Analysis of cellular behavior and cytoskeletal dynamics reveal a constriction mechanism driving optic cup morphogenesis
Figures
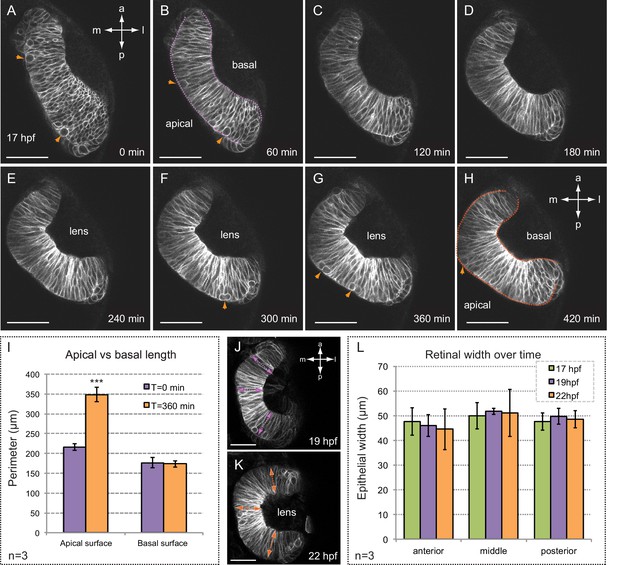
Folding of the retinal epithelium in zebrafish.
(A–H) Time series of optical sections show the progression of retinal morphogenesis starting at 17 hpf (dorsal view) in a tg(vsx2.2:GFP-caax) embryo. Arrowheads point to mitotic divisions at the apical surface. Apical and basal edges are indicated at 60 (purple) and 420 (orange) min. See also Video 1. (I) Quantification of the perimeter of the apical and basal edges between 18 and 24 hpf. (J–L) Retinal width remains constant throughout retinal folding as revealed in tg(vsx2.2:GFP-caax) embryos. Error bars indicate s.d. of the mean. (n = 3; T-test). Antero-posterior and medio-lateral axes are indicated. Scale bars = 50 µm.
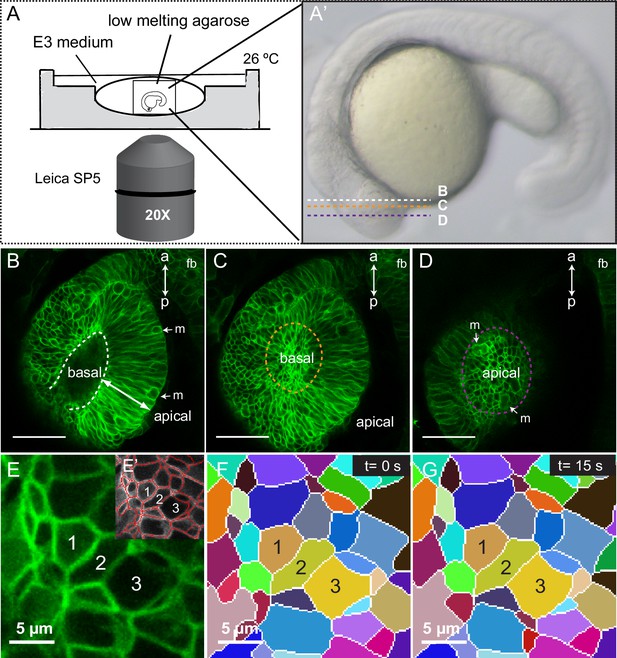
Imaging setup and segmentation.
(A, A’) Schematic representation of the imaging setup. Confocal planes for panels B–D are indicated in A’. (B–D) Optical sections through a 20 hpf tg(vsx2.2:GFP-caax) retina showing basal (orange in C) and apical (purple in D) planes. Mitotic figures (m) and antero-posterior axis (a–p) are indicated. fb = forebrain. (E–G) Automatic cell segmentation (E–E’) and manual tracking of the segmented cells through time (F, G) are shown. Scale bars = 50 µm in B–D and 5 µm in E–G.
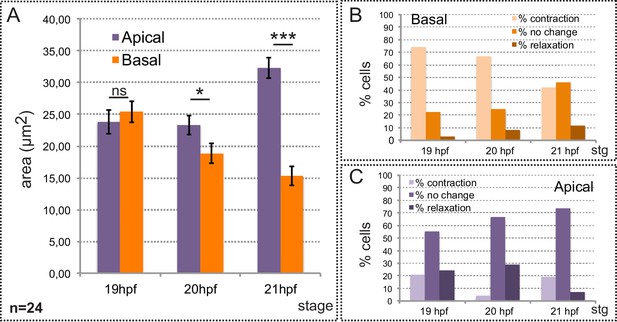
Neuroblasts’ area quantification during eye morphogenesis.
(A) Quantification of average cell areas at the apical and basal sides at 19, 20 and 21 hpf. A total of 24 cells from three different embryos were recorded either at the apical or at the basal side. Error bars indicate SE of the mean (n = 24). Statistical significance was determined after T-test. (B–C) The percentage of cells showing a contraction, or relaxation larger than 20% over a 25 min period is indicated for the three different stages. A total of 24 cells were monitored at both basal (B) and apical (C) surfaces.
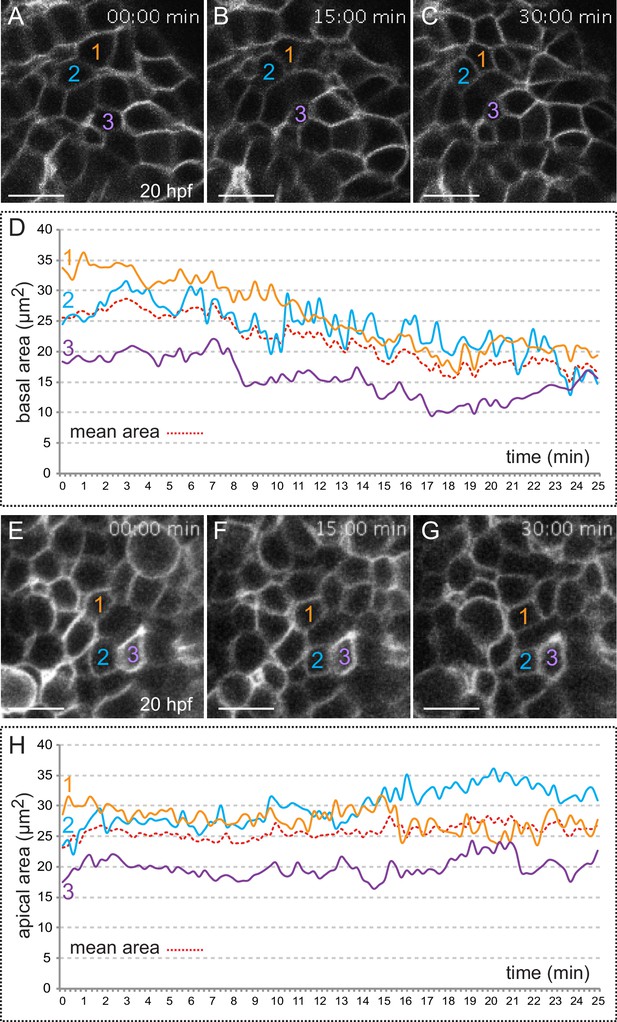
Quantitative analysis of membrane oscillations in tg(vsx2.2:GFP-caax) embryos.
Cell area dynamics at the basal (A–-D) and apical (E–H) surfaces is shown for three individual cells (color coded). Absolute basal (D) and apical (H) areas in µm2 are represented versus time for the individual cells. The mean area indicates a progressive constriction of the basal, but not apical surfaces over time (D, H). Scale bars = 10 µm.
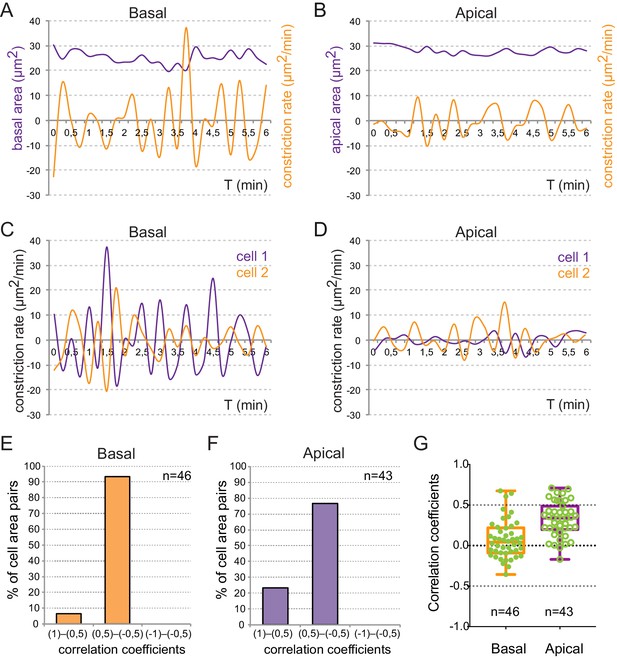
Quantitative analysis of cell pulses.
(A–-B) Single cell recordings of area variations in µm2 (purple) and constriction changes in µm2/min (orange) at the basal (A) and apical (B) surfaces are represented over time. (C—D) The evaluation of constriction rates in adjacent cells shows asynchronous pulsing. (E–G) Distribution of correlation coefficients between neighboring cell pairs is represented as bins for basal (E) and apical (F) oscillations, as well as in a box plot (G).
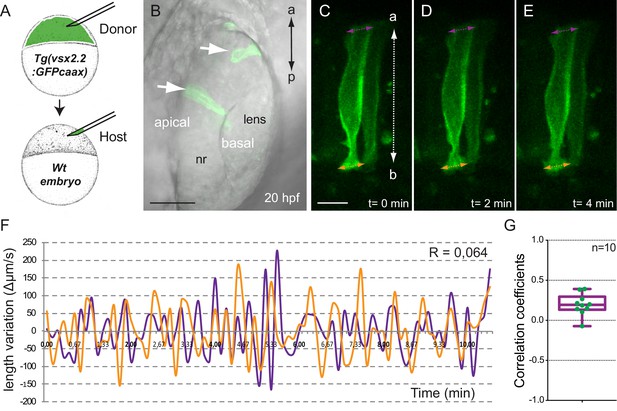
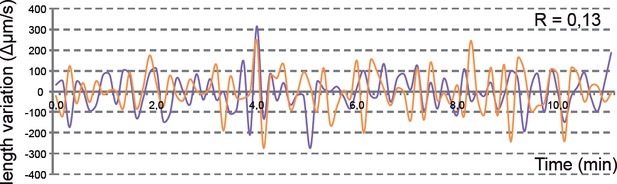
Analysis of tg(vsx2.2:GFP-caax) clones show uncoupled oscillations at apical and basal surfaces.
(A) Scheme of transplantation experiment at sphere stage. (B) Confocal microscopy image showing transmitted light and GFP expression for transplanted clones (white arrows) at 20 hpf. Antero-posterior (a–p) axis is indicated. (C–E) Confocal microscopy time-lapse images show length variation of basal (orange) and apical (purple) edges through time in a transplanted clone. The orientation of the apico-basal (a–b) axis is indicated. Scale bars = 50 µm in B and 10 µm in C–E. (F) Quantification of the basal (orange) and apical (purple) length variation for an individual clone showing no correlation between the oscillations (R = 0064). (G) Box plot showing the distribution of apical vs basal oscillations correlation coefficients for 10 transplanted neuroblasts from five different retinas.
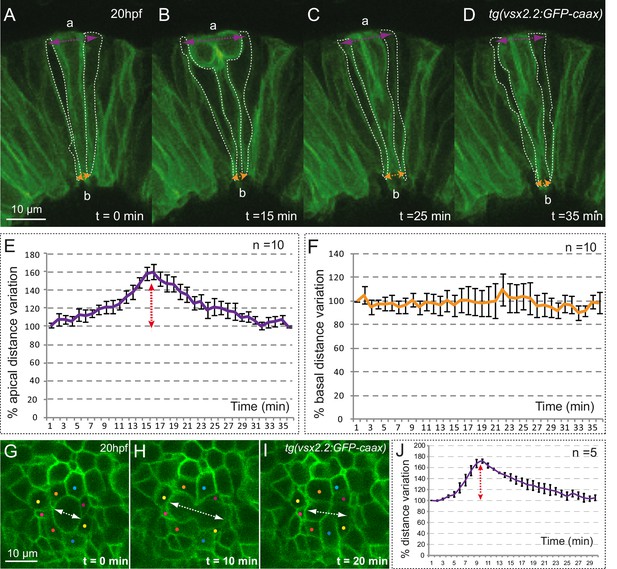
Mitotic rounding impact on basal constriction and apical expansion.
(A–D) Confocal microscopy time-lapse images showing a mitosis in a tg(vsx2.2:GFP-caax) retina at 20 hpf. Dashed white lines highlight flanking cells. Arrows indicate apical (purple) and basal (orange) distance variation. The orientation of the apico-basal (a–b) axis is indicated. (E–F) The graphs show the quantification of distance variation (%) for the apical (E) and basal (F) sides. The mitotic event (red arrow) results only in a transient expansion of the apical domain. Error bars indicate standard error of the mean (n = 10, from three different retinas). (G–I) Confocal microscopy time-lapse images showing a mitosis occurring in the apical plane in a tg(vsx2.2:GFP-caax) retina at 20 hpf. Dashed white arrows indicate apical distance variation along the mitotic axis. Neighboring cells are indicated with colored dots. (J) Quantification of apical distance variation (%) along the mitotic axis for five different cells confirms a transient expansion of the apical domain. Scale bars = 10 µm.
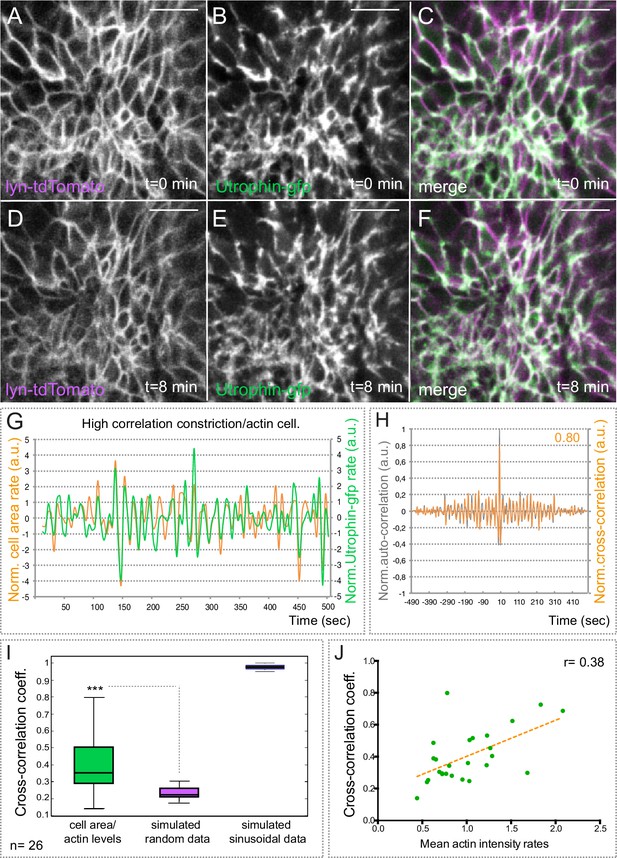
Basal actin dynamics in constricting retinal cells.
(A–F) Actin dynamics, as revealed by utrophin-gfp, and membrane oscillations were simultaneously examined by time lapse in the line tg(vsx2.2:lyn-tdTomato) at 20 hpf (see Video 4). Note that F-actin localizes mainly at the cellular cortex. Scale bars = 10 µm. (G) Normalized basal area rate (orange) and normalized utrophin-gfp rate (green) are shown over time for a cell displaying a high correlation between actin oscillations and membrane expansion. Area rate and Utrophin-gfp rate were normalized dividing by the mean of their absolute values. (H) Normalized auto-correlation (grey line) and cross-correlation (orange) are shown for cell represented in G. Maximum cross-correlation (0.8) is indicated. (I) Box plot comparison of cross-correlation results between actin vs. membrane oscillations, simulated random and simulated sinusoidal signals shows a significant (p<0.001; T-test; n = 26) positive correlation between actin accumulation and basal area expansion. (J) Scattered plot showing the dependency of cross-correlation coefficients (n = 26) on mean actin intensity rates. Linear regression line (orange) and linear correlation coefficient (0.38) are indicated.
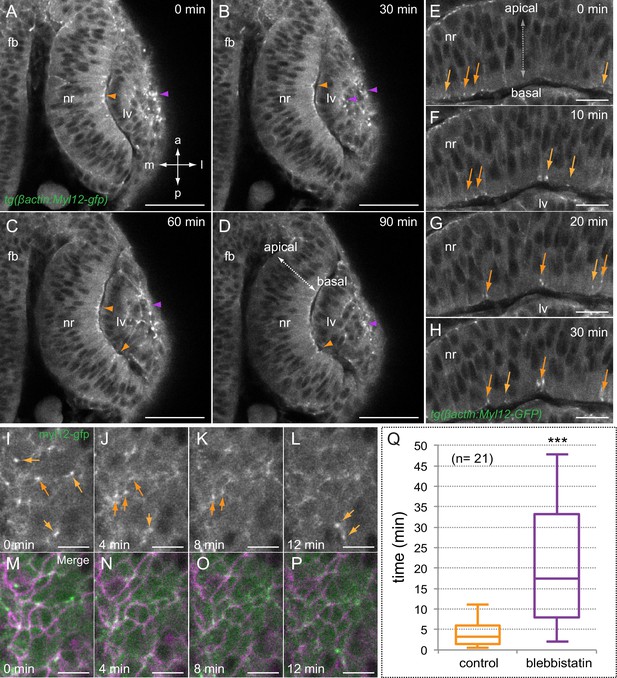
Myosin accumulates in basal foci during optic cup morphogenesis.
(A–D) Live-imaging analysis of tg(actb1:myl12.1-eGFP) embryos reveals myosin accumulation at the apical lens (purple arrowheads) and basal retina (orange arrowheads) between 19 and 20.5 hpf. Antero-posterior (a–p) and medio-lateral (m-l) axes are indicated. (E–H) Myosin accumulates in transient foci (orange arrows) at the basal cortex. (I–P) Time-lapse analysis of myosin foci at the basal surface plane in embryos injected with lyn-tdTomato RNA reveals that the protein accumulates at the peripheral cortex in scattered cells. (Q) The box plot shows a significant difference in foci stability between control and blebbistatin (150 µM) treated embryos (T-test, n = 21). fb = forebrain; nr = neural retina; lv = lens vesicle. Scale bars = 50 µm in A–D, 20 µm in E–H, and 10 µm in I–P.
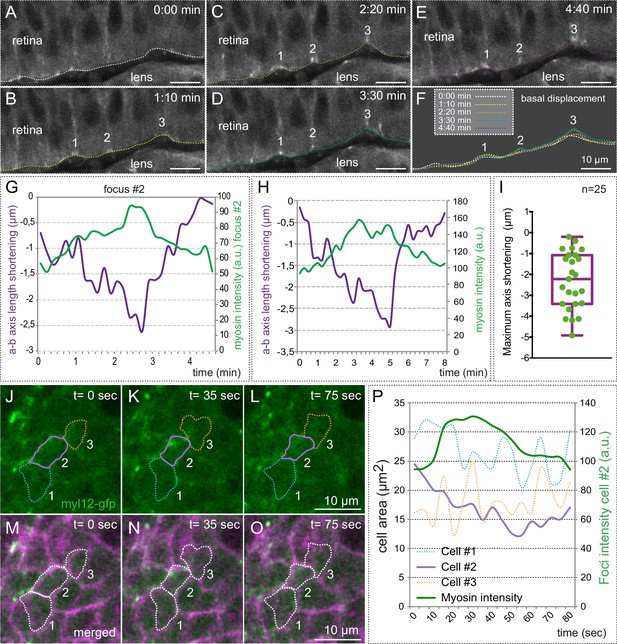
Myosin accumulation correlates with basal membrane displacement.
(A–E) Time series of optical sections from tg(actb1:myl12.1-eGFP) embryos show discrete myosin foci (labeled 1, 2, 3) and basal surface displacement. (F) Basal edges were color-coded for each time point and overlapped to illustrate the transient indentations of the basal surface associated to myosin foci. (G–H) Quantitative recording over time of myosin intensity and apico-basal axis shortening for a couple of representative foci. The focus in G is #2 in A–F. (I) Box plot showing the maximum shortening of the a-b axis for 25 foci from 12 different retinas. (J–O) Correlative analysis of basal area (revealed by lyn-tdTomato) and myosin dynamics is shown for three neighbor cells (color-coded). (P) Quantitative analysis of cell area changes and myosin intensity for the three neighboring cells. Note that only the cell accumulating myosin contracts. Scale bars =10 µm.
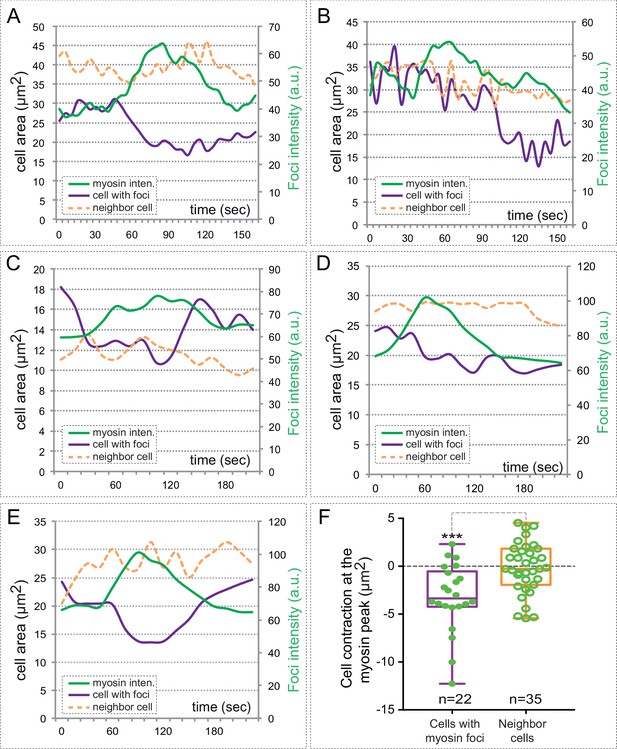
Myosin accumulation correlates with basal contraction.
(A–E) Quantitative analysis of myosin intensity (green lines) and cell area changes for five cells containing myosin foci (purple lines) and their neighboring cells (orange dashed lines). Note the contraction of the cells upon myosin accumulation. (F) Box plot showing average cell contraction (µm2) at the peak of myosin accumulation for 22 different cells containing myosin foci (purple) and 35 neighboring cells. Myosin accumulating cells undergo a significant contraction of their basal area, as determined by T-test.
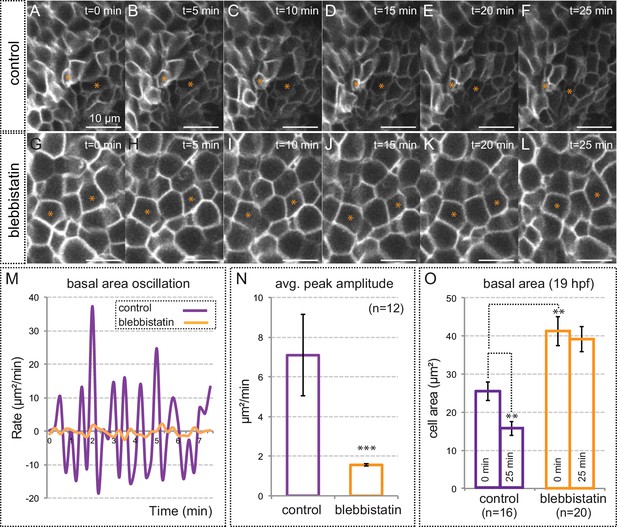
Myosin inhibition impairs basal constriction.
(A–L) Live-imaging analysis of cell area dynamics in control (A–F) and blebbistatin-treated (G–L) tg(vsx2.2:GFP-caax) embryos. Progressive constriction is observed in individual cells (asterisk) in control, but not in blebbistatin-treated tissue. (M) Basal area variation rate is shown for representative control and blebbistatin-treated cells. (N) Average peak amplitude of the cell area rate is considerably reduced in treated cells (T-test, n = 12). (O) Blebbistatin treatment significantly inhibited basal constriction over a considered period of 25 min, blocking the cells in a relaxed state (T-test). Scale bars = 10 µm.
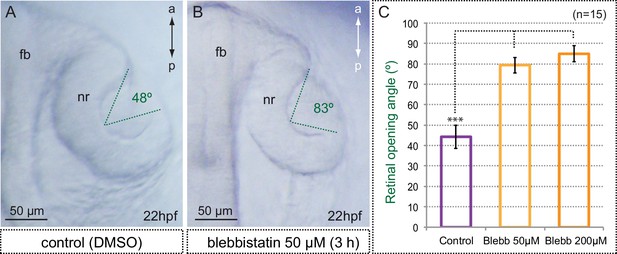
Myosin inhibition interferes with optic cup folding.
(A,B) Optic cup folding is also impaired in blebbistatin-treated embryos as assessed by the retinal opening angle (indicated with green dashed lines). (C) Quantitative analysis of retinal opening angles show a significant delay in optic cup folding in embryos treated with 50 and 200 µM blebbistatin (one-way ANOVA followed by Tukey test, n = 15). fb = forebrain; nr = neural retina. Scale bars = 50 µm.
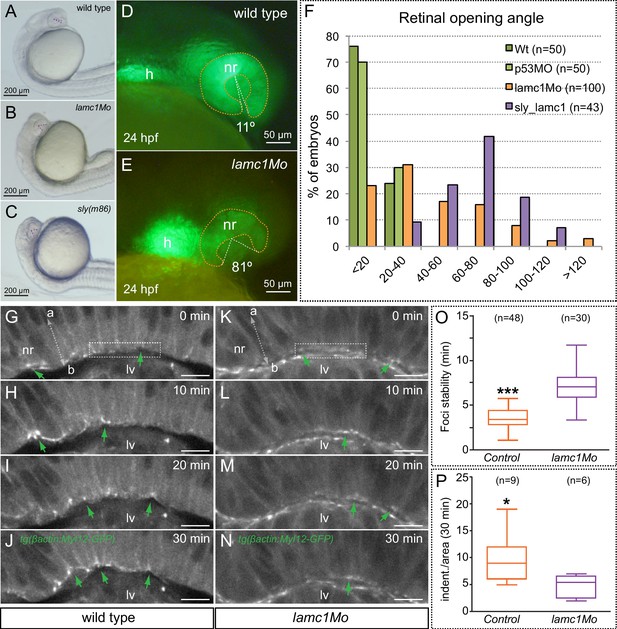
Optic cup folding, basal contractility and myosin dynamics depend on lamc1 function.
(A–C) General embryo morphology for wild type, lamc1 morphants and sly (lamc1-/-) mutants at 24 hpf. Retinal opening is indicated with a dashed line. (D–E) Retinal morphology in tg(vsx2.2:GFP-caax) both wild type and lamc1Mo-injected embryos, at 24 hpf. Ventral opening angle (white) and retinal contour (orange) are indicated with dashed lines. (F) Frequency distribution of retinal opening angles is shown for controls (either wild type or p53Mo-injected), lamc1Mo injected, or sly mutants. (G–N) Time-lapse analysis of tg(actb1:myl12.1-eGFP) wild type and lamc1Mo-injected embryos show dynamic accumulation of myosin foci (green arrows) at the basal surface. (O) Analysis of myosin foci reveals that they are significantly more stable in lamc1Mo-injected embryos (T-test). (P) The box plot shows that transient indentations of the basal surface are significantly diminished in lamc1Mo-injected embryos (T-test). h = heart; nr = neural retina; lv = lens vesicle. Scale bars = 200 µm in A–C, 50 µm in D–E, and 10 µm in G–N.
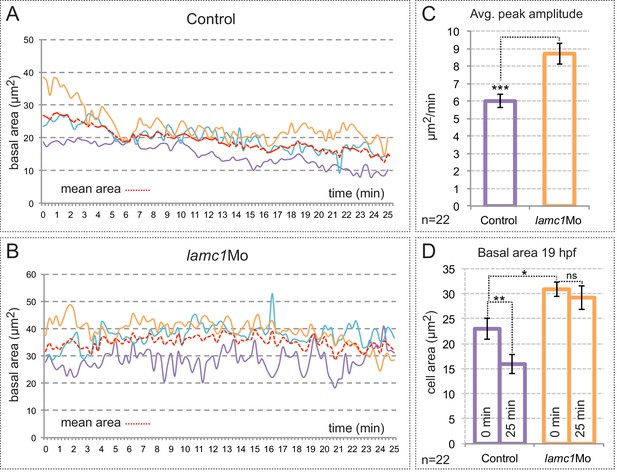
Analysis of membrane oscillations reveal impaired basal constriction in lamc1 morphant embryos.
(A–B) Cell area dynamics in control (A) and lamc1Mo (B) tg(vsx2.2:GFP-caax) embryos is shown for three representative cells. The mean area of the three cells is shown as red dotted lines. (C) Average peak amplitude of the cell area rate is significantly increased in lamc1 morphant cells (T-test, n = 22). (D) Basal feet area is larger and basal constriction, over the recorded period of 25 min, appears significantly inhibited in lamc1Mo retinas (T-test, n = 22). Mean ± SEM is represented.
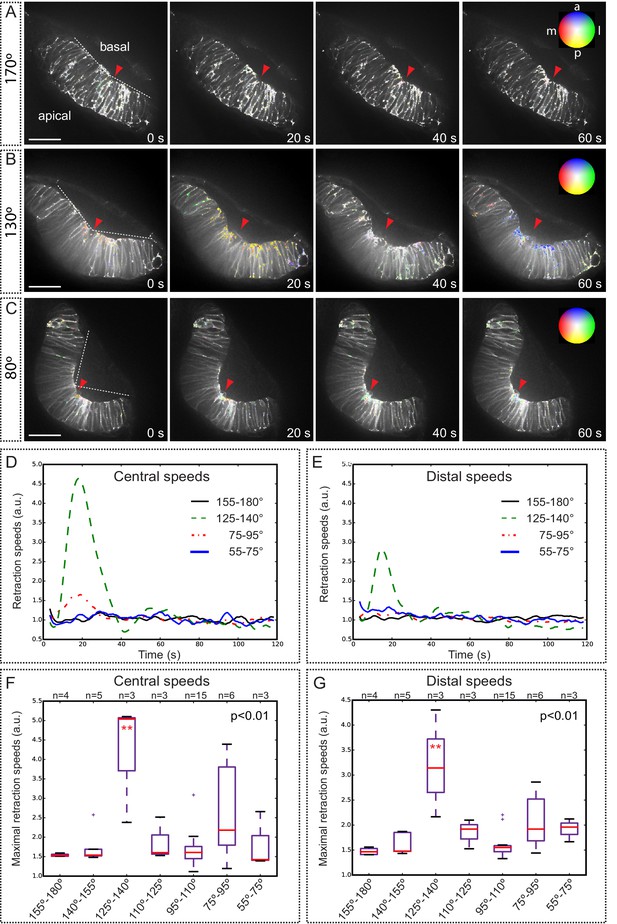
Optical flow analysis of tissue displacement upon laser ablation at different stages of folding.
(A–C) Analysis of pixel displacement after laser ablation at the basal surface is shown for retinas at 170°, 130°, and 80° of bending. Red arrowheads indicate the ablation point. Particles’ motion vectors are indicated with a color code: Colors correspond to the direction of the displacement and color intensity to its magnitude. Note maximum displacement 20 s after ablation in 130°-stage retina. Scale bar = 50 µm. See Video 12. (D–E) Average tissue retraction speed profiles over time are shown for different stages of optic cup folding (represented as angle bins), both at the central (D) or distal (E) positions in the retina. (F–G) Box plot representation of maximal retraction speeds at the different stages, represented as angle bins. For each stage, median values (red bars) and sample sizes are indicated. One-way ANOVA analysis followed by Dunnett’s multiple comparison tests show significant differences (p<0.01**) only at 125–140º-stage.
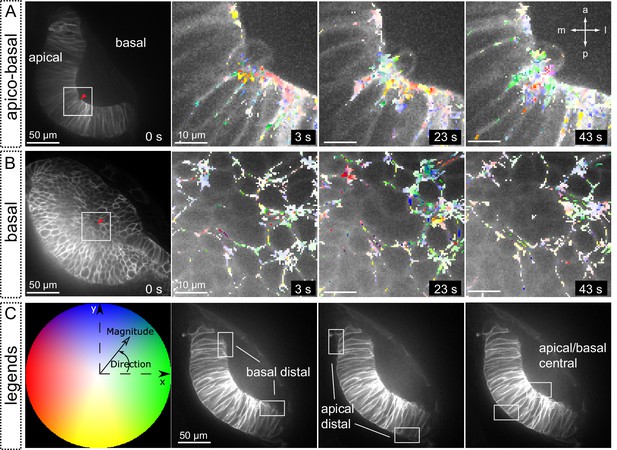
Tissue local relaxation upon laser ablation: Optical flow analysis of tissue displacement.
(A–B) Laser ablation experiments at the basal surface of the retina imaged along the apico-basal axis (A) and basal plane (B) Red arrowheads indicate the ablation point. Time 0 corresponds to the first frame after the ablation. Tissue reaction through time is shown at higher magnification (A–B) and particles’ motion is indicated with a color code. (C) Different colors correspond to the direction of the displacement and color intensity to its magnitude. Regions selected for optical flow quantification in Figure 8 are indicated.
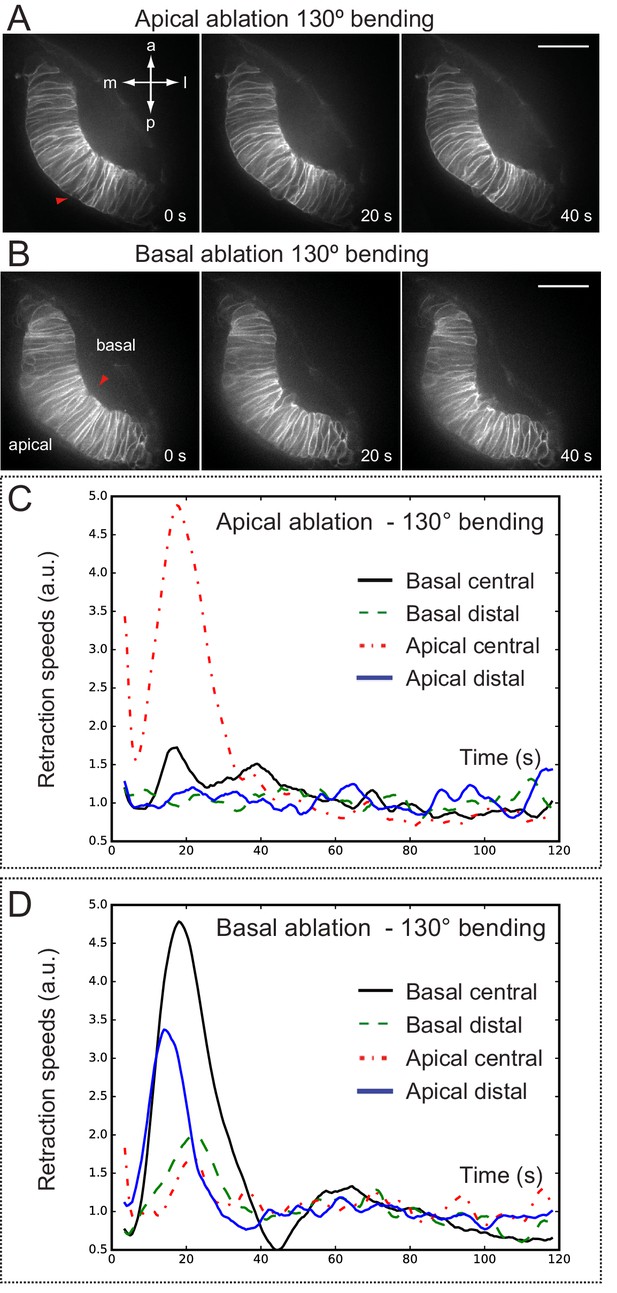
Optical flow analysis of retinal tissue displacement upon apical vs basal laser ablation.
(A–B) Laser ablation experiments at the apical (A) or basal (B) surfaces of the retina in wild-type embryos. Red arrowheads indicate the ablation point. Scale bar = 50 µm. (C–D) Tissue retraction speed profiles at different retinal positions (color-coded) are represented for apical (C) vs basal (D) ablations.
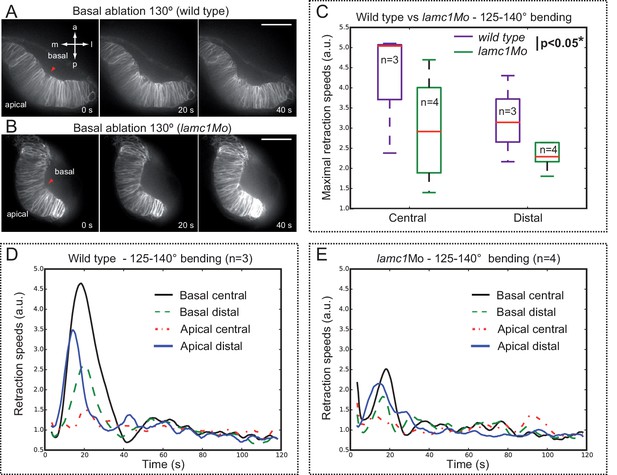
Optical flow analysis of tissue displacement upon laser ablation in wild type vs. lamc1_Mo tissues.
(A–B) Laser ablation experiments at the basal surface of the retina in wild type (A) and lamc1Mo (B) tissues. Red arrowheads indicate the ablation point. Scale bar = 50 µm. (C) Box plot representation of maximal retraction speeds for control and morphant tissues both at the central and distal (peripheral) retina. For each stage, median values (red bars) and sample sizes are indicated. Two-way ANOVA analysis shows that retraction speeds are significantly reduced in lamc1 morphants (p<0.05*). (D–E) Tissue retraction speed profiles at different retinal positions (color-coded) are represented over time for wild type (D) and lamc1Mo (E).
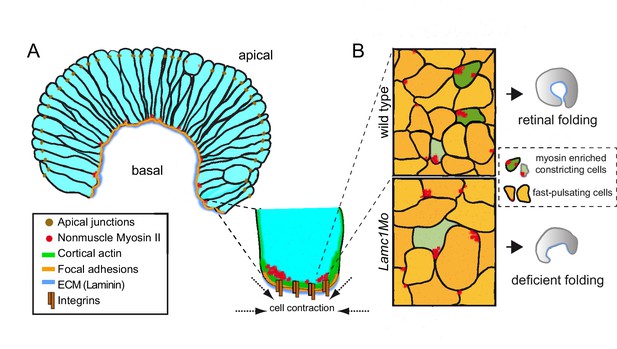
A working model for the basal constriction of the retinal epithelium.
(A) Representation of the retinal epithelium during eye morphogenesis showing the distribution of cortical actomyosin, integrins and ECM at the basal surface of the tissue. Apical junctions and focal adhesion components have been included as a reference for apico-basal polarity. (B) Schematic diagram representing the condensation of nonmuscle myosin II foci at the basal surface in wild type and lamc1Mo retinas. Both fast pulsating cells (orange) and myosin-enriched constricted cells (green) are depicted. Weakly constricting neuroblast feet are represented in pale green. The final form of the organ is also shown for wild type and lamc1 deficient embryos.

Videos
Time lapse of zebrafish optic cup folding.
Optical section from a tg(vsx2.2:GFP-caax) embryo showing the folding of the retinal tissue. Imaging starts at 17 hpf. Antero-posterior and medio-lateral axes are indicated. See also Figure 1.
Membrane oscillations at the basal and apical surfaces.
Maximum projection of 3 z-stacks (over a total of 1 µm) at the basal and apical surfaces in a tg(vsx2.2:GFP-caax) retina show the oscillatory behavior of the cell membranes over a period of 35 min. Images were acquired every 5 s. Scale bars = 10 µm. See also Figure 2.
Analysis of tg(vsx2.2:GFP-caax) clones show uncoupled oscillations at apical and basal surfaces.
Maximum projection of 3 z-stacks (over a total of 1 µm) along the apico-basal axis shows the oscillatory behavior of apical and basal edges simultaneously in tg(vsx2.2:GFP-caax) clones. Images were acquired every 8 s. See also Figure 3.
Actin dynamics in constricting retinal cells.
(Upper panel) Maximum projection of 3 z-stacks (over a total of 1 µm) along the apico-basal axis shows actin oscillatory activity in tg(vsx2.2:lyn-tdTomato) embryos at 20 hpf. Retinal basal surface (region within the square) is magnified in lower panels. (Lower panels) Time lapse shows the simultaneous recording of membrane behavior, as revealed by lyn-tdTomato (left panel), and actin dynamics, as revealed by Utrophin-GFP (right panel). Images were acquired every 5 s. Scale bars = 10 µm. See also Figure 4.
Myosin dynamics during optic cup morphogenesis.
Live imaging analysis of tg(actb1:myl12.1-eGFP) embryos reveal myosin accumulation at apical lens and basal retina epithelia. Movie starts at 19 hpf. Antero-posterior (a-p) axis is indicated. Images were acquired every 20 s. Scale bar 50 µm. See also Figure 5
Myosin foci dynamics at the basal surface.
Live-imaging analysis of myosin distribution at the basal surface in 20 hpf tg(actb1:myl12.1-eGFP) embryos shows cortical localization of myosin foci in scattered cells (left and middle panels). Membrane oscillations were simultaneously examined by injection of lyn-tdTomato RNA (merged in left panel with myl12gfp). Treatment of tg(actb1:myl12.1-eGFP) embryos with blebbistatin (150 µM) severely blocks myosin dynamics at the basal surface (right panel). Images were acquired every 5 s. Scale bar 10 µm. See also Figure 5.
Myosin foci dynamics and basal membrane indentations upon blebbistatin treatment.
Live-imaging analysis of myosin dynamics at the basal surface both in control (upper panel) and blebbistatin treated (150 µM; lower panel) 20 hpf embryos from the line tg(actb1:myl12.1-eGFP). Note the increased stability of the myosin foci and the reduced contractility of the basal surface in the retina of the blebbistatin-treated embryos. Images were acquired every 10 s Scale bar = 10 µm. See also Figure 6.
Membrane oscillations at the basal surface in control and blebbistatin-treated embryos.
Maximum projection of 3 z-stacks (over a total of 1 µm) at the basal surface in tg(vsx2.2:GFP-caax) retinae show cell membranes oscillatory behavior over a period of 25 min in control (left panel) and blebbistatin treated (150 µM; right panel) embryos. Note that blebbistatin treatment abolishes the oscillatory behavior and blocks the cells in a relaxed state. Images were acquired every 5 s. Scale bars = 10 µm. See also Figure 6—figure supplement 1.
Myosin foci dynamics and basal membrane indentations in wild-type and lamc1 morphants.
Live-imaging analysis of myosin dynamics at the basal surface both in control (upper panel) and lamc1Mo-injected (lower panel) 20 hpf embryos from the line tg(actb1:myl12.1-eGFP). Note the increased stability of the myosin foci and the reduced contractility of the basal surface in lamc1 morphants. Images were acquired every 10 s Scale bar = 10 µm. See also Figure 7.
Laser ablation experiments at the basal surface of the retina through optic cup folding.
Local cell ablations were carried out in tg(vsx2.2:GFP-caax) retinae at different stages. Ablation points are indicated with green arrowheads. Retinal folding angles are indicated. Note the global tissue relaxation upon ablation at 130º. Images were acquired every seconds. Scale bar = 50 µm.
Comparative analysis of focal ablations at the apical or basal surface of the retina.
Ablations were carried out in tg(vsx2.2:GFP-caax) retinas with a 130° opening. Ablation points are indicated with green arrowheads. Peripheral tissue displacement is indicated with white arrowheads. Scale bar = 50 µm.
Optical flow analysis of tissue displacement upon laser ablation at different stages of optic cup folding.
Ablation points are indicated with white arrowheads. Particles’ motion vectors are indicated with a color code. Images were acquired every seconds. Scale bar = 50 µm.
Membrane oscillations in an optical section from a tg(vsx2.2:GFP-caax) embryo and Packing Analyzer v2.0 automatic cell edge detection (represented by unique RGB codes) are shown in parallel movies.
Scale bar = 10 µm. See also Figure 1_figure supplement 1.





