope of the dose response curve (t(7) = 2.14, p = 0.07).
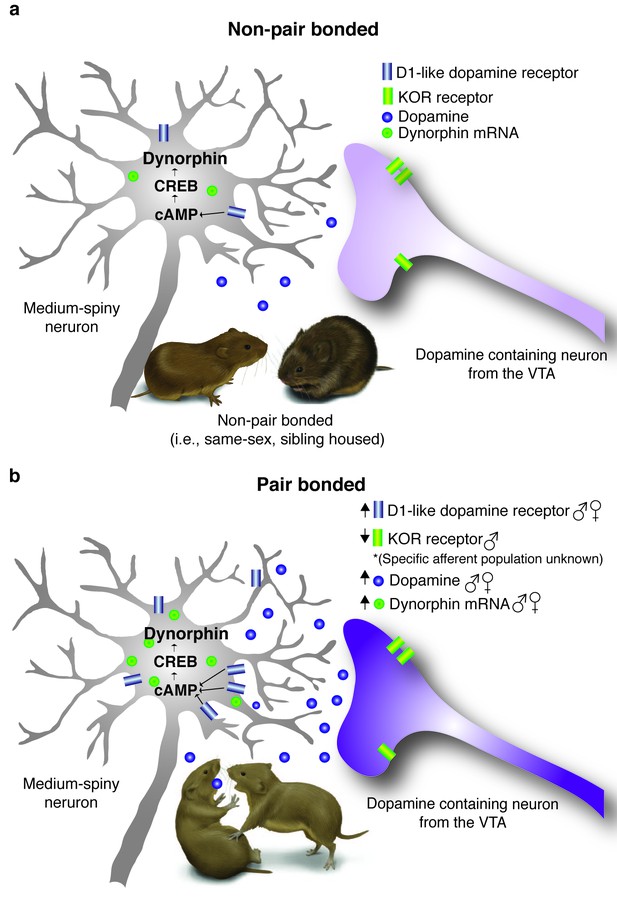
Pair bonding alters DA and dynorphin/KOR systems within the ventral striatum.
(a) Non-pair bonded prairie voles readily approach novel conspecifics and have lower levels of stimulated DA release as well as Drd1 and Pdyn mRNA expression within the ventral striatum. (b) Following the establishment of a pair bond, male and female prairie voles aggressively reject novel conspecifics and the ventral striatum undergoes a dramatic reorganization. Specifically, pair bonding enhances DA release within the NAc shell as well as up-regulates Drd1 as well as Pdyn within the ventral striatum of both males and females. Pair bonded males also show an additional decrease in KOR binding within the NAc shell.
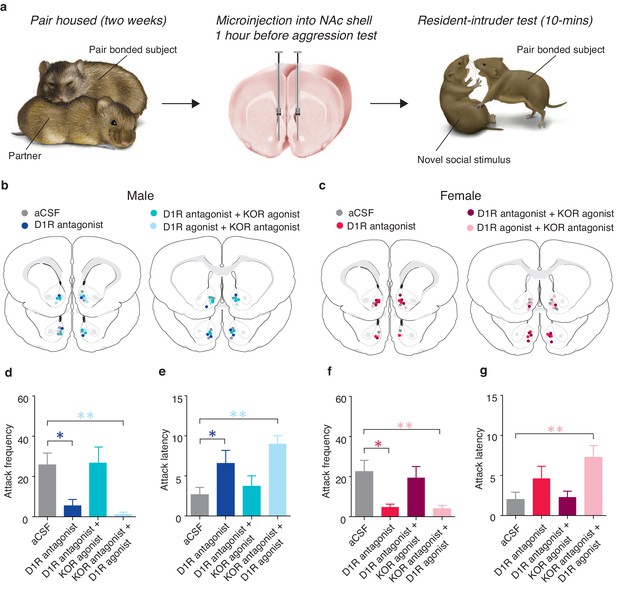
Interactions between D1-like and KORs mediate pair bond maintenance.
(a) Experimental Design. (b,c) Histological location of injection sites in (b) males and (c) females. (d) Compared to control pair-bonded males that received site-specific infusions of aCSF prior to resident-intruder testing (n = 6), males that received site-specific infusions of a D1-like receptor antagonist into the NAc shell showed attenuated levels of selective aggression as well as (e) increased attack latency toward intruders (n = 6). However, aggression levels and attack latencies were returned to normal when the antagonist for the D1-like receptor was administered in combination with a KOR agonist (n = 7) suggesting that D1-mediated aggression occurs through downstream activation of KORs. This interaction was confirmed by the ability of the KOR antagonist to attenuate selective aggression even when it was administered in combination with the D1-like receptor agonist (n = 7). (f,g) Similar to males, blockade of D1-like receptors within the NAc shell of paired females (n = 6) attenuated selective aggression compared to aCSF controls (n = 6). Aggression frequency was returned to the level of paired female controls when the D1-like receptor antagonist was administered in combination with a KOR agonist (n = 7). Finally, the attenuation of attack frequency and the increase in attack latency mediated by a KOR antagonist was maintained even in the presence a D1-like receptor agonist (n = 6). Summary data are presented as mean ± SEM. *p<0.05, **p<0.005.
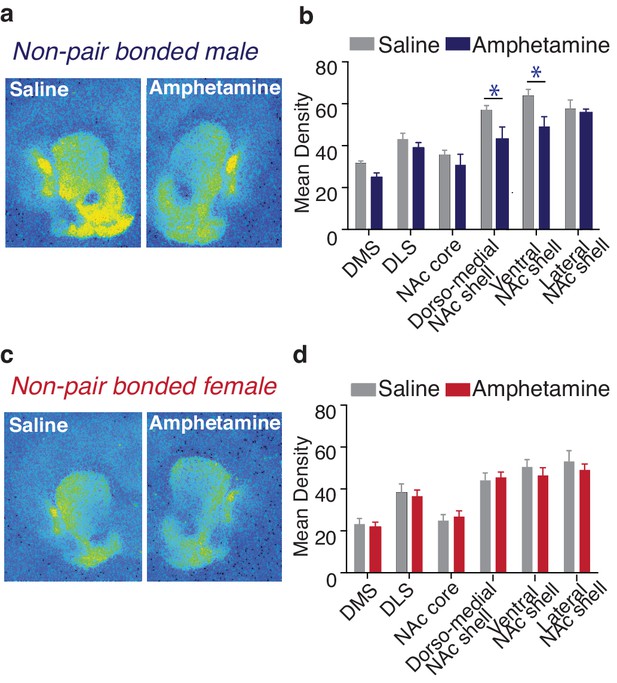
Amphetamine decreases KOR binding within the striatum of males.
(a,b) AMPH decreased KOR binding within the dorso-medial and ventral NAc shell of non-pair bonded males (n = 7–8/group). (c,d) Similar to pair bonding, AMPH did not impact striatal KOR binding in females (n = 7–8/group). Summary data are presented as mean ± SEM. *p<0.05.
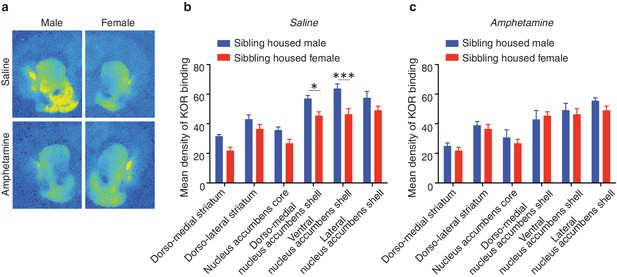
Sex differences in prairie vole KOR binding density following amphetamine exposure.
(a) Representative autoradiographs from saline (top) and amphetamine (1 mg/kg) (bottom) conditioned male (left) and female (right) prairie voles. For all subjects, saline (control) or amphetamine was administered once per day for 3 consecutive days. (b) KOR binding density varied as a function of sex and treatment (two-way ANOVA, F(1,96) = 39.80, p = 0.0001). Specifically, control males had significantly higher levels of KOR binding within the dorso-medial NAc shell (p = 0.03) as well as the ventral region of the NAc shell (p = 0.0002). (c) Interestingly, similar to pair bonding, repeated amphetamine exposure eliminated these sex differences in KOR binding density between sibling housed males and females (two-way ANOVA, F(1,90) = 1.86, p=0.17) (n = 7–10/group). *p<0.05, 4***p<0.0005
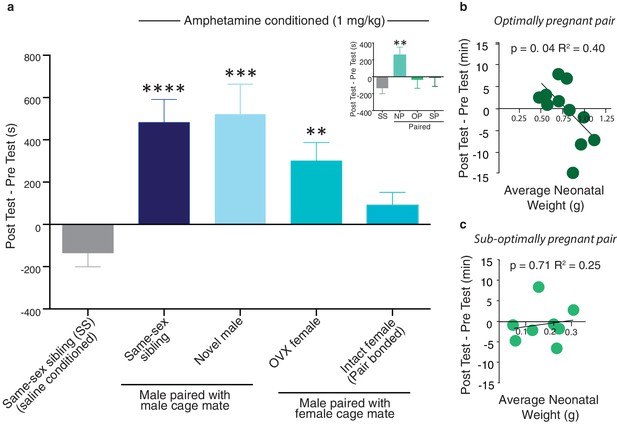
Neural protection against drug reward is specific to pair bonding in males.
(a) Male prairie voles were housed with a familiar cage mate, a novel male, an ovariectomized (OVX) female, or an intact female for two weeks prior to AMPH conditioning. Compared to saline treated males, all groups except males housed with an intact female formed a preference for the AMPH paired chamber (n = 6–33/group). To determine if pregnancy status influenced the rewarding properties of AMPH, males housed with an intact female were further classified by the pairs pregnancy status (inset). Only males paired with a female that became pregnant (suboptimally (SP) or optimally (OP)) during the 2-week pairing period were protected against the rewarding properties of amphetamine as males paired with females that were not pregnant (NP) formed a preference for the AMPH paired chamber. (b) The establishment of an optimal pregnancy strongly influenced the rewarding properties of AMPH as there was a negative correlation between the duration of time spent in the AMPH paired chamber and the gestational stage of the female for optimally pregnant pairs (n = 11). (c) In contrast, there was no relationship between pregnancy stage and AMPH preference for sub-optimally pregnant pairs (n = 8). Summary data are presented as mean ± SEM. *p<0.05, **p<0.005, ***p<0.0005.
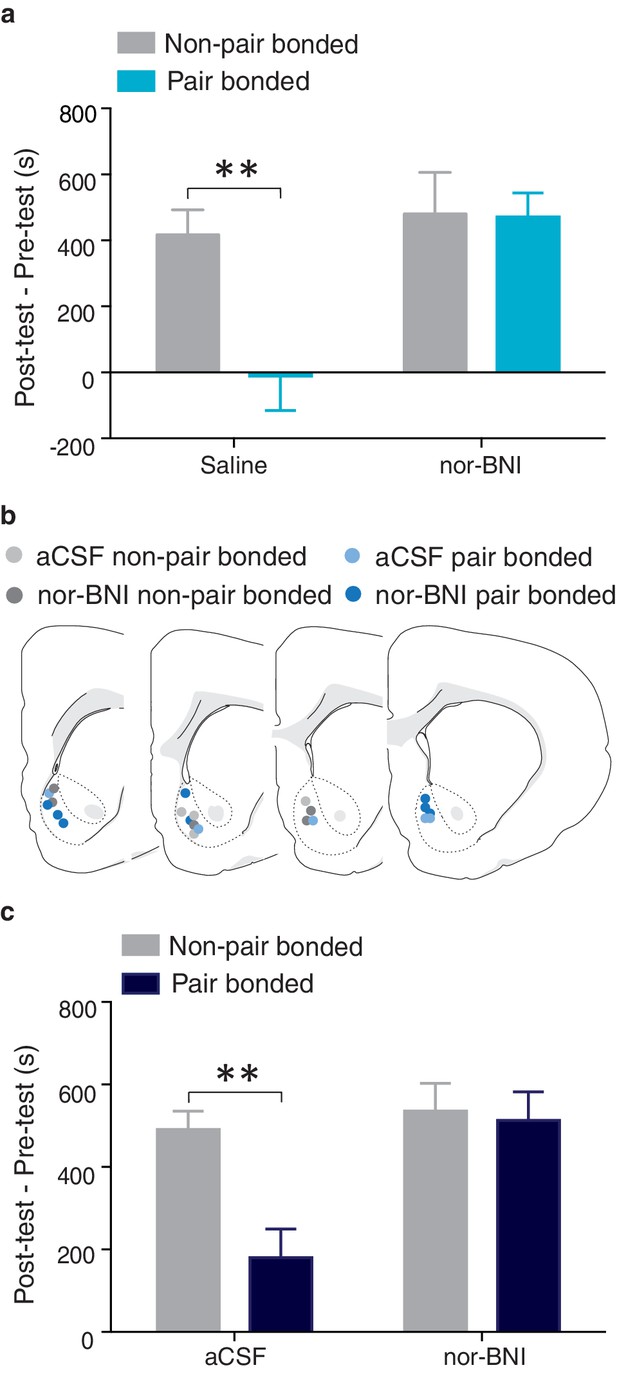
NAc shell KORs mediate the protective effects of pair bonding.
(a) Peripheral administration of nor-BNI restored the rewarding properties of AMPH for paired males (n = 6–14/group). (b) Histological location of injection sites. (c) Site-specific blockade of NAc shell KORs was sufficient to alleviate pair bond induced attenuation of AMPH reward (n = 4–7/group). Summary data are presented as mean ± SEM. **p<0.005.
Tables
Non-significant statistics for mRNA comparisons in the ventral striatum.
Sex | ||
|---|---|---|
| Gene | Male | Female |
| Pdyn | NA | NA |
| Penk | t(24) = 1.80, p = 0.09 | t(26) = 1.92, p = 0.07 |
| Oprk1 | t(24) = 1.99, p = 0.06 | t(11) = 0.36, p = 0.72 |
| Oprm1 | t(24) = 0.13, p = 0.90 | t(26) = 0.70, p = 0.49 |
| Drd1 | NA | NA |
| Drd2 | t(24) = 0.10, p = 0.33 | t(37) = 1.57, p = 0.13 |
| Drd3 | t(24) = 1.58, p = 0.13 | t(26) = 0.75, p = 0.46 |
| Oxtr | t(24) = 1.72, p = 0.10 | t(37) = 1.12, p = 0.27 |
| Avpr1a | t(24) = 0.82, p = 0.43 | t(37) = 0.25, p = 0.81 |
| Nadh | t(23) = 1.23, p = 0.23 | t(28) = 0.79, p = 0.44 |
Non-significant statistics for mRNA comparisons in the dorsal striatum.
Sex | ||
|---|---|---|
| Gene | Male | Female |
| Pdyn | t(26) = 0.80, p = 0.43 | t(26) = 0.21, p = 0.83 |
| Penk | t(26) = 0.56, p = 0.58 | t(26) = 0.13, p = 0.90 |
| Oprk1 | t(26) = 0.17, p = 0.86 | t(26) = 1.19, p = 0.24 |
| Oprm1 | t(26) = 0.63, p = 0.53 | t(26) = 0.05, p = 0.96 |
| Drd1 | t(26) = 1.15, p = 0.26 | t(26) = 0.88, p = 0.39 |
| Drd2 | t(26) = 0.18, p = 0.86 | NA |
| Drd3 | NA | t(26) = 1.69, p = 0.10 |
| Oxtr | t(26) = 0.20, p = 0.84 | t(26) = 1.50, p = 0.15 |
| Avpr1a | t(26) = 0.56, p = 0.58 | t(26) = 0.85, p = 0.40 |
| Nadh | t(26) = 0.97, p = 0.34 | t(26) = 0.005, p = 0.10 |
Non-significant statistics for comparisons of KOR binding density in paired versus unpaired males.
| Striatal sub-region | Bonferonni's post hoc test |
|---|---|
| Dorso-medial striatum | p>0.99 |
| Dorso-lateral striatum | p>0.99 |
| NAc core | p = 0.41 |
| Nac dorso-medial shell | p = 0.07 |
| NAc lateral shell | p = 0.99 |
Confidence intervals for IC50’s in KOR agonist dose response study.
| Group | 95% Confidence interval |
|---|---|
| Male non-pair bonded | 4.761818 to 13.906848 |
| Male pair bonded | −1.2093417 to 4.4570177 |
| Female non-pair bonded | 0.73751 to 18.11349 |
| Female pair bonded | −0.73394 to 10.70294 |
Non-significant statistics for comparisons of KOR binding density in saline versus amphetamine treated males.
| Striatal sub-region | Bonferonni's post hoc test |
|---|---|
| Dorso-medial striatum | p>0.99 |
| Dorso-lateral striatum | p>0.99 |
| NAc core | p>0.99 |
| Dorso-lateral striatum | p>0.99 |





