Direct modulation of GFAP-expressing glia in the arcuate nucleus bi-directionally regulates feeding
Figures
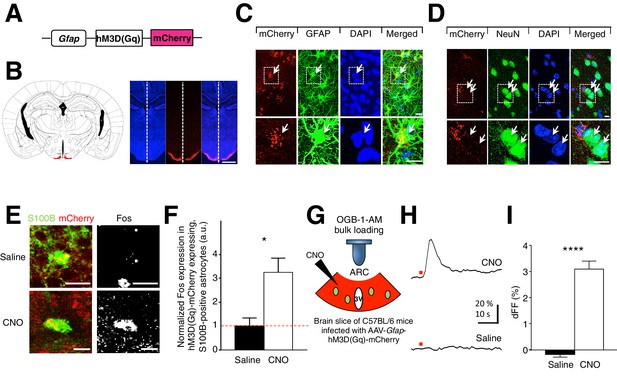
CNO-dependent activation of hM3D(Gq)-mCherry expressing ARC glia evokes increased Fos immunoreactivity and elevated intracellular Ca2+.
(A) Design of the AAV construct expressing hM3D(Gq)-mCherry under the Gfap promoter. (B) (Left) Schematic drawing showing the location of ARC (red) in a coronal brain slice (Right) An example DAPI-stained (blue) coronal brain slice containing hM3D(Gq)-mCherry (red) expressing glia in ARC. Scale bar, 1 mm. (C–D) DAPI, anti-GFAP and anti-NeuN immunohistochemistry showing expression of hM3D(Gq)-mCherry in glia but not in neurons. (Inset) Magnified image of a hM3D(Gq)-mCherry expressing glial cell and non hM3D(Gq)-mCherry expressing neurons. Scale bars, 10 µm. (E) In vivo injection of CNO but not saline induces Fos immunoreactivity in hM3D(Gq)-mCherry-expressing (red), S100B-positive (green) ARC astrocytes. Animals were perfused for Fos quantification 2 hr post injection. Scale bar, 10 µm. (F) Population mean of normalized Fos expression in astrocytes following saline or CNO injection. (G) Configuration of calcium imaging of OGB-1-AM loaded, hM3D(Gq)-mCherry expressing astrocytes in ARC coronal slices during CNO application. (H) Local application of CNO but not saline (red dot; 10 mM, 20 psi, 200 ms) evoked a robust Ca2+ increase in ARC astrocytes. (I) The population average of mean% fluorescence change (dFF) of astrocytes when CNO or saline was applied. *p<0.05, ****p<0.0001. Error bars represent SEM. See also Figure 1—figure supplement 1.
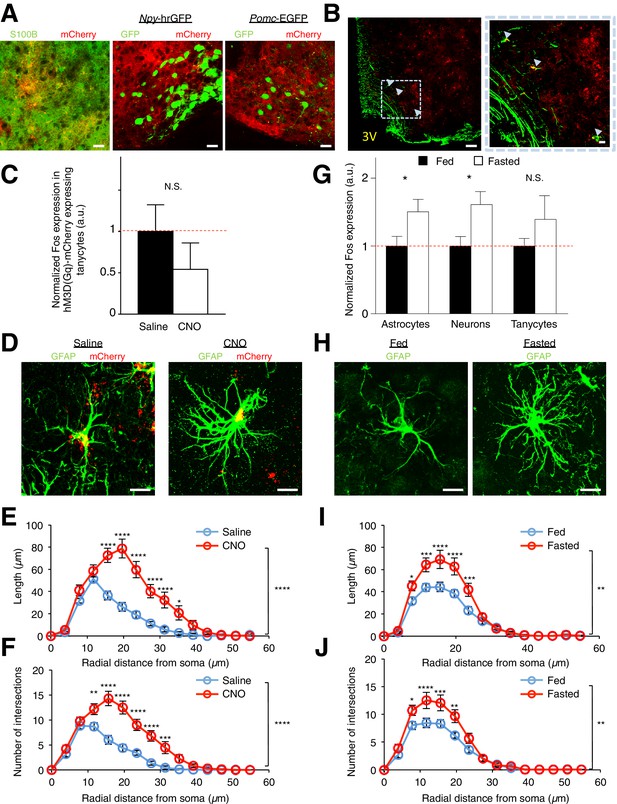
DREADDs are specifically expressed in glial cells. Astrocytes, but not tanycytes, are activated by DREADDs and fasting. Morphological changes in astrocytes are also induced by DREADDs and fasting.
(A) Expression of hM3D(Gq)-mCherry (red) (Left) co-localizes with anti-S100B (green) immunohistochemistry but did not co-localize with (Middle) GFP (green) in Npy-hrGFP and (Right) Pomc-EGFP slices. Scale bars, 20 µm. (B) Anti-vimentin immunohistochemistry showing expression of hM3D(Gq)-mCherry in some of the vimentin-expressing tanycytes lining the third ventricle (3V). Scale bars, 50 µm (left), 10 µm (right). (C) Population mean of normalized Fos expression in hM3D(Gq)-mCherry-expressing tanycytes 2 hr after saline or CNO (0.3 mg/kg) injection. n = 51 mCherry-expressing tanycytes in 6 saline-injected animals, n = 25 mCherry-expressing tanycytes in 4 CNO-injected animals, p=0.364, unpaired t-test, comparing averaged responses across tanycytes in each saline- and CNO-injected animal (D) anti-GFAP immunohistochemistry (green) revealing the morphology of hM3D(Gq)-mCherry (red) expressing ARC astrocytes 2 hr after in vivo injection of (Left) saline and (Right) CNO (0.3 mg/kg). Scale bars, 10 µm. (E) Sholl analysis of length and (F) intersections made by processes of hM3D(Gq)-mCherry ARC astrocytes revealed greater complexity in astrocytic morphology after in vivo injection of CNO compared to that of saline. n = 15 mCherry-expressing astrocytes in 5 animals in each group. (G) Fasting induces greater Fos immunoreactivity in S100B-positive ARC astrocytes and S100B-negative putative ARC neurons (but not in tanycytes) as compared to fed animals. Population mean of normalized Fos expression in ARC astrocytes, neurons and tanycytes of fasted and fed animals is shown. Fed: n = 440 astrocytes, 442 neurons and 244 tanycytes in 11 animals, Fasted: n = 400 astrocytes, 402 neurons and 226 tanycytes in 10 animals, p=0.0393/ p=0.0164/ p=0.279, unpaired t-test, comparing averaged responses of astrocytes/neurons/tanycytes between the fasted and fed animals. (H) anti-GFAP immunohistochemistry revealing the morphology of ARC astrocytes in (Left) fed and (Right) fasted animals. Scale bars, 10 µm. (I) Sholl analysis of length and (J) intersections made by the processes of ARC astrocytes revealed greater complexity in astrocytic morphology in fasted as compared to fed animals. n = 18 astrocytes in 6 animals in each group. In Figure 1—figure supplement 1G–J, fasted and fed animals were perfused 16–18 hr post initiation of fasting in fasted animals. Fed animals were given ad libitum access to food. In Figure 1—figure supplement 1E–F,1I–J, two-way ANOVA followed by Bonferroni post hoc tests was used. *p<0.05, **p<0.01, ***p<0.001, ****p<0.0001. N.S., not significant. Error bars represent SEM.
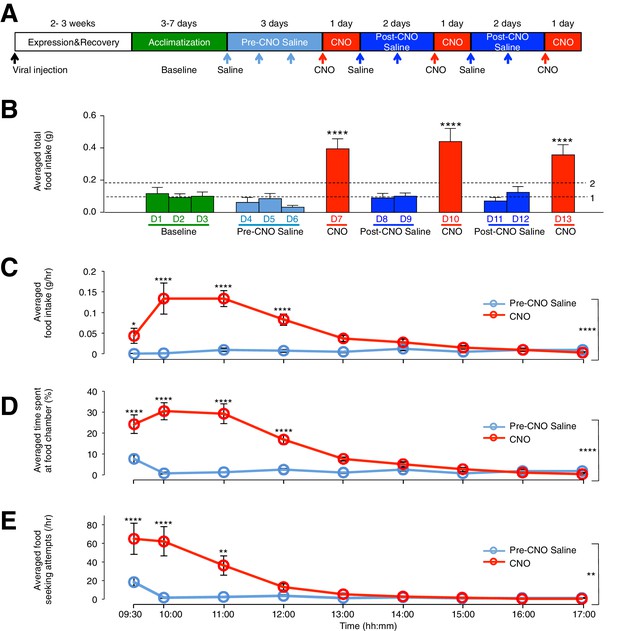
CNO-dependent activation of hM3D(Gq)-mCherry expressing ARC glia evokes increased day food intake, time spent at food chamber and food seeking attempts in C57BL/6 mice.
(A) Schematic of experimental paradigm. The mice were allowed to recover and for hM3D(Gq)-mCherry to be expressed 2–3 weeks post viral injection before acclimatization in custom cages for 3–7 days (baseline) and to saline injection for three days (pre-CNO saline). CNO injections were repeated for three days, each separated by two days of saline injection (post-CNO saline) to allow the CNO effects to clear. All injections were performed at 09:00 while the food intake was measured at specific time points between 09:00–17:00. All mice were housed in custom cages between 09:00–17:00 and returned to the standard cages after 17:00 daily. (B) Total food intake between 09:00–17:00 during baseline, pre-CNO saline, CNO and post-CNO saline averaged across animals. Dotted line 1 refers to the averaged total food intake across baseline, pre- and post- CNO saline while dotted line 2 refers to two folds of this average. (C) Food intake during hourly time points from 09:00 to 17:00 (except from 09:00–09:30 and 09:30–10:00 where 30 min time points were used) during pre-CNO saline and CNO administration. (D) The percentage of time mice spent at food chamber relative to other cage areas during specific time points following pre-CNO saline and CNO administration. (E) The frequency of attempts made to access the food chamber during specific time points following pre-CNO saline and CNO administration. In Figure 2C–E, values between 09:00–09:30 and 09:30–10:00 were normalized to hourly values. Pre-CNO saline and CNO values were averaged across three days of repeats before computing the average across animals. Two-way ANOVA followed by Bonferroni post hoc tests was used. *p<0.05, **p<0.01, ****p<0.0001. Error bars represent SEM. See also Figure 2—figure supplements 1–6.
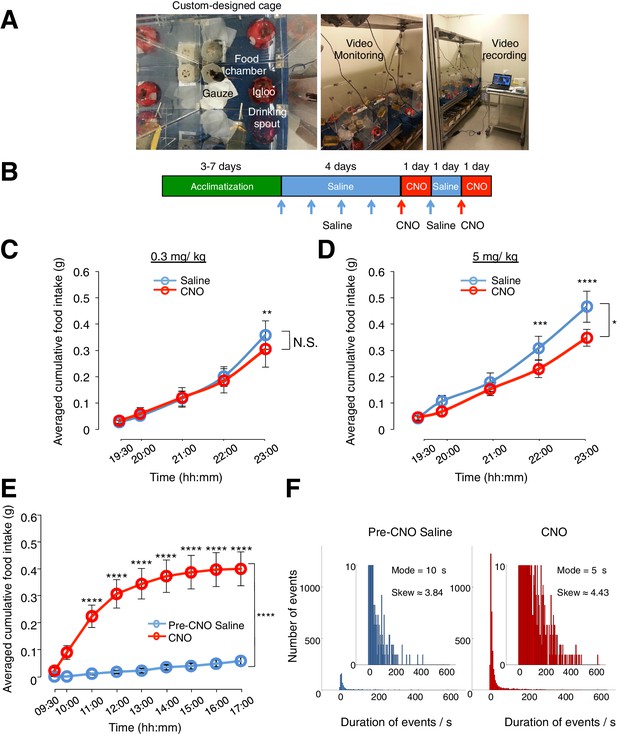
CNO administered at 5 mg/kg (but not at 0.3 mg/kg) induces non-specific effects on feeding in animals without DREADD expression. CNO(0.3 mg/kg) evoked robust feeding behavior in animals with hM3D(Gq)-mCherry-expressing ARC glia.
(A) Setup of custom-designed, open-top cages comprising a food chamber, a drinking spout/water bottle, mouse igloo and sterile gauzes. The mouse igloo was positioned opposite to the food chamber in each cage to serve as an alternative location for rest and play while the animal was not feeding at the food chamber. The bedding was removed and replaced with sterile gauzes to prevent contamination of food in the chamber and therefore ensure accurate measurement of food intake. Video monitoring of the food behavior was enabled with web-cameras positioned above the cages and connected to a laptop for data storage. (B–D) Administration of CNO at 5 mg/kg but not 0.3 mg/kg induces non-specific decrease in feeding in C57BL/6 animals without hM3D(Gq)-mCherry expression. (B) Schematic of experimental paradigm. Mice were acclimatized to custom cages before saline injections were performed for four days. CNO injections were repeated for two days, separated by one day of saline injection to allow the CNO effects to clear. All injections were performed at 19:00 while the food intake was measured at specific time points between 19:00–23:00. All mice were housed in custom cages between 19:00–23:00 and returned to the standard cages after 23:00 daily. Cumulative food intake in C57BL/6 animals was assessed from 19:00–23:00 at the specific time points, averaged across five days of saline and two days of CNO before taking the average across animals. (C) Animals were injected with CNO at lower dose (0.3 mg/kg). n = 8 animals, ANOVA, Drug: p>0.5, F (1, 7) = 0.4824; Time: p<0.0001, F (4, 28) = 30.17; Interaction between drug and time: p<0.02, F (4, 28) = 3.481, comparing the population and trial averaged cumulative food intake at specific time points between 19:00–23:00 during saline and CNO administration. (D) Animals were injected with CNO at higher dose (5 mg/kg). n = 7 animals, ANOVA, Drug: p<0.03, F (1, 6) = 8.59; Time: p<0.0001, F (4, 24) = 80.2; Interaction between drug and time: p=0.0005, F (4, 24) = 7.41, comparing the population and trial averaged cumulative food intake at specific time points between 19:00–23:00 during saline and CNO administration. (E–F) CNO administration (0.3 mg/kg), when compared to saline administration, induces an increase in food intake, frequency of shorter-duration feeding attempts and maximum duration of feeding episodes in animals with hM3D(Gq)-mCherry-expressing ARC glia. (E) Cumulative food intake from 09:00 to17:00 at the specific time points, averaged across three days of pre-CNO saline and three days of CNO administration before taking the average across animals. n = 11 animals, ANOVA, Drug: p<0.0001, F(2,24) = 19.5; Time: p<0.0001, F(8,192) = 39.2; Interaction between drug and time: p<0.0001, F(16,192) = 21.5, comparing the population and trial averaged cumulative food intake at specific time points between 09:00–17:00 during pre-CNO saline and CNO administration. (F) Frequency histograms of the food-seeking attempts of all animals during pre-CNO saline and CNO administration binned by the duration spent at the food chamber. Following CNO administration, the mode duration of food-seeking events was shorter while the skew of the distribution of duration of attempts tended towards more positive duration values than that following saline administration (p<0.0001, Kolmogorov-Smirnov test, comparing the distribution of durations following saline and CNO administration). In Figure 2—figure supplement 1C–E, two-way ANOVA followed by Bonferroni post hoc tests was used. *p<0.05, **p<0.01, ***p<0.001, ****p<0.0001. N.S., not significant. Error bars represent SEM.
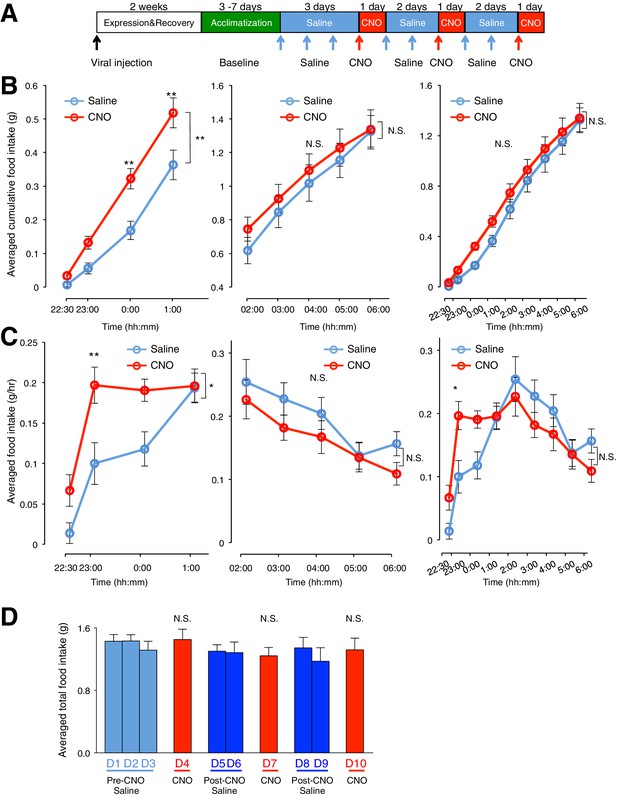
Activation of ARC glia evokes increased food intake within 3 hr after CNO application (0.3 mg/kg) but does not increase total food intake in the dark phase.
(A) Schematic of experimental paradigm. Mice were acclimatized to custom cages before saline injections were performed for three days. CNO injections were repeated for three days, separated by two days of saline injection to allow the CNO effects to clear. All injections were performed at 22:00 while the food intake was measured at specific time points between 22:00–06:00. All mice were housed in custom cages between 22:00–06:00 and returned to the standard cages after 06:00 daily. (B) Cumulative food intake from (Left) 22:00–01:00, (Middle) 02:00–06:00 and (Right) 22:00–06:00 at the specific time points, averaged across seven days of saline and three days of CNO before taking the average across animals. n = 6 animals, ANOVA, 22:00–01:00/02:00–06:00/22:00–06:00 - Drug: p=0.0099/0.5852/0.339, F(1,10) = 10.07/ F(1,10) = 0.318/ F(1,10) = 1.01; Time: p<0.0001/ p<0.0001/p<0.0001, F(3,30) = 195.4/F(4,40) = 180/ F(8,80) = 239; Interaction between drug and time: p=0.0036/0.341/0.739, F(3,30) = 5.60/ F(4,40) = 1.16/ F(8,80) = 0.643, comparing the population and trial averaged cumulative food intake at specific time points during saline and CNO administration. (C) Food intake at specific time points from (Left) 22:00–01:00, (Middle) 02:00–06:00 and (Right) 22:00–06:00, averaged across seven days of saline and three days of CNO administration before taking the average across animals. n = 6 animals, ANOVA, 22:00–01:00/02:00–06:00/22:00–06:00 - Drug: p=0.0228/0.150/0.717, F(1,10) = 7.22/ F(1,10) = 2.43/ F(1,10) = 0.139; Time: p<0.0001/ p<0.0001/ p<0.0001, F(3,30) = 38.2/ F(4,40) = 8.21/ F(8,80) = 17.2; Interaction between drug and time: p=0.0266/0.862/0.0007, F(3,30) = 3.53/ F(4,40) = 0.322/ F(8,80) = 3.85, comparing the population and trial averaged food intake at specific time points during saline and CNO administration. Food intake during 22:00–22:30 and 22:30–23:00 were normalized to hourly values. (D) Total food intake between 22:00–06:00 during pre-CNO saline, CNO and post-CNO saline averaged across animals. p>0.3, n = 6 animals, unpaired t-test comparing total food intake during three days of CNO administration with that during three days of pre-CNO saline and four days of post-CNO saline administration. In Figure 2—figure supplement 2B–C, two-way ANOVA followed by Bonferroni post hoc tests was used. *p<0.05, **p<0.05, N.S., not significant. Error bars represent SEM.
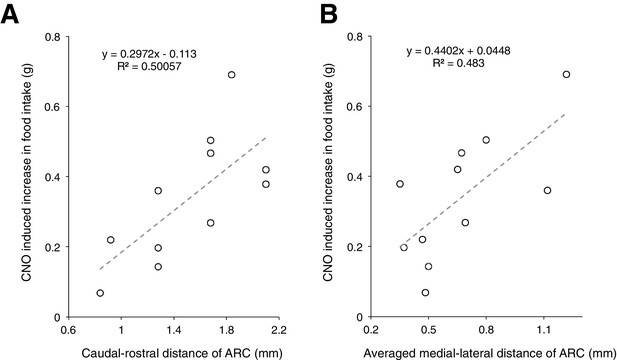
Analysis of viral expression showed that mCherry is specifically expressed within the ARC, although there were a few cases where the expression spread into adjacent areas in the hypothalamus.
(A–B) A trend where CNO induced greater increase in food intake with increased size of viral expression within the ARC was observed. CNO-induced increase in food intake was computed by taking the averaged total food intake across days with CNO administration subtracted by that across days with saline administration. The size of viral expression is quantified by the spread of viral expression along the (A) caudal-rostral axis and (B) medial-lateral axis of ARC.
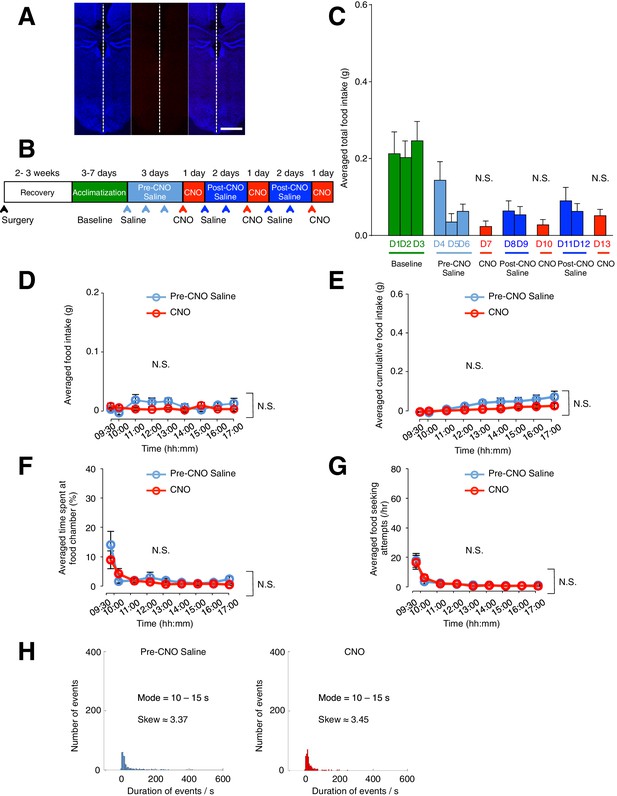
Administration of CNO (0.3 mg/kg) in mice lacking hM3D(Gq)-mCherry in GFAP-expressing ARC glia does not evoke increased food intake, frequency of feeding attempts and duration of feeding.
Viral injection of AAV-Gfap-hM3D(Gq)-mCherry was attempted in these animals but failed. All experimental procedures were identical in mice with and without successful viral injection. (A) Example DAPI-stained (blue) coronal brain slice where ARC glia did not express hM3D(Gq)-mCherry. (B) Schematic of experimental paradigm. The mice were allowed to recover after surgery before being acclimatized to custom cages for 3–7 days (baseline) and to saline injection for three days (pre-CNO saline). CNO injections were repeated for three days, each separated by two days of saline injection (post-CNO saline) to allow the CNO effects to clear. All injections were performed at 09:00 while the food intake was measured at specific time points between 09:00–17:00. All mice were housed in custom cages between 09:00–17:00 and returned to the standard cages after 17:00 daily. (C) Total food intake between 09:00–17:00 during baseline, pre-CNO saline, CNO and post-CNO saline averaged across animals. p>0.1, n = 8 animals, unpaired t-test comparing total food intake during three days of CNO administration with that during three days of baseline, three days of pre-CNO saline and 4 days of post-CNO saline administration. (D) Food intake during hourly time points (except from 09:00–09:30 and 09:30–10:00 where 30 min time points were used) averaged across three days of pre-CNO saline and three days of CNO administration before taking the average across animals. n = 8 animals, ANOVA, Drug: p>0.4, F(2,21) = 0.725; Time: p>0.3, F(8,168) = 1.16; Interaction between drug and time: p>0.1, F(16,168) = 1.33, comparing the population and trial averaged food intake at specific time points between 09:00–17:00 during pre-CNO saline and CNO administration. Food intake during 09:00–09:30 and 09:30–10:00 were normalized to hourly values. (E) Cumulative food intake from 09:00 to 17:00 at the specific time points, averaged across three days of pre-CNO saline and three days of CNO before taking the average across animals. n = 8 animals, ANOVA, Drug: p>0.3, F(2,21) = 1.08; Time: p<0.0001, F(8,168) = 17.8; Interaction between drug and time: p>0.2, F(16,168) = 1.29, comparing the population and trial averaged cumulative food intake at specific time points between 09:00–17:00 during pre-CNO saline and CNO administration. (F) The percentage of time mice spent at food chamber relative to other cage areas during specific time points averaged across three days of pre-CNO saline or three days of CNO before taking the average across animals. n = 8 animals, ANOVA, Drug: p>0.3, F(1,14) = 1.03; Time: p<0.0001, F(8,112) = 11.0; Interaction between drug and time: p>0.4, F(8,112) = 0.971, comparing the population and trial averaged percentage of time spent at specific time points between 09:00–17:00 during pre-CNO saline and CNO administration. (G) The frequency of attempts made to access the food chamber during specific time points averaged across three days of pre-CNO saline or three days of CNO before computing the average across animals. Values between 09:00–09:30 and 09:30–10:00 were normalized to hourly values. n = 8 animals, ANOVA, Drug: p>0.9, F(1,14) = 0.00391; Time: p<0.0001, F(8,112) = 20.6; Interaction between drug and time: p>0.9, F(8,112) = 0.203, comparing the population and trial averaged frequency of attempts at specific time points between 09:00–17:00 during pre-CNO saline and CNO administration. In Figure 2—figure supplement 4D–G, two-way ANOVA followed by Bonferroni post hoc tests was used. (H) Frequency histograms of food-seeking attempts of all animals during pre-CNO saline and CNO administration binned by the duration spent at the food chamber. p>0.1, Kolmogorov-Smirnov test, comparing the distribution of durations during saline and CNO administration. N.S., not significant. Error bars represent SEM.
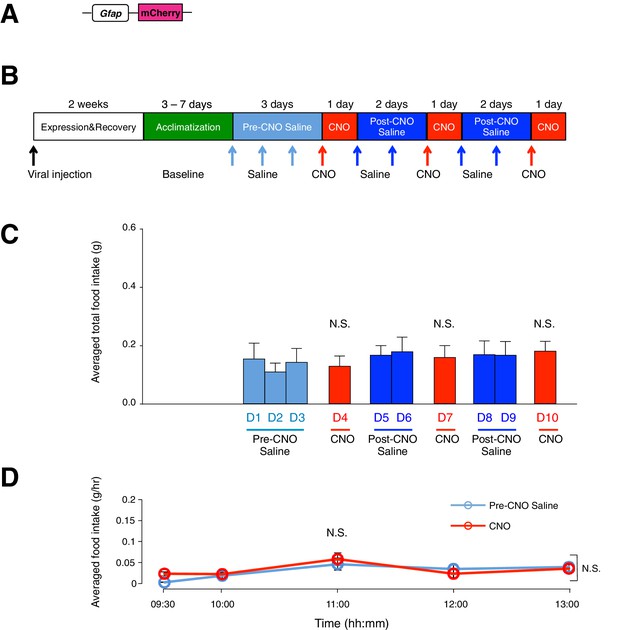
Administration of CNO (0.3 mg/kg) in control mice with mCherry-expressing ARC glia does not evoke increased food intake.
(A) Design of the AAV construct expressing mCherry under the Gfap promoter. (B) Schematic of experimental paradigm. The mice were allowed to recover after surgery before being acclimatized to custom cages for three days (baseline) and to saline injection for three days (pre-CNO saline). CNO injections were repeated for three days, each separated by two days of saline injection (post-CNO saline) to allow the CNO effects to clear. All injections were performed at 09:00 while the food intake was measured at specific time points between 09:00–13:00. All mice were housed in custom cages between 09:00–13:00 and returned to the standard cages after 13:00 daily. (C) Total food intake between 09:00–13:00 during pre-CNO saline, CNO and post-CNO saline averaged across animals. p>0.5, n = 7 animals, unpaired t-test comparing total food intake during three days of CNO administration with that during seven days of saline administration. (D) Food intake during hourly time points (except from 09:00–09:30 and 09:30–10:00 where 30 min time points were used) averaged across three days of pre-CNO saline and three days of CNO before taking the average across animals. n = 7 animals, ANOVA, Drug: p=0.701, F(1,12) = 0.154; Time: p<0.0001, F(4,48) = 8.90; Interaction between drug and time: p=0.225, F(4,48) = 1.47, comparing the population and trial averaged food intake at specific time points between 09:00–13:00 during pre-CNO saline and CNO administration. Food intake during 09:00–09:30 and 09:30–10:00 were normalized to hourly values. Two-way ANOVA followed by Bonferroni post hoc tests was used. N.S., not significant. Error bars represent SEM.
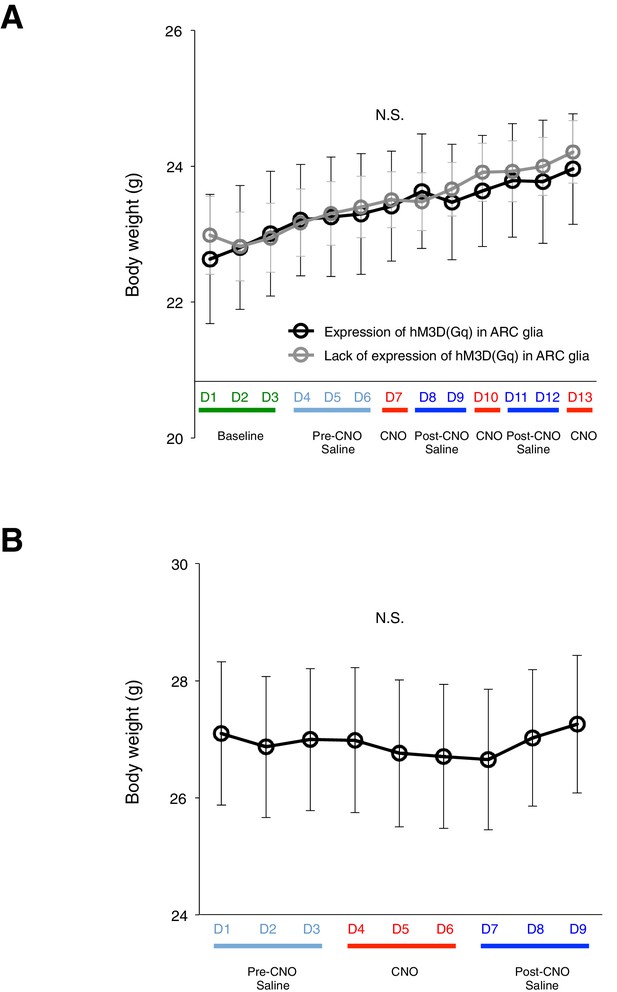
DREADD activation of glia does not lead to changes in body weight.
(A) Averaged body weight across animals with (black, n = 5 animals) or without (grey, n = 8 animals) expression of hM3D(Gq)-mCherry in ARC glia. Injections of CNO or saline were performed once a day at 09:00 (ANOVA, Without and with expression: p>0.9, F(1,11) = 0.0160; Time: p<0.0001, F(12,132) = 18.2; Interaction: p>0.8, F(12,132) = 0.578, comparing the population averaged body weight without and with hM3D(Gq)-mCherry expression. Two-way ANOVA followed by Bonferroni post hoc tests was used. (B) Averaged body weight across animals with expression of hM3D(Gq)-mCherry in ARC glia. Injections of CNO or saline were performed twice a day at 09:00 and 17:00. n = 8 animals, p>0.06, paired t-test comparing body weight measured during days of pre-CNO saline and CNO injections. N.S., not significant. Error bars represent SEM.
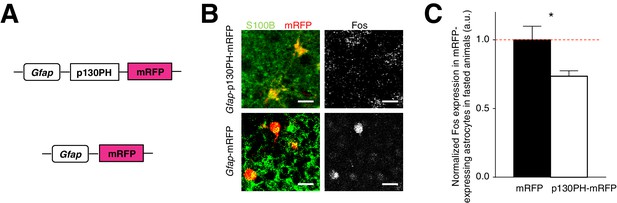
Disruption of Ca2+ signaling in ARC glia with selective expression of p130PH-mRFP leads to decreased Fos immunoreactivity.
(A) Design of the AAV constructs expressing Gfap promoter-driven p130PH-mRFP and mRFP (control) respectively. (B) Fos immunoreactivity in mRFP-expressing (red), S100B-positive (green) ARC astrocytes is lower in fasted animals injected with AAV-Gfap-p130PH-mRFP than in control animals injected with AAV-Gfap-mRFP. Animals were perfused for Fos quantification 16–18 hr post initiation of fasting. Scale bar, 20 µm. (C) Population mean of normalized Fos expression in ARC astrocytes of AAV-Gfap-p130PH-mRFP and AAV-Gfap-mRFP injected animals. *p<0.05. Error bars represent SEM. See also Figure 3—figure supplement 1.
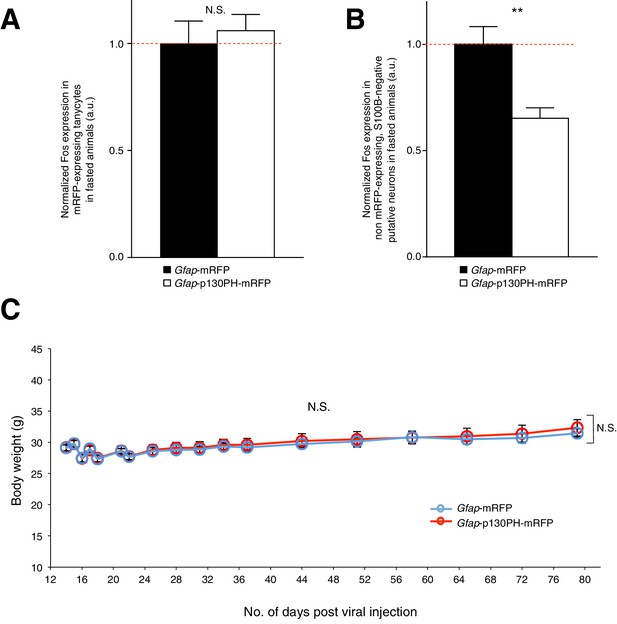
Disruption of Ca2+ signaling in ARC glia with selective expression of p130PH-mRFP leads to decreased Fos immunoreactivity in neurons (but not in tanycytes) with no change in the body weight of fasted animals.
(A) Fos immunoreactivity in mRFP-expressing tanycytes is not significantly different in fasted animals injected with AAV-Gfap-p130PH-mRFP than in control animals injected with AAV-Gfap-mRFP. Animals were perfused for Fos quantification 16–18 hr post initiation of fasting. Population mean of normalized Fos expression in ARC tanycytes of AAV-Gfap-p130PH-mRFP and AAV-Gfap-mRFP injected animals. p130PH: n = 22 tanycytes in 8 animals, Control: n = 24 tanycytes in 8 animals, p=0.651, unpaired t-test, comparing averaged responses across tanycytes in AAV-Gfap-p130PH-mRFP and AAV-Gfap-mRFP injected animals. (B) Fos immunoreactivity in non mRFP-expressing, S100B-negative ARC putative neurons is lower in fasted animals injected with AAV-Gfap-p130PH-mRFP than in control animals injected with AAV-Gfap-mRFP. Population mean of normalized Fos expression in ARC putative neurons of AAV-Gfap-p130PH-mRFP and AAV-Gfap-mRFP injected animals. p130PH: n = 718 putative neurons in 8 animals, Control: n = 600 putative neurons in 8 animals, p=0.0030, unpaired t-test, comparing averaged responses across putative neurons in AAV-Gfap-p130PH-mRFP and AAV-Gfap-mRFP injected animals. (C) Averaged body weight across AAV-Gfap-p130PH-mRFP (red, n = 8 animals) and AAV-Gfap-mRFP (blue, n = 8 animals) injected animals, measured after viral injection surgery. ANOVA, Gfap-p130PH-mRFP and Gfap-mRFP: p=0.805, F(1,14) = 0.0630; Days: p<0.0001, F(17,238) = 32.6; Interaction: p=0.980, F(17,238) = 0.420, comparing the population averaged body weight of AAV-Gfap-p130PH-mRFP and AAV-Gfap-mRFP injected animals. Two-way ANOVA followed by Bonferroni post hoc tests was used. **p<0.01, N.S., not significant. Error bars represent SEM.
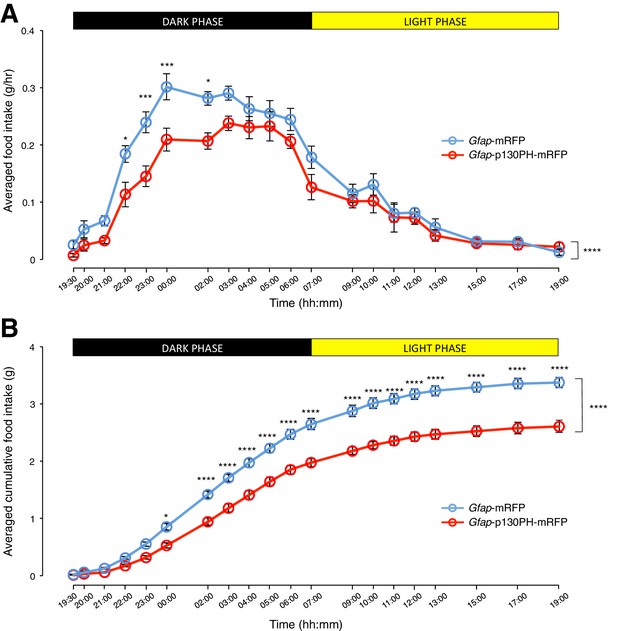
Disruption of Ca2+ signaling in ARC glia with selective expression of p130PH-mRFP leads to decreased food intake in C57BL/6 mice during the dark phase.
(A) Food intake during hourly time points (except from 19:00–19:30 and 19:30–20:00 where 30 min time points were used) in AAV-Gfap-p130PH-mRFP or AAV-Gfap-mRFP injected animals. Values between 19:00–19:30 and 19:30–20:00 were normalized to hourly values. (B) Cumulative food intake from 19:00–19:00 (24 hr, both dark and light phases) at the specific time points in AAV-Gfap-p130PH-mRFP or AAV-Gfap-mRFP injected animals. Values were averaged across three days of repeats before computing average across animals. Two-way ANOVA followed by Bonferroni post hoc tests was used. *p<0.05, ***p<0.001, ****p<0.0001. Error bars represent SEM.
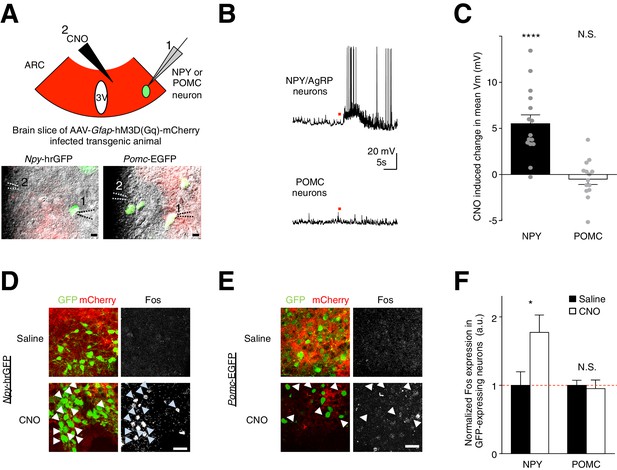
CNO activation of hM3D(Gq)-expressing glia evokes facilitatory responses in NPY but not POMC neurons.
(A) (Top) Configuration of whole-cell patch-clamp recordings of GFP-labeled NPY or POMC neurons in the ARC of coronal brain slices infected with AAV-Gfap-hM3D(Gq)-mCherry during CNO application. (Bottom) Merged green fluorescence (GFP), red fluorescence (mCherry) and differential interference contrast images of (Left) a GFP positive NPY neuron and (Right) a GFP positive POMC neuron patched in viral injected Npy-hrGFP and Pomc-EGFP slices respectively. Relative positions of (1) patch pipette and (2) CNO drug pipette were as indicated. Scale bars, 10 µm. (B) Local CNO application (red dot; 10 mM, 20 psi, 200 ms) evoked depolarizing response in (Top) an NPY (Bottom) but not in POMC neuron. (C) A population average of mean membrane potential (Vm) of NPY or POMC neurons when CNO was applied. (D–E) In vivo injection of CNO induced greater Fos immunoreactivity in NPY neurons than saline injection. This was not observed in POMC neurons. Scale bars, 20 µm. (F) Population mean of normalized Fos expression in NPY and POMC neurons following saline or CNO injection. *p<0.05, ****p<0.0001, N.S., not significant. Error bars represent SEM. See also Figure 5—figure supplement 1–2
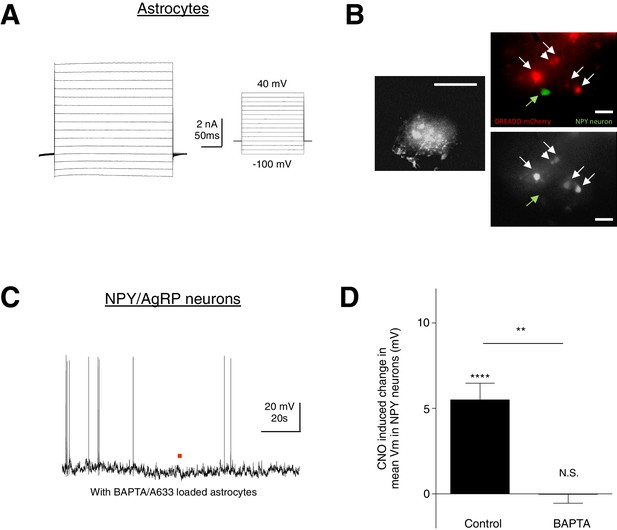
CNO does not evoke any response in NPY neurons when calcium is chelated in hM3D (Gq)-mCherry-expressing ARC astrocytes.
(A) Electrophysiological characteristics of an astrocyte with the I-V curve showing absence of active membrane currents. (B) (Left) Spread of A633 (and BAPTA) in the syncytium within 30–45 mins of patching an astrocyte identified by its small round soma with thin radiating processes. Scale bar, 200 µm. (Right Top) NPY neurons were patched immediately after BAPTA/A633 dialysis within the hM3D(Gq)-mCherry-expressing astrocyte syncytium. (Right Bottom) Individual hM3D(Gq)-mCherry-expressing astrocytes were loaded with A633 (and BAPTA). Scale bar, 20 µm. (C) Local CNO application (red dot; 10 mM, 20 psi, 200 ms) did not evoke any response in NPY neurons after BAPTA/A633 dialysis of adjacent astrocytes through whole cell patch clamping of a passive astrocyte. (D) Population average of mean Vm of NPY neurons when CNO was applied in the absence and presence of BAPTA/A633 dialysis of adjacent astrocytes (p=1.18 E-5 and p=0.957, unpaired t-test, comparing CNO-induced responses with null responses in the absence and presence of BAPTA dialysis respectively). **p<0.01, ****p<0.0001, N.S., not significant. Error bars represent SEM.
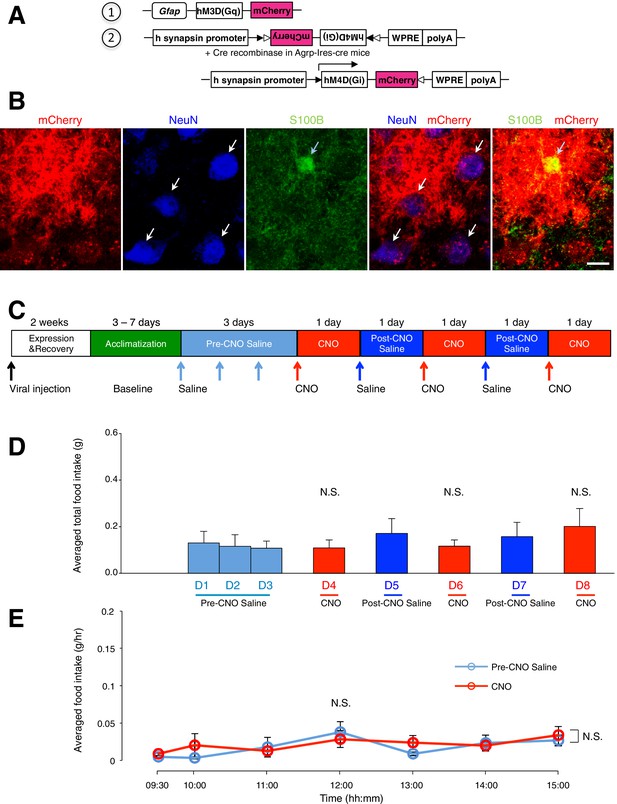
Administration of CNO (0.3 mg/kg) in Agrp-Ires-cre mice with hM4D(Gi)-mCherry-expressing AgRP neurons and hM3D(Gq)-mCherry-expressing ARC glia does not evoke any change in food intake.
(A) Design of the 2 AAV constructs (1) AAV construct expressing hM3D(Gq)-mCherry under the Gfap promoter (2) AAV construct expressing hM4D(Gi)-mCherry using FLEX switch strategy to induce Cre-mediated transgene inversion and expression in AgRP neurons. (B) Expression of mCherry (red) co-localizes with anti-NeuN (blue, presumably AgRP/NPY neurons) and anti-S100B (green) immunohistochemistry. Scale bar, 10 µm. (C) Schematic of experimental paradigm. Mice were allowed to recover after surgery before being acclimatized to custom cages (baseline) and to saline injection for three days (pre-CNO saline). CNO injections were repeated for three days, each separated by a day of saline injection (post-CNO saline) to allow the CNO effects to clear. All injections were performed at 09:00 while the food intake was measured at specific time points between 09:00–15:00. (D) Total food intake between 09:00–15:00 during pre-CNO saline, CNO and post-CNO saline administration, averaged across animals. p>0.2, n = 9 animals, unpaired t-test comparing total food intake during three days of CNO administration with that during five days of saline administration. (E) Food intake during hourly time points (except from 09:00–09:30 and 09:30–10:00 where 30 min time points were used) averaged across three days of pre-CNO saline and three days of CNO before taking the average across animals. n = 9 animals, ANOVA, Drug: p=0.684, F(1,16) = 0.172; Time: p=0.0072, F(6,96) = 3.15; Interaction between drug and time: p=0.520, F(6,96) = 0.870, comparing the population and trial averaged food intake at specific time points between 09:00–15:00 during saline and CNO administration. Food intake during 09:00–09:30 and 09:30–10:00 were normalized to hourly values. Two-way ANOVA followed by Bonferroni post hoc tests was used. N.S., not significant. Error bars represent SEM.
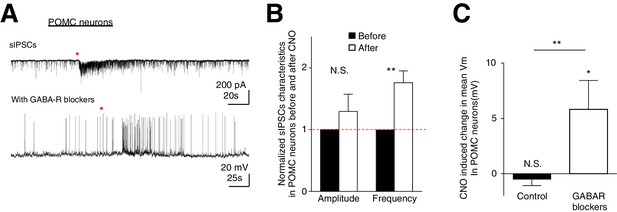
CNO activation of hM3D(Gq)-expressing glia evokes increased frequency of sIPSCs in POMC neurons.
In the presence of GABAergic blockers, hM3D(Gq)-expressing glia activated depolarization in POMC neurons is revealed. (A) (Top) Local CNO application (red dot; 10 mM, 20 psi, 200 ms) evoked increased frequency of sIPSCs in POMC neurons patched in high chloride internal solution in the presence of bath application of NBQX. (Bottom) Local CNO application (red dot; 10 mM, 20 psi, 200 ms) evoked a facilitatory response in POMC neurons patched in the presence of picrotoxin and SCH-50911 (see Materials and methods: Slice physiology). (B) Population average of mean normalized amplitude and frequency of sIPSCs in POMC neurons before and after CNO application. (C) Population average of mean Vm of POMC neurons when CNO was applied in the absence and presence of GABAergic blockers (p=0.370 and p=0.0460; unpaired t-test, comparing CNO-induced responses with null responses). *p<0.05, **p<0.01, N.S., not significant. Error bars represent SEM.
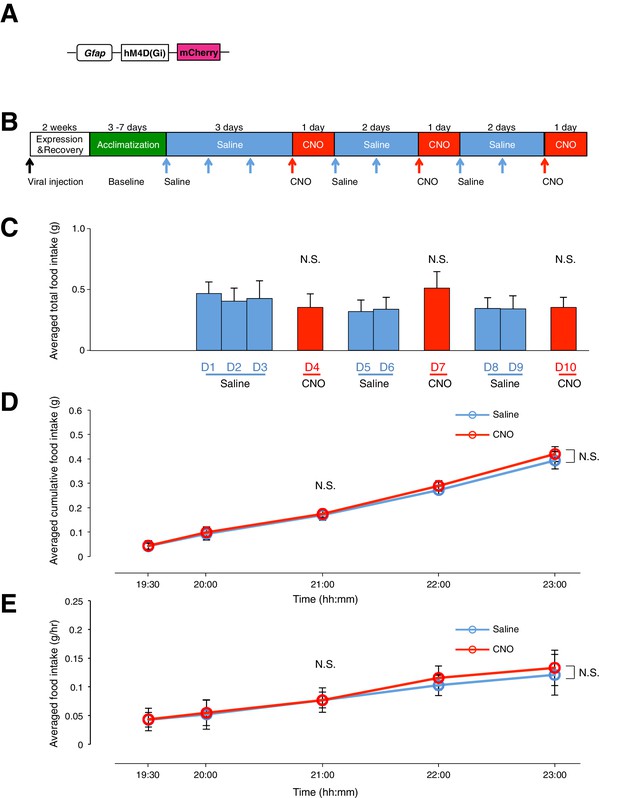
Administration of CNO (0.3 mg/kg) in mice with hM4D(Gi)-mCherry-expressing ARC glia does not evoke any change in food intake.
(A) Design of the AAV construct expressing hM4D(Gi)-mCherry under the Gfap promoter. (B) Schematic of experimental paradigm. Mice were allowed to recover after surgery before being acclimatized to custom cages (baseline) and to saline injection for three days. CNO injections were repeated for three days, each separated by two days of saline injection to allow the CNO effects to clear. All injections were performed at 19:00 while the food intake was measured at specific time points between 19:00–23:00. All mice were housed in custom cages between 19:00–23:00 and returned to the standard cages after 23:00 daily. (C) Total food intake between 19:00–23:00 during saline and CNO administration, averaged across animals. p>0.2, n = 7 animals, unpaired t-test comparing total food intake during three days of CNO administration with that during seven days of saline administration. (D) Cumulative food intake from 19:00–23:00 at the specific time points, averaged across seven days of saline and three days of CNO administration before taking the average across animals. n = 7 animals, ANOVA, Drug: p=0.888, F(1,12) = 0.0207; Time: p<0.0001, F(4,48) = 27.8; Interaction between drug and time: p=0.998, F(4,48) = 0.0333, comparing the population and trial averaged cumulative food intake at specific time points between 19:00–23:00 during saline and CNO administration. (E) Food intake during hourly time points (except from 19:00–19:30 and 19:30–20:00 where 30 min time points were used) averaged across seven days of saline and three days of CNO before taking the average across animals. n = 7 animals, ANOVA, Drug: p=0.854, F(1,12) = 0.0354; Time: p<0.0001, F(4,48) = 12.4; Interaction between drug and time: p=0.983, F(4,48) = 0.0980, comparing the population and trial averaged food intake at specific time points between 19:00–23:00 during saline and CNO administration. Food intake during 19:00–19:30 and 19:30–20:00 were normalized to hourly values. Two-way ANOVA followed by Bonferroni post hoc tests was used. N.S., not significant. Error bars represent SEM.





