The molecular basis for ANE syndrome revealed by the large ribosomal subunit processome interactome
Figures
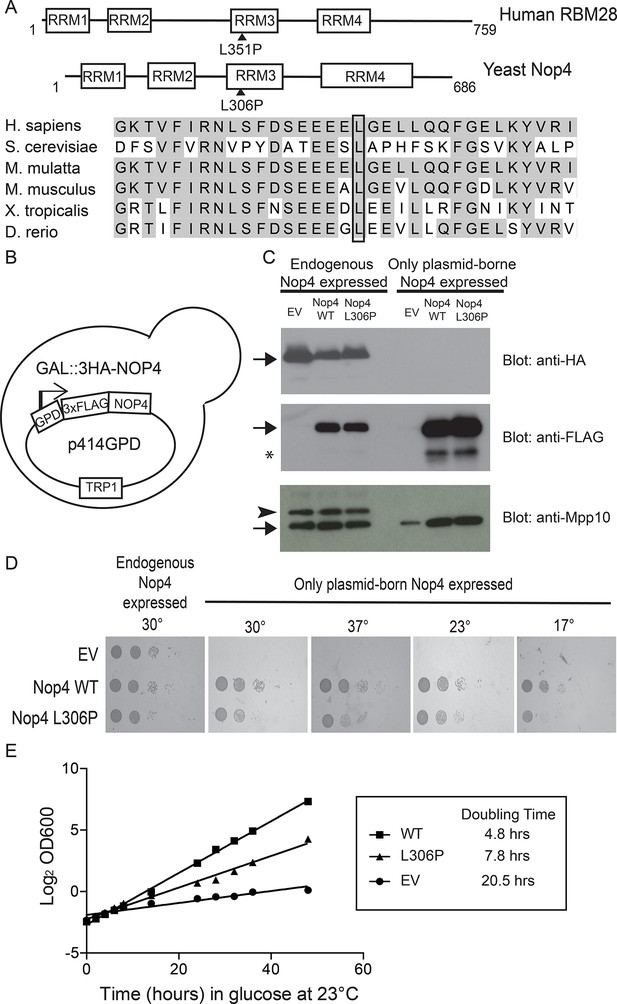
The ANE syndrome mutation confers a growth defect in yeast.
(A) The leucine that is mutated in ANE syndrome is highly conserved. Top: Diagram of the domain structure for human RBM28 and its yeast ortholog, Nop4. The boxes represent RNA Recognition Motifs (RRMs). Arrowheads indicate the approximate location of the mutated amino acid, L351P in humans and L306P in yeast. Bottom: Multiple sequence alignment of the portion of RRM3 containing the mutated leucine. Shaded amino acids in are conserved. A box outlines the conserved leucine that is mutated in ANE syndrome. (B) Schematic of the yeast strain used for testing the ANE syndrome mutation in Nop4. Endogenous Nop4 was placed under the control of the inducible GAL4 promoter in haploid yeast. FLAG-tagged unmutated Nop4 WT or Nop4 L306P was expressed constitutively from the p414GPD plasmid. (C) Nop4 WT and Nop4 L306P are expressed at equivalent levels from the yeast expression vector p414GPD-3xFLAG-GW. The depletion of endogenous Nop4 protein was confirmed by western blot using an HRP-conjugated monoclonal antibody against the 3xHA tag. Expression of Nop4 WT or Nop4 L306P from p414GPD-3xFLAG-GW was analyzed by western blot using a monoclonal antibody against the 3xFLAG tag. As a loading control, a western blot using α-Mpp10 was performed. The expression levels of Nop4 WT and Nop4 L306P relative to Mpp10 were quantitated and normalized to Nop4 WT: Nop4 WT = 1, Nop4 L306P = 0.96. EV = empty vector. The arrows indicate the expected bands. The arrowhead indicates an Mpp10 species only observed when yeast are grown in galactose and raffinose. The asterisk denotes degradation. (D) The ANE syndrome mutation in Nop4 impairs growth on solid medium. Serial dilutions of yeast expressing the indicated Nop4 constructs were grown on solid medium for 3 days at 30°C and 37°C or for 5 days at 23°C and 17°C. Three biological replicates were performed starting with transformation of the plasmids into the yeast strain. (E) The Nop4 ANE syndrome mutation impairs growth in liquid medium. Yeast expressing the indicated Nop4 constructs were transferred from SG/R-Trp to SD-Trp and 23°C to deplete the endogenous Nop4. Growth was monitored for 48 hr by measuring the absorbance at OD600. The log2 of the OD600 was plotted over time and the slope was used to estimate the doubling time. Four biological replicates were performed starting with transformation of the plasmids into the yeast strain.

Multiple sequence alignment of RRM3 from RBM28.
The amino acids corresponding to RRM3 from human RBM28, yeast Nop4 and RBM28 from M. mulatta, M. musculus, X. tropicalis and D. rerio were aligned. The conserved structural elements of a canonical RRM are indicated above the alignment. The shaded amino acids are conserved. The conserved leucine that is mutated in ANE syndrome is outlined with a box.
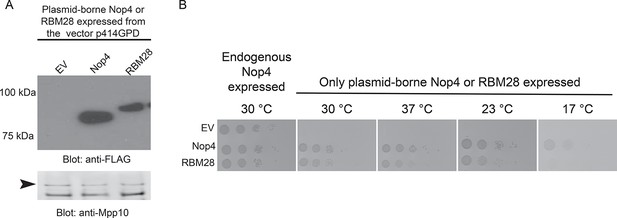
RBM28 complements the growth defect in yeast observed upon depletion of its essential ortholog, Nop4.
(A) Nop4 and RBM28 are expressed from the yeast expression vector p414GPD-3xFLAG-GW. Expression of Nop4 (78 kDa) or RBM28 (86 kDa) from p414GPD-3xFLAG-GW was analyzed by western blot using a monoclonal antibody against the 3xFLAG tag. As a loading control, a western blot using α-Mpp10 was performed. The arrowhead indicates an Mpp10 species only observed when yeast are grown in galactose and raffinose. The expression levels of Nop4 and RBM28 relative to Mpp10 were quantitated and normalized to Nop4: Nop4 = 1, RBM28 = 0.4. (B) RBM28 complements growth on solid medium. Serial dilutions of yeast expressing either Nop4 or RBM28 were grown on solid medium for 3 days at 30°C and 37°C or for 5 days at 23°C and 17°C.

Amino acid sequence alignment of human RBM28 and its yeast ortholog Nop4.
The complete amino acid alignment of human RBM28 protein and its yeast ortholog, Nop4, was created using ClustalX. The conserved leucine that is mutated to proline in ANE syndrome, L351 in RBM28 and L306 in Nop4, is circled in black. RRM domains are indicated by the labeled boxes. Below the alignment, the conservation score for each column is plotted. A high score indicates high conservation whereas a low score indicates low conservation.
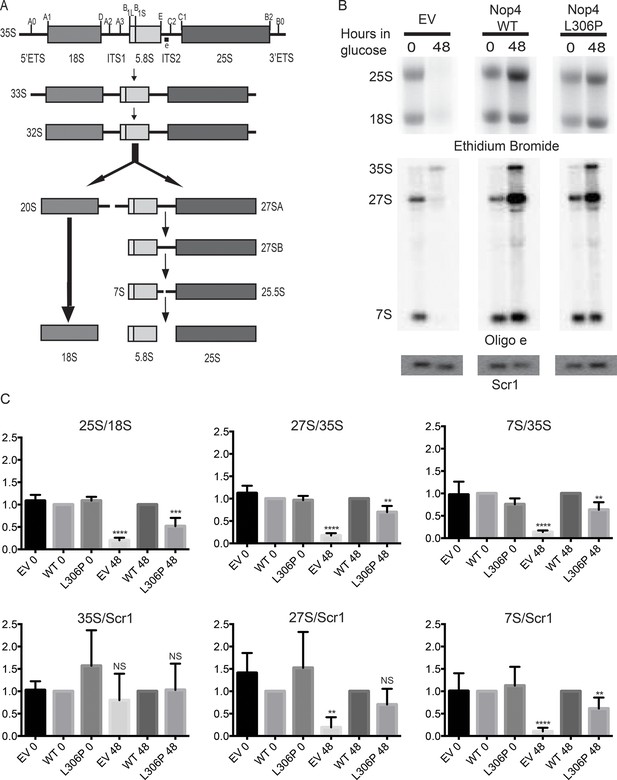
The ANE syndrome mutation disrupts pre-rRNA processing in yeast.
(A) Simplified diagram depicting the pre-rRNA processing steps in yeast. The pre-rRNA is transcribed as a 35S polycistronic precursor. The external transcribed spacers (5´ and 3´ ETS) and the internal transcribed spacers (ITS1 and 2) are removed through a number of cleavage steps to produce the mature 18S, 5.8S and 25S rRNAs. Oligonucleotide probe e, which is complementary to ITS2 and detects all 27S and 7S pre-rRNAs (indicated on top line), was used for northern blotting. (B) The ANE syndrome mutation in Nop4 impairs pre-rRNA processing in yeast. Top panel: Ethidium bromide staining of total RNA extracted from yeast expressing no Nop4 (empty vector; EV), Nop4 WT or Nop4 L306P after depletion of endogenous Nop4 for the indicated time. Bottom panel: Northern blots of total RNA using radio-labeled oligonucleotide probe e to detect 35S, 27S, and 7S pre-rRNAs and an oligonucleotide probe complementary to Scr1 as a loading control. (C) The ratios of the mature rRNAs (25S/18S), the ratios of the precursors (27S/35S and 7S/35S) and the ratios of the precursors to the loading control Scr1 (35S/Scr1, 27S/Scr1 and 7S/Scr1) were calculated from four replicate experiments and were plotted with error bars representing the standard deviation. The significance of the ratios of Nop4 depleted yeast (empty vector; EV) or Nop4 L306P compared to WT was evaluated using one-way ANOVA. ****indicates a p value < 0.0001. ***indicates a p value < 0.001. **indicates a p value <0.01. NS = not significant. Four biological replicates were performed.
-
Figure 2—source data 1
Quantitation and statistical analyses for Figure 2C.
- https://doi.org/10.7554/eLife.16381.008
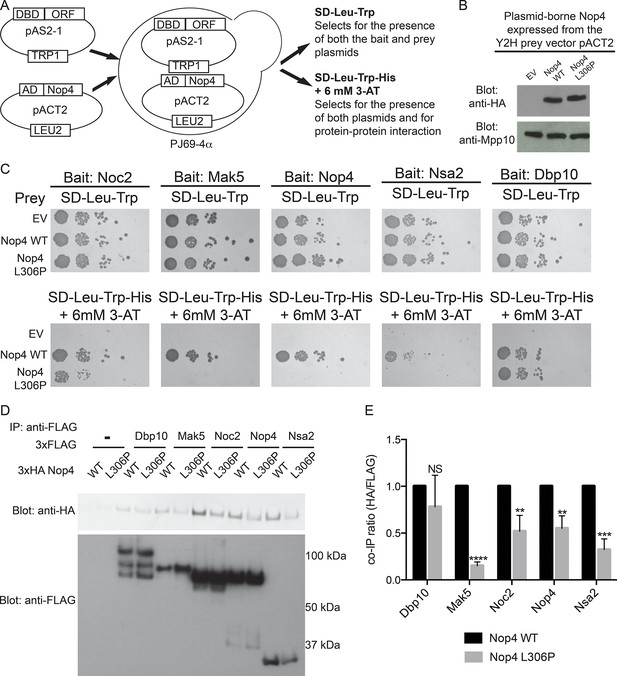
The ANE syndrome mutation in Nop4 disrupts protein-protein interactions.
(A) Schematic of Y2H analysis. Nop4 WT or Nop4 L306P were cloned into the prey vector (pACT2) while five Nop4 interacting proteins (Noc2, Mak5, Nop4, Nsa2 and Dbp10) were cloned into the bait vector (pAS2-1). Each bait was individually co-transformed into the yeast strain PJ69-α with empty vector (EV), Nop4 WT or Nop4 L306P prey and spotted onto medium to confirm the presence of both Y2H plasmids (SD-Leu-Trp) and onto medium to test for protein-protein interactions (SD-Leu-Trp-His + 6 mM 3-AT). (B) Nop4 WT and Nop4 L306P are expressed at equivalent levels from the Y2H vector pACT2. Total protein was extracted from PJ69-4α yeast transformed with EV or expressing Nop4 WT or Nop4 L306P from the Y2H prey vector, pACT2. Nop4 WT and Nop4 L306P are expressed as fusions with the GAL4 activation domain and a 3xHA tag. Protein extracts were separated by SDS-PAGE and analyzed by α-HA western blot. As a loading control, a western blot using α-Mpp10 was performed. The expression levels of Nop4 WT and Nop4 L306P relative to Mpp10 were quantitated and normalized to Nop4 WT: Nop4 WT = 1, Nop4 L306P = 1.1 (C) Y2H analysis by serial dilution reveals that the ANE syndrome (L306P) mutation disrupts some Nop4 protein-protein interactions. Two biological replicates of a subset of interacting proteins were performed starting with co-transformation of the bait and prey plasmids into the Y2H strain. (D) The ANE syndrome (L306P) mutation reduces protein-protein interactions as determined by co-immunoprecipitation. Yeast extract was generated from yeast expressing either Nop4 WT or Nop4 L306P and one of its interacting partners and incubated with α-FLAG resin. Co-immunoprecipitations were assessed by α-HA western blot. The expected molecular weights of the Nop4 interacting proteins are: Dbp10 = 113 kDa, Mak5 = 87 kDa, Noc2 = 82 kDa, Nop4 = 78 kDa and Nsa2 = 30 kDa. (E) The ratio of co-purified 3xFLAG tagged Nop4 WT or Nop4 L306P to co-immunoprecipitated 3xHA tagged interacting partner was calculated from three replicate experiments and plotted with error bars representing the standard deviation. The significance of the co-immunoprecipitation ratio of Nop4 L306P compared to WT for each interacting partner was evaluated using a t-test. ****indicates a p value < 0.0001. ***indicates a p value < 0.001. **indicates a p value <0.01. NS = not significant. Three biological replicates were performed.
-
Figure 3—source data 1
Quantitation and statistical analyses for Figure 3E.
- https://doi.org/10.7554/eLife.16381.010
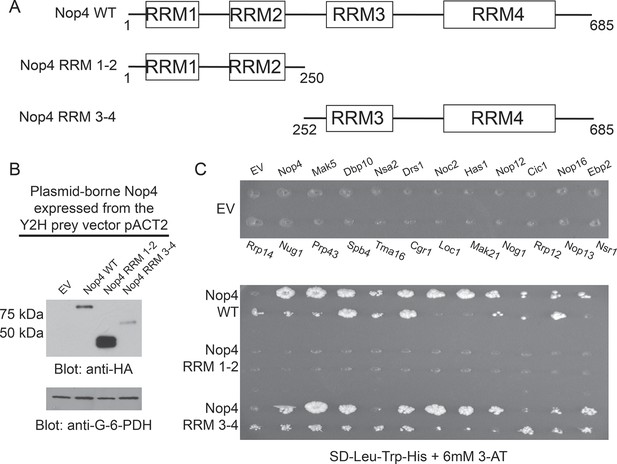
RRM3 and RRM4 of Nop4 mediate protein-protein interactions.
(A) Schematic representation of Nop4 RRM domains and the N- and C-terminal fragments containing RRMs 1 and 2 (RRM 1–2; residues 1–250) or 3 and 4 (RRM 3–4; residues 252–685), respectively. (B) Nop4 WT and the Nop4 fragments are differentially expressed from the Y2H vector pACT2. Total protein was extracted from PJ69-4α yeast transformed with empty vector (EV) or expressing Nop4 WT (78 kDa), Nop4 RRM 1–2 (28.3 kDa) or Nop4 RRM 3–4 (49.4 kDa) from the Y2H prey vector, pACT2. Nop4 WT, Nop4 RRM 1–2 and Nop4 RRM 3–4 are expressed as fusions with the GAL4 activation domain and a 3xHA tag. Protein extracts were separated by SDS-PAGE and analyzed by α-HA western blot. As a loading control, a western blot using α-Glucose-6-Phosphate Dehydrogenase (G-6-PDH) was performed. The expression levels of Nop4 WT, Nop4 RRM 1–2 and Nop4 RRM 3–4 relative to G-6-PDH were quantitated and normalized to Nop4 WT: Nop4 WT = 1, Nop4 RRM 1–2 = 7.4, Nop4 RRM 3–4 = 0.29. (C) Y2H analysis demonstrates that Nop4 RRM 3–4 mediates protein-protein interactions. Nop4 WT and the Nop4 fragments described in (a) were tested as preys for interaction with 23 Nop4 interacting proteins as baits. The baits are labeled for the empty vector (EV) control plate. Growth on selective medium (SD-Leu-Trp-His + 6 mM 3-AT) indicates an interacting bait-prey pair. Two biological replicates were performed starting with the transformation of the bait and prey plasmids into the Y2H strains.
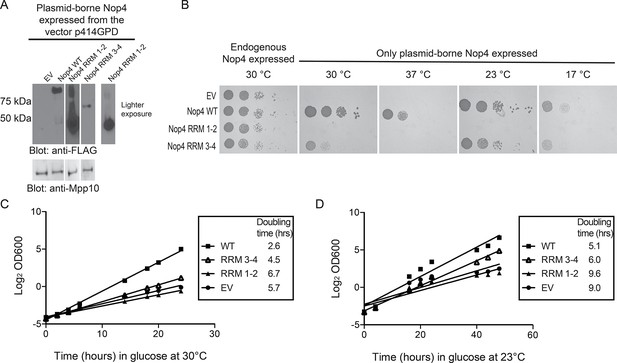
RRM3 and RRM4 of Nop4 are necessary and sufficient to complement the growth defect due to Nop4 depletion.
(A) Nop4 WT and the Nop4 fragments are differentially expressed from the yeast expression vector p414GPD-3xFLAG-GW. Total protein was extracted from YPH499 GAL::3xHA-NOP4 yeast transformed with empty vector (EV) or expressing Nop4 WT (78 kDa), Nop4 RRM 1–2 (28.3 kDa) or Nop4 RRM 3–4 (49.4 kDa) from the yeast expression vector, p414GPD-3xFLAG-GW. Protein extracts were separated by SDS-PAGE and analyzed by α-FLAG western blot. As a loading control, a western blot using α-Mpp10 was performed. The expression levels of Nop4 WT, Nop4 RRM 1–2 and Nop4 RRM 3–4 relative to Mpp10 were quantitated and normalized to Nop4 WT: Nop4 WT = 1, Nop4 RRM 1–2 = 4.9, Nop4 RRM 3–4 = 0.27. (B) Serial dilutions of yeast expressing the indicated Nop4 fragments were grown on solid medium for 3 days at 30°C and 37°C or for 5 days at 23°C and 17°C. (C) Yeast expressing the indicated Nop4 fragments were transferred from SG/R-Trp-Leu to SD-Trp-Leu to deplete the endogenous Nop4. Growth was monitored for 24 hr at 30°C by measuring the absorbance at OD600. The log2 of the OD600 was plotted over time and the slope was used to estimate the doubling time. Three biological replicates were performed starting with transformation of the plasmids into the yeast strain. (D) Yeast expressing the indicated Nop4 fragments were transferred from SG/R-Trp-Leu to SD-Trp-Leu to deplete the endogenous Nop4. Growth was monitored for 48 hr at 23°C by measuring the absorbance at OD600. The log2 of the OD600 was plotted over time and the slope was used to estimate the doubling time. Three biological replicates were performed starting with transformation of the plasmids into the yeast strain.
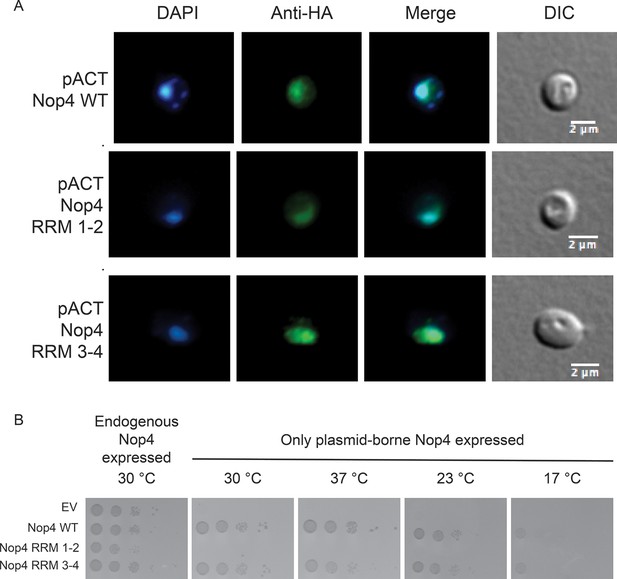
Nop4 RRM 1–2 fails to complement even when targeted to the nucleus.
(A) Expression of the Nop4 fragments from the yeast two-hybrid vector pACT2 ensures nuclear targeting. Yeast expressing 3xHA-Nop4 WT, 3xHA-Nop4 RRM 1–2 or 3xHA-Nop4 RRM 3–4 from the yeast two-hybrid vector, pACT2, after depletion of endogenous Nop4 for 48 hr at 23°C were stained with DAPI (blue) and HA antibodies (green). The immunofluorescence microscopy images were merged. Nop4 WT, Nop4 RRM 1–2 and Nop4 RRM 3–4 localize primarily to the nucleus. (B) Serial dilutions of yeast expressing the indicated Nop4 fragments from pACT2 were grown on solid medium for 3 days at 30°C and 37°C or for 5 days at 23°C and 17°C.
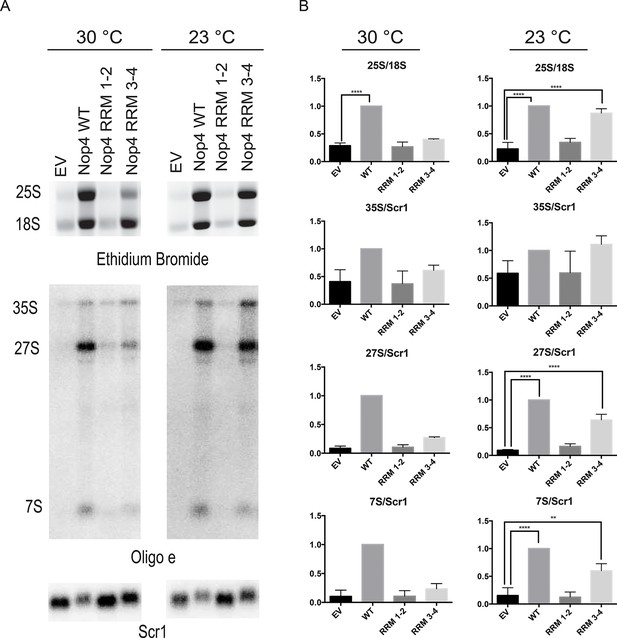
Nop4 RRM 3–4 is necessary and sufficient to complement the pre-rRNA processing defect after Nop4 depletion.
(A) Top panel: Total RNA extracted from yeast expressing the indicated Nop4 fragment after depletion of endogenous Nop4 for 24 hr at 30°C or for 48 hr at 23°C was visualized by ethidium bromide staining. Bottom panel: Northern blot analysis of total RNA using oligonucleotide probe e, which is complementary to a region of ITS2 of the pre-rRNA. As a loading control, we used an oligonucleotide complementary to the Scr1 RNA. Three biological replicates were performed. (B) The ratios of the mature rRNAs (25S/18S) and the ratios of the precursors to the loading control Scr1 (35S/Scr1, 27S/Scr1S and 7S/Scr1) were calculated from three replicate experiments and were plotted with error bars representing the standard deviation. The significance of the ratios of 25S/18S, 35S/Scr1, 27S/Scr1 and 7S/Scr1 of Nop4 WT, Nop4 RRM 1–2 or Nop4 RRM 3–4 compared to EV was evaluated using one-way ANOVA. ****indicates a p value < 0.0001. **indicates a p value < 0.01.
-
Figure 6—source data 1
Quantitation and statistical analyses for Figure 6B.
- https://doi.org/10.7554/eLife.16381.016
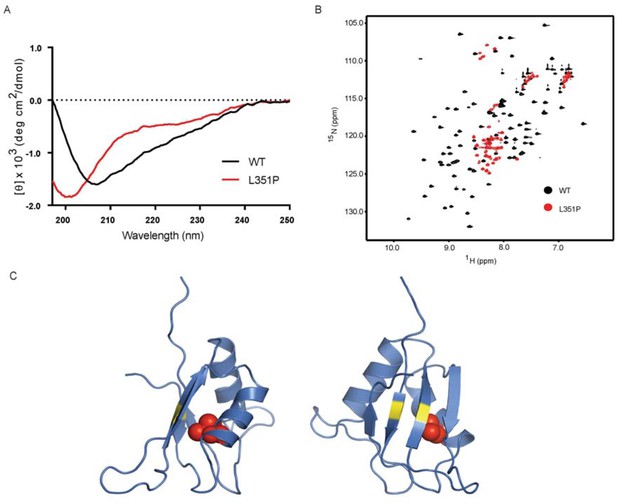
The ANE syndrome mutation, L351P, in human RBM28 disrupts RRM3 domain structure.
(A) Circular dichroism spectra of WT human RBM28 RRM3 (black) and L351P mutant (red). Four technical replicates were performed. (B) 15N-HSQC spectra of WT hRBM28 RRM3 (amino acids 330–419) (black) and L351P mutant (red) are superimposed and plotted at the same contour level. In addition to clustering of resonances around 8.0~8.5 ppm in the proton dimension, dispersion of glutamine and asparagine side chains (7.0~7.8 ppm in the 1H dimension and 111~114 ppm in the 15N dimension) is reduced considerably, consistent with protein backbone disruption. Thirty-two technical replicates were performed. (C) Ribbon diagram of a homology model of human RBM28 RRM3. The model including residues 330–419 was generated using the Phyre2 server (Kelley et al., 2015), and the best template was a solution structure of mouse RBM28 RRM3 (90% sequence identity with human RBM28, PDB ID 1X4H). L351 is shown with red space-filling spheres and typical RNA interacting residues in RNP motifs are colored yellow.

The ANE syndrome mutation (L306P) disrupts 25S production in yeast.
(A) Left panel: longer exposure of the ethidium bromide stained gel in Figure 2B. Total RNA was extracted from yeast expressing no Nop4 (EV), Nop4 WT or Nop4 L306P after depletion of endogenous Nop4 for the indicated time. Right panel: The ratios of the mature rRNAs (25S/18S) were calculated from four replicate EtBr experiments and were plotted with error bars representing the standard deviation. Significance compared to WT was evaluated using one-way ANOVA. (B) Left panel: Methylene blue stained membrane of the northern blot in Figure 2B. Total RNA was extracted from yeast expressing no Nop4 (EV), Nop4 WT or Nop4 L306P after depletion of endogenous Nop4 for the indicated time. Right panel: The ratios of the mature rRNAs (25S/18S) were calculated from four replicate methylene blue experiments and were plotted with error bars representing the standard deviation. Significance compared to WT was evaluated using one-way ANOVA. **** indicates a p value <0.0001. ***indicates a p value <0.001.
Tables
Nop4 interacts with 23 large subunit assembly factors with high confidence. The Nop4 interacting proteins were identified by yeast two-hybrid and were assigned a confidence score in (McCann et al., 2015).
| Nop4 Interacting Partner | Confidence Score (from McCann et al., 2015) |
|---|---|
| Nop4 | 92% |
| Loc1 | 92% |
| Ebp2 | 85% |
| Nop12 | 85% |
| Nsa2 | 85% |
| Mak5 | 84% |
| Cgr1 | 70% |
| Cic1 | 70% |
| Has1 | 70% |
| Noc2 | 70% |
| Nop13 | 70% |
| Nsr1 | 70% |
| Rrp12 | 70% |
| Rrp14 | 70% |
| Mak21 | 68% |
| Dbp10 | 63% |
| Drs1 | 63% |
| Nop16 | 63% |
| Nug1 | 63% |
| Prp43 | 63% |
| Spb4 | 63% |
| Tma16 | 63% |
| Nog1 | 53% |
Additional files
-
Supplementary file 1
Western blot quantitation source data.
- https://doi.org/10.7554/eLife.16381.018





