Temporal proteomic analysis of HIV infection reveals remodelling of the host phosphoproteome by lentiviral Vif variants
Figures
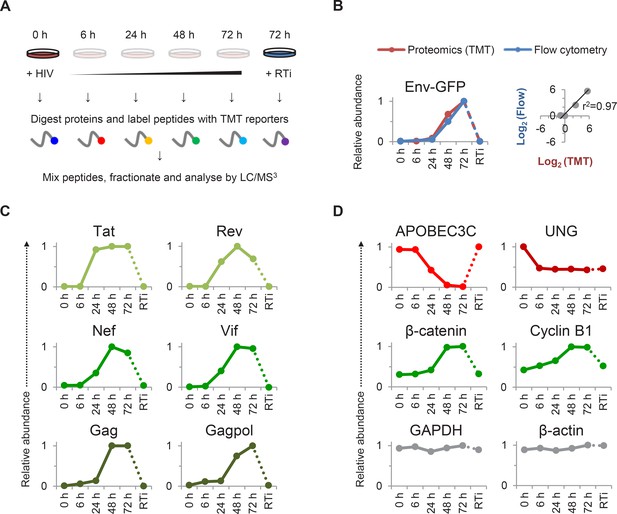
TMT-based proteomic time course analysis of HIV-infected cells.
(A) Workflow of 6-plex TMT-based whole cell proteomic time course experiment. CEM-T4 T cells were infected with NL4-3-dE-EGFP HIV at an MOI of 10. In subsequent figures timepoints 1–5 show protein abundance 0, 6, 24, 48 and 72 hr after HIV infection (where 0 hr = uninfected cells) and timepoint 6 shows protein abundance 72 hr after HIV infection in the presence of reverse transcriptase inhibitors (RTi). (B) Comparison of temporal profiles of Env-GFP obtained by proteomic (TMT) versus flow cytometric quantitation. Cells from (A) were analysed by flow cytometry. Relative abundance is expressed as a fraction of maximum TMT reporter ion or fluorescence intensity. For linear regression, log2 (fold change in protein abundance compared with uninfected cells) is shown. (C–D) Temporal profiles of viral proteins (C) and previously reported HIV targets (D). GAPDH and β-actin are included as controls. Relative abundance is expressed as a fraction of maximum TMT reporter ion intensity.
-
Figure 1—source data 1
Interactive spreadsheet of TMT time course data.
Interactive spreadsheet enabling generation of temporal profiles of protein abundance for any quantitated genes of interest ('Gene search and plots' worksheet). Detailed instructions are incorporated into the spreadsheet. The complete (unfiltered) TMT-based proteomic time course dataset ('Complete TMT time course data' worksheet) and a database of gene name aliases ('Gene name aliases' worksheet) are also included. Protein abundance is depicted on a colour scale (red = downregulated; green = upregulated). The number of unique peptides, peptides and peptide spectral matches are specified for each protein, along with ratio counts and variability for each TMT condition.
- https://doi.org/10.7554/eLife.18296.004
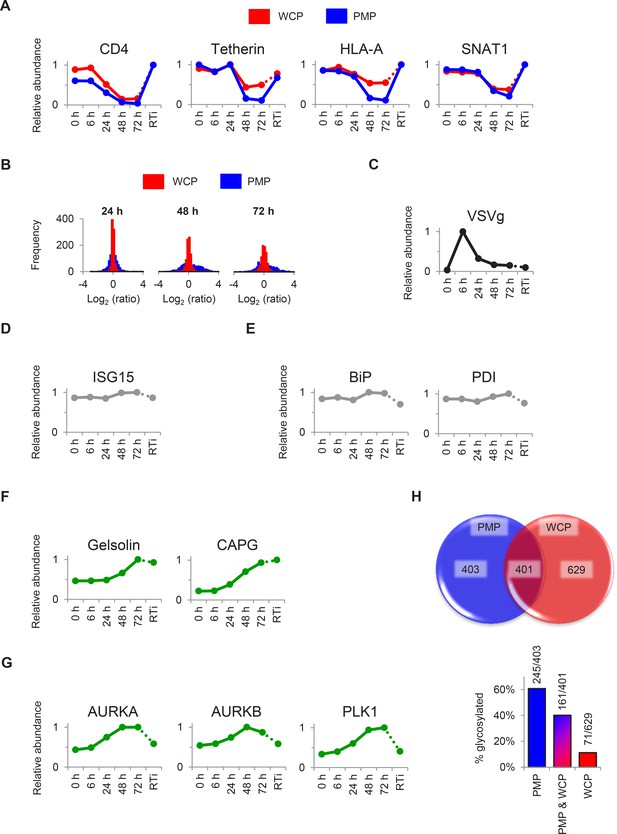
Additional temporal profiles and comparison with plasma membrane profiling.
(A) Comparison of temporal profiles of cell surface Nef- (CD4 and HLA-A) and Vpu- (CD4, tetherin and SNAT1) targets obtained by whole cell or plasma membrane proteomics. Expression levels from our whole cell proteomic analysis (WCP, red) and our previous plasma membrane profiling analysis ([Matheson et al., 2015]; PMP, blue) are shown. Relative abundance is expressed as a fraction of maximum TMT reporter ion intensity. (B) Dynamic range of protein regulation observed by whole cell or plasma membrane proteomics. Histograms show the frequencies of log2 (fold change compared with mock/uninfected cells) values for proteins quantitated in our whole cell proteomic analysis (WCP, red) and our previous PMP ([Matheson et al., 2015]; blue) at 24, 48 and 72 hr. All proteins identified by >1 unique peptide in both WCP and PMP experiments are shown. (C–G) Temporal profiles of selected control proteins (C–E), actin regulatory proteins (F) and mitotic kinases (G). Relative abundance is expressed as a fraction of maximum TMT reporter ion intensity. (H) Quantitation of plasma membrane proteins by whole cell or plasma membrane proteomics. The pie chart shows overlap of proteins with Gene Ontology Cellular Compartment annotations indicative of plasma membrane localisation quantitated in our whole cell proteomic analysis (WCP, red) and our previous PMP analysis ([Matheson et al., 2015]; blue). Protein numbers are indicated. The bar chart details the glycosylation status of proteins quantitated in WCP (red), PMP (blue) or both (tan) experiments. Glycosylation sites were identified from the UniProt Knowledgebase (accessed on 4/12/15 at http://www.uniprot.org). Protein numbers (glycosylated/total) are indicated.
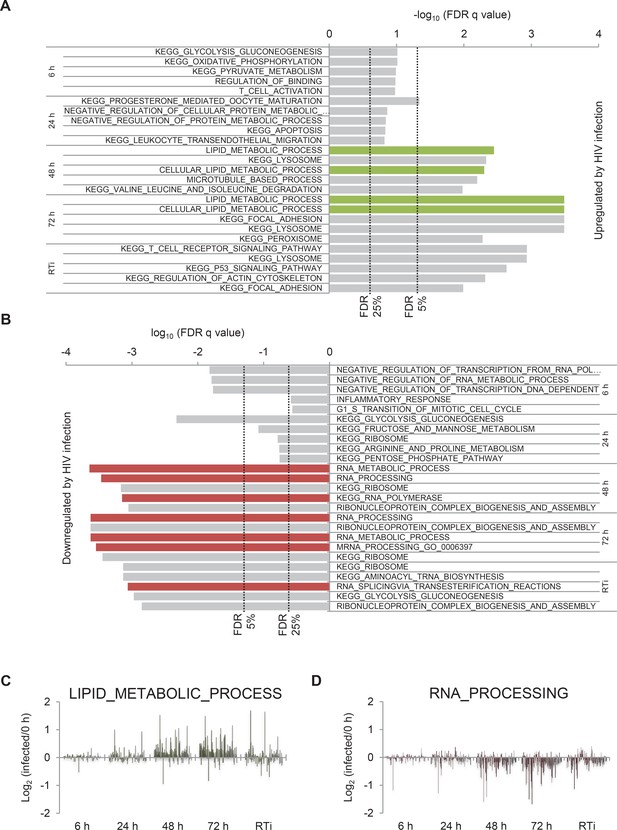
Gene set enrichment analysis of HIV infection.
(A) Pathways and processes upregulated by HIV infection. KEGG Pathway and Gene Ontology Biological Process gene sets enriched in infected cells at the indicated timepoints compared with uninfected cells were determined using GSEA. Indicative false discovery rate (FDR) thresholds for gene set enrichment are shown and gene sets related to lipid metabolism are highlighted (green). (B) Pathways and processes downregulated by HIV infection. KEGG Pathway and Gene Ontology Biological Process gene sets enriched in uninfected cells compared with infected cells at the indicated timepoints were determined using GSEA. Indicative FDR thresholds for gene set enrichment are shown and gene sets related to RNA processing are highlighted (green). (C–D) Expression levels of all quantitated proteins in Lipid_Metabolic_Process (C) and RNA_Processing (D) gene sets. Log2 (fold change in protein abundance compared with uninfected cells) is shown at the indicated timepoints.
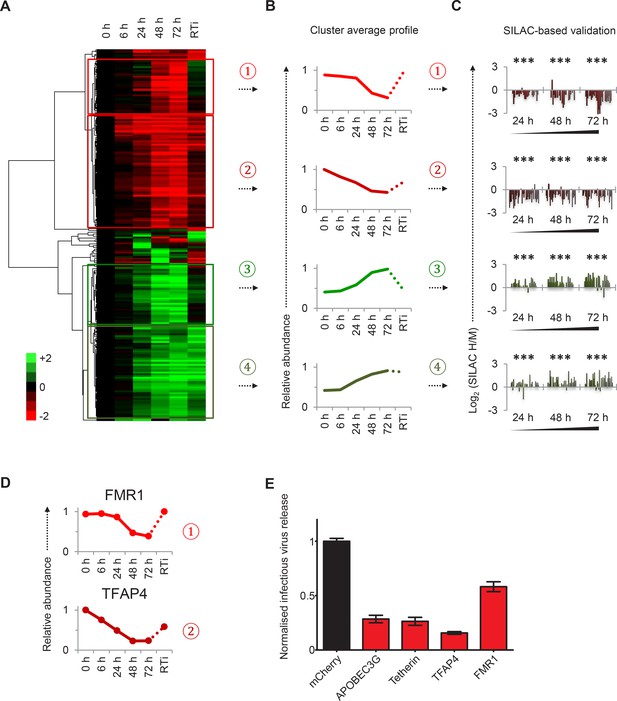
Identification and SILAC-based proteomic validation of novel HIV targets.
(A) Hierarchical cluster analysis of temporal profiles of proteins regulated by HIV. The heatmap shows log2 (fold change in protein abundance compared with uninfected cells) and clusters #1–4 are indicated. (B) Average temporal profiles of proteins in clusters #1–4. Relative abundance is expressed as a fraction of maximum TMT reporter ion intensity. (C) SILAC-based validation of novel HIV targets. CEM-T4 T cells pre-labelled with heavy amino acids were infected with NL4-3-dE-EGFP HIV at an MOI of 10, and control cells pre-labelled with medium amino acids were mock-infected without virus. Aliquots of HIV-infected (heavy; H) and mock (medium; M) cells were harvested sequentially at the indicated timepoints and subjected to SILAC-based whole cell proteomic analysis (Figure 2—figure supplement 1A). Log2 (H/M protein abundance) at 24, 48 and 72 hr is shown for proteins from clusters #1–4. ***p value<0.001. (D) Temporal profiles of novel HIV-1 targets FMR1 and TFAP4. Relative abundance is expressed as a fraction of maximum TMT reporter ion intensity. (E) Antagonism of HIV production by FMR1 and TFAP4. 293T cells were co-transfected with pNL4-3-dE-EGFP/pMD.G and either mCherry or the indicated cellular protein. 48 hr culture supernatants were assayed for infectious virus by infection of HeLa cells. Well-characterised restriction factors APOBEC3G and tetherin were included as controls, and infectious virus release normalised compared with mCherry. Mean values and standard errors are shown from at least 4 replicates. All four proteins significantly reduced viral release compared with mCherry in an ANOVA analysis with Bonferroni post-test, p values<0.001.
-
Figure 2—source data 1
Clusters #1–4 summary proteomic data.
Spreadsheet of all proteomic data for proteins in clusters #1–4. Each cluster is represented by a single worksheet, with proteins ranked according to the hierarchical cluster analysis shown (Figure 2A) and protein abundance depicted on a colour scale (red = downregulated; green = upregulated). As well as data from the TMT-based proteomic time course experiment (Figure 1 and Figure 1—source data 1), additional data from the SILAC-based proteomic validation (Figure 2C and Figure 2—figure supplement 1A) and TMT-based single timepoint experiment (Figure 4A and Figure 4—figure supplement 1A) are also included. The number of unique peptides is shown for each protein (TMT experiments) and each timepoint (SILAC experiment). q values for the TMT-based single timepoint experiment were determined using Limma with Benjamini-Hochberg adjustment for multiple testing, with q values < 0.01 highlighted in gold.
- https://doi.org/10.7554/eLife.18296.008
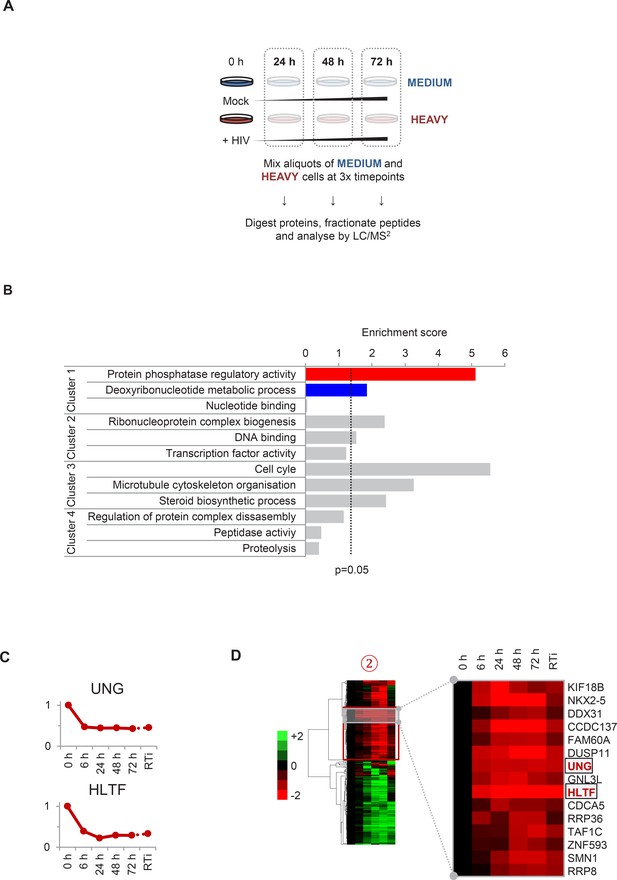
Workflow of SILAC-based proteomic time course experiment, functional analysis of clusters #1–4 and prediction of novel Vpr targets.
(A) Workflow of SILAC-based proteomic validation time course experiment. CEM-T4 T cells pre-labelled with heavy amino acids were infected with NL4-3-dE-EGFP HIV at an MOI of 10, and control cells pre-labelled with medium amino acids were mock-infected without virus. Aliquots of HIV-infected (heavy) and mock (medium) cells were harvested sequentially at the indicated timepoints and subjected to SILAC-based whole cell proteomic analysis. (B) Functional annotation clusters enriched amongst proteins from clusters #1–4. Enrichment of Gene Ontology Molecular Function and Biological Process terms against a background of all quantitated proteins was determined using DAVID. Functional annotation clusters with enrichment scores > 1.3 (equivalent to a geometric mean of all included enrichment p values<0.05) were considered significant. Representative Gene Ontology terms are indicated. (C) Temporal profiles of Vpr targets UNG and HLTF. Relative abundance is expressed as a fraction of maximum TMT reporter ion intensity. (D) Enlargement of part of cluster #2 from hierarchical cluster analysis (Figure 2A). The heatmap shows log2 (fold change in protein abundance compared with uninfected cells). Previously characterised Vpr targets UNG and HLTF are highlighted (red).
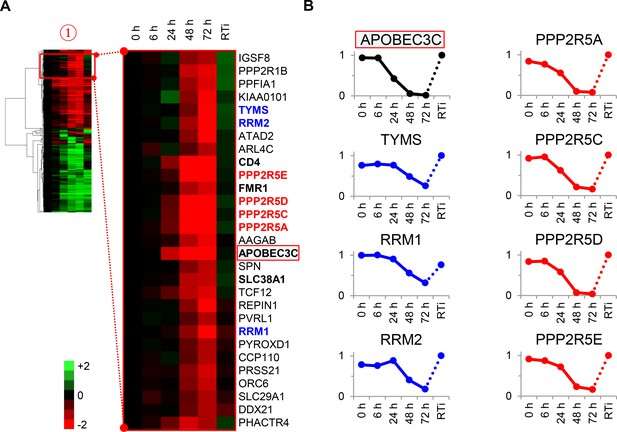
Cellular proteins progressively downregulated by HIV infection.
(A) Enlargement of cluster #1 from hierarchical cluster analysis (Figure 2A). The heatmap shows log2 (fold change in protein abundance compared with uninfected cells). Enzymes associated with deoxynucleotide metabolism (blue) and B56 family regulatory subunits of serine/threonine protein phosphatase PP2A (red) are highlighted, along with known Vif target APOBEC3C (boxed) and other proteins of interest (bold). (B) Temporal profiles of enzymes associated with deoxynucleotide metabolism (blue) and B56 family regulatory subunits of serine/threonine protein phosphatase PP2A (red). Relative abundance is expressed as a fraction of maximum TMT reporter ion intensity, and the temporal profile of APOBEC3C is shown for comparison.
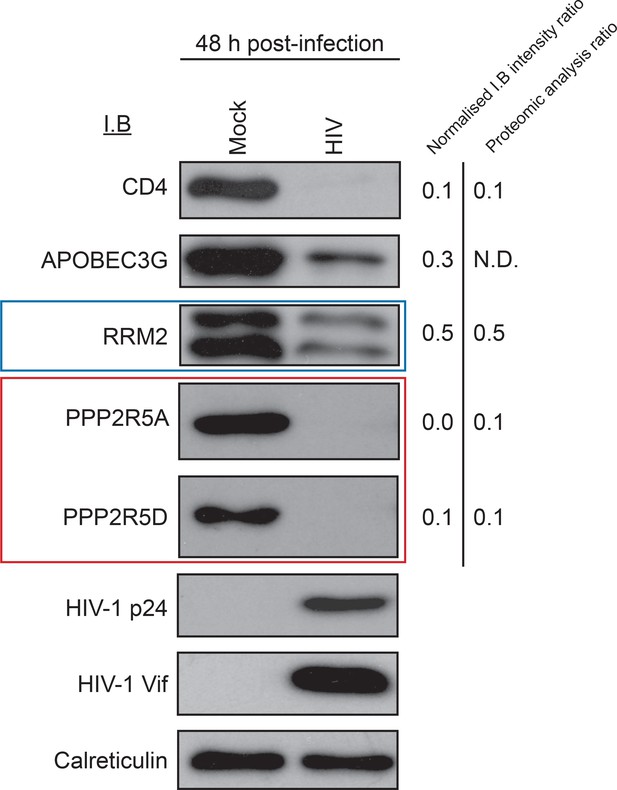
Immunoblot validation of novel HIV targets.
Depletion of proteins in cluster #1 by HIV-1 infection. CEM-T4s were infected with NL4-3-dE-EGFP HIV at an MOI of 10 and analysed by immunoblot (IB) 48 hr post-infection. Depletion of positive control (CD4 and APOBEC3G) and novel (RRM2 and PPP2R5A/D) HIV targets was confirmed. ImageJ (Schneider et al., 2012) was used to determine band intensities, which were normalised to calreticulin intensity and compared with TMT-based proteomic quantitation at 48 hr. p24 (capsid) and Vif are included as additional controls.
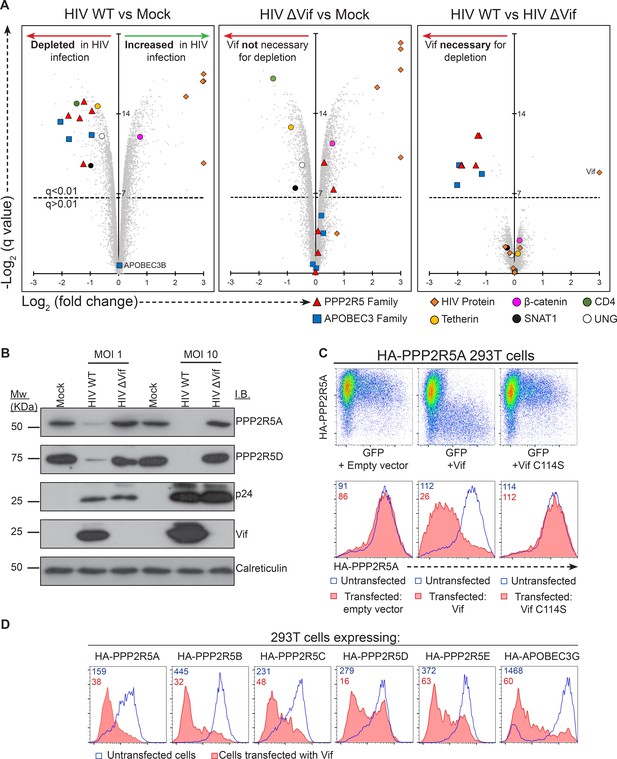
Vif-mediated depletion of PP2A-B56 family members PPP2R5A-E.
(A) Proteomic analysis of CEM-T4 T cells infected with WT and ΔVif HIV. Cells were infected with NL4-3-ΔE-EGFP viruses at an MOI of 1.5, and harvested 48 hr post-infection. Scatterplots display pairwise comparisons between WT, ΔVif and mock-infected cells. Each point represents a single protein, plotted by its log2 (fold change in abundance) versus the statistical significance of that change. q values were determined using Limma with Benjamini-Hochberg adjustment for multiple testing, with increasing −log2 (q value) indicating increasing significance. Points above the dotted line change with a q value < 0.01. HIV proteins and host proteins of interest are highlighted with different symbols (see key). (B) Depletion of PPP2R5A and PPP2R5D during HIV infection. CEM-T4 T-cells were infected with NL4-3-dE-EGFP WT and ΔVif viruses at an MOI of 1 or 10 and analysed by immunoblot (IB) 48 hr post-infection. p24 (capsid), Vif and calreticulin are included as controls. (C) Depletion of exogenous PPP2R5A by Vif. 293T cells stably expressing HA-PPP2R5A were co-transfected with GFP plus empty vector, NL4-3 Vif or NL4-3 Vif with a single amino acid mutation C114S and analysed by intracellular flow cytometry for HA 36 hr post-transfection. Histograms show GFP positive (transfected, red shading) and negative (untransfected, blue line) cells. Median fluorescence intensity (MFI) values are shown for GFP positive (red) and negative (blue) cells. (D) Depletion of PPP2R5A-E family members by Vif. 293T cells stably expressing HA-tagged PPP2R5A-E or APOBEC3G were co-transfected with GFP plus NL4-3 Vif expression vectors, and intracellular HA staining quantitated by flow cytometry 36 hr post transfection. Histograms show GFP positive (transfected, red shading) and negative (untransfected, blue line) cells. MFI values are shown for GFP positive (red) and negative (blue) cells.
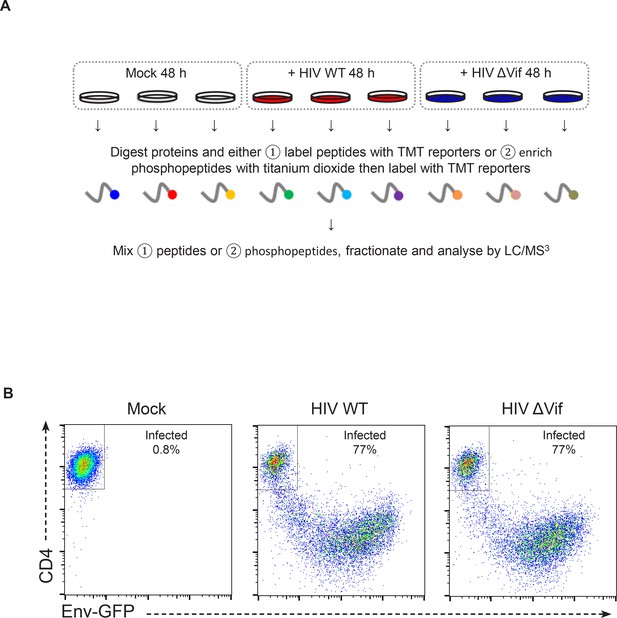
Workflow and controls for single timepoint proteomic/phosphoproteomic experiment.
(A) Workflow of TMT-based single timepoint whole cell proteomic and phosphoproteomic experiment. CEM-T4 T cells were mock-infected or infected with NL4-3-dE-EGFP WT or ΔVif HIV at an MOI of 1.5. Cells were harvested for proteomic analysis 48 hr post-infection. Samples were subjected to (1) whole cell proteome and (2) phosphopeptide analysis. Each condition was carried out in triplicate. (B) Quantitation of infected cells from experiment described in (A). Cells were analysed by flow cytometry for CD4 and GFP expression, with infected cells losing CD4 and gaining GFP. Example flow cytometric analysis of one replicate for each condition is shown, demonstrating the % infected cells. Across all replicates, cells infected with WT or ΔVif viruses were 74–78 % infected.
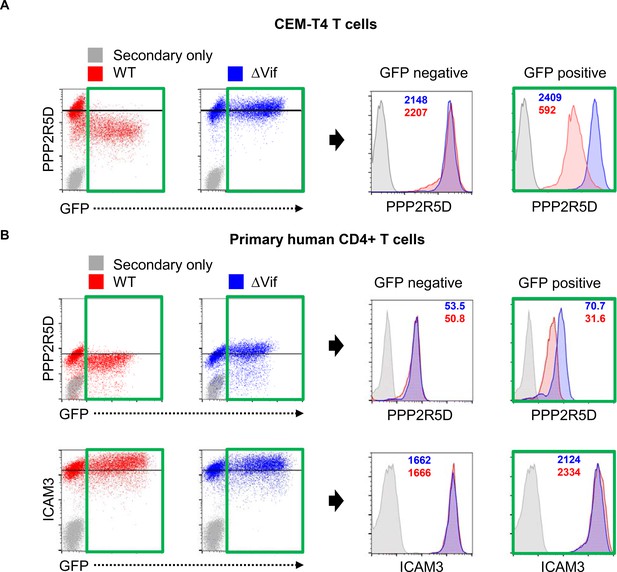
Depletion of endogenous PPP2R5D during HIV infection of primary cells.
(A–B) CEM-T4 T cells (A) or activated primary human CD4+ T cells (B) were infected with NL4-3-dE-EGFP WT and ΔVif viruses at an MOI of 1 and analysed by intracellular flow cytometry for PPP2R5D 48 hr post-infection. ICAM3 is included as a control. MFI values are shown for ΔVif (blue) and WT (red) viruses.
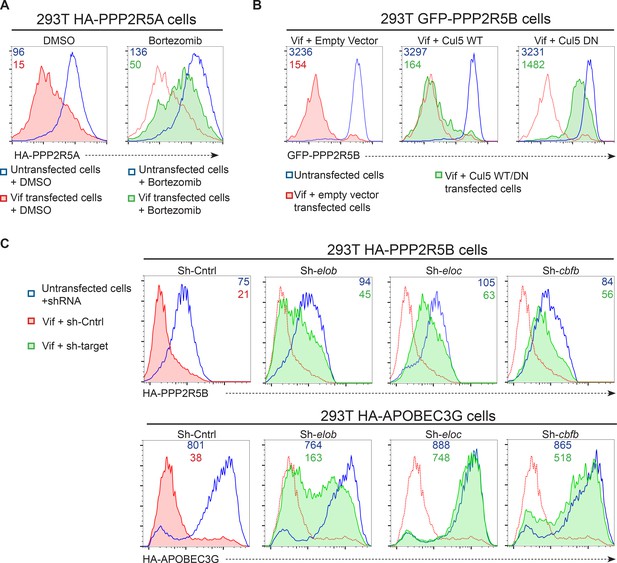
Mechanism of Vif-mediated degradation of PPP2R5A-E subunits.
(A) Proteasomal degradation. 293T cells stably expressing HA-PPP2R5A were transfected with NL4-3 Vif in the presence of DMSO (control) or the proteasome inhibitor bortezomib and analysed by intracellular flow cytometry for HA. (B) CUL5-dependent degradation. 293T cells stably expressing GFP-PPP2R5B were co-transfected with NL4-3 Vif plus empty vector, wildtype cullin-5 (CUL5 WT) or a dominant negative cullin-5 mutant (CUL5 DN) and analysed by flow cytometry for GFP. (C) CUL5 complex-dependent degradation. 293T cells stably expressing HA-PPP2R5B (upper panels) or HA-APOBEC3G (lower panels) were transduced with the indicated shRNA. Cells were then transfected with NL4-3 Vif and analysed by intracellular flow cytometry for HA. Green/red shading shows Vif-transfected cells in the indicated shRNA background. Red lines showing HA staining in cells transduced with control shRNA are included in each panel for reference. In all experiments, cells were analysed 36 hr post-transfection, and transfected cells determined by co-transfection with GFP (A and C) or mCherry (B). MFI values are shown for transfected (red/green) and untransfected (blue) cells.
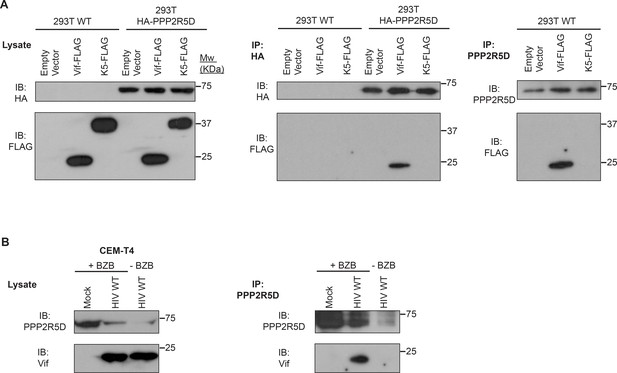
Co-immunoprecipitation of Vif and PPP2R5D.
(A) Co-immunoprecipitation in 293Ts. WT 293T cells or 293T cells stably expressing HA-tagged PPP2R5D were transfected with FLAG-tagged NL4-3 Vif, pre-treated with bortezomib (10 nM) for 16 hr, and analysed by immunoblot (IB) for HA-PPP2R5D and FLAG-Vif 48 hr post-infection (left panels). Lysates were subjected to immunoprecipitation (IP) with anti-HA (middle panels) or anti-PPP2R5D (right panels, WT 293Ts only) and re-analysed by immunoblot. 293T cells transfected with empty vector or FLAG-tagged K5 protein of Kaposi's sarcoma-associated herpesvirus (KSHV) were included as controls. (B) Co-immunoprecipitation during HIV infection of T cells. CEM-T4 T cells were infected with NL4-3-dE-EGFP HIV at an MOI of 1.5, pre-treated with bortezomib (10 nM) for 16 hr, and analysed by immunoblot (IB) for PPP2R5D and Vif 48 hr post-infection (left panels). Lysates were subjected to immunoprecipitation (IP) with anti-PPP2R5D (right panels) and re-analysed by immunoblot. Uninfected CEM-T4 T cells and infected CEM-T4 T cells without bortezomib pre-treatment were included as controls.
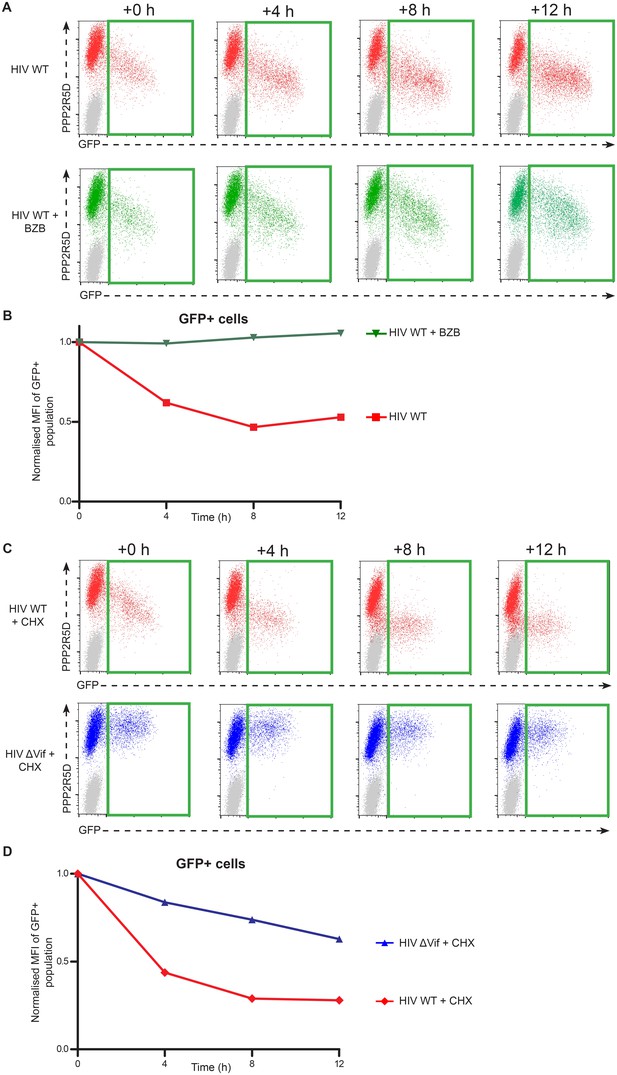
Time course analysis of endogenous PPP2R5D during HIV infection of T cells.
(A–B) Rescue by proteasome inhibition. CEM-T4 T cells were infected with NL4-3-dE-EGFP HIV at an MOI of 1.5 and analysed by flow cytometry (A) at the indicated timepoints in the presence (bottom panels) or absence (top panels) of bortezomib (BZB) added 24 hr post-infection (+0 hr). MFI values (PPP2R5D staining) for GFP positive (infected) cells are shown, normalized to values at +0 hr (B). (C–D) Cycloheximide chase. CEM-T4 T cells were infected with NL4-3-dE-EGFP WT (top panels) and ΔVif (bottom panels) viruses at an MOI of 1.5 and analysed by flow cytometry (C) at the indicated timepoints in the presence of cycloheximide (CHX) added 24 hr post-infection (+0 hr). MFI values (PPP2R5D staining) for GFP positive (infected) cells are shown, normalized to values at +0 hr (D). As predicted, the presence of CHX inhibited the production of new Env-EGFP protein.
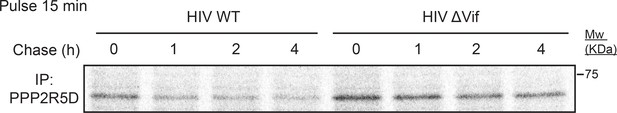
Pulse-chase analysis of endogenous PPP2R5D during HIV infection of T cells.
CEM-T4 T cells were infected with NL4-3-dE-EGFP WT or ΔVif viruses at an MOI of 1. 5 and pulsed with [35S]methionine/[35S]cysteine 48 hr post-infection. Cells were chased until the indicated timepoints, subjected to immunoprecipitation (IP) with anti-PPP2R5D and analysed by autoradiography.
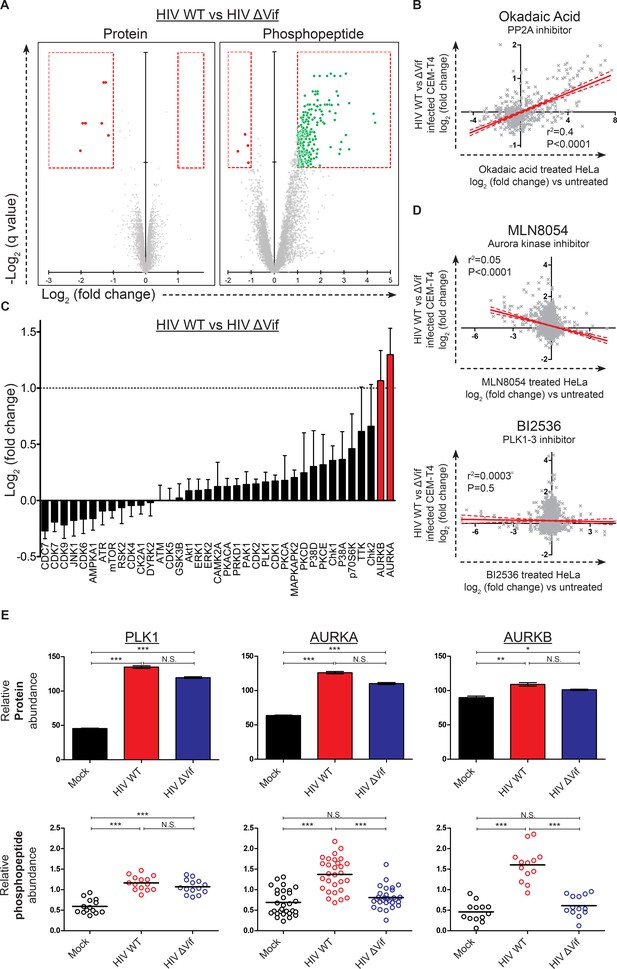
Global phosphoproteomic analysis of cells infected with WT or ∆Vif HIV.
(A) Vif-dependent changes in peptide and phosphopeptide abundance. CEM-T4 T cells from Figure 4A and Figure 4—figure supplement 1A were subjected to TMT-based phosphoproteomic analysis. Scatterplots display differences in protein (left panel, as in Figure 4A, right panel) and phosphopeptide abundance (right panel) between WT and ΔVif-infected cells. Each point represents a single protein or phosphopeptide, plotted by its log2 (fold change in abundance) versus the statistical significance of that change. q values were determined using Limma with Benjamini-Hochberg adjustment for multiple testing, with increasing −log2 (q value) indicating increasing significance. Proteins and phosphopeptides downregulated (red) or upregulated (green) with a fold change > 2 and q value < 0.01 are highlighted. (B) Comparison of changes in phosphopeptide abundance between WT and ΔVif-infected CEM-T4 T cells with previously published data for okadaic acid-treated HeLa cells (Kauko et al., 2015). Lines show linear correlation with associated 95% confidence areas, r2 values and p values of a non-zero correlation. (C) Analysis of changes in phosphopeptide abundance between WT and ΔVif-infected cells CEM-T4 T cells using the PhosphoSitePlus kinase-substrate database. Bars show log2 (fold change in phosphopeptide abundance) for peptides spanning known kinase substrate sites. Error bars show the standard error of the mean. (D) Comparison of changes in phosphopeptide abundance between WT and ΔVif-infected CEM-T4 T cells with previously published data for kinase inhibitor-treated HeLa cells (Kettenbach et al., 2011). At low concentrations, MLN8054 is a selective AURKA inhibitor, but at 5 μM (as shown) reduced activity of AURKB and PLK1 is also observed. Lines show linear correlation with associated 95% confidence areas, r2 values and p values of a non-zero correlation. (E) Vif-specific hyperphosphorylation of aurora kinase substrates. Protein abundances of PLK1, AURKA and AURKB were compared with normalised abundances of manually curated phosphopeptides targeted by the respective kinases. Abundances of kinase proteins were compared using Limma with Benjamini-Hochberg adjustment for multiple testing. Abundances of target phosphopeptides were compared by Repeated Measures ANOVA with Bonferroni post-test. N.S., p value>0.05; *p value<0.05; **p value<0.01; ***p value<0.001.
-
Figure 6—source data 1
Single timepoint phosphoproteomic data.
Spreadsheet of cellular phosphopeptides identified in mock, WT and ΔVif HIV-infected cells in the TMT-based single timepoint phosphoproteomic experiment (Figure 6, Figure 4—figure supplement 1A and Figure 6—figure supplement 1). Peptide sequence, details of the cognate protein and the position of the peptide within the protein are shown. The column 'Phosphosite Probabilities' indicates the probability that each serine, threonine or tyrosine within the peptide is phosphorylated. The amino acid is stated (S, serine; T, threonine; Y, tyrosine) with the position in the peptide in parentheses, followed by the probability (%). Each potential phosphosite is separated by a semicolon. Phosphosites with a probability of over 75% are listed in the column 'Modifications in Master Proteins', which shows a summary of the phosphorylated amino acids identified and their position in the protein. Log2 (fold change) compares phosphopeptide abundance normalized to total protein abundance, with abundance depicted on a colour scale (red = downregulated; green = upregulated). q values were determined using Limma with Benjamini-Hochberg adjustment for multiple testing, with q values < 0.01 highlighted in gold.
- https://doi.org/10.7554/eLife.18296.020
-
Figure 6—source data 2
Previously reported AURKA, AURKB and PLK1 targets.
AURKA, AURKB and PLK1 targets were manually curated from the literature. Peptides listed overlap reported sites of phosphorylation by AURKA, AURAKB or PLK1, or are the only phosphopeptide identified from a protein known to be phosphorylated by one of these kinases, but at an unknown site. Peptides where the identified phosphorylation site explicitly matches the one reported are plotted in Figure 6E (blue shading; excluded peptides highlighted in red text). Studies cited: (Asano et al., 2013; Dephoure et al., 2008; Hengeveld et al., 2012; Kettenbach et al., 2011; Santamaria et al., 2011; Welburn et al., 2010; Yu et al., 2005).
- https://doi.org/10.7554/eLife.18296.021
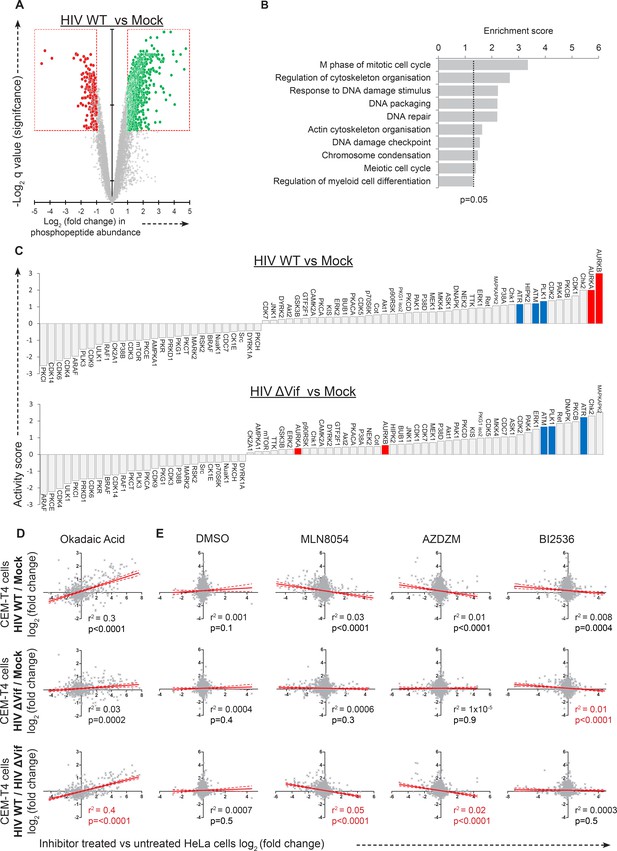
Further phosphoproteomic analysis.
(A) Remodelling of the cellular phosphoproteome by HIV infection. CEM-T4 T cells from Figure 4A and Figure 4—figure supplement 1A were subjected to TMT-based phosphoproteomic analysis. The scatterplot displays differences in phosphopeptide abundance between WT HIV and mock-infected cells. Each point represents a single phosphopeptide, plotted by its log2 (fold change in abundance) versus the statistical significance of that change. q values were determined using Limma with Benjamini-Hochberg adjustment for multiple testing, with increasing -log2 (q value) indicating increasing significance. Phosphopeptides downregulated (red) or upregulated (green) with a fold change > 2 and q value < 0.01 are highlighted. (B) Functional annotation clusters enriched amongst proteins hyperphosphorylated in the presence of HIV infection. Proteins containing phosphopeptides significantly upregulated (q values < 0.01) in cells infected with WT HIV compared with mock-infected cells were analysed. Enrichment of Gene Ontology Molecular Function and Biological Process terms against a background of all identified phosphoproteins was determined using DAVID. Functional annotation clusters with enrichment scores > 1.3 (equivalent to a geometric mean of all included enrichment p values<0.05) were considered significant. Representative Gene Ontology terms are indicated. (C) PhosFate analysis of kinase activity in HIV-infected versus mock-infected cells. Data are shown for WT HIV (upper panel) and ΔVif HIV (lower panel). A positive activity score indicates enhanced phosphorylation of kinase-specific phosphosites in infected cells. Aurora kinases A and B (AurA/AurB; red) and other control mitotic/checkpoint kinases (PLK1, ATR and ATM; blue) are highlighted. Kinases represented in the dataset by a single target phosphosite were excluded. (D–E) Comparison of phosphoproteomic dataset with previously published data for (D) okadaic acid-treated (Kauko et al., 2015) and (E) kinase inhibitor-treated (Kettenbach et al., 2011) HeLa cells. Each row shows a different pairwise comparison: top row, HIV WT versus mock; middle row, HIV ΔVif vs mock; bottom row, HIV WT vs HIV ΔVif. Each column shows a different inhibitor treatment. At low concentrations, MLN8054 is a selective AURKA inhibitor, but at 5 μM (as shown) reduced activity of AURKB and PLK1 is also observed. AZDZM indicates a combined analysis of selective AURKB inhibitors AZD1152 and ZM447439. BI2536 is a selective PLK1-3 inhibitor. Each scatterplot compares log2 (fold change) in WT/ ΔVif/mock-infected cells (y axis) with log2 (fold change) in inhibitor treated cells (x axis). Lines indicate linear correlation with 95% confidence areas, r2 values and p values of a non-zero correlation. For each column, the most significant correlation is highlighted (red).
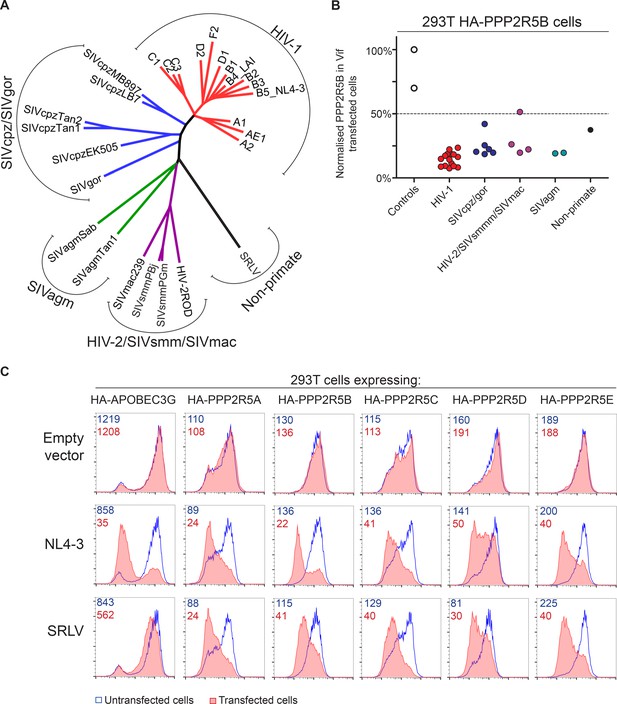
Phylogenetic conservation of PPP2R5A-E subunit degradation.
(A) Phylogenetic tree based on the amino acid alignment of Vif variants used in this study. (B) Conservation of PPP2R5B subunit degradation by phylogenetically diverse lentiviral Vif variants. 293T cells stably expressing HA-PPP2R5B were transfected with a panel of lentiviral Vif variants and analysed by intracellular flow cytometry for HA 36 hr post-transfection. The median fluorescence intensity of the transfected population is shown as a proportion of median fluorescence intensity of the untransfected population for each condition, normalized to the empty vector control. Datapoints represent mean values for different Vif variants obtained from up to four independent experiments. (C) Depletion of PPP2R5A-E subunits by small ruminant lentivirus Vif. 293T cells stably expressing HA-PPP2R5A-E or HA-APOBEC3G were transfected with NL4-3 (HIV-1) or SRLV Vif variants. Histograms show GFP positive (transfected, red shading) and negative (untransfected, blue line) cells. MFI values are shown for GFP positive (red) and negative (blue) cells.
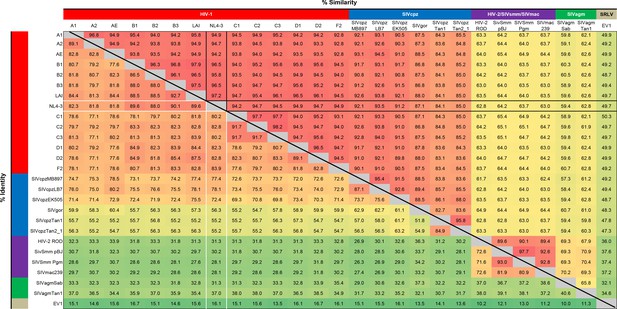
Identity/similarity matrix of lentiviral Vif variants.
Upper-right half of matrix shows pairwise similarity between Vif variants, lower-right half shows pairwise identity. For pairwise comparisons with NL4-3 Vif, relevant similarity values (black lines) and identity values (white lines) are highlighted. Matrix is based on a Vif amino acid alignment carried out with the PSI-Coffee variant of the T-Coffee alignment algorithm (Notredame et al., 2000) and the SIAS tool available at http://imed.med.ucm.es/Tools/sias.html using default settings.
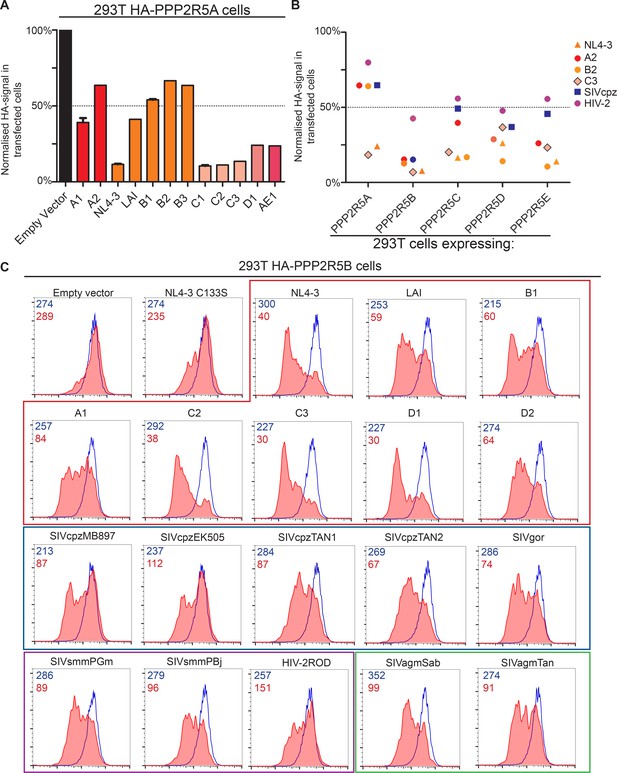
Additional data on phylogenetic conservation of PPP2R5A-E subunit degradation.
(A) Conservation of PPP2R5A subunit degradation by HIV-1 Vif variants. 293T cells stably expressing HA-PPP2R5A were transfected with the indicated Vif variants and analysed by intracellular flow cytometry for HA 36 hr post-transfection. The median fluorescence intensity of the transfected population is shown as a proportion of median fluorescence intensity of the untransfected population for each condition, normalized to the empty vector control. Mean values and standard errors are shown. (B) Conservation of PPP2R5A-E subunit degradation by phylogenetically diverse lentiviral Vif variants. 293T cells stably expressing different HA-tagged PPP2R5A-E subunits were transfected with the indicated Vif variants and analysed by intracellular flow cytometry for HA 36 hr post-transfection. Median fluorescence intensity of the transfected population is shown as a proportion of median fluorescence intensity of the untransfected population for each condition, normalized to the empty vector control. Each datapoint represents a different Vif variant. (C) Representative data for primate lentiviral Vif degradation of PPP2R5B. Data shown in Figure 7B was acquired in several experiments, one example is shown here. Histograms show GFP positive (transfected, red shading) and negative (untransfected, blue line) cells. MFI values are shown for GFP positive (red) and negative (blue) cells.
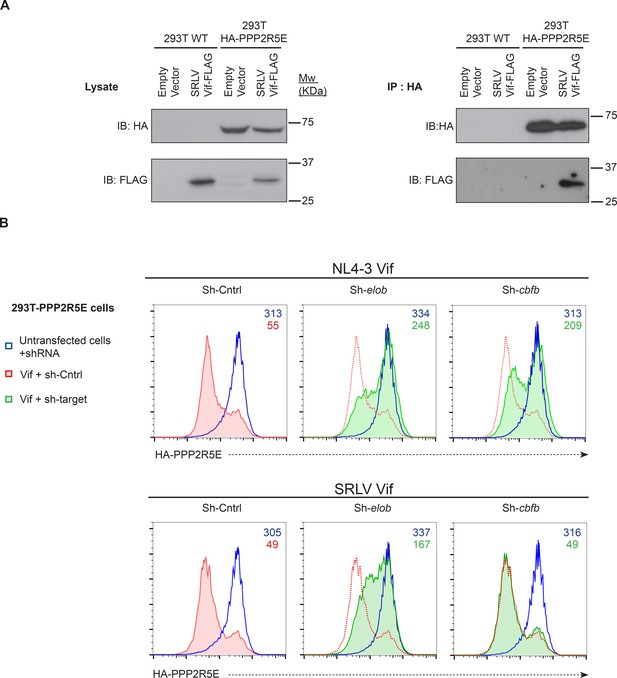
Mechanism of PPP2R5E degradation by SRLV Vif.
(A) Co-immunoprecipitation of SRLV Vif with PPP2R5E. WT 293T cells or 293T cells stably expressing HA-tagged PPP2R5E were transfected with FLAG-tagged SRLVE Vif, pre-treated with bortezomib (10 nM) for 16 hr, and analysed by immunoblot (IB) for HA-PPP2R5E and FLAG-Vif 48 hr post-infection (left panels). Lysates were subjected to immunoprecipitation (IP) with anti-HA and re-analysed by immunoblot (right panels). 293T cells transfected with empty vector were included as controls. (B) CBFβ-independent degradation of PPP2R5E by SRLV Vif. 293T cells stably expressing HA-PPP2R5E were transduced with the indicated shRNA. Cells were then transfected with NL4-3 Vif (upper panels) or SRLV Vif (lower panels) and analysed by intracellular flow cytometry for HA. Red/green shading shows Vif-transfected cells in the indicated shRNA background. Red lines showing HA staining in cells transduced with control shRNA are included in each panel for reference. Cells were analysed 36 hr post-transfection, and transfected cells determined by co-transfection with GFP. MFI values are shown for transfected (red/green) and untransfected (blue) cells.
Additional files
-
Supplementary file 1
Previous proteomic studies of HIV-infected cells.
- https://doi.org/10.7554/eLife.18296.027





