A novel optical microscope for imaging large embryos and tissue volumes with sub-cellular resolution throughout
Figures
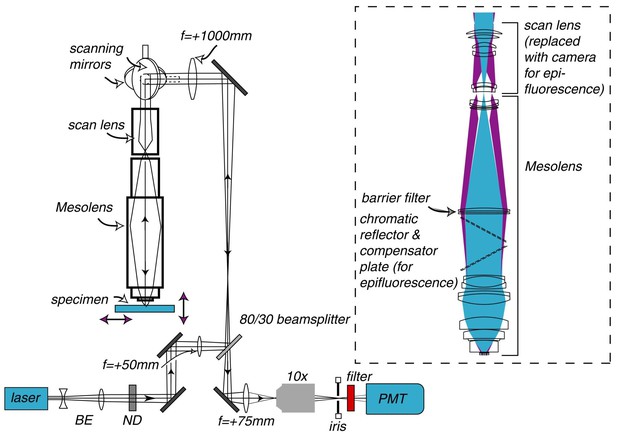
Schematic diagram of the laser scanning confocal Mesolens system.
BE=beam expander, ND= neutral density filter. Only one detection channel is shown here for ease of presentation. Inset: Illustration of the optical train of the Mesolens and scan lens. Also shown is the position of the compensator plates and camera when the Mesolens is used for wide-field epi-fluorescence imaging.
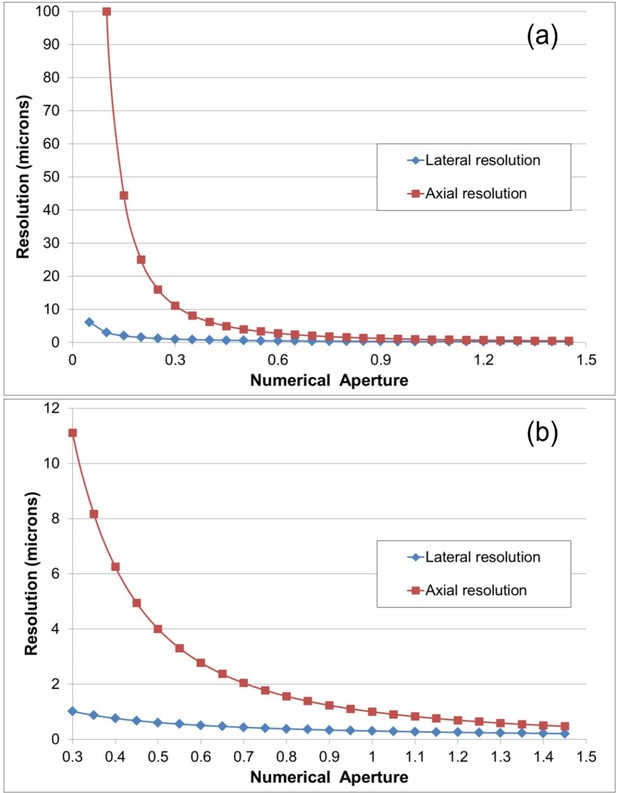
Lateral and axial resolution of a dry lens as a function of N.A.
for a wavelength of 500 nm, from Equations 1 and 2. (a) shows the full range of N.A. while (b) shows the higher N.A. range only. It is clear that below N.A.=0.45 the axial resolution becomes significantly poorer with decreasing N.A.
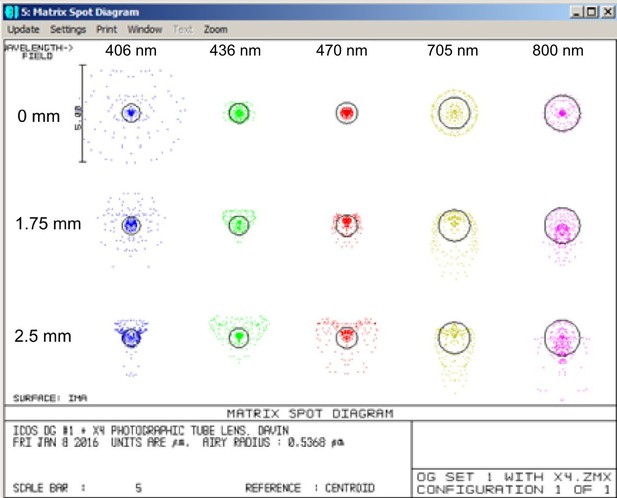
Illustration of the excellent chromatic correction of the Mesolens.
Zemax spot diagrams at wavelengths of 405, 436, 470, 705 and 800 nm and at distances of 0, 1.75 and 2.5 mm from the center of the field at the sample plane. The black circles are the Airy disk diameters calculated from the N.A. and wavelength.
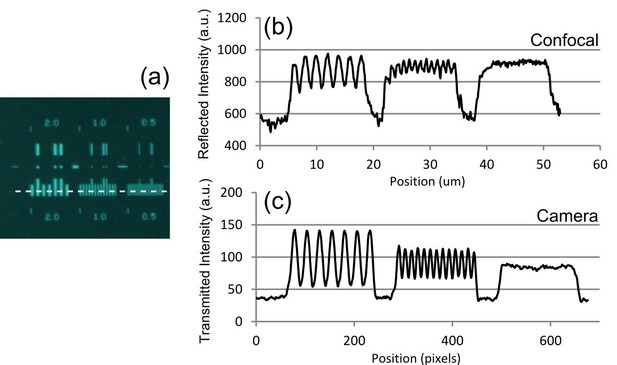
Optical resolution measurements for the Mesolens.
(a) shows an image of the specimen consisting of a series of gratings with periods of 2 µm, 1 µm, and 0.5 µm obtained with brightfield illumination and captured in wide-field mode using a camera. (b) and (c) show intensity linescan measurements of this region of the specimen obtained in (b) laser scanning confocal reflection mode using a 514 nm laser and (c) in camera mode. The intensity profiles in (b) and (c) are obtained from the region indicated by the dashed line in (a).
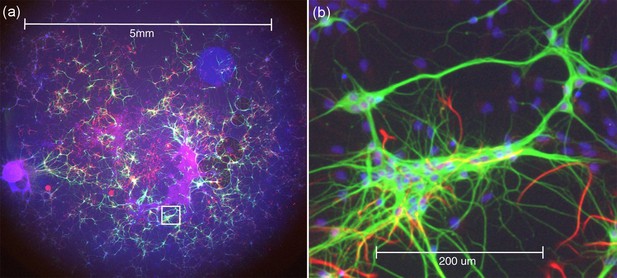
Wide-field epi-fluorescence imaging of an explant culture of rat embryonic brain as a colour merge of three camera images at the same focus position, demonstrating the excellent colour correction of the Mesolens.
The blue fluorescence is of nuclei stained with DAPI, neurons show green because of Alexa 488 conjugated to an antibody against beta-III tubulin and astrocytes are red because of Alexa 546 conjugated to anti-GFAP. (a) Full FOV (b) Software zoom of the area indicated in (a) (Video 1) to visualize individual nuclei and neurons.
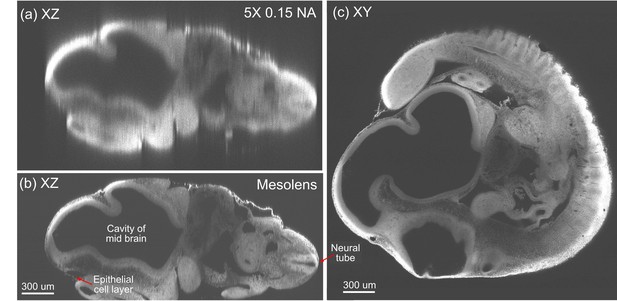
Optical sectioning of an optically-cleared and fluorescently-stained 10 day old mouse embryo.
(a) is the embryo imaged in XZ using a dry objective lens of similarly low magnification to the Mesolens (5x/0.15 N.A., HCX PL Fluotar, Leica Microsystems). (b) is the same embryo imaged at the same region in XZ using the Mesolens with oil immersion. The Mesolens can image throughout the entire 2 mm thick embryo with sub-cellular resolution: individual nuclei can be observed all the way through. This is in contrast to the image with the commercial low magnification, low N.A. objective, where the long needle-like vertical structures indicate a very poor z resolution. Figure 4c shows an XY cross section at a depth of around 1 mm into the same specimen imaged using the Mesolens. Full resolution versions of (b) and (c) are available as Figure 4—figure supplements 1 and 2.
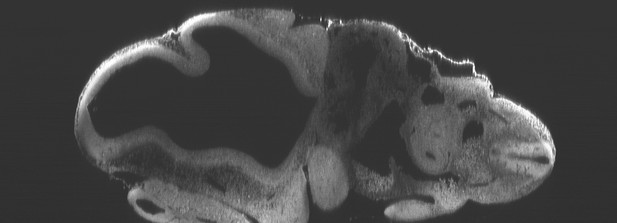
Optical sectioning of an optically-cleared and fluorescently-stained 10 day old mouse embryo imaged using the Mesolens.
XZ section. This is the high-resolution version of Figure 4b.
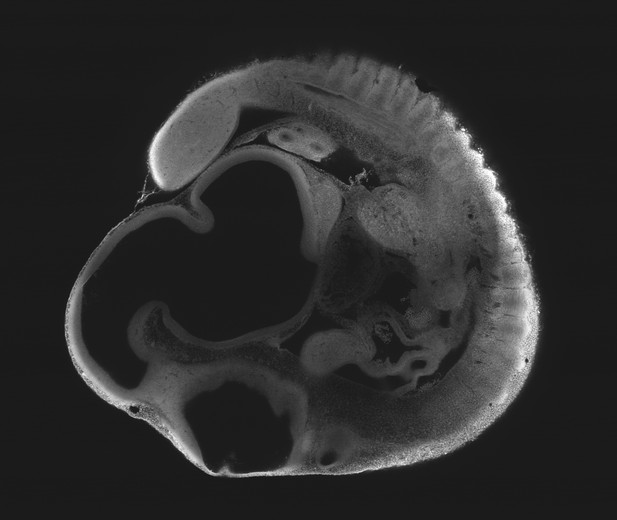
Optical sectioning of an optically-cleared and fluorescently-stained 10 day old mouse embryo imaged using the Mesolens.
XY cross section at a depth of around 1 mm. This is the high-resolution version of Figure 4c.
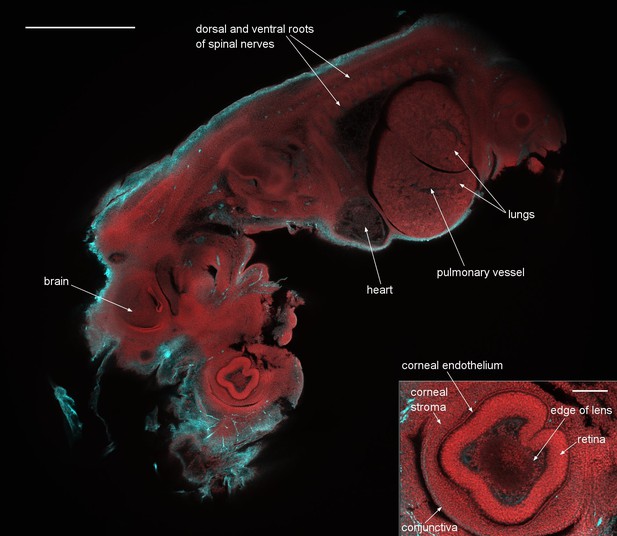
A dual-colour optical section of a 12.5 day old mouse embryo obtained with the Mesolens in laser scanning confocal mode.
The neuronal axons are stained with Alexa 594 (cyan) and nuclei with acridine orange (red). Scale bar=1 mm. The inset shows a blow-up of the eye region revealing the individual cell nuclei. The image plane of this inset is located 36 µm closer to the specimen surface than in the full image shown. The scale bar on the inset image is 300 µm. We can clearly identify fine structures throughout the embryo such as the developing heart muscle fibers and fine details in the eye such as the corneal endothelium. The cyan streak in the lower right corner of the blow-up indicates a stained nerve. A z-stack of the same specimen is presented in Video 2.
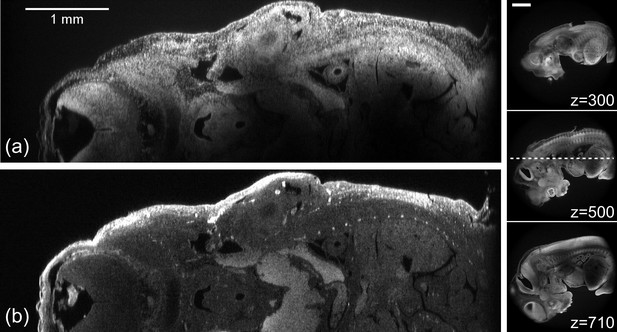
Illustration of the high z-resolution throughout the full 6 mm FOV.
(a) XZ section through 12.5 day old embryo detecting fluorescence from acridine orange. (b) The same XZ section detecting fluorescence from Alexa 594. Panels to the right show XY sections at different heights (indicated in the Fig. in microns). The dashed line in the central panel shows the location of the XZ plane. Scale bar = 1 mm.

Videos
Epi-fluorescence in an explant culture of rat embryonic brain as a colour merge of three camera images at the same focus position.
The blue fluorescence is from nuclei stained with DAPI, neurones show green because of Alexa 488 conjugated to an antibody against beta-III tubulin and astrocytes are red because of Alexa 546 conjugated to anti-GFAP. The zoom is generated by software only. Halfway into the movie we have changed to a smaller format camera mounted on an image magnifier to achieve fine enough sampling. Note that this will not enhance the optical resolution of the image but was necessary to overcome the lack of a camera able to record hundreds of megapixels in a single image.
The movie is a 3D rendering generated from a z-stack through the eye of a 12.5 day old mouse embryo (the same embryo as in Figure 5).
This embryo was stained with an antibody against axonal beta tubulin coupled to the fluorescent dye Alexa 594, to highlight nerve cells, and with acridine orange, here used chiefly as a nuclear marker. All frames in the rendering are from full 6 mm x 6 mm frames and only software zoom is used. The blow-up of the eye region reveals individual cell nuclei. The lens was visible as a slightly absorbing structure with a sharply defined outline, surrounded in turn by cells within the aqueous humour and then the strongly-stained retina. All these structures were enclosed in the corneal stroma, lined on the inside with the corneal endothelium and on the outside with the epithelial precursor of the conjunctiva.
This movie is a highly compressed and lower quality version of Video 2.
Video 3 has a much smaller file size for quick downloading and viewing.
Tables
Measured and calculated values (µm) for the FWHM of the PSF in laser scanning confocal mode and wide-field epi-fluorescence mode for a conventional 5x/0.15 N.A. lens and the 4x/0.47 N.A. Mesolens used with oil immersion. We have assumed emission λ=550 nm for the calculations.
| 5x/0.15 N.A. | Mesolens (4x/0.47 N.A.) | |||
|---|---|---|---|---|
| Theoretical | Measured | Theoretical | Measured | |
| XY FWHM, epi | 1.9 | 1.9 | 0.6 | 0.7 |
| Z FWHM, epi | 41 | 51 | 6.3 | 7 |
| XY FWHM, confocal | 1.8 | 1.7 | 0.6 | 0.8 |
| Z FWHM, confocal | 25 | 38 | 3.7 | 8 |





