A functional genomics screen in planarians reveals regulators of whole-brain regeneration
Figures
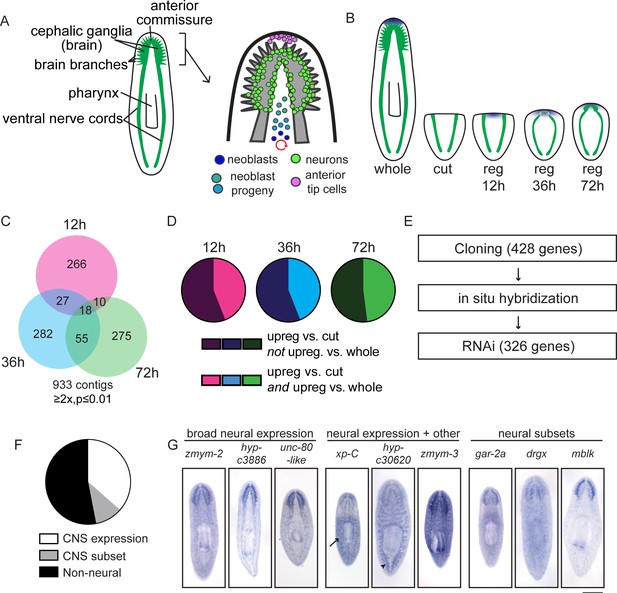
A transcriptional view of planarian head regeneration.
(A) Diagrams depicting the overall organization of the planarian CNS (left) and the principal cell types known to play roles in brain regeneration (right). (B) A schematic illustrating the samples used for RNA-Seq analysis. The CNS is shown in green and anterior polarity signals are represented in blue. Whole animals and animals killed immediately after postpharyngeal amputation were used for controls. (C) A Venn diagram depicts the 933 contigs upregulated during head regeneration compared to cut controls (≥2x upregulated, p≤0.01). Most identified genes were upregulated to this threshold at only one time point. (D) Pie charts showing the proportion of genes upregulated at each time point (vs. cut control) that are also upregulated when compared to uninjured (whole) animals (≥2x upregulated, p≤0.01). The fractions are 44% (12 hr, 36 hr) and 48% (72 hr). (E) Workflow depicting our characterization of upregulated genes. (F) Pie chart showing the percentage of upregulated genes with neural expression patterns. 362 genes gave clear expression patterns. Of these, 47% had expression in the central nervous system—132 genes with expression in the brain and 38 with expression in a subset of CNS cells. (G) Examples of expression patterns (by in situ hybridization, ISH) are shown for genes expressed in the CNS alone, in the CNS and other tissues (arrow indicates the parenchyma for xp-C and arrowhead indicates the gut for hyp-c30620), and in neural subsets. Scale bar: 500 µm.
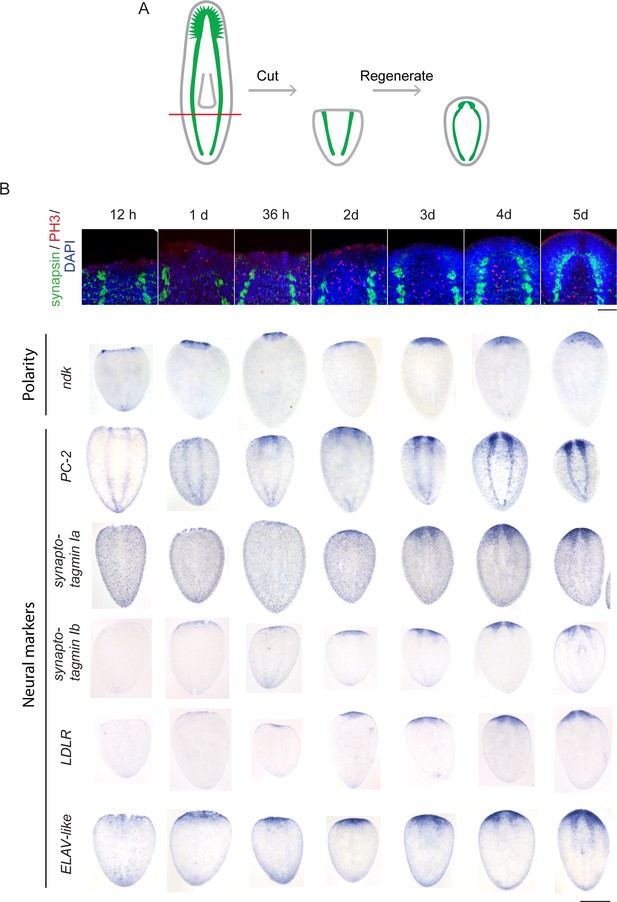
Time courses of anterior regeneration.
(A) Diagram of the postpharyngeal amputation used for the time courses in this figure, as well as the transcriptomic experiments. The CNS is diagrammed in green. (B) Time courses using immunofluorescence and ISH to illustrate the timing of events in head regeneration. Anterior markers appear early (e.g., nou darake, ndk). Neural gene expression initiates at around 36 hr (e.g., prohormone convertase 2, PC-2) and becomes stronger at 2 d post-amputation. By 3 d after amputation, a brain primordium is visible by DAPI staining and neural markers are all reexpressed. Synapsin staining indicating further maturation of the neurons continues to 5 d after amputation.The innervated pharynx is also visible 5 d after amputation. (Scale bars: 500 µm (B, bottom rows), 100 µm (B, top row).
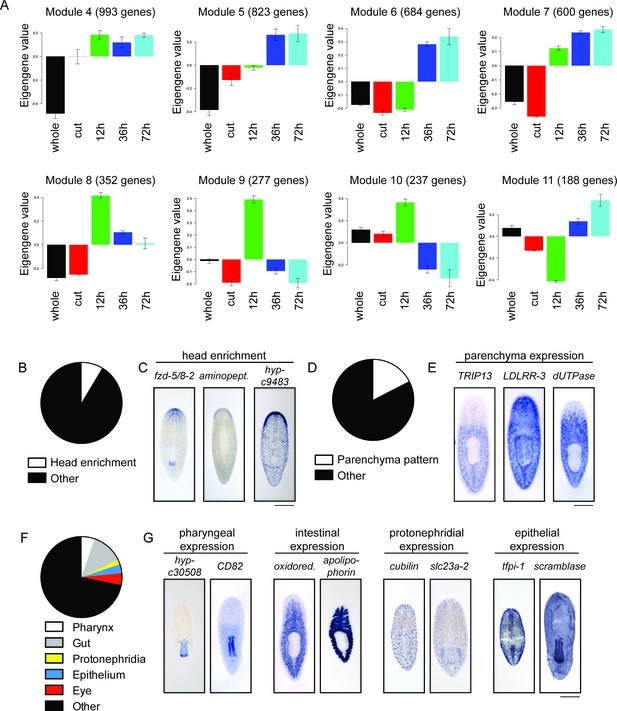
Expression patterns of genes upregulated during head regeneration.
(A) The eight largest clusters of upregulated genes. For each gene in the cluster, the mean expression across the five conditions is set to zero and relative expression is calculated for each experiment. The expression profile across all genes in the cluster is averaged to form a representation typical of the group. Each bar represents the average gene expression relative to the mean for that time point, with one unit representing one standard deviation from the mean across time points. (B) 31/362 upregulated genes (8.5%) show head enrichment. (C) Examples of genes enriched in the anterior of the animal are shown. (D) 63/362 upregulated genes (17.4%) are expressed during homeostasis in the parenchyma of the planarian body. (E) Examples of genes expressed in the parenchyma are shown. (F) Other expression patterns seen in our analysis are the pharynx (19 genes), intestine (46), protonephridia (7), epithelium (13) and eye (17). (G) Examples of genes with the aforementioned gene expression patterns are shown. Scale bars: 500 µm.
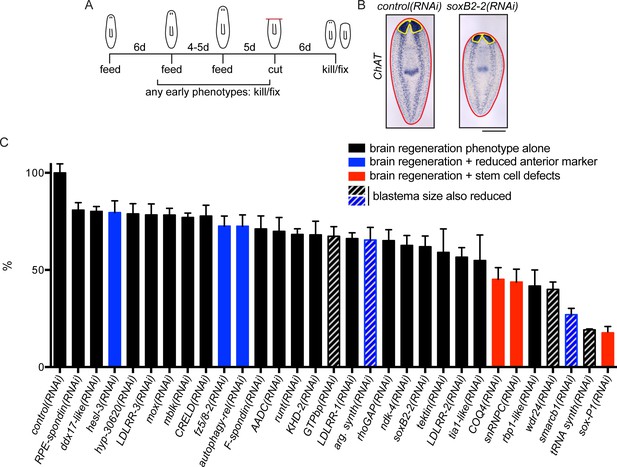
A screen for genes required for regeneration of the planarian brain.
(A) A diagram depicting the RNAi protocol used for our functional screen. For each of the 326 genes analyzed in our study, dsRNA was synthesized and fed to animals three times over 10–11 days. Five days after the final dsRNA feeding, animals were cut prepharyngeally and allowed to regenerate for 6 days. The animals were then killed and fixed for ISH to visualize the CNS. Animals were also monitored for behavioral defects prior to the termination of the experiment. When animals manifested phenotypes earlier than the conclusion of the experiment (e.g., lysis or curling), they were fixed when such a phenotype was first observed. (B) Example of RNAi animals used for brain area quantification. Control(RNAi) and soxB2-2(RNAi) animals were subjected to ISH with the choline acetyltransferase (ChAT) probe. After imaging, the body area (red line) and brain area (yellow line) were calculated for each sample. (C) Brain area/body area ratios were averaged across ~8 worms per sample and were normalized to control(RNAi) animals. All 30 genes for which RNAi caused a significant reduction in brain area are shown here, with error bars representing SEM. Genes for which RNAi also caused reduction in an anterior marker are indicated with blue bars. Red bars indicate genes for which RNAi also affected stem cell maintenance. Bars with grey diagonal lines indicate genes for which RNAi also caused small blastemas after amputation. Scale bar: 500 µm.
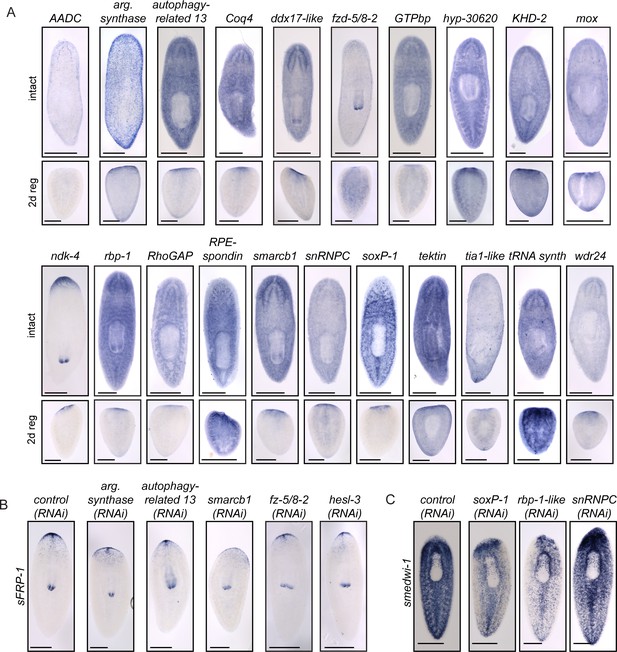
Further analysis of genes with functions in CNS regeneration.
(A) The expression patterns of the genes identified as having roles in CNS regeneration are shown in the main text (Figures 3, 4) or in this panel. Expression patterns of uninjured worms are shown in the top panels, while expression of each gene in a 2 d regenerating worm (cut postpharyngeally) is shown below. (B) Reestablishment of anterior polarity, using sFRP-1 as a marker, after gene knockdown with the indicated dsRNA. Knockdown of several genes caused reduction of sFRP-1 expression which ranged from minor to severe. (C) The stem cell pool was examined using smedwi-1 after RNAi targeting control or target genes. These three genes showed some decrease in smedwi-1 staining. Scale bars: 500 µm (A–C).
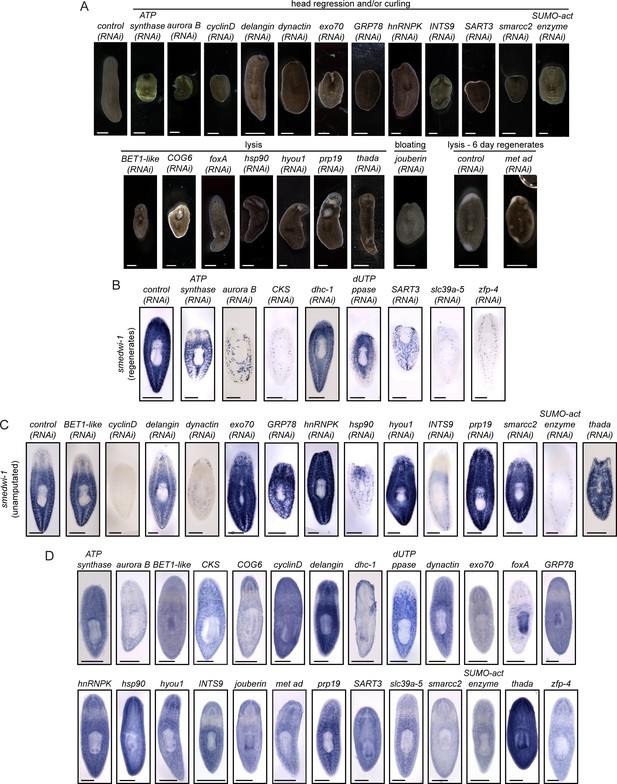
Upregulated genes play roles in tissue maintenance and stem cell biology.
(A) Knockdowns of the indicated genes show phenotypes including head regression, curling, and lysis. Live animals are shown after dsRNA feedings prior to amputation or after regeneration as indicated. (B–C) smedwi-1 expression was used to determine the effect of each RNAi knockdown on the stem cell pool. Animals were prepared for ISH either after regeneration (B) or in uninjured animals (C) depending on the timing of phenotype presentation. Many genes identified in this study perturb smedwi-1 expression and likely disrupt stem cell maintenance and/or function. (D) Gene expression patterns of the genes with roles in homeostasis are shown. Scale bars: 500 µm.
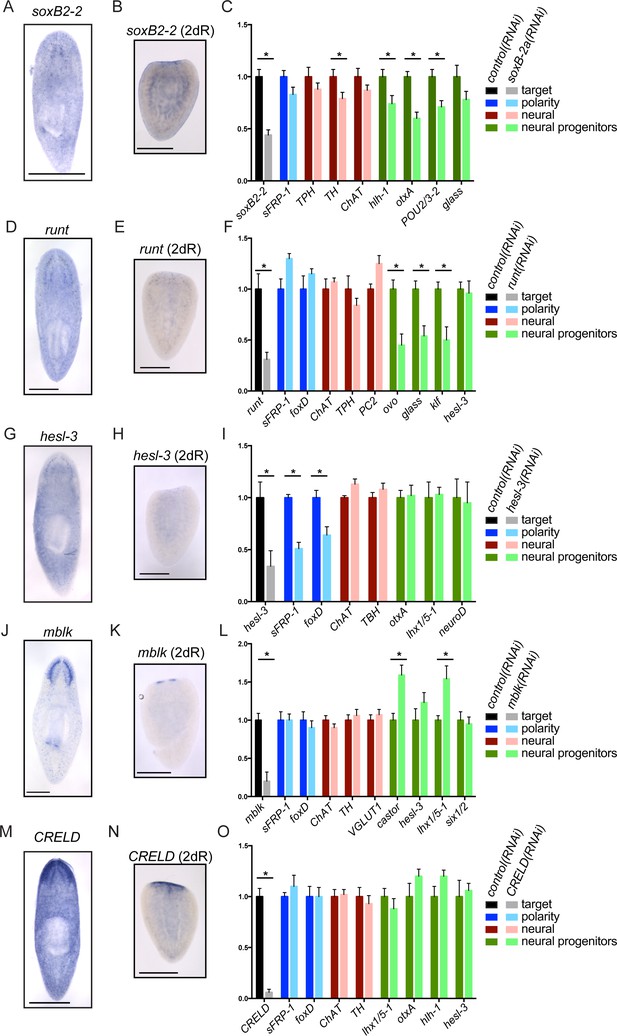
Genes important for brain regeneration exert their effects through multiple mechanisms.
(A) Expression of soxB2-2 by ISH during homeostasis. soxB2-2 cells are scarce, but mostly localized near the brain. (B) soxB2-2 expression was also determined by ISH in animals cut postpharyngeally and allowed to regenerate for two days. (C) RNA was prepared from whole control(RNAi) and soxB2-2(RNAi) animals at the end of a treatment course depicted in Figure 2A. qPCR was used to examine the expression of 134 genes after RNAi. Selected qPCR results are shown here. The bar graph is color coded to indicate the expression of the gene targeted by RNAi (black/grey), anterior polarity genes (blue), genes important for neurotransmitter biogenesis (red/pink), and genes with suspected roles in neural differentiation or neural progenitors (dark/light green). soxB2-2 was knocked down to 44% of control(RNAi) levels after RNAi. Significant decreases in hlh-1, otxA, and POU2/3-2 were also seen after soxB2-2(RNAi). Similarly, expression patterns and downstream effects were determined for runt (D–F), hesl-3 (G–I), mblk (J–L), and CRELD (M–O). runt also affected neural progenitor genes, while hesl-3 primarily affected polarity genes. No downregulated genes were identified after mblk(RNAi) or CRELD(RNAi). Targets were knocked down to 31% (runt), 34% (hesl-3), 20% (mblk) and 6% (CRELD) of control(RNAi) levels. Asterisks mark genes with a significant difference in expression (p≤0.05) after RNAi. Scale bars: 500 µm.
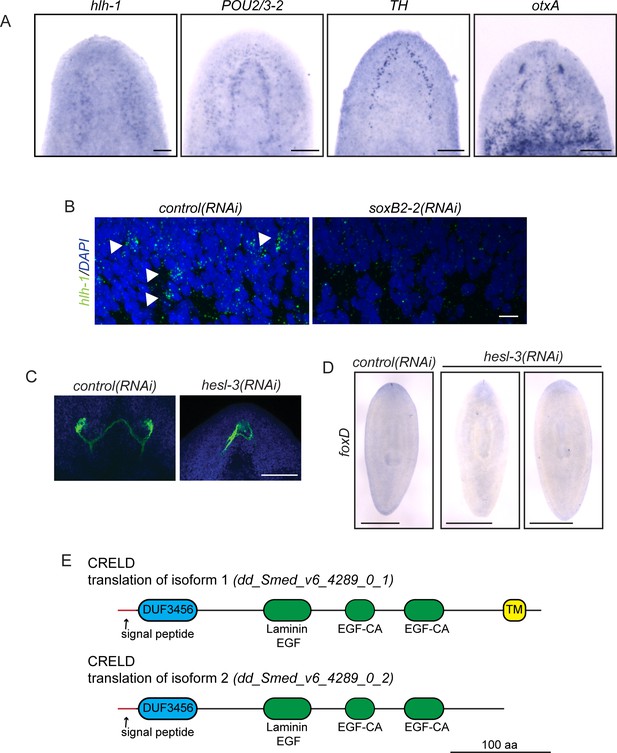
Upregulated genes play roles in diverse processes to promote regeneration.
(A) hlh-1, POU2/3–2, TH, and otxA were detected in subsets of cells in the planarian CNS by ISH. (B) hlh-1 FISH (green) revealed cells near the neuropil in control(RNAi) worms but not soxB2-2(RNAi) worms. Background speckles are present due to background because of the low abundance of the hlh-1 transcript. DAPI is shown in blue. (C) Photoreceptor neurons were visualized after control(RNAi) and hesl-3(RNAi) treatment using VC-1 antibody (green). Eyespots were fused medially after hesl-3(RNAi). DAPI is shown in blue. (D) By in situ hybridization (ISH), foxD expression at the anterior-most point of the planarian body is reestablished after control(RNAi) but is diminished or absent after hesl-3(RNAi). (E) Diagrams depict translations of the two isoforms of CRELD supported by transcriptomics (Brandl et al., 2016). TM marks a predicted transmembrane domain. Epidermal growth factor-like domains (EGF) and a domain of unknown function (DUF) are also marked. Scale bars: 100 µm (A, C), 10 µm (B), 500 µm (D).
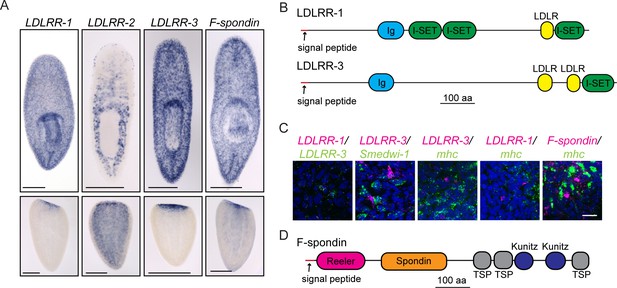
Secreted molecules with roles in planarian brain regeneration.
(A) Expression patterns of LDLRR-1, LDLRR-2, LDLRR-3, and F-spondin were determined by ISH for uninjured worms (top) and tail fragments two days after amputation (bottom). (B) The domain architectures of LDLRR-1 and LDLRR-3. (C) Coexpression of the indicated genes by fluorescent in situ hybridization (FISH). All LDLRR-3+ cells express LDLRR-1 (16/16), but only about half of LDLRR-1+cells express LDLRR-3 (16/31). LDLRR-3+ cells do not express smedwi-1 (0/34), although these cells are often found in proximity to one another. LDLRR-1, LDLRR-3, and F-spondin are not expressed in mhc+ muscle cells (0/8, 0/23, and 0/6 respectively). LDLRR FISH experiments were performed on whole worms and F-spondin FISH was performed on 2d regenerating animals. (D) The domain architecture of F-spondin. Scale bars: 500 µm (A), 20 µm (C).
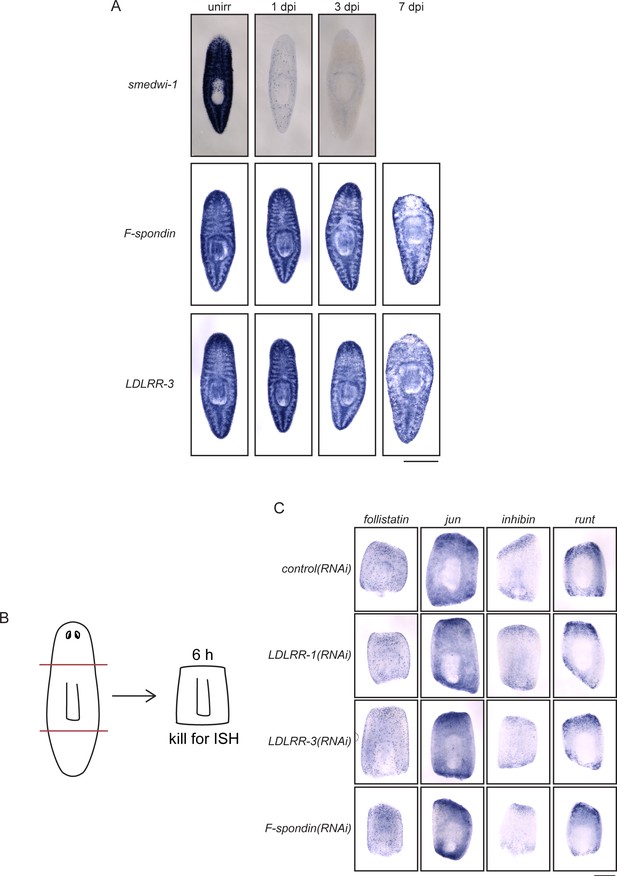
Parenchymal genes are irradiation-insensitive and do not influence wound response.
(A) Planarians were subjected to ISH with smedwi-1, F-spondin, and LDLRR-3 probes after mock irradiation as well as 1, 3, and 7 days post-irradiation. While smedwi-1 transcript is lost quickly, F-Spondin and LDLRR-3 transcripts are still present at 7 dpi, when most animals were lysing and dying. (B) Diagram of amputation schema for wound-response genes. Animals were fed dsRNA three times before amputation as in Figure 2A. (C) Wound-response genes follistatin, jun, inhibin, and runt were examined by ISH after RNAi and amputation. All genes were upregulated despite knockdown of the genes indicated, suggesting that LDLRR and F-spondin genes do not inhibit the general wound response. Scale bars: 500 µm (A, C).
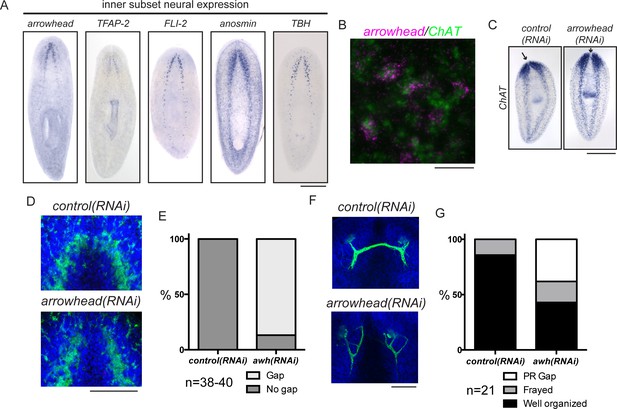
arrowhead is required for regeneration of medial structures in the planarian CNS.
(A) Five upregulated genes were shown to be expressed medially in the CNS. These expression patterns were not overlapping (data not shown). (B) arrowhead is expressed in neurons, many of which are ChAT+ (12/27). (C) arrowhead(RNAi) did not cause a small brain phenotype, but ISH with ChAT revealed a gap between the cephalic ganglia after regeneration. Arrows denote the location between cephalic ganglia in both samples. (D) The gap at the midline in arrowhead(RNAi) regenerates was visualized with an anti-synapsin antibody that stains the neuropil of the CNS. (E) 86.8% of arrowhead(RNAi) animals showed a gap at the midline, while this gap was not visible in any control(RNAi) animals (N = 38 and 40, respectively). (F) Photoreceptor neurons were visualized with the anti-arrestin (VC-1) antibody after regeneration of control(RNAi) and arrowhead(RNAi) animals. (G) Photoreceptor neurons were disorganized (19%) and often failed to cross the midline (38%) in arrowhead(RNAi) animals (N = 21). Scale bars: 500 µm (A, C), 20 µm (B), 100 µm (D, F).
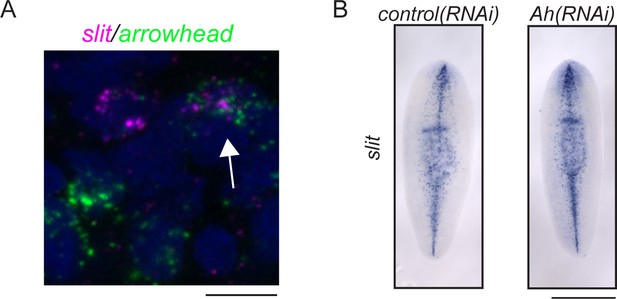
arrowhead and slit at the midline of the planarian brain.
(A) arrowhead and slit are coexpressed in some cells near the anterior midline of the cephalic ganglia (arrow). (B) slit expression is not visibly altered in arrowhead(RNAi) animals vs. controls. Scale bars: 10 µm (A), 500 µm (B).
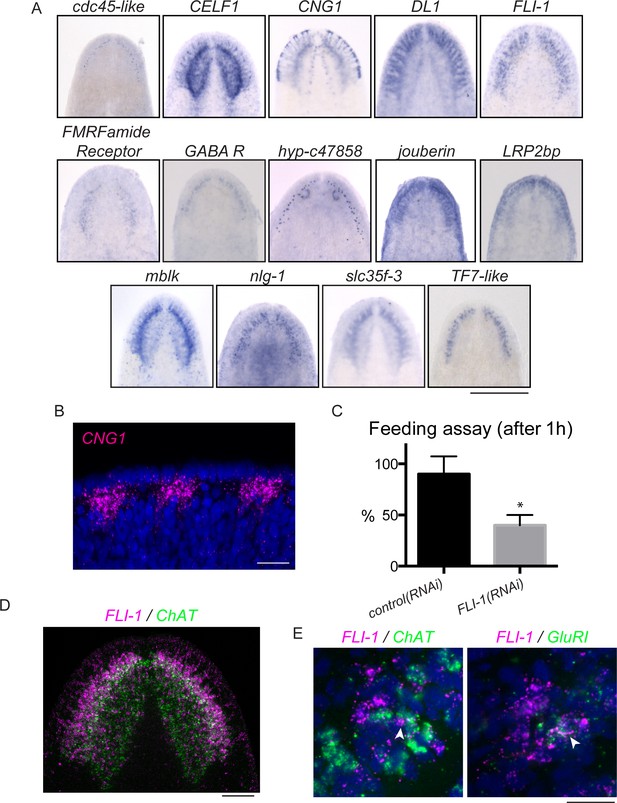
FLI-1 is expressed in the brain branches and functions in reacquisition of chemosensory behavior during regeneration.
(A) Fourteen upregulated genes were expressed in the brain branches of the planarian head. Expression patterns ranged from single rows of cells to broad expression in these structures. (B) CNG1 is expressed in large, sub-epithelial regions of the planarian head margin. (C) Knockdown of FLI-1 prior to regeneration caused a reduction of feeding in a maze-based experiment (shown in Figure 6—figure supplement 1E–H). Only 40% of FLI-1(RNAi) animals ate in this assay, compared to 90% of control(RNAi) animals (9–10 animals each condition for each replicate, 3 replicates). Error bars depict SEM; results were significant with p=6.8 × 10−5. (D) FLI-1 is expressed in lateral structures of the planarian brain (here marked with ChAT). (E) Some FLI-1+ cells express ChAT and GluR1 (7/42 and 12/44 respectively). Scale bars: 500 µm (A), 20 µm (B, E), 100 µm (D).
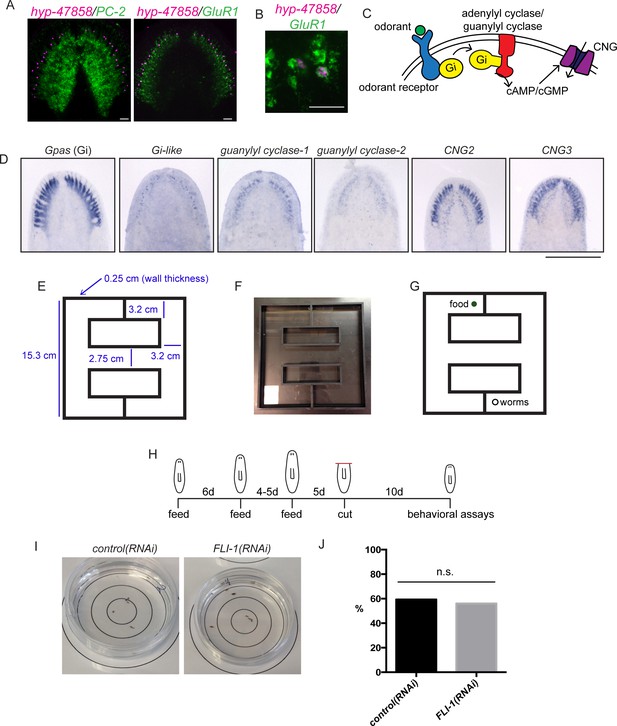
Assaying chemosensory function in the brain branches.
(A) Upper panel: brain branch markers like hyp-c47858 are expressed lateral to the main cephalic ganglia structures (here marked with PC-2). Lower panel: brain branch structures are labeled with GluR1 and newly identified brain branch genes are expressed in these structures. (B) Some hyp-c47858+ cells express GluR1 (12/44). (C) CNG channels function downstream of cyclic AMP or cyclic GMP signaling (reviewed in Pifferi et al., 2006). (D) Conserved components of olfactory signaling are also expressed in cells of the brain branches. (E–G) Diagrams and photo of the maze used for feeding assay that relies on chemosensory behavior. Dimensions are shown in (E) and the placement of food and animals is shown in (G). (H) The time course used for behavioral RNAi experiments. (I) control(RNAi) and FLI-1(RNAi) animals were placed in the interior-most 1.5 cm of a 6 cm petri dish and were allowed 15 min to move about the petri dish. At the conclusion of the time period, the number of worms outside the inner-most circle were counted; 59.2% of control(RNAi) animals (16/27) were outside the circle at this point and 56% of FLI-1(RNAi) animals were outside the circle in this assay, indicating similar movement after FLI-1(RNAi) (J). The difference between samples was not significant. Scale bars: 500 µm (D), 40 µm (A, B).
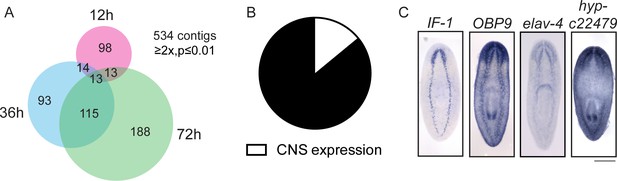
Expression profiling of genes downregulated during head regeneration.
(A) Venn diagram showing the 534 genes significantly downregulated compared to cut controls (≥2x downregulated, p≤0.01). The number of downregulated genes increased over time. (B) Pie chart depicting the fraction of downregulated genes examined that are expressed in the CNS (8 of 58 with clear expression patterns). (C) ISH showing expression of several downregulated genes in the planarian CNS. IF-1 is expressed in a pattern closely resembling the neuropil, while other markers (OBP9, elav-4, and hyp-22479) are typical of neural expression. Scale bars: 500 µm (C).
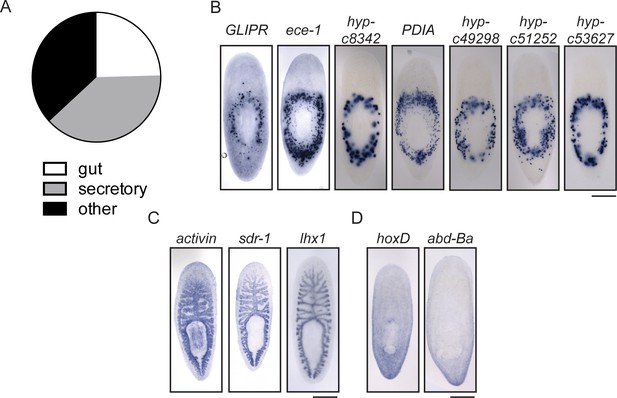
Expression patterns of genes downregulated during head regeneration.
(A) The predominant gene expression patterns of downregulated genes were in the peripharyngeal secretory region (20/58, 34.4%) and gut (16/58, 27.6%). Secretory expression patterns are shown in (B) and intestinal expression patterns are shown in (C). Some genes were also expressed in posterior patterns (D). Scale bars: 500 µm.
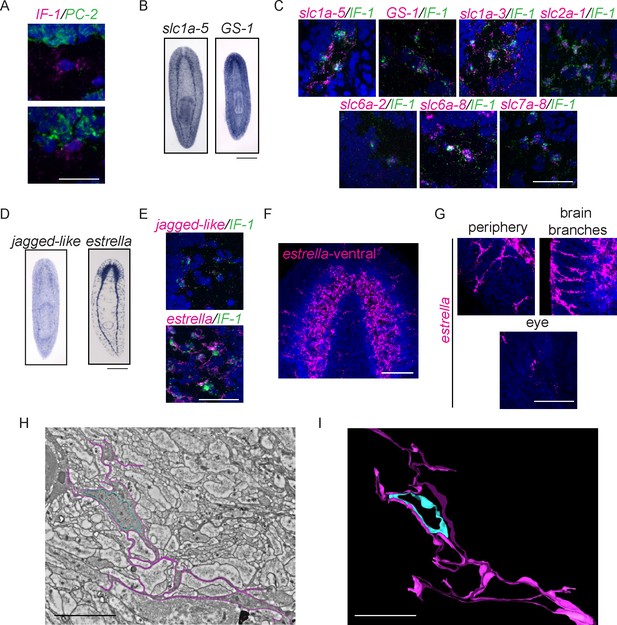
Intermediate Filament (IF-1) gene expression marks planarian glial cells.
(A) FISH was used to show that IF-1 is not coexpressed with prohormone convertase-2 (PC-2, 0/25 IF-1+ cells were PC-2+). (B) slc1a-5 and glutamine synthetase 1 (GS-1), genes required for glutamate uptake and conversion to glutamine are also expressed in an IF-1-like neuropil pattern. (C) IF-1 is co-expressed with slc1a-5 and GS-1 (46/62 IF-1+ cells were slc1a-5+ and 56/56 IF-1+ cells were GS-1+). IF-1+cells also express the solute carrier genes slc1a-3 (33/33), slc2a-1 (76/76), slc6a-2 (31/35), slc6a-8 (48/48) and slc7a-8 (63/63). Overlap was near perfect for some genes (all slc7a-8+ cells express IF-1), but other genes are more widely expressed (only 48/347 slc6a-8+ cells express IF-1). (D) Two genes from module 2 (a downregulated gene cluster) are also expressed in a neuropil-like pattern. jagged-like is expressed in puncta and a novel gene, estrella, is expressed strongly in the neuropil and in cells around the planarian body. (E) IF-1+ cells express jagged-like and estrella (48/48 and 61/61, respectively). (F–G) FISH with the estrella probe marks cells with extensive cytoplasmic projections in the neuropil, in the periphery of the animal body, near the brain branches, and in the eye, suggesting that estrella+ cells surround many components of the planarian nervous system. (H) An electron micrograph of a putative planarian glial cell in the neuropil of the ventral nerve cord. The cell membrane (determined by images acquired at higher magnification) is marked in magenta and the nuclear envelope is shown in cyan. The cytoplasm is more electron dense than that in surrounding axons. The nuclear morphology is elongated, with heterochromatin at the nuclear periphery; these features are not present in neurons visualized by EM. (I) A partial reconstruction of the cell in (H) through ten serial sections. The cell membrane and nuclear envelope are colored as above. Scale bars: 500 µm (B, D), 20 µm (A, C, E, G), 100 µm (F), 5 µm (H–I).
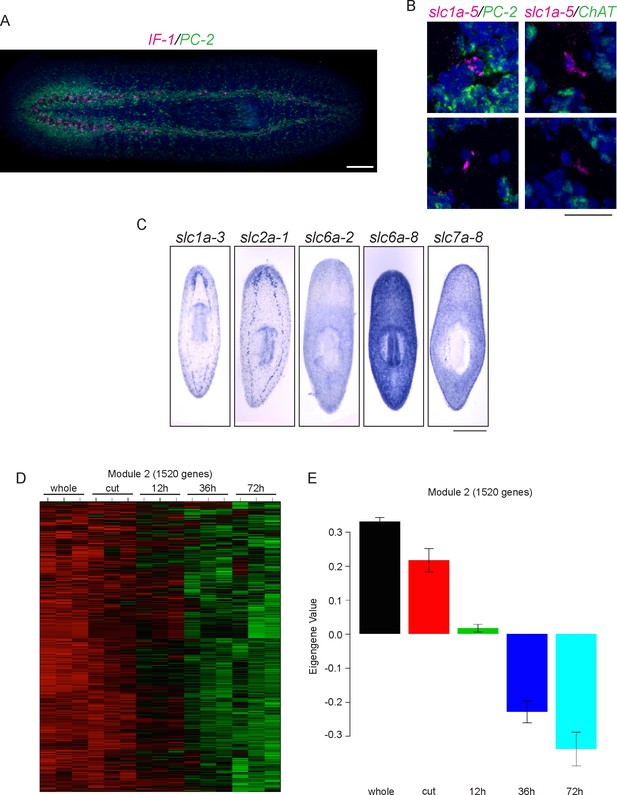
Characterization of planarian glial cells.
(A) A whole-animal double FISH showing the expression of IF-1 in PC-2- cells throughout the neuropil of the animal. (B) slc1a-5 is also expressed in cells that do not express PC-2 or ChAT (0/8 for each gene). (C) ISH showing solute carrier genes (slc) expressed in glial cells. (D) Module 2 from our clustering analysis contains IF-1 and many other downregulated genes. High gene expression is shown in red while downregulated gene expression is shown in green. (E) The average expression levels of genes in module 2 across samples is shown. This graph is configured as in Figure 1—figure supplement 2A. Scale bars: 100 µm (A), 20 µm (B), 500 µm (C).
Videos
Live imaging of control(RNAi) animals.
https://doi.org/10.7554/eLife.17002.013Live imaging of soxB2-2(RNAi) animals.
https://doi.org/10.7554/eLife.17002.014Electron micrographs of the glial cell shown in Figure 8H through serial sections.
https://doi.org/10.7554/eLife.17002.023Rotation of the reconstruction shown in Figure 8I.
https://doi.org/10.7554/eLife.17002.024Close-up from serial sections showing electron-dense projections of glial cells among axons in the neuropil.
https://doi.org/10.7554/eLife.17002.025Additional files
-
Supplementary file 1
All gene expression data from this study.
Read mapping and expression levels are given for each contig in our transcriptome. Column explanations are listed in the second tab in this spreadsheet.
- https://doi.org/10.7554/eLife.17002.026
-
Supplementary file 2
Clustering analysis for gene expression during regeneration.
Mapped reads were analyzed independently for weighted gene correlation network analysis (WGCNA). Column explanations are listed in the second tab in this spreadsheet, but the modules for each differentially expressed contig are in column C. A greater number of genes were identified as being significantly upregulated in this analysis compared to our initial study because this list also includes genes differentially regulated in either direction (upregulation or downregulation) between any two samples. Also, the thresholds for fold-change and significance were not as strict.
- https://doi.org/10.7554/eLife.17002.027
-
Supplementary file 3
(A) Expression patterns of upregulated genes cloned in this study. Expression patterns are as follows: BB- brain branch; Ep- epithelial; Ey- eyespots; G- gut; GC- germ cell; H- head; L- lateral margin; M- mesenchymal (parenchymal) expression pattern (could include muscle-like or neoblast-like patterns); N- neural; N+- neural plus another expression pattern (including neural with background); NS- neural subset; Ph- pharynx; Pol- polar expression; Pr- protonephridia; Pu- punctate; U- ubiquitous. (B) Genes with functions in regeneration. Includes genes with small brain regeneration phenotypes, plus arrowhead and SP6 which functions in eye regeneration. The results of ISH experiments, phenotype (including whether significant result was found in brain regeneration quantification), and follow-up RNAi experiments with sFRP-1 and smedwi-1 are shown. For sFRP-1 and smedwi-1 experiments, the results are summarized as follows: P- signal present; R- signal reduced; A- signal absent. For each contig, results from clustering analysis, expression in FACs analysis (LabbÉLabbé et al., 2012), and time points for significant upregulation vs. cut control (≥2x, p≤0.01) are also shown. BLAST hits and other annotation are also presented. (C) Genes with functions in homeostasis. Includes 26 genes that caused phenotypes during homeostasis in RNAi experiments. The table includes similar information given in Supplementary file 3B. (D) Genes with no phenotypes. Summary of all other genes from this analysis, including those that gave no detectable phenotype after RNAi. (E) List of genes used for qPCR experiments in this study. (F) Total preliminary and final qPCR results from Figure 3. Each tab includes results for one gene in our study. (G) Homologs of genes from this study. (H) Gene expression pattern for downregulated genes. Here we present a list of 71 downregulated genes from this study, including the expression patterns for these genes. (I) Annotation information and primers for genes described in this study.
- https://doi.org/10.7554/eLife.17002.028





