Autophagy inhibition overcomes multiple mechanisms of resistance to BRAF inhibition in brain tumors
Figures
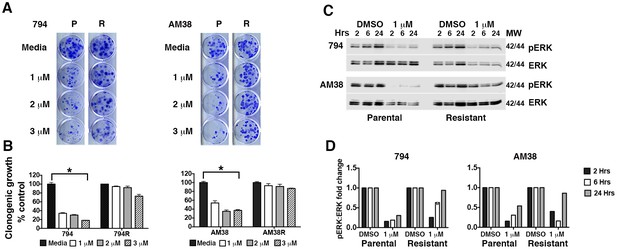
Brain tumor cell lines develop resistance to pharmacologic inhibition of BRAFV600E.
(A) Comparison of parental (P) and isogenic resistant (R) cell line response long-term growth following BRAFi for 72 hr. Representative image shown. (B) Quantification of clonogenic growth shown in A. Two way ANOVA; mean ± s.e.m, n = 3. *p<0.05. (C) Representative Western blot demonstrating decreased pERK suppression in resistant cells compared to parental cells following BRAFi. (D) Quantification of pERK:ERK ratios shown in C.
-
Figure 1—source data 1
Quantification of long-term clonogenic growth assays in 794 and AM38 parental and resistant cells treated with increasing doses of vemurafenib.
- https://doi.org/10.7554/eLife.19671.004
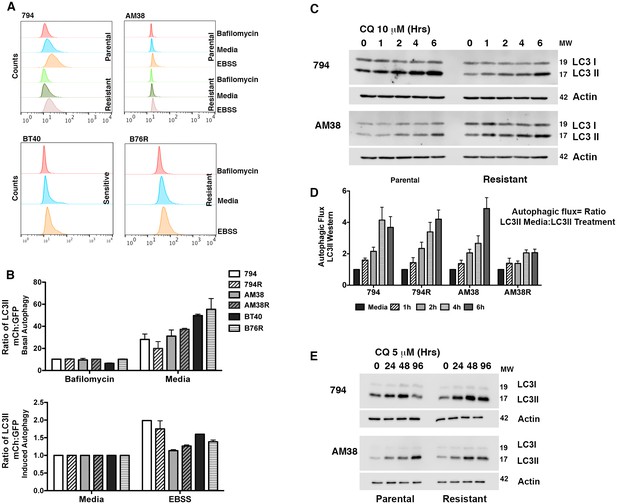
Parental and resistant BRAFV600E CNS tumor cells have similar levels of autophagy.
(A) Representative histogram comparison of parental and resistant cell line autophagy. Cells with mCh-GFP-LC3 were exposed to either standard media or starvation EBSS media for 4 hr and analyzed by flow cytometry for the change in ratio of mCh to GFP signal as a measure of autophagic flux. (B) Quantification of basal and induced autophagy as measured in A (mean ± s.e.m, n = 3). There was no significant increase of autophagic flux in resistant over parental cell lines. (C) Representative westerns and (D) quantification of samples showing accumulation of LC3II in the presence of CQ as a measure of autophagic flux (mean ± s.e.m, n = 3). There was no significant increase of autophagic flux in resistant over parental cell lines. (E) Western blot showing inhibition of autophagy with IC50 CQ dose.
-
Figure 2—source data 1
Quantification of autophagic flux by (A) Flow cytometry) and (D) Western blotting.
- https://doi.org/10.7554/eLife.19671.006
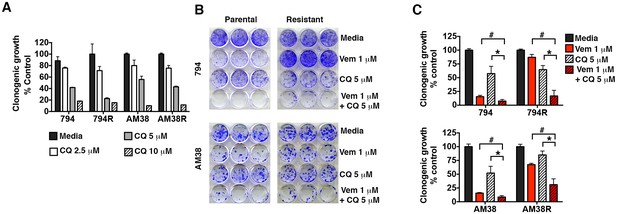
Pharmacologic inhibition of autophagy overcomes BRAFi resistance.
(A) Long-term growth assay of parental and resistant cells in response to continuous autophagy inhibition (mean ± s.e.m, n = 3). (B) Representative and (C) quantified long-term growth of parental and resistant cells following continuous autophagy inhibition (CQ), BRAF inhibition (Vem), or combination therapy. Two way ANOVA; mean ± s.e.m, n = 3. *p<0.05, # p<0.001.
-
Figure 3—source data 1
Quantification of long-term clonogenic growth assays in 794 and AM38 parental and resistant cells treated with (A) increasing doses of CQ and (B–C) vemurafenib, CQ, or a combination of the two drugs.
- https://doi.org/10.7554/eLife.19671.008
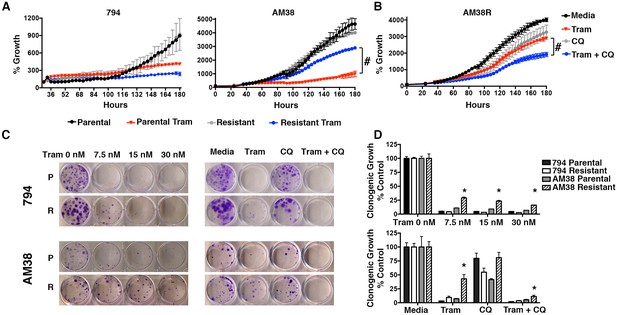
Autophagy inhibition overcomes cross-resistance to MEKi.
(A) Percent growth over time in BRAFi resistant cell lines treated with 5 nM trametinib, a MEK inhibitor (Tram). Growth measured by continuous Incucyte monitoring (Two way ANOVA, mean ± s.e.m., n = 3, # p<0.001). (B) Percent growth over time in AM38R cells treated with MEK inhibition (Tram), autophagy inhibition (CQ), or combination therapy. Growth measured by continuous Incucyte monitoring (Two way ANOVA, mean ± s.e.m., n = 3, # p<0.001). (C) Representative and (D) quantified long-term growth assay of parental and BRAFi resistant cells following continuous MEK inhibition (Tram), autophagy inhibition (CQ 5 μM), or combination therapy. One way ANOVA; mean ± s.e.m, n = 3. *p<0.001.
-
Figure 4—source data 1
Quantification of (A) % growth over time for 794 and AM38 parental and vemurafenib resistant cells treated with trametinib alone or (B) cells treated with trametinib, CQ or a combination of the two drugs.
(D) Quantification of clonogenic growth assays of 794 and AM38 parental and vemurafenib resistant cells treated with either increasing doses of trametinib, or with trametinib, CQ or a combination of the two drugs.
- https://doi.org/10.7554/eLife.19671.011
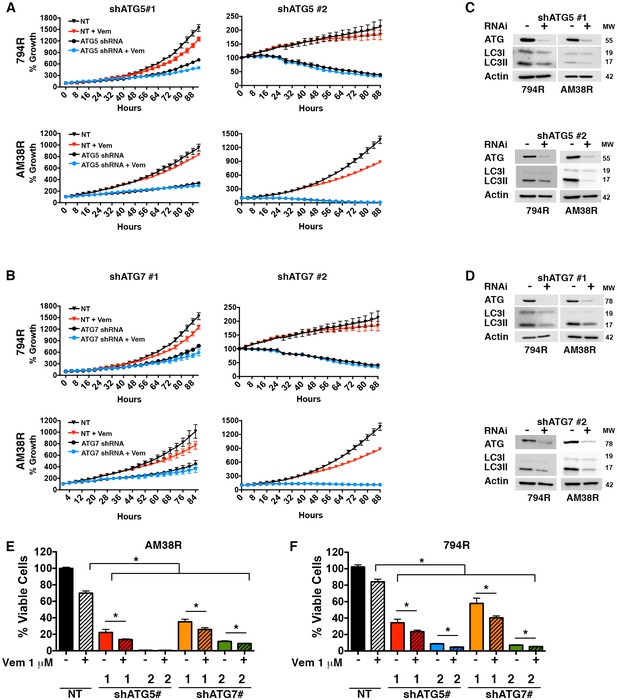
Genetic inhibition of autophagy overcomes BRAFi resistance.
(A–B) Percent growth over time in resistant cell lines with control non-targeted (NT) RNAi compared to autophagy inhibition through RNAi against (A) ATG5 or (B) ATG7, required autophagy proteins. Growth measured by continuous Incucyte monitoring (mean ± s.e.m (n = 3) (C–D) Representative westerns showing effectiveness of (C) ATG5 and (D) ATG7 RNAi and resultant decrease of LC3II. (E–F) Percent viable cells, by Cell Titer-Glo (compared to control NT) following 72 hr of vemurafenib (Vem) drug therapy with and without RNAi of essential autophagy proteins ATG5 and ATG7. One way ANOVA; mean ± s.e.m (n = 3). *p<0.05.
-
Figure 5—source data 1
Incucyte timecourse and endpoint survival data.
(A and B) Quantification of % growth over time for 794R and AM38R cells treated with RNAi to ATG5 #1, ATG5#2, ATG7#1 and ATG7#2 with and without vemurafenib. (E) Percent viable cells following RNAi to ATG5 #1, ATG5#2, ATG7#1 and ATG7#2 with and without vemurafenib.
- https://doi.org/10.7554/eLife.19671.013
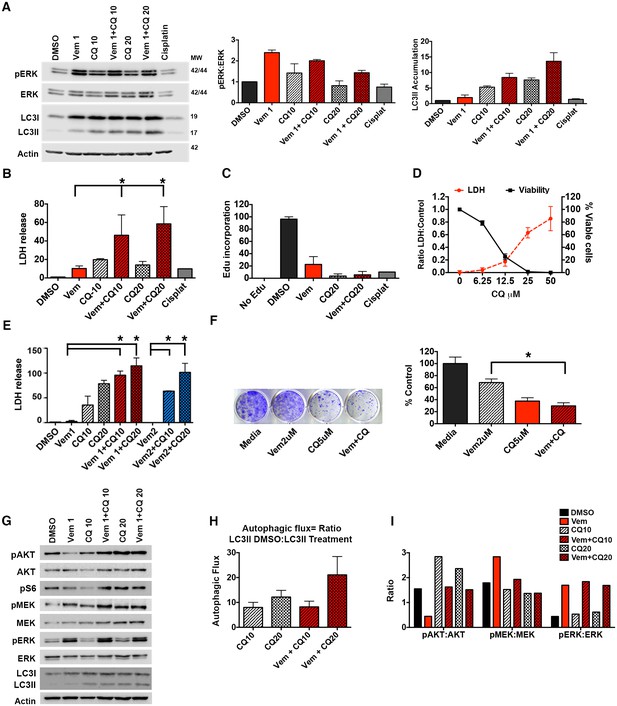
Autophagy inhibition improves clinically acquired BRAFi resistance.
(A) Ex vivo slice culture of Patient #1 tumor showing up-regulation of pERK:ERK and inhibition of autophagic flux as indicated by LC3II accumulation by Western blot with quantification of triplicate samples, mean ± s.e.m, n = 3. (B) Cumulative LDH release and (C) EdU incorporation as a measure of cytotoxicity and decreased cell proliferation in Patient #1 treated slice culture samples; One way ANOVA; mean ± s.e.m. *p<0.05. (D) In vitro cell line derived from Patient #1 showing retained response to pharmacologic inhibition of autophagy with decreasing viability and contrasting increase in LDH release with increasing doses of chloroquine (CQ). (E) LDH release as a measure of cytotoxicity in Patient #1 cell line treated for 72 hr as indicated; vemurafenib (Vem) at 1 or 2 μM, CQ at 10 or 20 μM; Unpaired two-tailed Student’s t-test; mean ± s.e.m, n = 3. (F) Long-term growth assay of Patient #1 cell line following autophagy inhibition (CQ), BRAFi (Vem), or combination therapy. Quantified collated data for triplicate experiments. Unpaired two-tailed Student’s t-test; mean ± s.e.m, n = 3. *p<0.05. (G) Western blot analysis of pAKT, AKT, pS6, pMEK, MEK, pERK, ERK, LC3I and LC3II in Patient #1 slice culture samples. Actin included as loading control. (H) Quantification of slice culture samples showing accumulation of LC3II in the presence of CQ as a measure of autophagic flux (mean ± s.e.m, n = 3). There was no significant difference of autophagic flux between the treatment groups. (I) Quantified densitometry ratios of phosphorylated proteins to total proteins shown in (G) for AKT, MEK, and ERK.
-
Figure 6—source data 1
Western quantifications, LDH and survival data.
(A) Densitometry quantification of Western blotting of slice culture samples. (B) Normalized LDH measures of Patient #1 slice culture samples. (C) EdU quantification by flow cytometry of Patient #1 slice culture samples. (D) LDH and cell viability of Patient #1 cell line treated with increasing doses of CQ. (F) Quantification of long-term clonogenic growth assays in Patient #1 cell line treated with vemurafenib, CQ or a combination of the two drugs. (H) Quantification of autophagy flux in Patient #1 slice culture samples. (I) Quantification of phosphorylated to total protein for AKT, MEK and ERK in Patient #1 slice culture samples.
- https://doi.org/10.7554/eLife.19671.015

Caspase 3/7 activation occurs in the presence of BRAF and autophagy inhibition in cells with acquired BRAFi resistance.
Patient #1 primary cells were treated with BRAFi (Vem 1 μM), autophagy inhibition (CQ 10 μM) or a combination of the two drugs. Cell growth and caspase 3/7 activation was monitored every four hours using Incucyte monitoring for 72 hr. The area under the curve (AUC) for caspase 3/7 activation was normalized to the AUC of cell numbers over time (mean ± s.e.m., n = 3).
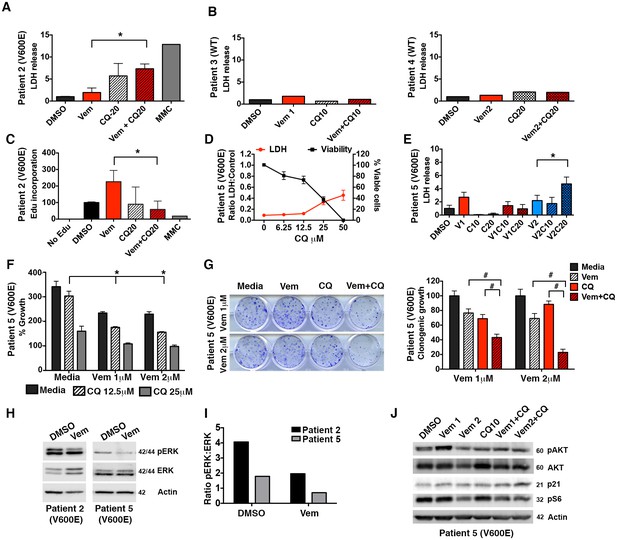
Autophagy inhibition is effective in a variety of BRAFV600E tumor models.
(A) Slice culture evaluation showing cytotoxicity as measured by LDH release in Patient #2 V600E mutant tumor. (B) No significant cytotoxicity as measured by LDH release is seen in Patients #3 and #4 with wild type (WT) BRAF tumors. (C) Decrease in EdU incorporation in Patient #2 V600E mutant tumor with combination BRAF (Vem) and autophagy (CQ) inhibition. (D) Cell line derived from Patient #5 with V600E mutant tumor showing retained response to pharmacologic inhibition of autophagy with decreasing viability and contrasting increase in LDH release with increasing doses of CQ. (E) LDH release in Patient #5 V600E mutant tumor cells treated with vemurafenib (Vem) at 1 or 2 μM, CQ at 10 or 20 μM autophagy inhibition (CQ), BRAFi (Vem), or combination therapy for 72 hr as indicated. Unpaired two-tailed Student’s t-test; mean ± s.e.m, n = 3. *p<0.05. (F) Short-term (five day) growth assay demonstrating percent growth of Patient #5 cell line following autophagy inhibition (CQ), BRAFi (Vem), or combination therapy. (G) Representative long-term (fourteen day) clonogenic assay and quantified collated data for cells treated with combination drug therapy as indicated; Vem at 1 or 2 μM, CQ at 10 μM; Unpaired two-tailed Student’s t-test; mean ± s.e.m. # p<0.001, n = 3. (H) Representative Western blot and (I) quantification demonstrating pERK response in resistant primary patient samples following BRAFi (Vem). (J) Western blot of PTEN downstream effectors in Patient #5 V600E mutant tumor cells, known to carry a PTEN mutation. No significant protein changes with BRAFi (Vem), autophagy inhibition (CQ), or combination therapy.
-
Figure 7—source data 1
Western quantifications, LDH and survival data.
(A) Normalized LDH release of Patient #2 slice culture samples. (C) EdU quantification by flow cytometry of slice culture samples. (D) LDH and cell viability of Patient #5 cell line treated with increasing doses of CQ. (E) Normalized LDH release of Patient #5 cell line treated with vemurafenib, CQ, or a combination of the two drugs. (G) Quantification of long-term clonogenic growth assays in Patient #5 cell line treated with vemurafenib, CQ, or a combination of the two drugs.
- https://doi.org/10.7554/eLife.19671.018
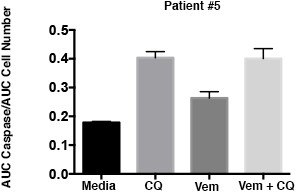
Caspase 3/7 activation occurs in the presence of BRAF and autophagy inhibition in cells with inherent BRAFi resistance.
Patient #5 primary cells were treated with BRAFi (Vem 1 μM), autophagy inhibition (CQ 10 μM) or a combination of the two drugs. Cell growth and caspase 3/7 activation was monitored every four hours using Incucyte monitoring for 72 hr. The area under the curve (AUC) for caspase 3/7 activation was normalized to the AUC of cell numbers over time. mean ± s.e.m., n = 3).
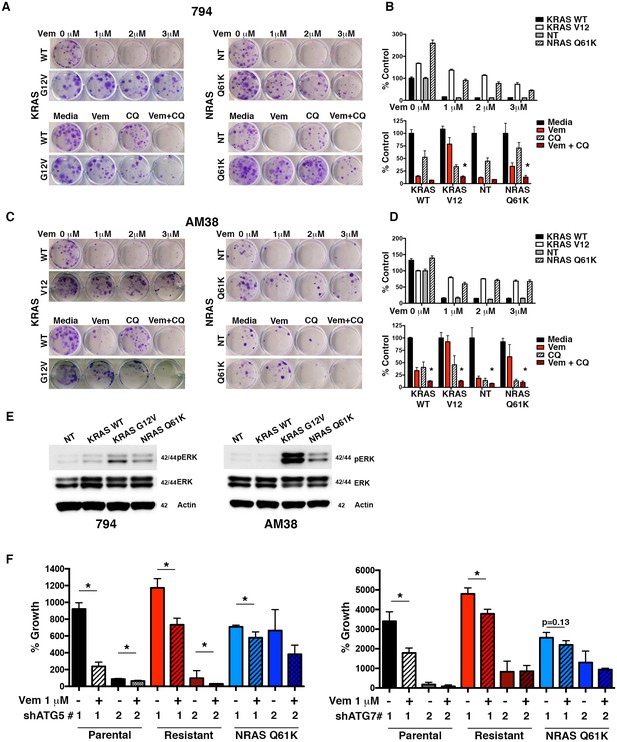
Autophagy inhibition overcomes molecularly distinct mechanisms of BRAFi resistance.
(A) to (D) Representative long-term clonogenic assays (A and C) and quantified collated data (B and D) for cells treated with combination drug therapy as indicated; Vem with an increasing dose of 1, 2 or 3 μM, CQ at 5 μM; or a combination of Vem 1 μM and CQ 5 μM. Two way ANOVA; mean ± s.e.m. # p<0.001, n = 3. (E) Representative Westerns showing increased pERK expression in cells with KRASG12V and NRASQ61K compared to NT or KRASWT. (F) Percent growth at 140 hr in AM38 (parental), AM38R (resistant) and AM38 NRASQ61K (resistant) cell lines treated with autophagy inhibition through RNAi against ATG5, ATG7 or a combination of RNAi and vemurafenib. Growth measured by continuous IncuCyte monitoring. mean ± s.e.m, n = 3.
-
Figure 8—source data 1
Long term growth assay quantifications and incucyte timecourse data.
(B and D) Quantification of long-term clonogenic growth assays in for 794R and AM38R cells with and without inserted mechanisms of resistance treated with increasing doses of vemurafenib and vemurafenib, CQ, or a combination of the two drugs. (F) Quantification of % growth over time for AM38, AM38R and AM38 NRASQ61K cells treated with RNAi to ATG5 #1, ATG5#2, ATG7#1 and ATG7#2 with and without vemurafenib.
- https://doi.org/10.7554/eLife.19671.022
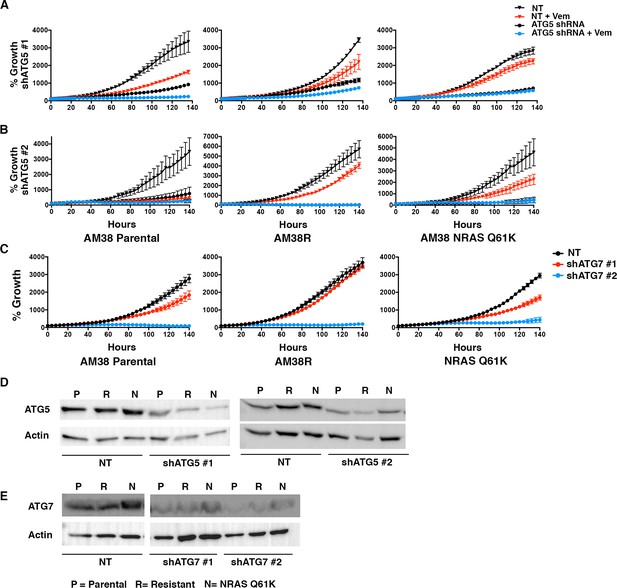
Autophagy inhibition overcomes molecularly distinct mechanisms of BRAFi resistance.
(A–B) Percent growth over time in AM38 parental, AM38R (resistant) and AM38 NRASQ61K (resistant) cell lines treated with control non-targeted (NT) RNAi, autophagy inhibition through RNAi against ATG5 (required autophagy protein) or a combination of RNAi and vemurafenib. Growth measured by continuous IncuCyte monitoring. mean ± s.e.m, n=3. (C) Percent growth over time in AM38 parental, AM38R (resistant) and AM38 NRASQ61K (resistant) cell lines treated with control non-targeted (NT) RNAi, autophagy inhibition through RNAi against ATG7. Growth measured by continuous IncuCyte monitoring. mean ± s.e.m, n=3. (D–E) Western blot analysis demonstrating level of knockdown in NT, shATG5 and shATG7 treated cells.
-
Figure 8—figure supplement 1—source data 1
Full image of ATG7 Western with associated actin blot for control to demonstrate shATG5 bands cut out of image.
All ATG7 bands shown were run and developed on the same blot.
- https://doi.org/10.7554/eLife.19671.024
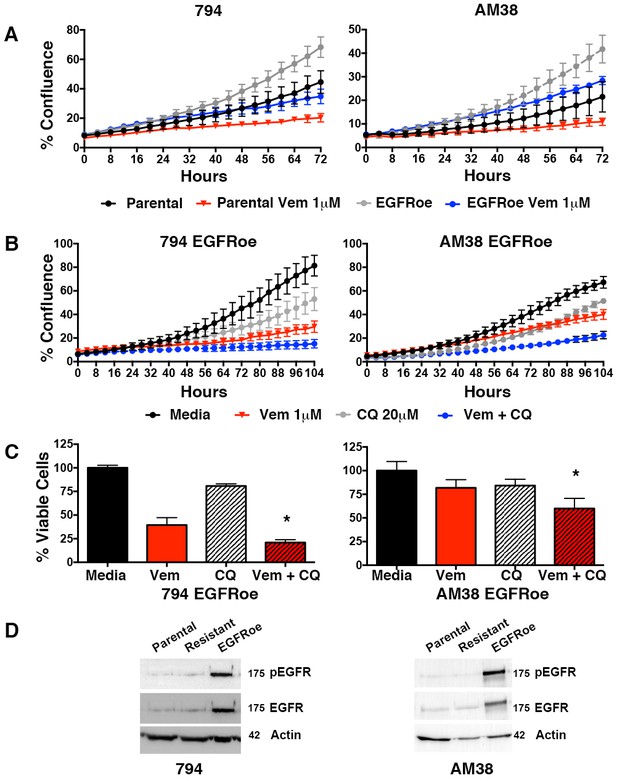
Autophagy inhibition overcomes BRAFi resistance due to escape through EGFR.
(A) Percent confluence over time in parental and EGFR overexpression (EGFRoe) cell lines treated with BRAFi (Vem 1 μM), autophagy inhibition (CQ 20 μM) or a combination of the two. Growth measured by continuous IncuCyte monitoring (mean ± s.e.m., n = 3). (B) Measure of percent viable cells (compared to media control) following 4 days of BRAFi (Vem 1 μM), autophagy inhibition (CQ 20 μM) or a combination of the two. One way ANOVA; mean ± s.e.m (n = 6), *p<0.05. (C) Percent viable cells, by Cell Titer-Glo (compared to control NT) following four days of vemurafenib (Vem) drug therapy with and without CQ autophagy inhibition in EGFRoe resistant cells. One way ANOVA; mean ± s.e.m (n = 3). *p<0.05. (D) Western blot demonstrating EGFR and pEGFR overexpression in 794 and AM38 EGFRoe cells compared to parental and polyclonal resistant isogenic cells.
-
Figure 9—source data 1
Incucyte timecourse and endpoint survival data.
(A–B) Quantification of % growth over time for 794 and AM38 parental and EGFRoe cells treated with vemurafenib, CQ or a combination of the two drugs. (C) 794 and AM38 EGFRoe percent viable cells treated with vemurafenib, CQ or a combination of the two drugs.
- https://doi.org/10.7554/eLife.19671.027

Combination BRAF and autophagy inhibition results in fewer cells with EGFR overexpression.
Representative phase contrast images showing confluence of cells following four days of therapy as indicated.
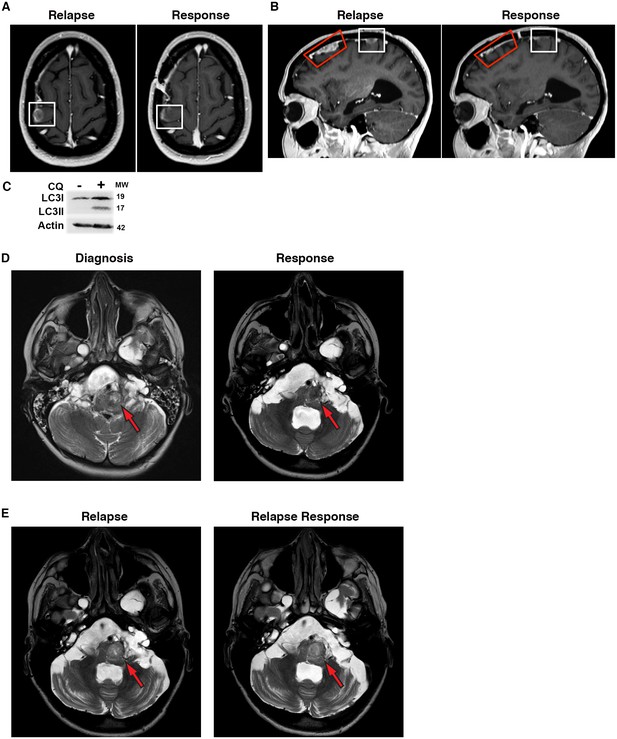
Autophagy inhibition decreases growth of metastatic glioblastoma in a patient resistant to BRAF inhibition.
(A) Contrast-enhanced axial T1-weighted MR image demonstrates relapse of tumor over the right precentral gyrus (Relapse, white box) while receiving single drug BRAFi therapy. Significant response noted on the 6 month interval MRI following 6 months of combination autophagy and BRAF inhibition (Response, white box). (B) Contrast-enhanced sagittal T1-weighted MR image illustrates tumor relapse over the right frontal lobe (red and white boxes). Significant response was noted following 6 months of combination autophagy and BRAF inhibition in both radiated (Response, red box) and non-radiated (Response, white box) tumor. (C) Demonstration of clinical autophagy inhibition as measured by LC3II accumulation in white blood cells from Patient 1. (D) Axial T2-weighted MR image demonstrates a left anterolateral medullary ganglioglioma (red arrow) at diagnosis and following 1 year of vemurafenib and vinblastine therapy. (E) Axial T2-weighted MR image demonstrates a progressive left anterolateral medullary ganglioglioma (red arrow) at relapse and stable tumor following vemurafenib and CQ therapy.
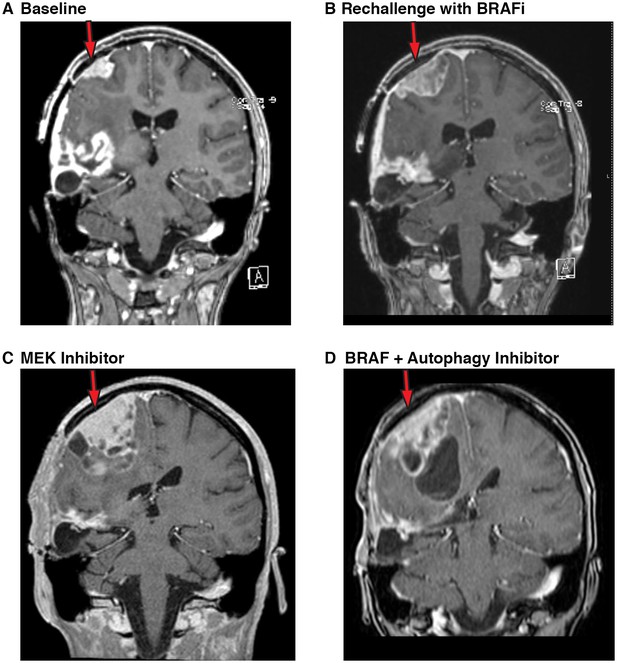
Autophagy inhibition decreases growth of metastatic glioblastoma in a patient resistant to BRAF inhibition.
(A) Contrast-enhanced coronal T1-weighted MR image demonstrates relapse of tumor over the right precentral gyrus (red arrow) while on palliative care. (B) Contrast-enhanced coronal T1-weighted MR image demonstrates the progression of tumor (red arrow) following re-initiation of vemurafenib (BRAFi) single agent therapy. (C) Contrast-enhanced coronal T1-weighted MR image demonstrates the progression of tumor (red arrow) following trametinib (MEK inhibition) single agent therapy. (D) Contrast-enhanced coronal T1-weighted MR image demonstrates response of tumor with reduction in solid tumor mass (red arrow) following re-initiation of combination there with vemurafenib (BRAFi) and CQ (autophagy inhibitor).
Tables
Combination index values for long-term growth assays in parental and resistant cells.
Cell line | Vemurafenib 1 μM + CQ 5 μM |
|---|---|
794 | 0.41 |
794R | 0.74 |
AM38 | 0.15 |
AM38R | 0.85 |
-
R= drug induced resistance; Value > 1 antagonistic,=1 additive,<1 synergistic.
Mutation analysis of studied samples.
Sample | BRAF status | Additional mutations identified |
|---|---|---|
Patient 1 Sensitive | BRAF c. 1799T>A; p.V600E | None |
Patient 1 Resistant | BRAF c. 1799T>A; p.V600E | None |
Patient 5 Resistant | BRAF c. 1799T>A; p.V600E | PTEN c.74T>C; p.L25S TP53 c.743G>A; p. R248Q |
794 | BRAF c. 1799T>A; p.V600E | None |
794R | BRAF c. 1799T>A; p.V600E | None |
AM38 | BRAF c. 1799T>A; p.V600E | None |
AM38R | BRAF c. 1799T>A; p.V600E | None |
Combination index values for long-term growth assays in RAS driven resistant cells.
Cell line | Vemurafenib 1 μM + CQ 5 μM |
|---|---|
794 KRASV12 | 0.61 |
794 NRAS61K | 0.73 |
AM38KRASV12 | 0.54 |
AM38NRAS61K | 0.01 |
-
R= drug induced resistance; Value > 1 antagonistic,=1 additive,<1 synergistic.





