Adequate immune response ensured by binary IL-2 and graded CD25 expression in a murine transfer model
Figures
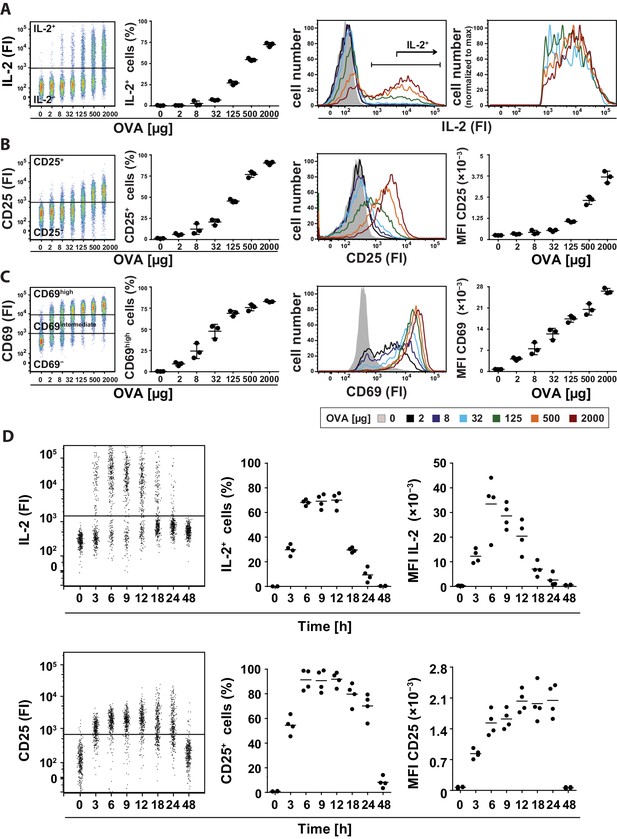
Binary IL-2 secretion of adoptively transferred Th cells in vivo after immunization with increasing amounts of antigen and kinetics of IL-2 secretion.
BALB/c mice, adoptively transferred with OVA-specific T cells from DO11.10 mice, were immunized intravenously with the indicated amounts of OVA and 10 μg LPS as adjuvant and analyzed 14 hr later (A–C) or with 2 mg OVA + 10 µg LPS and analyzed over time (D). IL-2 secretion of OVA-TCR+ CD4+ T cells was analyzed using the IL-2 secretion assay. Data from gated transgenic T cells (live B220− CD4+ OVA-TCR+ Foxp3−) were concatenated (3 mice per antigen dose). (A) IL-2 production was plotted against the amount of OVA used for immunization: Very left dot plot: fluorescence intensities of IL-2 per cell; left graph: frequencies of IL-2 producing cells; right histogram overlay: IL-2 fluorescence intensities (FI) for all gated OVA-TCR+ CD4+ T cells; very right histogram overlay: IL-2 producing OVA-TCR+ CD4+ T cells. (B) Similar analysis for the expression of CD25 on gated OVA-TCR+ CD4+ T cells. In the very right graph the mean fluorescence intensity (MFI) of CD25 is plotted against the amount of OVA. (C) Similar analysis for the expression of CD69 on gated OVA-TCR+ CD4+ T cells. CD69 expressing T cells were distinguished in antigen-specific activated CD69high and bystander-activated CD69intermediate expressing T cells. The gates for CD69intermediate and CD69high T cells were set according to unimmunized and non-transfer immunized controls. (D) IL-2 production (top row) and CD25 expression (bottom row) in OVA-TCR+ CD4+ T cells are plotted against time after immunization. Dot plots (left) depict fluorescence intensities per cell; the middle graphs depict frequencies of positive cells; the right graphs depict MFIs. Statistics: mean and standard deviation were plotted in all graphs. Data are representative of three independent experiments.
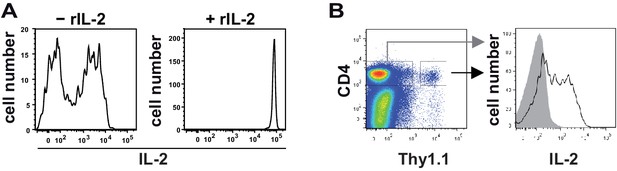
The cytokine capture matrix is not saturated by the endogenous IL-2 secretion.
C57BL/6 mice with adoptively transferred sorted naive (CD62Lhi) CD4+ T cells from OT-II mice were immunized intravenously with 2 mg OVA and 10 µg LPS as adjuvant and analyzed at 7 hr. (A) At the end of the secretion phase, 0.4 µg/ml exogenous recombinant IL-2 was added (right panel). Samples without recombinant IL-2 served as controls (left panel). (B) Transgenic IL-2-secreting CD4+ T cells were determined using the cytokine secretion assay.
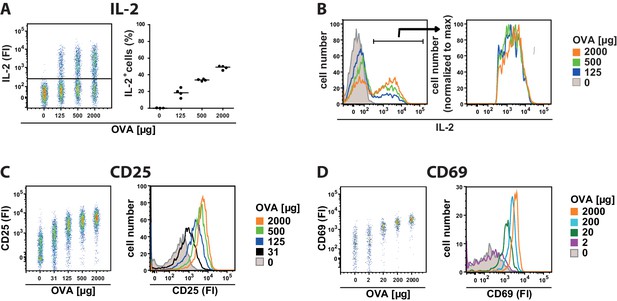
Binary IL-2 secretion of adoptively transferred OT-II Th cells in vivo after immunization with increasing amounts of antigen.
C57BL/6 mice, adoptively transferred with sorted naive (CD62Lhi) OVA-specific CD4+ T cells from OT-II mice, were immunized intravenously with the indicated amounts of OVA and 10 μg LPS as adjuvant and analysed at 14 hr (A–D). IL-2 secretion of Thy-1.1+ CD4+ T cells was analyzed using the IL-2 secretion assay. Data from gated transgenic T cells (live B220− CD4+ Thy-1.1+) were concatenated (4 mice per antigen dose in A+B and 3 mice per antigen dose in C+D). (A) IL-2 production is plotted against the amount of OVA used for immunization: left dot plot: fluorescence intensities (FI) of IL-2 per Th cell; right figure: frequencies of IL-2 producing Th cells. (B) Histogram overlays of IL-2 fluorescence intensities for all gated transgenic Thy-1.1+ CD4+ T cells (left) as well as only for IL-2 producing Thy-1.1+ CD4+ T cells (right) are depicted. (C) CD25 expression is plotted against the amount of OVA used for immunization: fluorescence intensities of CD25 per Th cell in a dot plot (left) or a histogram overlay (right). (D) CD69 expression is plotted against the amount of OVA used for immunization: fluorescence intensities of CD69 per Th cell in a dot plot (left) or a histogram overlay (right). Data are representative of three independent experiments.
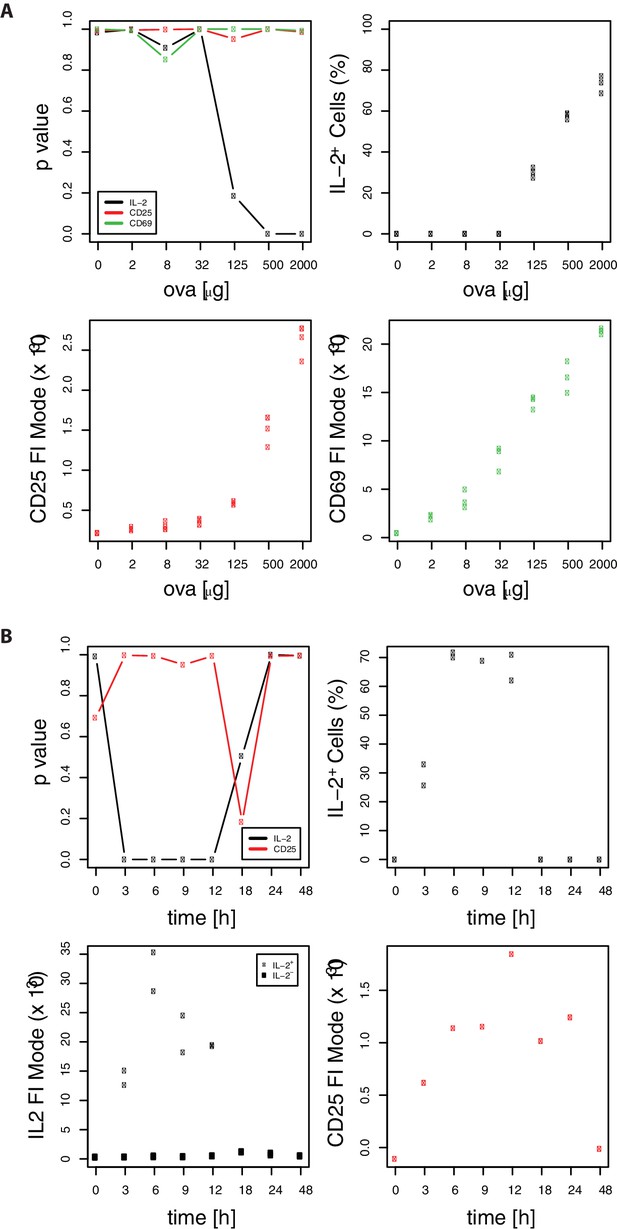
Confirmation of binary IL-2 expression using Gaussian mixture models.
Mode expression values are extracted as mean values of the components of a fitted Gaussian mixture model taking data of Figure 1, in (B) only 2 mice per group are depicted. Models with two mixture components are used only in case of significant bimodality (p<0.05). (A) Plotted are significance of digital expression (upper left) and percentage of IL-2+ cells (upper right) as well as mode expression of CD25 (lower left) and CD69 (lower right) for the indicated amounts of OVA and 10 μg LPS after 14 hr. (B) Plotted are significance of digital expression (upper left) and percentage of IL-2+ cells (upper right) as well as mode expression of IL-2+/− (lower left) and CD25 (lower right) for the indicated time points after immunization with 2 mg OVA + 10 μg LPS. The low p-values for IL-2 indicate digital expression for strong immunization (OVA > 125 μg) and between 3 and 12 hr after immunization. The subpopulations of IL-2+/− expression correspond to the mixture components with higher/lower mean. The percentage of IL-2+ cells corresponds to the relative weight of the mixture component with the higher mean. To test for digital expression we performed Hartigan's dip test statistic for unimodality on log-transformed FACS data using the R package ‘diptest’ (https://cran.r-project.org/package=diptest). The Gaussian mixture model fits were performed using the R package ‘mixtools’ (https://cran.r-project.org/package=mixtools).
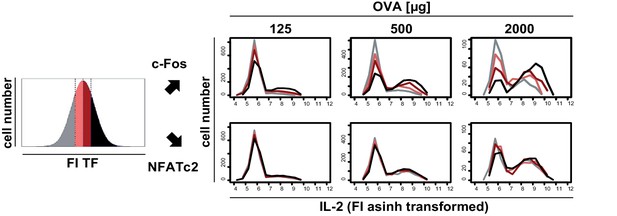
Transcriptional regulation of IL-2 secretion in adoptively transferred Th cells.
C57BL/6 mice with adoptively transferred sorted naive (CD62Lhi) CD4+ T cells from OT-II mice were immunized intravenously with increasing amounts of OVA and 10 μg LPS as adjuvant and analyzed at 7 hr. IL-2 secretion of Thy-1.1+ cells was analyzed using the IL-2 secretion assay. Splenocytes were stained and analyzed by flow cytometry. Transgenic T cells were gated (live B220− CD4+ Thy-1.1+), data from gated transgenic T cells were concatenated (4 mice per antigen dose), and the fluorescence intensity (FI) of c-Fos or NFATc2 was divided into quartiles having the same cell number (left: schematic diagram). Histogram overlays show the FI of IL-2 for each quartile and each transcription factor in samples from mice with increasing amounts of OVA (right). Data of one representative experiment out of two are shown.
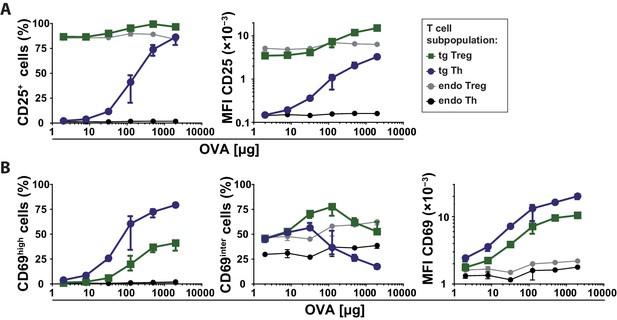
Expression levels of CD25 and CD69 on T cell subpopulations after immunization.
BALB/c mice with adoptively transferred sorted OVA-TCR+ CD4+ T cells from DO11.10 mice were immunized intravenously with increasing amounts of OVA and 10 μg LPS as adjuvant and analyzed at 14 hr. Data from gated endogenous and transgenic T cells (live B220− CD4+ OVA-TCR+/− Foxp3+/−) were concatenated (3 mice per antigen dose). (A) CD25 expression on T cell subpopulations plotted against the amount of OVA used for immunization. Left graph shows frequencies of CD25+ T cells and right graph the mean fluorescence intensity of CD25 expression on T cells. (B) CD69 expression on T cell subpopulations plotted against the amount of OVA used for immunization. Left graph shows frequencies of CD69high T cells representing antigen-specifically activated T cells, middle graph shows frequencies of CD69intermediate T cells representing bystander activated T cells, and right graph shows the mean fluorescence intensity (MFI) of CD69 expression on T cells. The gates for CD69intermediate and CD69high T cells were set according to unimmunized and non-transfer immunized controls as shown in Figure 1C. Statistics: mean and standard deviation were plotted in all graphs. Data are representative of three independent experiments.
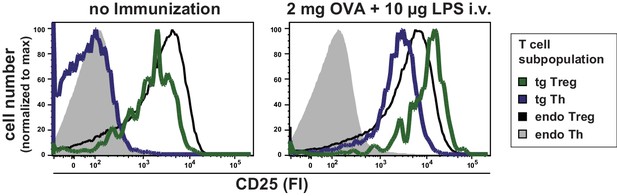
Expression levels of CD25 on T cell subpopulations at rest and after immunization.
BALB/c mice with adoptively transferred sorted OVA-TCR+ CD4+ T cells from DO11.10 mice were immunized intravenously with 2 mg OVA and 10 μg LPS as adjuvant and anaylzed at 14 hr or left unimmunized. CD25 expression levels were analyzed by histogram overlays of gated T cells (live B220− CD4+ OVA-TCR+/− Foxp3+/−) without immunization (left) and after immunization (right). Data of three individual mice per antigen dose were concatenated. One representative experiment out of three is shown.
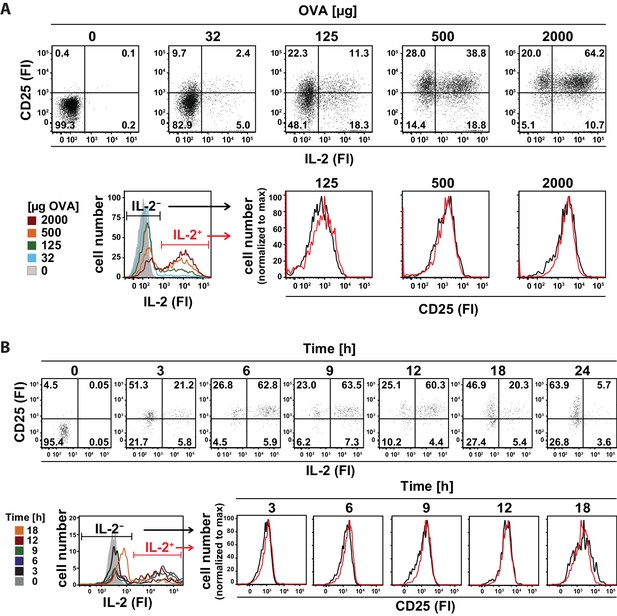
In vivo kinetics of IL-2 secretion and CD25 expression in adoptively transferred Th cells after immunization and their correlation upon titration of antigen amount as well as over time.
BALB/c mice, adoptively transferred with OVA-specific T cells from DO11.10 mice, were immunized intravenously with increasing amounts of OVA and 10 μg LPS as adjuvant and analyzed at 14 hr (A) or 2 mg OVA + 10 μg LPS and analyzed over time (B). IL-2 secretion of OVA-TCR+ CD4+ T cells was analyzed using the IL-2 secretion assay. Data from gated OVA-TCR+ CD4+ T cells (live B220− CD4+ OVA-TCR+ Foxp3−) were concatenated (3 mice per antigen dose or time point). (A) IL-2 production and CD25 expression (fluorescence intensities per cell) are depicted for increasing amounts of OVA used for immunization (top row, dot plots). A histogram overlay shows the IL-2 fluorescence intensities of gated OVA-TCR+ CD4+ T cells for different antigen amounts for direct comparison (bottom row, left). Histogram overlays of CD25 fluorescence intensities in IL-2 producing (red) and non-producing (black) OVA-TCR+ CD4+ T cells for individual OVA amounts are shown for direct comparison of CD25 expression (bottom row, right). (B) IL-2 production and CD25 expression (fluorescence intensities per cell) are depicted over time (top row, dot plots). A histogram overlay shows the IL-2 fluorescence intensities of gated OVA-TCR+ CD4+ T cells over time for direct comparison (bottom row, left). Histogram overlays of CD25 fluorescence intensities in IL-2 producing (red) and non-producing (black) OVA-TCR+ CD4+ T cells for individual time points are shown for direct comparison of CD25 expression (bottom row, right). Statistics: mean and standard deviation were plotted in all graphs. Data are representative of three independent experiments.
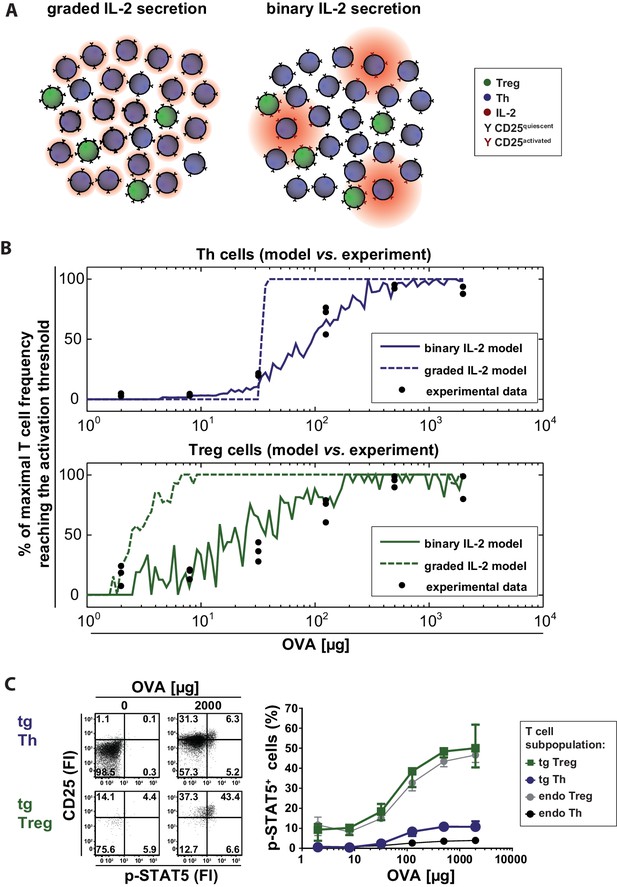
Model and experimental validation of binary versus graded IL-2 production.
(A) Schematic representation for the effects of different modes of IL-2 secretion (red) by Th (blue) on surrounding Th (blue) and Treg (green) cells. Graded IL-2 secretion at low antigen dose (left) is compared to binary IL-2 secretion (right). IL-2 mediated T cell activation is depicted via conversion of quiescent CD25 (black Y) to activated CD25 (red Y). (B) Model and comparison to experiment: Diffusion of IL-2 and interaction of IL-2 and IL-2R is modeled for a population of T cells in the spleen. The figure depicts the fraction of T cells that have more than a critical amount IL-2 bound to IL-2R on their surface (and thus reach their activation threshold) and compares it to the fraction of p-STAT5+ T cells in the experiment. (C) BALB/c mice, adoptively transferred with OVA-specific T cells from DO11.10 mice, were immunized intravenously with increasing amounts of OVA and 10 µg LPS as adjuvant. CD25 expression and STAT5 phosphorylation were analyzed after 14 hr on gated CD4+ T cells (live B220− CD4+ OVA-TCR+/− Foxp3+/−). Left figure: Dot plots of p-STAT5 versus CD25 expression for gated transgenic Th and Treg cells without immunization or after immunization with 2 mg OVA + 10 µg LPS. Right graph: Percentage of p-STAT5+ signal in different T cell subpopulations versus amount of OVA used for immunization (3 mice per antigen dose). Statistics: mean and standard deviation were plotted. Data are representative of two independent experiments.
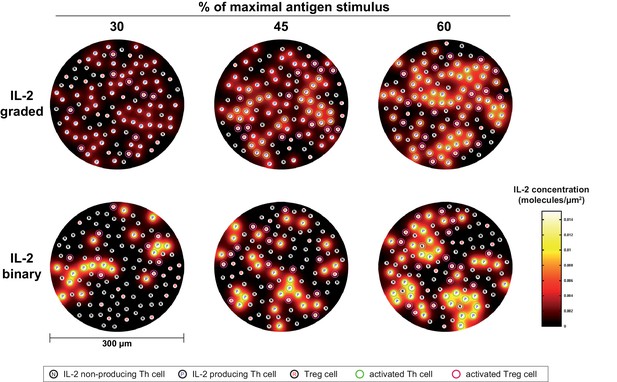
Simulation model of binary versus graded IL-2 production.
Model of IL-2 mediated Th and Treg cell activation via CD25. IL-2 producing (P), IL-2 non-producing (N) T helper cells and regulatory T cells (R) are randomly distributed in a grid. The level of available IL-2 in the intercellular space is represented by a heatmap. T helper cells that become activated via the IL-2/CD25 axis, i.e. reaching the activation threshold, appear green, and regulatory T cells that become activated via the IL-2/CD25 axis appear pink. Graded and binary IL-2 secretion are simulated for increasing percentages of maximal antigen stimulus.
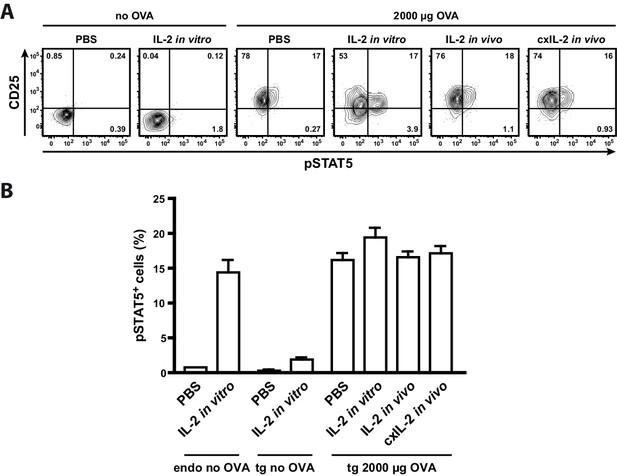
Treatment of mice with recombinant IL-2 does not increase STAT5 activation.
C57BL/6 mice, adoptively transferred with OVA-specific T cells from OT-II mice, were immunized intravenously with 2000 µg OVA and 10 µg LPS or were left unimmunized. After 12 hr mice received either 20 µg recombinant IL-2, 2.5 µg IL-2 complexed with 10 µg anti-IL-2 (JES6-5H4; targets IL-2 to CD122), or PBS as control. Mice were sacrificed 1 hr later. Cells from one part of the spleen were incubated for 25 min in vitro with 200 ng recombinant IL-2. (A) Representative original data (gated on transgenic CD4+ T cells) are shown in the flow cytometric plots. (B) Percentage of p-STAT5+ cells within either endogenous CD4+ T cells (including FoxP3+ cells) or transgenic FoxP3-negative CD4+ T cells are depicted in bar graphs. Pooled data are from two independent experiments (together six mice per group). Error bars represent SEM.
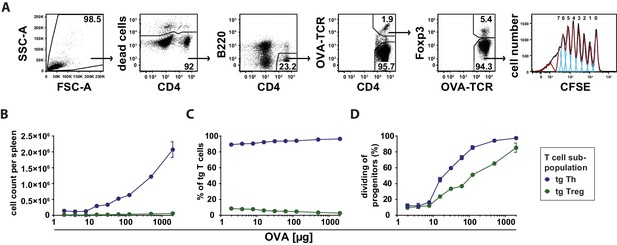
Dependency of in vivo proliferation rate of adoptively transferred Th and Treg cells on amount of antigen.
BALB/c mice, adoptively transferred with CFSE-labeled OVA-specific T cells from DO11.10 mice, were immunized intravenously with increasing amounts of OVA and 10 μg LPS as adjuvant and analyzed at 72 hr. Gated OVA-TCR+ CD4+ T cells (live B220− CD4+ OVA-TCR+ Foxp3+/−) were analyzed for CFSE dilution (2 mice per antigen dose). (A) Gating strategy for proliferation analysis: after doublet exclusion in FSC-A/FSC-H and SSC-A/SSC-H plots (not shown), cells were gated according to scatter characteristics, dead cells (Pacific Orange positive) were excluded, followed by gating on B220− CD4+ OVA-TCR+ Foxp3+/− T cells and analysis of CFSE dilution. (B) The total T cell count per spleen was calculated and plotted against amount of antigen for OVA-TCR+ Foxp3− Th cells (blue circles) and OVA-TCR+ Foxp3+ Treg cells (green circles). (C) The relative frequencies of Foxp3− Th cells (blue circles) and Foxp3+ Treg cells (green circles) in all transgenic OVA-TCR+ T cells were plotted against amount of antigen. (D) Furthermore, the percentage of dividing cells of the progenitor cells (generation 0) was calculated and plotted against the amount of OVA used for immunization. Statistics: mean and standard deviation were plotted in all graphs. Data are representative of four independent experiments.
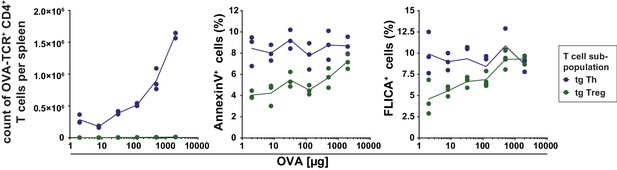
Antigen-specific Treg cells show less apoptosis then conventional Th cells at low amounts of antigen.
BALB/c mice with adoptively transferred sorted OVA-TCR+ CD4+ T cells from DO11.10×DEREG mice were immunized intravenously with increasing amounts of OVA and 10 μg LPS as adjuvant and analyzed at 72 hr. Gated OVA-TCR+ CD4+ T cells (DAPIlow B220− CD4+ OVA-TCR+ Foxp3-GFP+/−) were analyzed for AnnexinV and FLICA binding (3 mice per antigen dose). The total T cell count per spleen was calculated and plotted against the amount of antigen for OVA-TCR+ Foxp3− Th cells (blue circles) and OVA-TCR+ Foxp3+ Treg cells (green circles). The percentage of apoptotic cells (AnnexinV+ or FLICA+) amongst transgenic Th and Treg cells was plotted against the increasing amounts of OVA used for immunization. Statistics: individual data points and mean connecting line were plotted in both graphs. Data are representative of two independent experiments.
Additional files
-
Supplementary file 1
Parameter estimates for the mathematical model.
Parameter names, symbols and values used for the mathematical modeling are given.
- https://doi.org/10.7554/eLife.20616.015





