Dissection of the host-pathogen interaction in human tuberculosis using a bioengineered 3-dimensional model
Figures
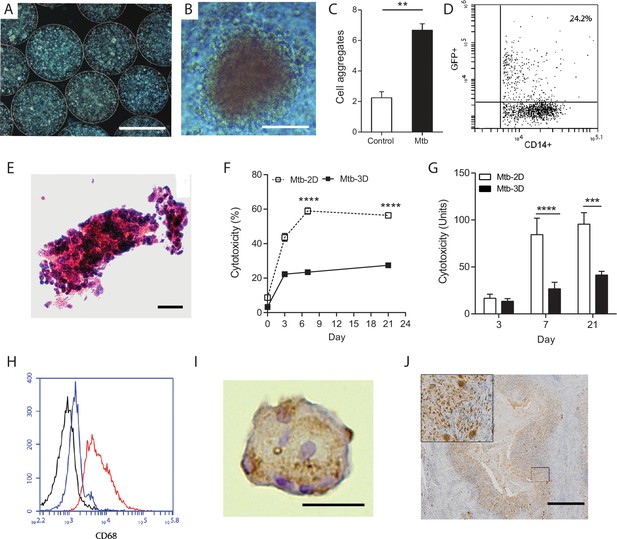
Primary human cells have greater survival in 3-D and aggregate, differentiate and fuse into multinucleate giant cells.
(A) Phase contrast microscopy with overlay of Hoeschst 33256 (blue) at Day seven demonstrates PBMCs forming aggregates within microspheres. Scale 300 µm. (B) Cell aggregation in Mtb-infected PBMC-collagen-alginate microspheres at Day 7. Scale 50 µm. (C) Cell aggregation is greater in Mtb-infected microspheres than uninfected microspheres. Cells aggregates were defined as eight or more cells viewed under 20x magnification. Data are representative of a minimum of 10 fields of view per group. (D) Cells were infected with GFP+ Mtb and then released by decapsulation. At 24 hr after infection, 24.2% monocytes had phagocytosed GFP-expressing Mtb by flow cytometric analysis. (E) Haematoxylin and eosin staining of paraffin-fixed microspheres demonstrates cell aggregates in Mtb-infected microspheres at day 14. Scale 20 µm. (F) Host cell survival is significantly greater in 3-D microspheres than 2-D cell culture as demonstrated by LDH assay. Clear box 2-D cell culture, filled box 3-D culture; an equal number of cells killed with digitonin (30 µg/ml) in the respective 3D and 2D culture was used as denominator. Mean ± SE values (n = 4). (G) Cytotoxicity measured by CytoTox Glo assay is significantly lower in 3D culture than 2D culture. (H) CD68 expression is increased in macrophages in Mtb-infected microspheres analyzed by flow cytometry. Black isotype control, blue uninfected cells, red Mtb-infected cells. (I) Multinucleate giant cells form within microspheres at day 14, immunostained with CD68 (brown) and counterstained with Haematoxylin (blue). Scale 20 µm. (J) In patients with pulmonary TB, similar giant cells are observed in pulmonary granulomas. A low power image of human pulmonary granuloma (G), with numerous multinucleate giant cells surrounding caseous centre (box, magnified area). Scale bar: 1000 μm.

Equipment set-up within containment level three tuberculosis laboratory.
(A) Bioelectrosprayer housed within a double sized class I/III Microbiological Safety Cabinet. (B) Close-up of the bioelectrosprayer cleaned after an experiment, with a syringe driver on the top. The external doors must be closed for microsphere generation to commence, providing the first level of containment, with the airflow of the class I MSC providing the second level of containment. The cabinet is formalin-fumigated after each experiment.
Microspheres placed in a 12 well tissue culture plate immediately after generation demonstrating non-magnified appearance.
https://doi.org/10.7554/eLife.21283.005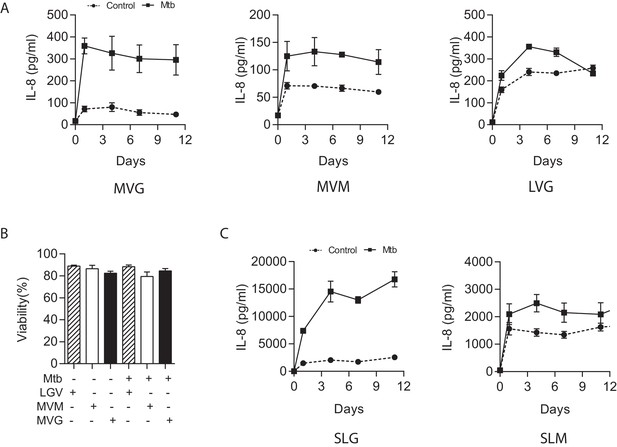
Medium viscosity guluronate (MVG) alginate is the optimal alginate for immunological studies.
(A) IL-8 secretion from microspheres composed of different alginates measured by ELISA. MVM: medium viscosity mannuronate, LVG: low viscosity guluronate. In addition, LVG had very poor biophysical properties for biolelectrospraying. (B) Cell viability analyzed by LDH release was not different between the different alginate types. (C) In sterile alginate, IL-8 secretion was higher in microspheres containing SLG compared to SLM. SLG: Sterile guluronate, SLM: sterile mannuronate. Squares, filled lines: Mtb-infected microspheres, circles and broken lines: uninfected microspheres.

Viability of PBMCs remains over 90% after decapsulation.
PBMCs were encapsulated into alginate-collagen microspheres. After 1 hr incubation at 37°C in HBSS, they were decapsulated in solution of 55 mM of sodium citrate and 5 mM EDTA prepared in HBSS (pH = 7.4) for 10 min. The cells were then washed with HBSS twice before suspension in 5 ml of RPMI and staining with propidium iodide (1 mg/ml) at 1:100 dilution and analysis by flow cytometry. For comparison, the viability of PBMCs kept in RPMI at 4°C during this period was used. The data are a representative of two independent experiments.
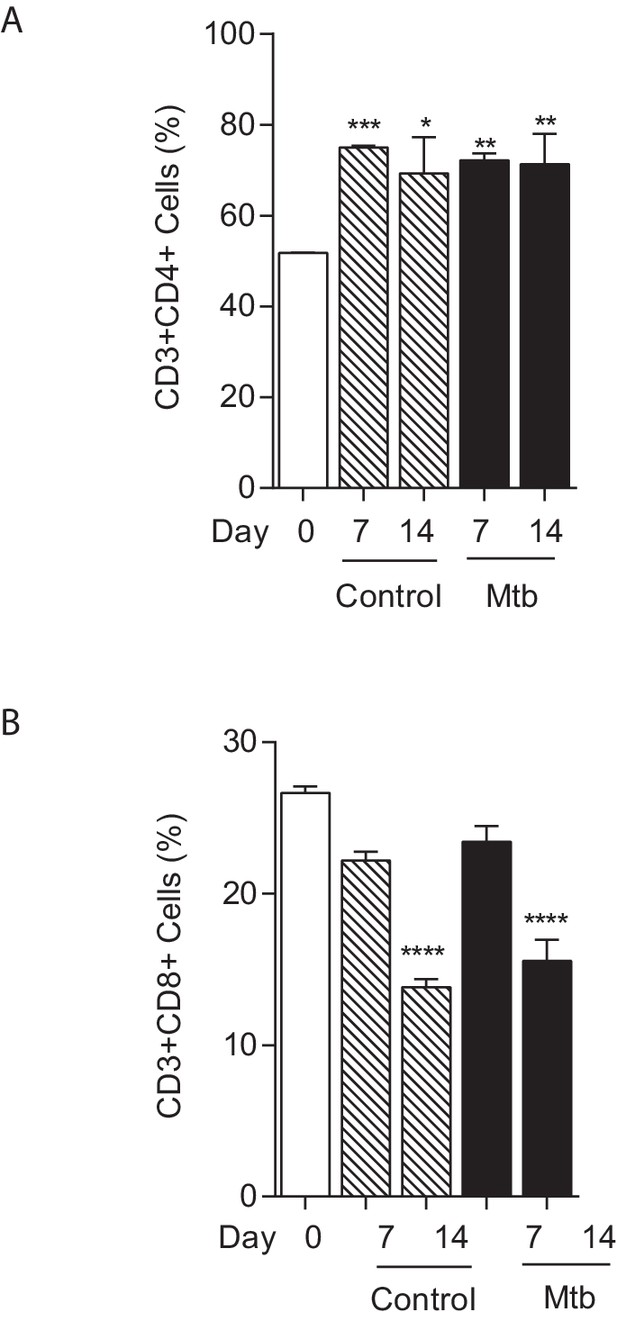
T cell composition of microspheres.
The proportion of CD4+ T cells increases between day 0 and day seven in both uninfected and infected microspheres (A), while the proportion of CD8+ T cells falls (B). *p<0.05, **p<0.01, ***p<0.001, ****p<0.0001 compared to day 0.
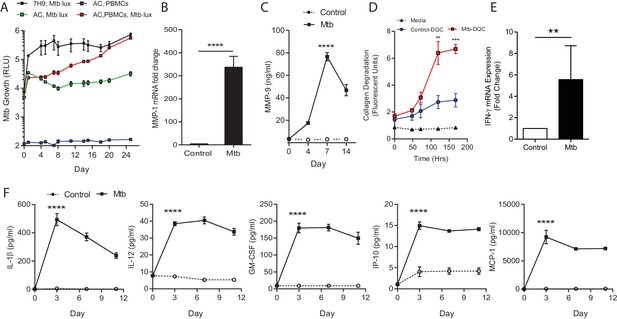
Mtb grows within microspheres containing PBMCs and upregulates MMP and cytokine expression.
(A) Mtb proliferates slowly in microspheres with no cells (green line), but progressively in microspheres containing PBMCs (red line), reaching similar luminescence to Middlebrook 7H9 broth culture at 24 days (black line). Blue line, uninfected microspheres. (B) Mtb infection upregulates MMP-1 gene expression and (C) MMP-9 secretion in microspheres. (D) MMP upregulation has a functional effect, causing collagen degradation. DQ Collagen breakdown is higher in Mtb-infected microspheres (red line) than uninfected (blue line). Triangles, microspheres with no PBMCs. (E) Mtb infection increases cellular IFN-γ mRNA accumulation relative to uninfected cells at day four in microspheres (n = 4). (F) Secretion of cytokines by Mtb-infected microspheres (squares) is significantly higher than in microspheres containing uninfected PBMCs (circles). ****p<0.0001 by t-test (B and E) and ANOVA (A, C, D, F).

Mtb infection upregulates secretion of multiple growth factors, cytokines and chemokines from microspheres measured by Luminex array.
*p<0.05, **p<0.01, ***p<0.001, ****p<0.0001 compared to uninfected microspheres at each time point.
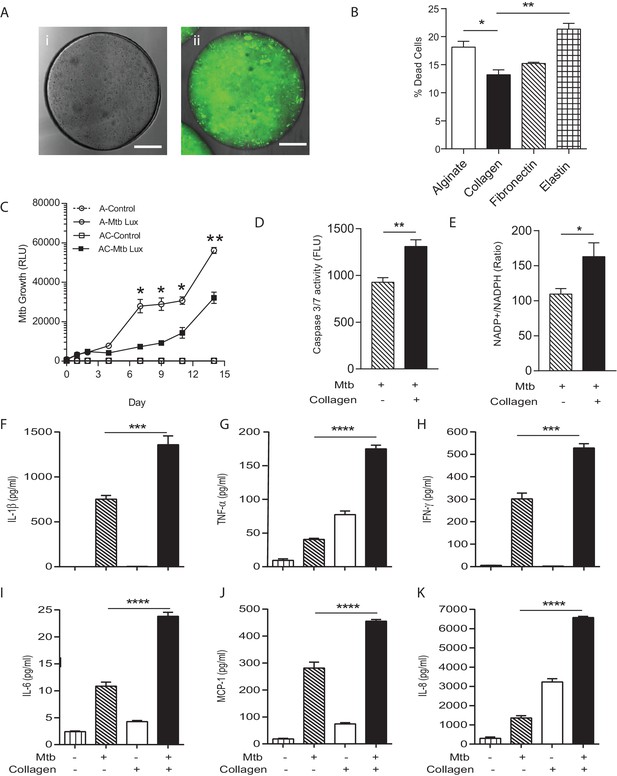
Incorporation of collagen into microspheres limits Mtb growth and increases host cell survival.
(A) Microspheres were created without collagen (i), or incorporating FITC-labelled collagen (ii) to demonstrate distribution. Immediately after bioelectrospraying, collagen is homogenous throughout the microspheres. (B) Incorporation of Type I collagen into microspheres improves cell survival at 72 hr after Mtb infection, whereas elastin did not, analyzed by CytoTox-Glo assay. (C) PBMCs control Mtb growth in microspheres containing collagen (squares) better than cells without collagen (circles). Open squares, uninfected PBMCs. (D) The level of apoptosis and NADP+/NADPH ratio (E) are higher in microspheres containing collagen at day 7. Collagen incorporation caused increased secretion of IL-1β (F), TNF-α (G), IFN-γ (H), IL-6 (I), MCP-1 (J) and IL-8 (K) at day 7. Each experiment was performed a minimum of 2 times and charts represent mean values + SEM of a representative experiment performed in triplicate. *p<0.05, **p<0.01, ***p<0.001, ****p<0.0001.
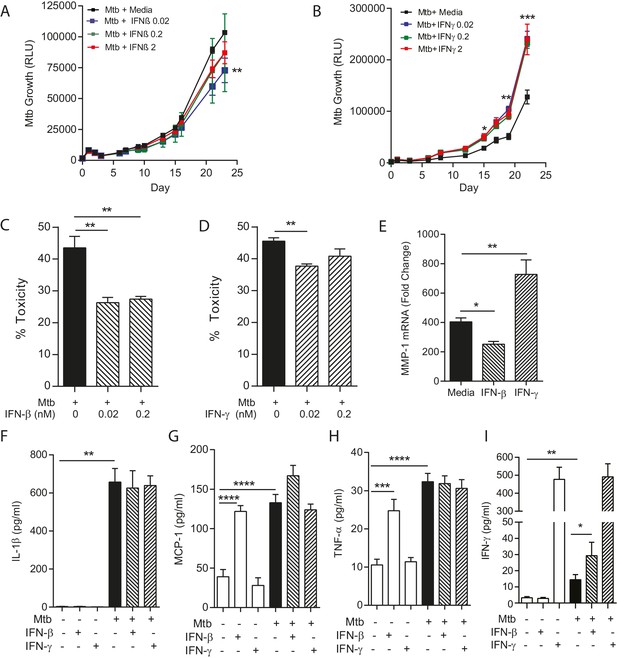
IFN-β and IFN-γ have divergent effects on bacterial growth within microspheres.
(A) Exogenous IFN-β suppresses Mtb growth after 24 days culture. Black line represents Mtb infected PBMCs. IFN-β supplementation at 0.02 nM (blue), 0.2 nM (green) and 2 nM (red) suppresses Mtb luminescence. (B) IFN-γ increases Mtb growth compared to infected PBMCs without additional cytokine. Exogenous IFN-γ at 0.02 nM (blue), 0.2 nM (green) and 2 nM (red) increases Mtb luminescence above Mtb-infected PBMCs without cytokine supplementation (black line). (C, D) Both IFN-β and IFN-γ reduce toxicity in Mtb-infected PBMCs, analyzed by CytoTox-Glo assay. (E) IFNs divergently regulate MMP-1, with IFN-β suppressing gene expression in infected microspheres while IFN-γ increases MMP-1 expression. (F–H). Mtb upregulates cytokine secretion but this is not modulated by IFNs. IFN-β drives TNF-α and MCP-1 as a single stimulus (E and G), but has no significant synergistic effect with Mtb. (I) Mtb upregulates IFN-γ secretion, and this is further increased by the addition of IFN-β. Mean + SEM of a representative experiment performed in triplicate is shown, and are representative of a minimum of 2 experiments done in triplicate. *p<0.05, **p<0.01, ***p<0.001 and ****p<0.0001.
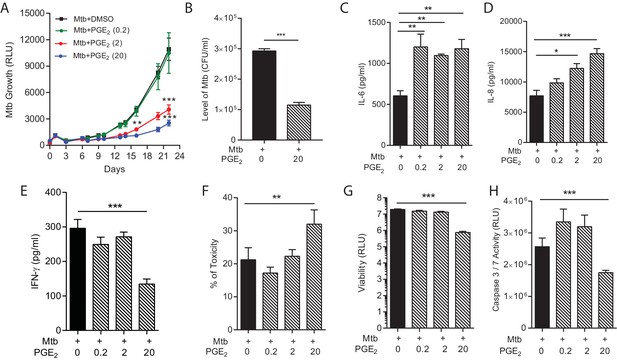
PGE2 augmentation limits bacterial growth but increases pro-inflammatory cytokine secretion and cellular toxicity.
(A) Addition of exogenous PGE2 suppresses Mtb growth in microspheres in a dose-dependent manner. Mtb-infected PBMCs (black line), 0.2 µg/ml PGE2 (green line), 2 µg/ml PGE2 (red line), 20 µg/ml PGE2 (blue line). (B) Colony counts of microspheres decapsulated at day 24 and then plated on Middlebrook 7H11 agar correlate with luminescence. (C, D and E) PGE2 increases secretion of IL-6 and IL-8, but significantly decreases IFN-γ secretion, from Mtb-infected microspheres. (F) Cellular toxicity is increased in PGE2 treated microspheres at day 3, analyzed by LDH release, and (G) total cell viability was reduced at day 7, analyzed by CytoTox-Glo assay. (H) PGE2 reduces caspase 3/7 activity at day 7. *p<0.05, **p<0.01, ***p<0.001.
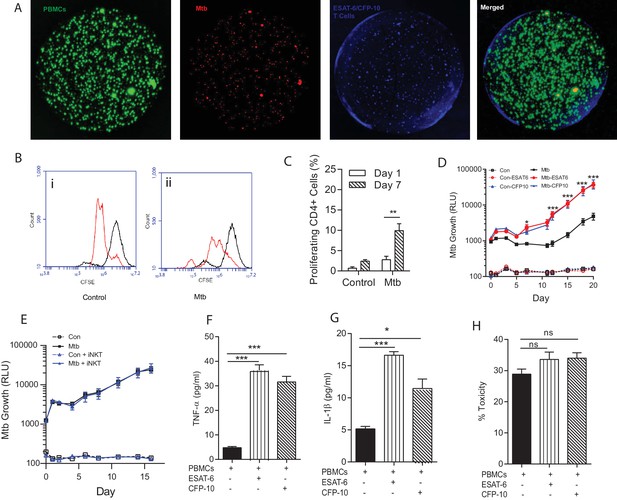
Immunoaugmentation with Mtb-specific T cells increases Mtb growth.
(A) Microspheres imaged after 4 days show early granuloma formation. (i) PBMCs labelled with CellTrace CFSE (green), (ii) Mtb expressing mCherry (red), (iii) autologous ESAT-6 specific T cells labelled with CellTracker Blue, (iv) Merged image shows granulomas containing Mtb, PBMCs and augmented T cells (yellow). (B) Cellular proliferation is increased in infected microspheres with immunoaugmented autologous T cells, analysed by CFSE staining. Day 1, black line; Day 7, red line; (i) Uninfected, (ii) Mtb-infected. (C) Quantitative analysis of the proliferative capacity of ESAT-6 augumented PBMCs at Day one and Day 7. The bars show percentage of proliferating CD4 cells after gating on CD3+CD4+ lymphocytes. Differences between Day 1 and 7 were assessed for three experiments by t-test. (D) Addition of either ESAT-6 responsive T cells (red) or CFP-10 responsive T cells (blue) increases Mtb growth compared to infected PBMCs without supplemented T cells (black). Open symbols, uninfected microspheres. (E) Supplementation with an iNKT autologous T cell line (blue triangle) did not significantly affect Mtb growth compared to infected PBMCs alone (black square). (F, G) Secretion of TNF-α and IL-1β is increased in immunoaugmented microspheres at day 7. (H) Immunoaugmentation did not significantly modulate cell toxicity in infected microspheres at day three analysed by LDH release.
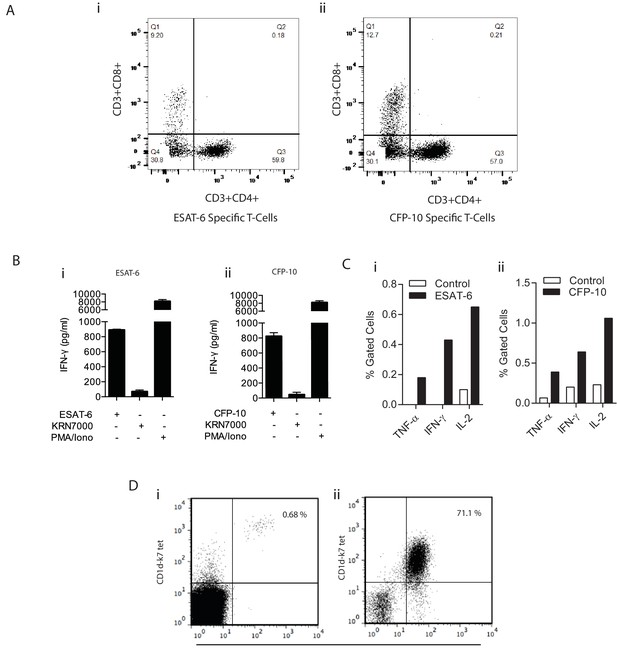
Confirmation of specificity of in vitro expanded T cells.
(A) Dot plot shows the percentage of T cells that are either CD4+/CD8+, CD4+/CD8-, CD4-/CD8+, CD4-/CD8- from ESAT-6 (i) and CFP-10 (ii) specific T cell lines. (B) PBMC were cultured with ESAT-6 (i) and CFP-10 (ii) peptides and after two weeks’ culture, T cell specificity was confirmed by IFN-γ release after exposure to their respective antigen. The iNKT antigen KRN7000 was used as a negative control and PMA/Ionomycin as a polyclonal T cell mitogen. Data is representative of three donors with each co-culture performed in triplicate. (C) Percentage of ESAT-6 (i) and CFP-10 (ii) specific T cells measured by intracellular cytokine staining. Monocyte derived dendritic cells (moDCs) were pulsed with ESAT-6 or CFP-10 peptide for 12 hr prior to the addition of T cells. After 8 hr of stimulation with their corresponding antigen, each line was stained for intracellular cytokines and cells were acquired on a FACSaria. (D) Dot plots representing in vitro antigen induced iNKT expansion in one donor. Live CD3+ CD1d-K7 tet+ Vα24 + iNKT cells were stained (i) ex vivo and (ii) two weeks post stimulation with KRN7000. Plots show % of iNKT cells ex vivo (0.68%) and after in vitro culture (71.1%) confirming their expansion.
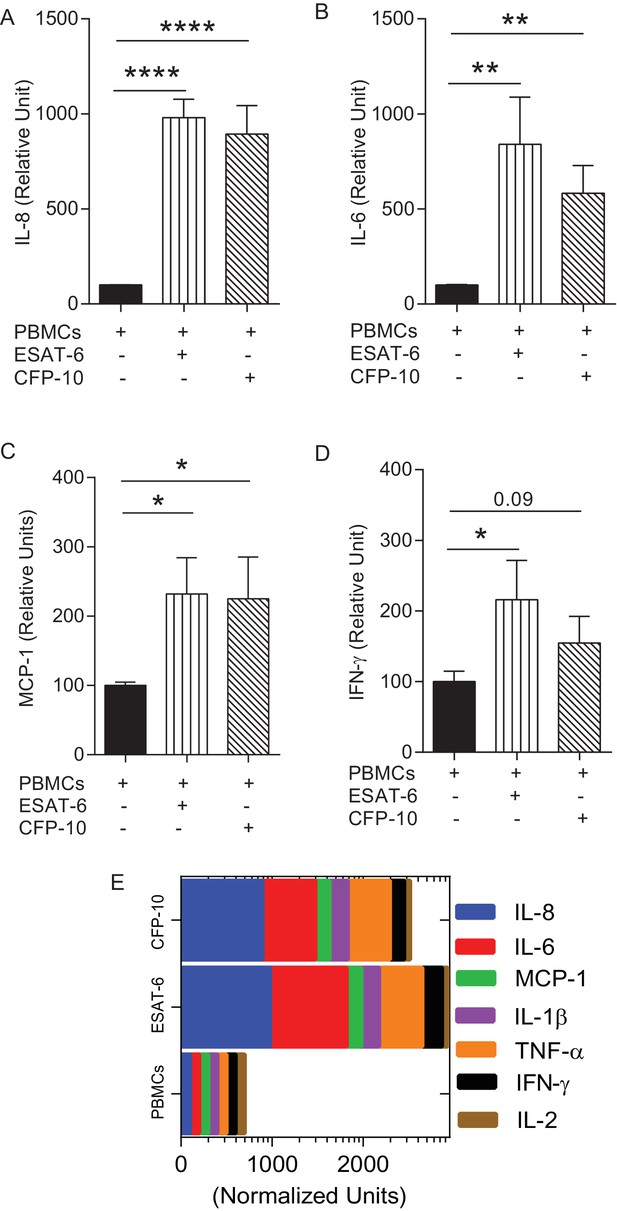
Augmentation of PBMCs with ESAT-6/CFP-10 specific T cell lines in microspheres causes differential secretion of cytokines after Mtb infection compared to PBMCs alone.
(A–D) Cytokine secretion in CFP-10 and ESAT-6 specific T cell supplemented microspheres was normalized to the secretion from infected PBMCs within PBMC-only microspheres and results are expressed as relative units. (E) Graphic representation of the cytokine profile (n = 3). Data show mean ± SEM of three independent experiments done in triplicate. *p<0.05, **p<0.01, ***p<0.001, p<0.0001.
Videos
Generation of microspheres.
During the bio-electrospray process, a Phantom v7 high-speed camera, capable of capturing 150000 fps in conjunction with a long-distance microscope lens, was triggered simultaneously with a fibre optic lighting system. Relative video speed 0.15 s.





