Tumor-induced MDSC act via remote control to inhibit L-selectin-dependent adaptive immunity in lymph nodes
Figures
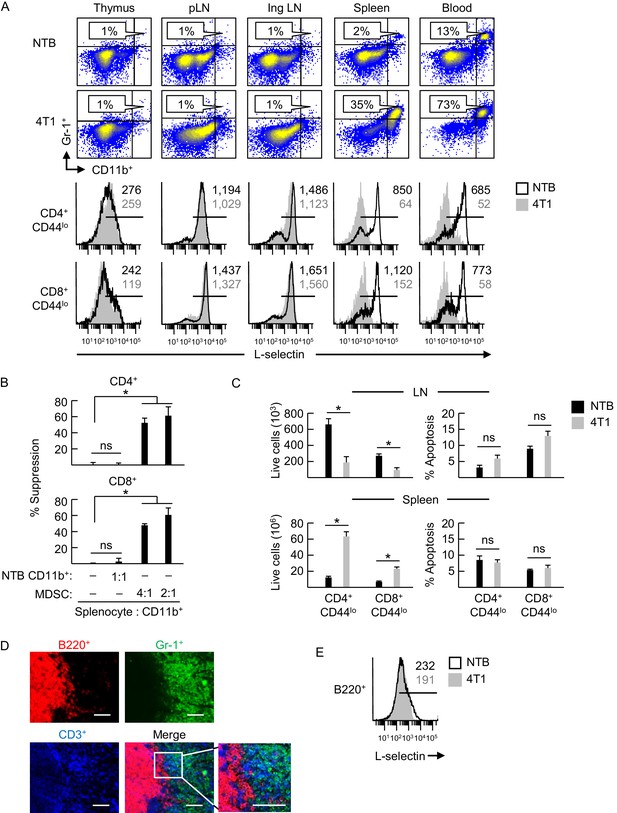
L-selectin downregulation on naïve T cells is restricted to specific anatomical compartments associated with MDSC accumulation.
(A) Flow cytometric analysis demonstrating CD11b+Gr-1+ MDSC accumulation (% CD45+ leukocytes, top) and L-selectin expression on CD4+CD44lo and CD8+CD44lo naïve T cells (below) in the indicated lymphoid organs (thymus, peripheral lymph node (pLN), inguinal (Ing) LN, tumor-draining Ing LN, spleen, and blood) of non-tumor bearing (NTB) mice or 4T1-bearing mice (tumor volume 1150 ± 150 mm3). (B) MDSC-T cell suppression assay. Splenic CD11b+ cells either from NTB mice or 4T1-bearing mice (tumor volume 2600 ± 380 mm3) were co-cultured with CFSE-labeled target splenocytes from NTB mice at the indicated splenocyte:myeloid cell ratios. Proliferation (based on CFSE dilution) in T cell subsets was measured 72 hr after addition of anti-CD3/CD28 antibody-conjugated activation beads. Percent suppression is for one experiment (mean±s.e.m, n = 3 replicates per condition) and is representative of three independent experiments. (C) Total numbers of viable naïve CD4+CD44lo and CD8+CD44lo T cell subsets (left) and percentages of annexin V+ early apoptotic T cells (right) were quantified by flow cytometric analysis from peripheral lymph nodes and spleens of NTB or 4T1-bearing mice (tumor volume 1340 ± 242 mm3). Data (mean±s.e.m.) are of one experiment (n = 3 mice per group) and are representative of two independent experiments. (B,C) *p<0.05; ns, not significant; data were analyzed by unpaired two-tailed Student’s t-test. (D) Splenic cryosections from NTB and 4T1-bearing mice stained for B220+, Gr-1+ and CD3+ cells; parallel fluorocytometric analysis (as in A) confirmed that >90% of splenic Gr-1+ cells co-expressed CD11b. Scale bar, 50 µm. (E) L-selectin expression on splenic B220+ cells of NTB and 4T1–bearing mice. (A,E) Horizontal lines in histograms indicate positively stained cells; numbers are mean fluorescence intensity. (A,B,D–E) Data are for one experiment and are representative of ≥ three independent experiments (n = 3 replicates or mice per group). pLN, peripheral lymph node; Ing LN, inguinal lymph node; NTB, non-tumor bearing.
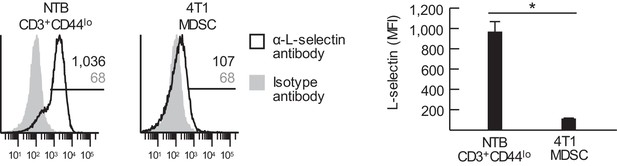
MDSC express low levels of L-selectin.
Flow cytometric analysis of L-selectin expression on splenic CD3+CD44lo naïve T cells from non-tumor bearing (NTB) mice and CD11b+Gr-1+ MDSC from 4T1-bearing mice (tumor volume 1050 ± 150 mm3). Horizontal lines in histograms indicate positively stained cells; numbers are mean fluorescence intensity (MFI). Data (mean±s.e.m.) are of one experiment (n = 3 mice per group) and are representative of two independent experiments. *p<0.05; data were analyzed by unpaired two-tailed Student’s t-test. NTB, non-tumor bearing; MFI, mean fluorescence intensity.
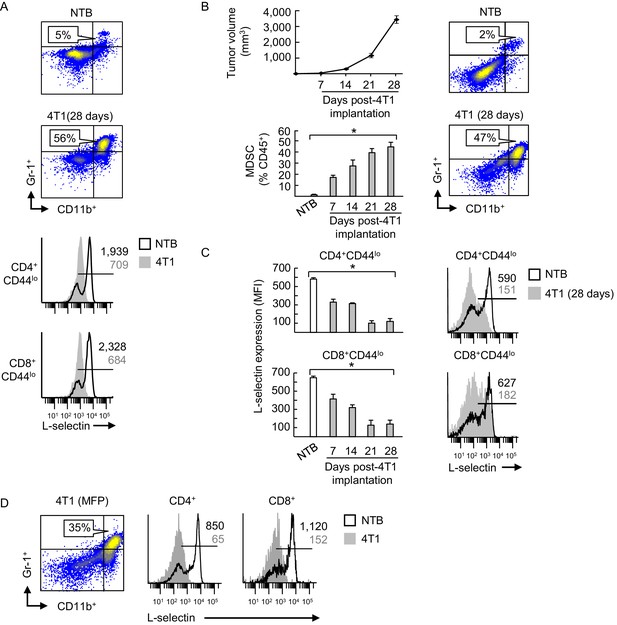
Inverse correlation between MDSC expansion and L-selectin expression on naive CD4+ and CD8+ T cells during 4T1 tumor progression.
(A) Fluorocytometric analysis of CD11b+Gr-1+ MDSC accumulation (% CD45+ leukocytes, top) and L-selectin expression on splenic CD4+CD44lo and CD8+CD44lo naïve T cells (below) of non-tumor bearing (NTB) or 4T1-bearing male mice (tumor volume ~2000 mm3). (B) Tumor volume (top left) and accumulation of splenic CD11b+Gr-1+ MDSC (bottom left) in 4T1-tumor-bearing female mice were measured over time. Representative flow cytometric analysis is shown for CD11b+Gr-1+ cell frequency (% CD45+ leukocytes) of NTB controls compared to 28 days post-4T1 implantation (right). (C) L-selectin on splenic CD4+CD44lo and CD8+CD44lo naïve T cells in 4T1-bearing female mice was assessed during tumor progression and compared with NTB mice (left). Representative L-selectin profiles are shown for NTB and 4T1-bearing mice at 28 days post-tumor implantation (right). (B,C) Data (mean±s.e.m.) are for one representative experiment (n = 3 mice per group); *p<0.05; data were analyzed by unpaired two-tailed Student’s t-test. (D) Flow cytometric analysis of CD11b+Gr-1+ MDSC (% CD45+ leukocytes, left) and L-selectin expression profiles on CD4+ and CD8+ T cells (right) in spleens of female mice implanted with orthotopic 4T1 tumors in the mammary fat pad (MFP; tumor volume >1500 mm3). (A,C–D) Horizontal lines in histograms indicate positively stained cells; numbers are mean fluorescence intensity (MFI). (A–D) Data are of one experiment (n = 3 mice per group) and are representative of > three independent experiments. NTB, non-tumor bearing; MFP, mammary fat pad; MFI, mean fluorescence intensity.
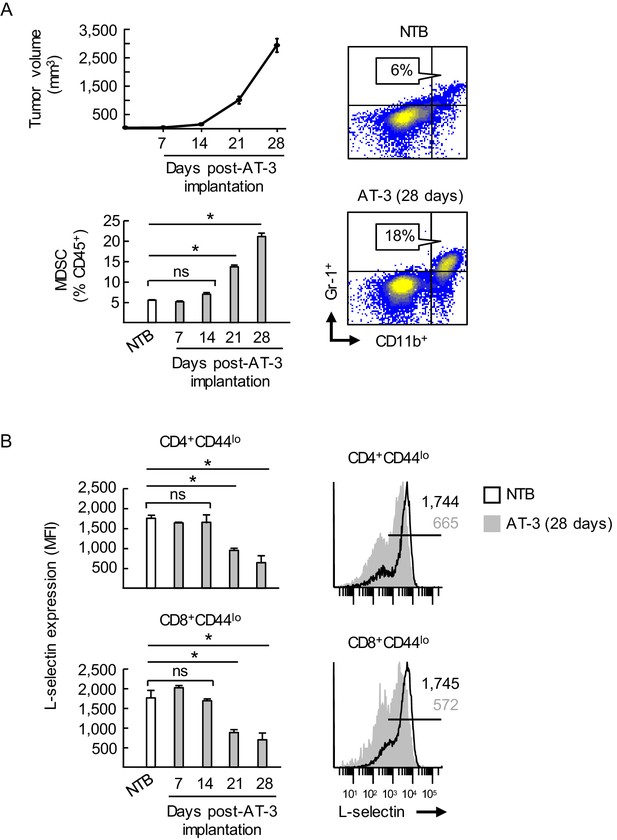
MDSC expansion coincides with L-selectin downregulation on naive CD4+ and CD8+ T cells during AT-3 tumor progression.
(A) Tumor volume (top left) and accumulation of splenic CD11b+Gr-1+ MDSC (bottom left) in AT-3-tumor-bearing mice were measured over time. Representative flow cytometric analysis is shown (right) for CD11b+Gr-1+ cell frequency (% CD45+ leukocytes) in the spleens of non-tumor bearing (NTB) controls compared with AT-3 tumors-bearing mice (28 days post-tumor implantation). (B) L-selectin on splenic CD4+CD44lo and CD8+CD44lo naïve T cells in AT3-bearing mice was compared to L-selectin levels on T cells from NTB mice (left). Representative L-selectin profiles of NTB and AT-3-bearing mice are shown (right). Horizontal lines in histograms indicate positively stained cells; numbers are mean fluorescence intensity (MFI). (A,B) Data (mean±s.e.m.) are from one experiment (n = 3 mice per group) and are representative of > three independent experiments. *p<0.05; ns, not significant; data were analyzed by unpaired two-tailed Student’s t-test. NTB, non-tumor bearing; MFI, mean fluorescence intensity.
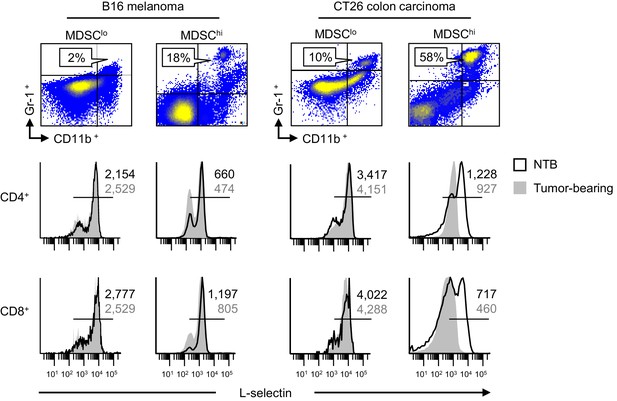
L-selectin down-modulation is associated with MDSC expansion in different tumor types.
Flow cytometric analysis of CD11b+Gr-1+ MDSC (% CD45+ leukocytes, top) and L-selectin expression profiles on CD4+ and CD8+ T cells (bottom) in spleens of mice implanted with subcutaneous B16 melanoma and CT26 colorectal tumors (in C57BL/6 mice or BALB/c mice, respectively). Tumor volume >1500 mm3 for all tumor models. CD11b+Gr-1+ cells in non-tumor bearing (NTB) controls were <3% of CD45+ leukocytes (data not shown). Comparative analysis of L-selectin expression is shown for tumor-bearing mice and NTB controls; horizontal lines in histograms indicate positively stained cells; numbers are mean fluorescence intensity. Data are representative of ≥ three independent experiments. NTB, non-tumor bearing.
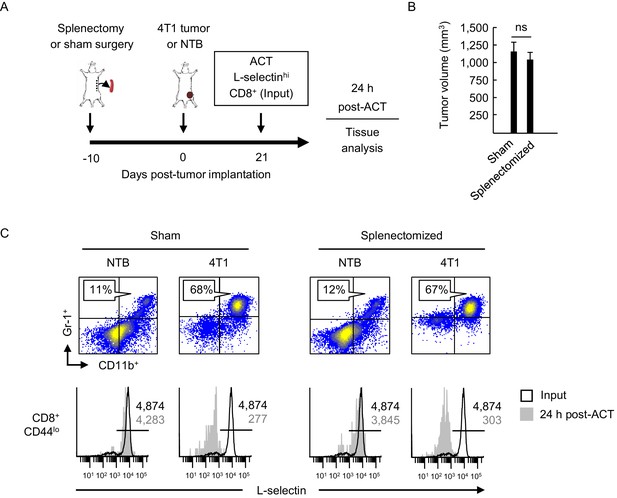
L-selectin loss occurs on naïve T cells within the MDSC-enriched blood compartment of splenectomized mice.
(A) Experimental design in which splenectomy or sham surgery was performed in non-tumor bearing (NTB) mice. Mice were then inoculated with 4T1 tumor or maintained as NTB controls. Fluorescently-labeled L-selectinhi CD8+ T cells isolated from NTB mice (input) were used for intravenous adoptive cell transfer (ACT) into tumor-bearing mice or in NTB controls. (B) 4T1 tumor volume of sham and splenectomized mice at 21 days post-4T1 implantation. Data (mean±s.e.m.) are for a single representative experiment (n = 3 mice per group); ns, not significant; data were analyzed by unpaired two-tailed Student’s t-test. (C) Representative flow cytometric analysis showing accumulation of CD11b+Gr-1+ cells (% CD45+ leukocytes, top) and L-selectin expression (bottom) on CD8+CD44lo T cells before ACT (input) and 24 hr post-ACT in the blood of sham or splenectomized NTB and 4T1-bearing recipient mice. Horizontal lines in histograms indicate positively stained cells; numbers are mean fluorescence intensity. (A–C) Data are representative of three independent experiments (n = 3 mice per group). NTB, non-tumor bearing; ACT, adoptive cell transfer.
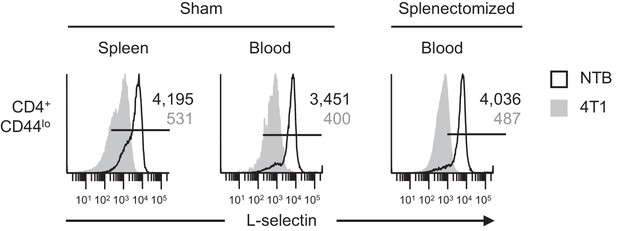
L-selectin downregulation on naïve CD4+CD44lo T cells occurs in the MDSC-enriched peripheral blood compartment of splenectomized mice.
Mice underwent sham surgery or splenectomy 10 days prior to 4T1 tumor inoculation as described for Figure 2. L-selectin expression was then analyzed 22 days after tumor inoculation (tumor volume ~1000 mm3) on endogenous naïve CD4+CD44lo T cells in the spleen or blood of non-tumor bearing (NTB) and 4T1-bearing sham and splenectomized mice. Data are from one experiment (n = 3 mice per group) and are representative of three independent experiments. Horizontal lines in histograms indicate positively stained cells; numbers are mean fluorescence intensity. NTB, non-tumor bearing.
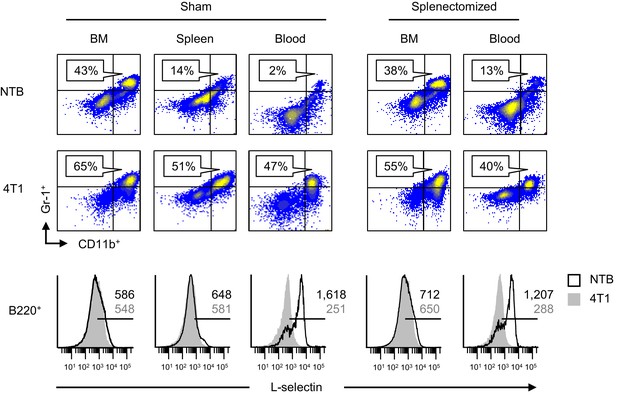
L-selectin downregulation on B cells occurs exclusively in the peripheral blood.
Splenectomy or sham surgery was performed 10 days prior to 4T1 tumor inoculation and tissues were evaluated for MDSC expansion and L-selectin expression 22 days after tumor implantation (tumor volume ~1000 mm3). Flow cytometric analysis of CD11b+Gr-1+ cell burden (% CD45+ leukocytes; top) and L-selectin expression on endogenous B220+ B cell populations (bottom) in the indicated organs (BM, bone marrow; spleen; blood) of sham or splenectomized non-tumor bearing (NTB) and 4T1-tumor bearing mice. Horizontal lines on histograms indicate positively stained cells; numbers are mean fluorescence intensity. Data are representative of ≥ three independent experiments (n = 3 mice per group); NTB, non-tumor bearing; BM, bone marrow.
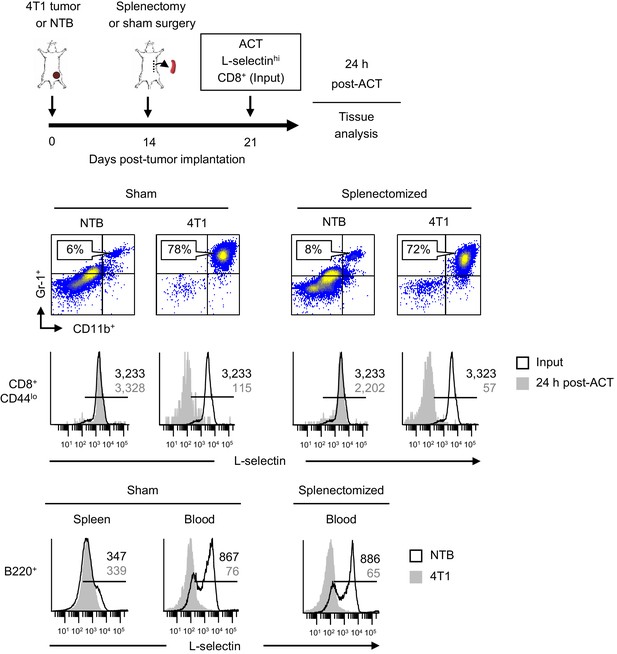
L-selectin downregulation on T and B cells occurs in the MDSC-enriched peripheral blood compartment of splenectomized mice.
Schematic is shown for experimental design in which 4T1-bearing mice underwent splenectomy or sham surgery 14 days-post tumor implantation. Fluorescently-labeled CD8+ T cells isolated from NTB mice (input) were intravenously transferred 21 days post-4T1 inoculation (tumor volume 1499 ± 449 mm3, n = 3 mice). Representative flow cytometric dot plots demonstrating CD11b+Gr-1+ MDSC accumulation in blood (% of CD45+ leukocytes, top) and histograms (below) for L-selectin expression on CD8+CD44lo T cells before adoptive cell transfer (ACT) (input) and on circulating transferred T cells 24 hr post-ACT in sham and splenectomized NTB recipients or 4T1-bearing recipients. L-selectin profiles are also shown for endogenous B220+ B cells in spleen and blood of sham and splenectomized NTB mice and 4T1-tumor bearing mice (bottom histograms). Data are from one experiment (n = 3 mice per group). Horizontal lines in histograms indicate positively stained cells; numbers are mean fluorescence intensity. NTB, non-tumor bearing; ACT, adoptive cell transfer.
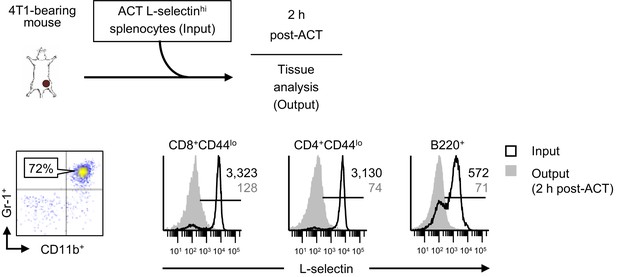
L-selectin loss on naïve T and B lymphocytes occurs rapidly in the blood of tumor-bearing mice.
Schematic is shown for experiment design in which fluorescently-labeled, L-selectinhi splenocytes isolated from non-tumor bearing donors (input) were adoptively transferred into 4T1-bearing recipient mice (tumor volume 1026 ± 54 mm3, n = 2 mice). Representative flow cytometric dot plots demonstrating CD11b+Gr-1+ MDSC accumulation in blood (% CD45+ leukocytes, left) and histograms for L-selectin expression (right) on CD8+CD44lo, CD4+CD44lo, and B220+ splenocytes before adoptive cell transfer (ACT) (input) and on circulating transferred lymphocyte populations 2 hr post-ACT into 4T1-bearing recipient mice. Data are from one experiment (n = 2 mice per group). Horizontal lines in histograms indicate positively stained cells; numbers are mean fluorescence intensity. NTB, non-tumor bearing; ACT, adoptive cell transfer.
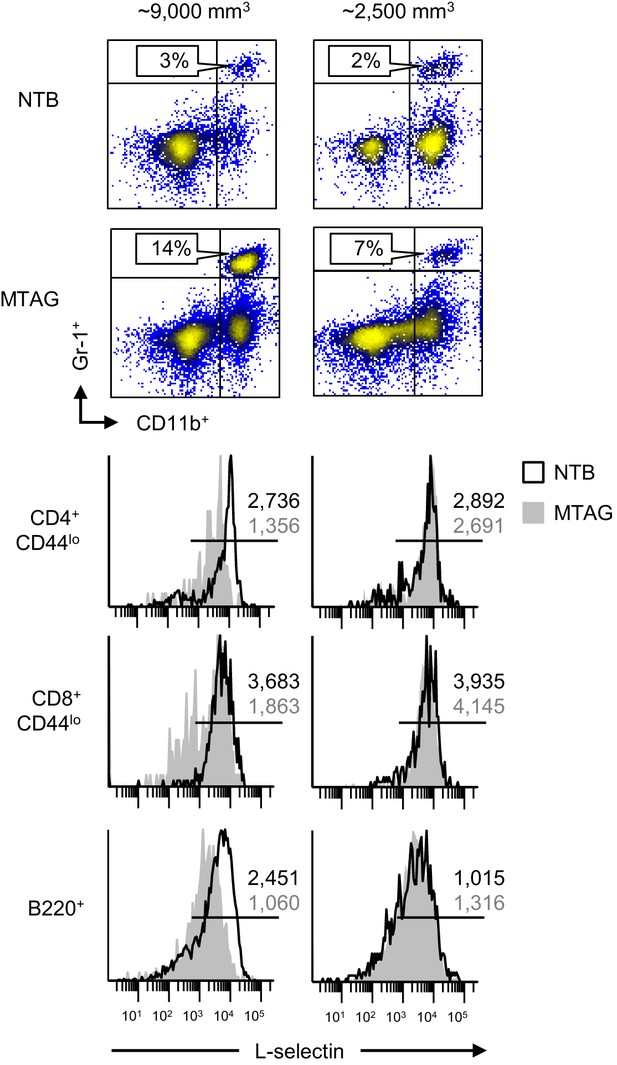
MDSC-associated downregulation of L-selectin on naïve T and B lymphocytes occurs in autochthonous mammary carcinoma MTAG mice.
Blood was collected from individual transgenic MTAG mice (with the indicated total tumor volume) and from age-matched non-tumor bearing (NTB) wildtype littermates. Flow cytometric analysis is shown for CD11b+Gr-1+ cell accumulation (% CD45+ leukocyte, above) and L-selectin expression on naive CD4+CD44lo and CD8+CD44lo T cells and B220+ B cells (below). Data are from one experiment (n ≥ 3 mice per group) and are representative of three independent experiments. Horizontal lines in histograms indicate positively stained cells; numbers are mean fluorescence intensity. NTB, non-tumor bearing.
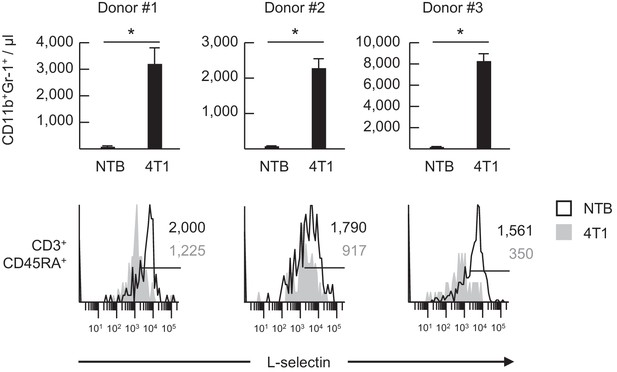
L-selectin on human naïve T cells is downregulated following transfer into 4T1 tumor-bearing mice.
Normal human-donor peripheral blood lymphocytes were adoptively transferred into non-tumor bearing (NTB) severe-combined immunodeficient (SCID) mice or 4T1-bearing SCID mice at 21 days-post tumor implantation (average tumor volume for three experiments, 1190 ± 197 mm3). The burden of CD11b+Gr-1+ cells in the blood of NTB and 4T1-bearing SCID mice was measured 24 hr after adoptive cell transfer (top) and was quantified based on blood volume for T- and B-cell deficient SCID mice. Data (mean±s.e.m.) are from three independent experiments for three different lymphocyte donors (n = 3 mice per group in each experiment). *p<0.05; data were analyzed by unpaired two-tailed Student’s t-test. Flow histograms depict L-selectin expression of individual donor CD3+CD45RA+ T cells at 24 hr post-ACT in NTB or 4T1 tumor-bearing recipient SCID mice (bottom). Horizontal lines in histograms indicate positively stained cells; numbers are mean fluorescence intensity. NTB, non-tumor bearing.
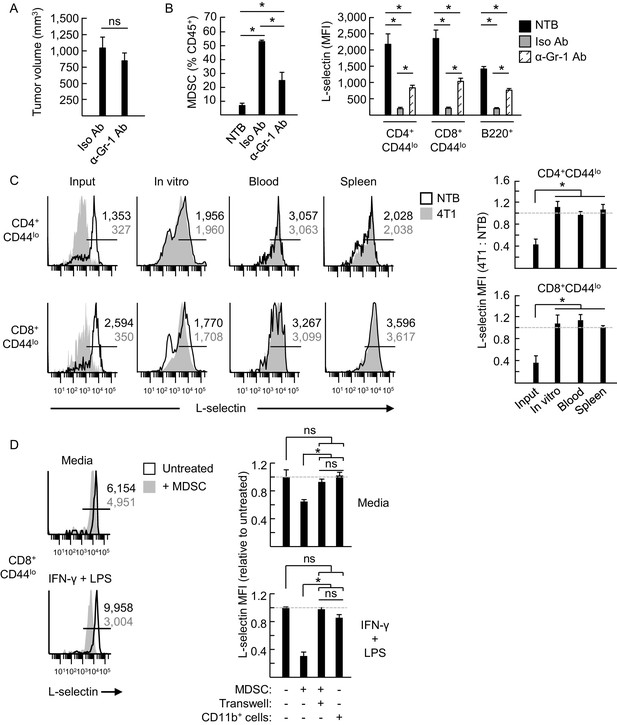
MDSC induce L-selectin loss on T and B lymphocytes in 4T1 tumor-bearing mice via a contact-dependent mechanism.
(A) 4T1-tumor-bearing mice were treated with anti-Gr-1 antibodies (α-Gr-1 Ab) or isotype control antibodies (Iso Ab) every 3 days for three weeks starting at three days post-tumor implantation. Endpoint tumor volumes are shown. (B) CD11b+Gr-1+ MDSC burden (% CD45+ leukocytes, left) and L-selectin expression (mean fluorescence intensity, MFI) on endogenous CD4+CD44lo, CD8+CD44lo, and B220+ lymphocytes (right) were measured in the blood of NTB or in 4T1-bearing mice treated with Iso Ab or anti-Gr-1 Ab. (C) Splenocytes from NTB or 4T1-bearing mice were depleted of CD11b+ cells by magnetic bead isolation (94.8 ± 1.8% depletion, n = 3 mice). These cell populations were then fluorescently-labeled with different tracking dyes, co-mixed at a 1:1 ratio, and cultured in vitro or adoptively transferred into NTB recipients. Representative flow cytometric L-selectin profiles are shown for naïve CD4+CD44lo and CD8+CD44lo T cells before culture or adoptive cell transfer (ACT) (input) and four days after in culture (in vitro) or for cells recovered from blood and spleen post-ACT (left). Quantification of L-selectin modulation (right) is based on a ratio of the MFI for T cells from 4T1-bearing mice relative to NTB mice; dashed lines indicate NTB control. (D) MDSC or CD11b+ control cells were isolated from 4T1-tumor bearing mice (tumor volume >1000 mm3) or NTB mice, respectively. Myeloid cells were then co-cultured with fluorescently-labeled splenocytes from NTB mice (10:1 ratio) in media alone or with IFN-γ (20 U/mL) and LPS (100 ng/mL). MDSC and splenocytes were separated by transwell inserts (0.4 μm pore size) in the indicated co-cultures. After 24 hr, L-selectin expression on viable naive CD8+CD44lo T cells was analyzed by flow cytometry; representative profiles are shown (left). Relative changes in L-selectin expression were normalized to untreated CD8+CD44lo T cells (indicated by dashed lines; right). (A–D) Data (mean±s.e.m.) are from one experiment (n = 3 mice per group or ≥3 replicates per group) and are representative of ≥ two independent experiments. *p<0.05; ns, not significant; data were analyzed by unpaired two-tailed Student’s t-test. (C,D) Horizontal lines in histograms indicate positively stained cells; numbers are MFI. NTB, non-tumor bearing; Ab, antibody; Iso, isotype; MFI, mean fluorescence intensity.
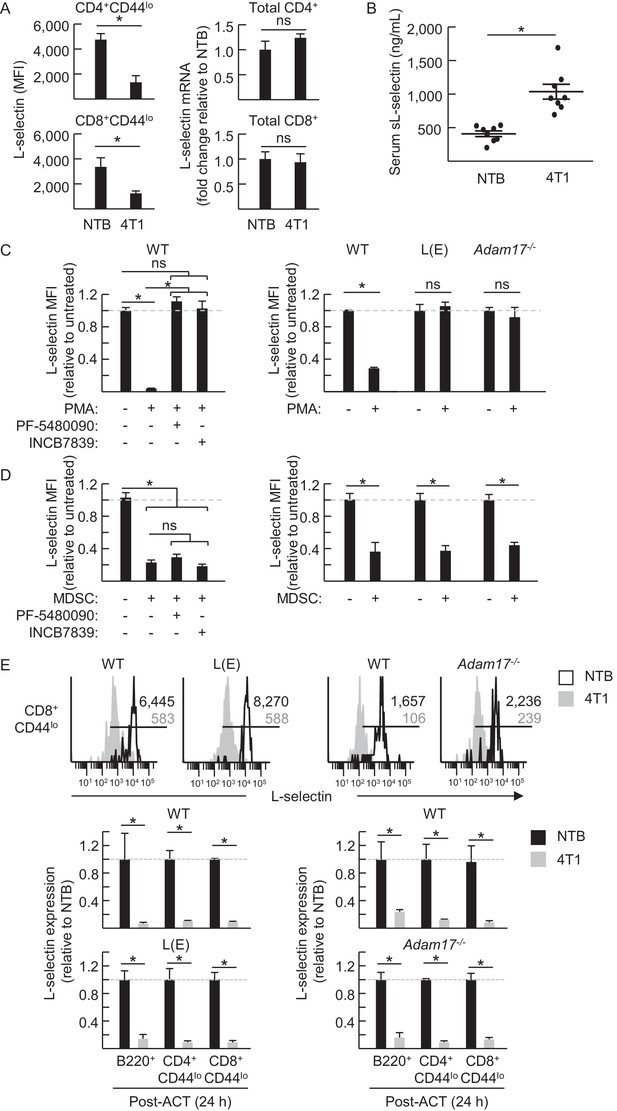
MDSC-induced L-selectin downregulation is post-transcriptional and does not depend on the ADAM17 metalloprotease.
(A) Flow cytometric analysis of surface L-selectin (mean fluorescence intensity; MFI) on splenic naïve CD4+CD44lo and CD8+CD44lo T cells of non-tumor bearing (NTB) and 4T1-bearing mice is shown (left). L-selectin mRNA expression in splenic CD4+ and CD8+ T cells from NTB and 4T1-bearing mice was determined by qRT-PCR with fold-change normalized with β-actin (right). (B) Soluble (s)L-selectin in serum of individual NTB and 4T1-bearing mice was assessed by ELISA. (A,B) Data are from three independent experiments (n ≥ 2 mice per group in each experiment; tumor volume >1500 mm3; average frequency of splenic CD11b+Gr-1+ cells (% CD45+ leukocytes) in tumor-bearing mice was ~40%). (C) Splenocytes were isolated from NTB wildtype (WT) C57BL/6 mice, L(E)-selectin transgenic mice, Adam17flox/flox/Vav1-Cre mice (Adam17−/−), or age-matched WT littermate controls. WT splenocytes were pretreated for 30 min with the ADAM17-specific inhibitor PF-5480090 (10 μM) or the ADAM17/10-specific inhibitor INCB7839 (20 μM). WT, L(E), and Adam17−/−splenocytes were then cultured 2 hr with or without phorbol myristate acetate (PMA, 100 ng/mL). Surface L-selectin (MFI) on viable naïve CD8+CD44lo T cells relative to untreated controls was determined by fluorocytometric analysis. (D) Splenic CD11b+Gr-1+ MDSC were purified from 4T1-bearing mice; splenocytes were from various NTB mice as described in (C). MDSC and WT splenocytes were both pretreated for 30 min with or without PF-5480090 or INC7839. MDSC and splenocytes from WT, L(E), or Adam17-/- mice were then co-cultured at a 10:1 ratio for 24 hr in media containing IFN-γ (20 U/mL) and LPS (100 ng/mL). L-selectin on viable naïve CD8+CD44lo T cells was assessed by flow cytometric analysis. (E) Fluorescently-labeled WT, L(E), and Adam17−/− splenocytes (i.e., from NTB mice) were adoptively transferred into NTB severe-combined immunodeficient (SCID) mice or 4T1-bearing SCID mice at 21 days-post tumor implantation (average tumor volume for all experiments, 1102 ± 191 mm3; average circulating CD11b+Gr-1+ frequencies in NTB SCID recipients, 75 ± 8 cells/µL blood, and 4T1-bearing SCID recipients, 4081 ± 876 cells/µL blood). After 24 hr post-ACT, L-selectin was assessed by flow cytometry on transferred splenocytes recovered from the blood of NTB and 4T1-bearing SCID mice. Representative flow histograms depict L-selectin expression on naïve CD8+CD44lo T cells (above); horizontal lines indicate positively stained cells, numbers are mean fluorescence intensity. Normalized data for L-selectin expression on B220+, CD4+CD44lo, CD8+CD44lo cells 24 hr post-adoptive transfer are for one representative experiment (n ≥ 2 mice per group) (below). (A–E) *p<0.05; ns, not significant; all data (mean±s.e.m.) were analyzed by unpaired two-tailed Student’s t-test. (C–E) Data are representative of ≥ two independent experiments (n ≥ 2 replicates or mice per group) and are normalized to untreated or NTB controls (indicated by dashed lines). NTB, non-tumor bearing; WT, wildtype; MFI, mean fluorescence intensity; sL-selectin, soluble L-selectin; ACT, adoptive cell transfer.
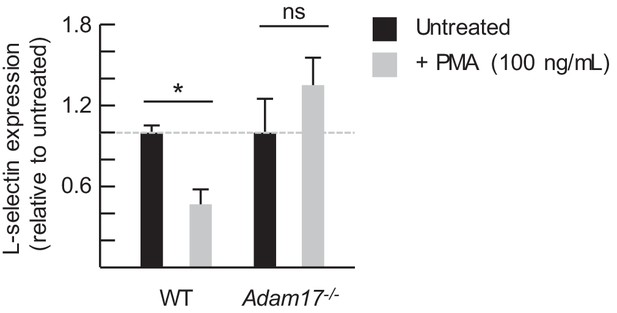
PMA-induced loss of L-selectin depends on cis-acting ADAM17.
Wildtype (WT) and Adam17−/− splenocytes were labeled with different fluorescent dyes prior to co-culture at a 10:1 ratio for 2 hr with or without phorbol myristate acetate (PMA, 100 ng/mL). L-selectin on viable WT and Adam17−/− CD8+CD44lo T cells was assessed by flow cytometric analysis and compared to untreated controls (indicated by dashed line). Data (mean±s.e.m.) are from one experiment (n = 2 replicates per group). *p<0.05; ns, not significant; data were analyzed by unpaired two-tailed Student’s t-test. WT, wildtype.
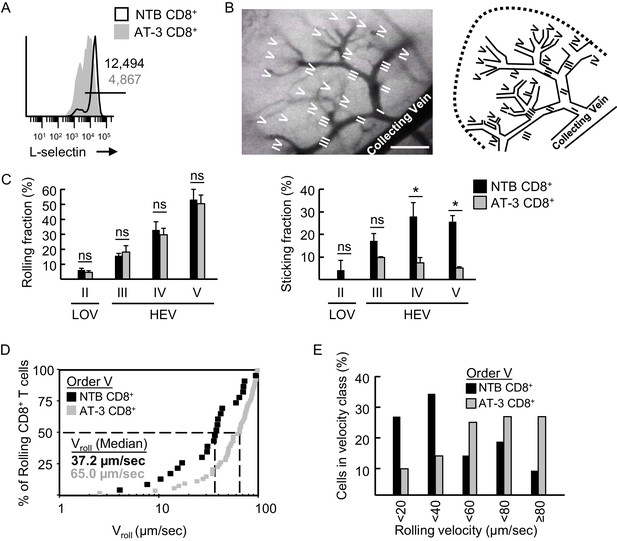
L-selectin-deficient CD8+ T cells from AT-3-bearing mice exhibit reduced firm adhesion and faster rolling velocity in LN HEV.
(A) Flow cytometric analysis of L-selectin expression prior to intravenous adoptive transfer of CD8+ T cells from non-tumor bearing mice (NTB CD8+) or CD8+ T cells from AT-3–bearing mice (AT-3 CD8+). Horizontal line in histogram indicates positively stained cells; numbers are mean fluorescence intensity. (B) Representative photomicrograph (left) and schematic (right) of the postcapillary vascular tree visualized by epifluorescence intravital microscopy in inguinal LN of NTB recipient mice. Hierarchical branches of venular orders I and II (low-order venules, LOV) and III-V (high-order venules corresponding to HEV) are labeled. Direction of blood flow in post-capillary venules is from order V to order I venules which directly empty into collecting veins. Scale bar, 100 µm. (C) Rolling fraction and sticking fraction of fluorescently-labeled CD8+ T cells isolated from NTB or AT-3–bearing mice following adoptive transfer into NTB recipients. Data (mean±s.e.m.) are from three independent experiments; n ≥ 3 mice per group. *p<0.05; ns, not significant; data were analyzed by unpaired two-tailed Student’s t-test. (D) Cumulative rolling velocity curve was generated by measuring the velocities of transferred CD8+ T cells in order V venules of inguinal LN in three independent experiments. Comparison of cumulative rolling velocity plot data was performed by unpaired two-tailed Student’s t-test; L-selectinhi NTB CD8+ T cells versus L-selectinint/lo AT-3 CD8+ T cells, *p<0.01. (E) Distributions of rolling velocities in velocity histograms in order V venules were evaluated by a nonparametric Mann-Whitney U test; L-selectin+ NTB CD8+ T cells versus L-selectinint/lo AT-3 CD8+ T cells, *p<0.01. NTB, non-tumor bearing.
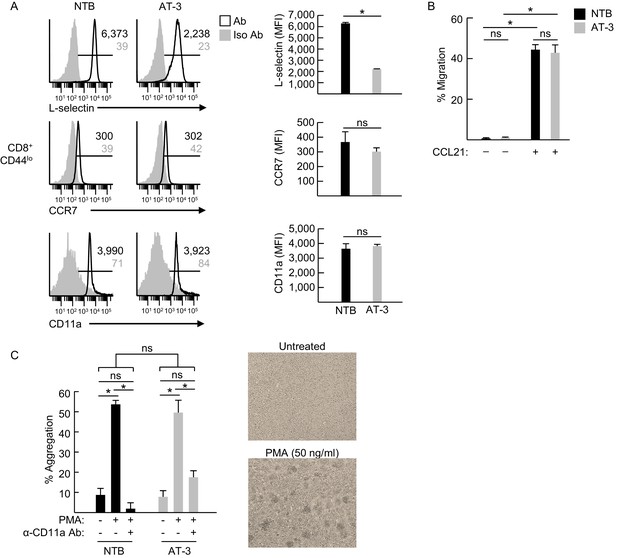
Adhesion molecule expression and function on CD4+ and CD8+ T lymphocytes.
(A) Single parameter fluorocytometric histograms (left) depicting expression of L-selectin, CCR7, and CD11a (i.e., the αL subunit of LFA-1) on CD8+CD44lo splenic T cells derived from non-tumor bearing (NTB) mice or AT-3-bearing mice (three weeks post-AT-3 implantation, tumor volume ~4000 mm3). Horizontal lines in histograms indicate positively stained cells; numbers are mean fluorescence intensity (MFI); trafficking molecule expression (MFI) is quantified for n = 3 mice (right). (B) Transwell chemotaxis assays compared migration of purified splenic CD8+ T cells from NTB and AT-3-bearing mice in response to recombinant CCL21 (70 nM). (C) LFA-1-dependent homotypic aggregation of CD8+ T cells was induced by phorbol myristate acetate (PMA). Purified CD8+ splenic T cells from NTB and AT-3-bearing mice were pretreated with or without α-CD11a antibody (10 ug/mL) for 15 min. Cells were then cultured with and without PMA (50 ng/mL) for 18 hr. Percent aggregation was quantified based on total number of input cells (left); representative photomicrographs of untreated and PMA-treated CD8+ T cells are shown (right). (A–C) Data (mean±s.e.m.) are representative of ≥ two independent experiments (n ≥ 2 mice per treatment group with ≥3 replicates per condition). *p<0.05; ns, not significant; data were analyzed by unpaired two-tailed Student’s t-test. NTB, non-tumor bearing; Iso, isotype; Ab, antibody; MFI, mean fluorescence intensity.
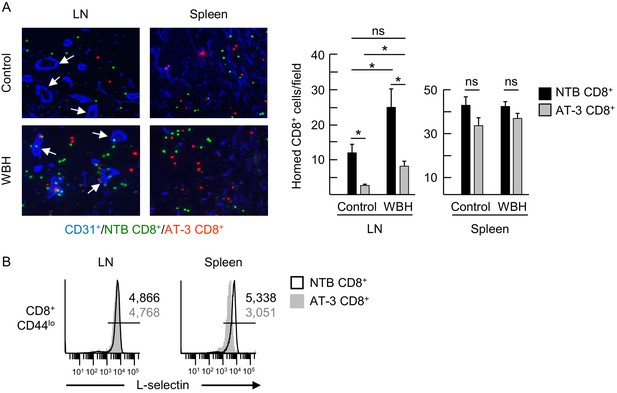
L-selectin down-modulation on CD8+ T cells of AT-3-bearing mice inhibits trafficking across LN HEV.
(A) Competitive homing studies used a 1:1 ratio of splenic CD8+ T cells isolated from non-tumor bearing mice (NTB CD8+, green) and AT-3–bearing mice (AT-3 CD8+, red). One hour after intravenous adoptive transfer of CD8+ T cells, the extent of homing of fluorescently-tagged transferred cells was examined in LN or spleens either of NTB controls (i.e., homeostatic trafficking) or in an inflammatory model in which the core body temperature of tumor-free recipient mice was elevated by whole body hyperthermia (WBH, 39.5 ± 0.5°C for 6 hr) prior to T cell transfer. Representative photomicrographs of fluorescently-labeled homed cells in histological LN and splenic cryosections; counterstaining with CD31 antibody identified cuboidal high endothelial venules (HEV, denoted by white arrows, left) in LN. Scale bar, 50 µm. Note the majority of T cells detected in images extravasated across HEV and were located in the LN parenchyma. Data (mean±s.e.m.; right) are from one experiment (n = 3 mice per group) and are representative of three independent experiments. *p<0.05; ns, not significant; data were analyzed by unpaired two-tailed Student’s t-test. (B) L-selectin expression profiles of CD8+ T cells (originating from NTB mice or AT-3–bearing mice) that were recovered 1 hr after adoptive transfer in LN or spleen of NTB recipients. Horizontal lines in histograms indicate positively stained cells; numbers are mean fluorescence intensity. NTB, non-tumor bearing; WBH, whole body hyperthermia.
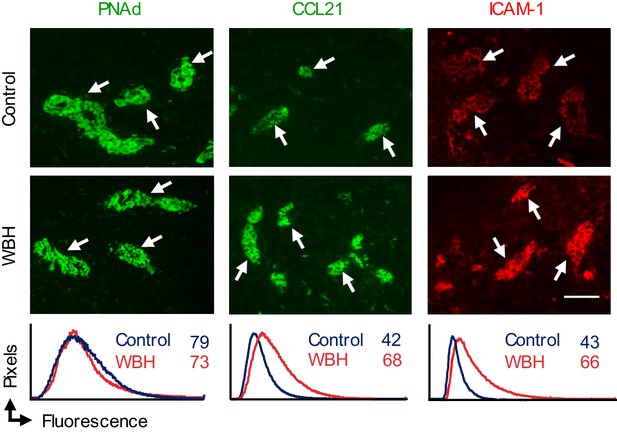
Adhesion molecule expression on LN HEV.
Lymph node cryosections of NTB mice were stained for peripheral lymph node addressin (PNAd, green), CCL21 (green), or intercellular adhesion molecule-1 (ICAM-1, red) in non-tumor bearing mice maintained under homeostatic control conditions or in inflamed high endothelial venules (HEV) of mice pre-treated with whole body hyperthermia (WBH). White arrows denote position of cuboidal HEV determined by counterstaining tissues with PNAd or CD31-specific Ab (not shown). Trafficking molecule expression on HEV was assessed by quantitative image analysis for immunofluorescence staining intensity in cuboidal HEV and depicted by histograms. The x-axis indicates fluorescence intensity and the y-axis indicates the number of pixels with each intensity; numbers in histograms denote mean fluorescence intensity. Data are representative of > three independent experiments (n ≥ 2 mice per treatment group). Scale bar, 50 µm. PNAd, peripheral lymph node addressin; WBH, whole body hyperthermia.
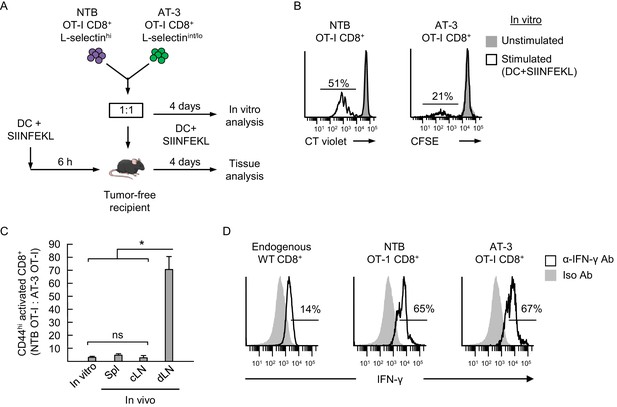
Antigen-driven activation of CD8+ OT-I T cells is compromised by poor L-selectin-dependent trafficking in lymph nodes.
(A) Schematic of competitive activation assays. CD8+ T cell populations (>95% CD8+) were isolated from non-tumor bearing OT-I mice (NTB OT-I CD8+ L-selectinhi) and from AT-3-bearing OT-1 mice (AT-3 OT-I CD8+ L-selectin intermediate-to-low; L-selectinint/lo; tumor volume 3825 ± 123 mm3 for n = 4 mice). T cells (depleted of CD11b+ MDSC) from NTB or tumor-bearing mice were then labeled ex vivo with different proliferation dyes (CellTrace Violet or CellTrace CFSE, respectively), co-mixed at a 1:1 ratio, and assessed for functional responses to cognate antigen (SIINFEKL) after four days in competitive activation assays in vitro and in vivo. (B) Flow cytometric analysis of proliferation of NTB OT-I CD8+ and AT-3 OT-I CD8+ T cells after activation by SIINFEKL-loaded dendritic cells (DC) for four days in vitro. Horizontal lines on histograms indicate percent proliferating cells. (C) Competitive in vivo activation assay in which NTB recipient mice were vaccinated (via footpad) with SIINFEKL-loaded DC 6 hr before adoptive transfer of a 1:1 mixture of L-selectinhi NTB OT-I and L-selectinint/lo AT-3 OT-I CD8+ T cells. After four days, the ratios of the adoptively transferred cells were assessed in the following lymphoid compartments: spleen (Spl), contralateral popliteal lymph node (cLN), and draining popliteal lymph node (dLN). Data (mean±s.e.m.) are from one experiment (n = 4 mice per group) and are representative of 2 independent experiments. *p<0.05; ns, not significant; data were analyzed by unpaired two-tailed Student’s t-test. (D) IFN-γ expression profiles for endogenous CD8+ T cells and adoptively transferred NTB OT-I CD8+ and AT-3 OT-I CD8+ T cells recovered in dLN of DC-vaccinated mice. Horizontal lines on histograms indicate positively stained cells; data are representative of 2 mice per group. NTB, non-tumor bearing; DC, dendritic cell; Iso, Isotype; Ab, antibody.
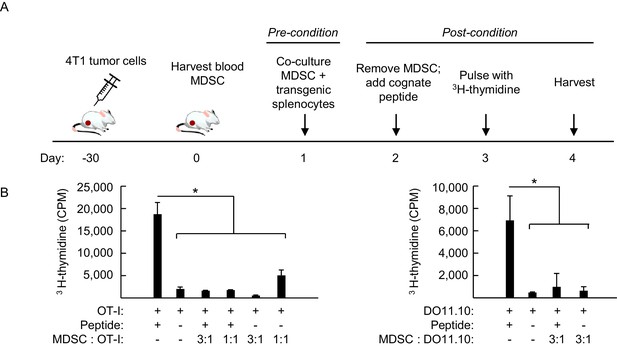
Preconditioning of antigen-inexperienced TcR-transgenic CD8+ and CD4+ T cells with MDSC in vitro suppresses responsiveness to subsequent antigen challenge.
(A) Schematic of MDSC preconditioning protocol. MDSC were isolated from the blood of 4T1-bearing mice (≥93% CD11b+Gr-1+) and co-cultured with splenocytes from non-tumor bearing (NTB) OT-I and DO11.10 T cell receptor (TcR) transgenic mice. After 16 hr, MDSC were depleted by magnetic separation using biotinylated anti-Gr-1 antibody and streptavidin beads. Remaining splenocyte populations contained 0.4–4% CD11b+Gr-1+ cells. Cognate peptide was then added to each culture. After three days in culture, splenocytes were pulsed with 3H-thymidine and harvested 16 hr later. (B) Proliferative responses are shown for OT-I CD8+ (left) and DO11.10 CD4+ (right) T cells based on 3H-thymidine incorporation (CPM). Ratios denote the relative proportion of purified MDSC:OT-I or MDSC:DO11.10 splenocytes used in the preconditioning phase. Data (mean±s.d.) are of six replicate cultures in a single experiment and are representative of three independent experiments. *p<0.002; unpaired two-tailed Student’s t-test. CPM, counts per minute; TcR, T cell receptor; NTB, non-tumor bearing.
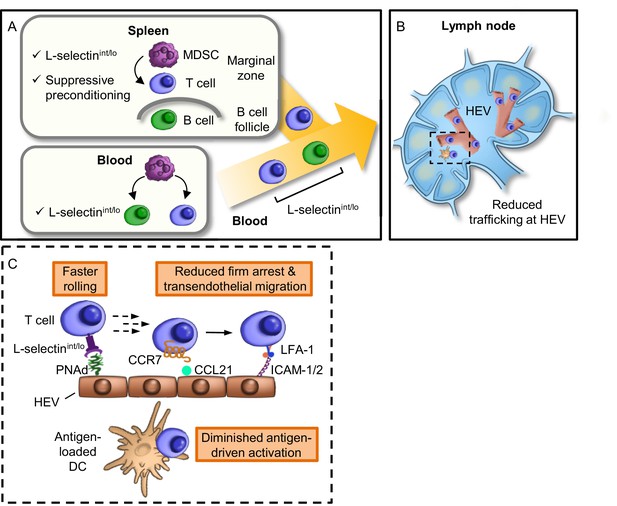
Model for MDSC actions at remote sites that compromise adaptive immunity in the LN compartment.
(A) Restricted localization of MDSC in the splenic marginal zone leads to preferential, downregulation of L-selectin (i.e., intermediate-to-low phenotype, L-selectinint/lo), on naïve CD4+ and CD8+ T cells, but not on B220+ B cells. MDSC in the splenic marginal zone also precondition CD4+ and CD8+ T cells which leads to suppressed responsiveness to antigen outside the splenic environment. L-selectin on circulating T and B cells can be independently targeted by MDSC within the blood compartment, leading to significantly elevated levels of circulating soluble L-selectin. MDSC-mediated downregulation of L-selectin is contact-dependent and occurs post-transcriptionally, but is independent of the major L-selectin sheddase, ADAM17. (B) Diminished L-selectin expression reduces trafficking of blood-borne lymphocytes across high endothelial venules (HEV) in the lymph node compartment. Boxed region is shown in more detail in inset. (C) Inset of lymph node region showing that moderate L-selectin loss (L-selectinint/lo phenotype) results in faster rolling of T cells on lymph node HEV which, in turn, reduces the transition to firm arrest and subsequent transendothelial migration into the underlying parenchyma. Diminished trafficking in HEV, in combination with sustained immunosuppression caused by MDSC preconditioning in the spleen, profoundly compromises the generation of effector T cells in response to cognate antigen presented by dendritic cells (DC). L-selectinint/lo, L-selectin intermediate-to-low expression; PNAd, peripheral lymph node addressin; LFA-1, leukocyte function-adhesion molecule-1; ICAM-1/2, intercellular adhesion molecule-1 and -2.
Videos
Real-time intravital imaging of L-selectinhi CD8+ T cells trafficking in lymph node HEV.
Intravital imaging of fluorescently-labeled L-selectinhi CD8+ T cells from a non-tumor bearing mouse undergoing transient rolling interactions and firm arrest within postcapillary venules of an inguinal lymph node in a non-tumor bearing mouse. The initial injection of transferred CD8+ cells occurred ~12 min prior to capture of images.
Intravital imaging of L-selectinlo CD8+ T cells trafficking in lymph node HEV.
Impaired sticking of calcein-labeled L-selectinlo CD8+ T cells from an AT-3-bearing mouse within postcapillary venules of an inguinal lymph node in a non-tumor bearing mouse. The initial injection of transferred CD8+ cells occurred ~13 min prior to capture of images.
Additional files
-
Supplementary file 1
Supporting information for antibodies used in current study.
App, application; FC, flow cytometry; IF, immunofluorescence histology; Activ, T cell activation; Mag, magnetic isolation or depletion; Dep; in vivo antibody-mediated depletion.
- https://doi.org/10.7554/eLife.17375.026





