The Chd1 chromatin remodeler shifts hexasomes unidirectionally
Figures
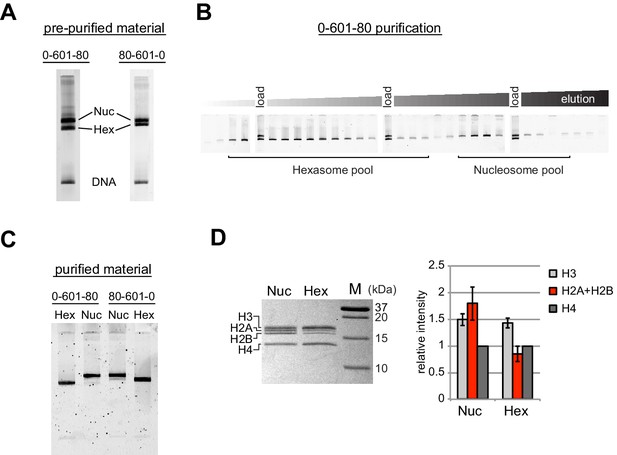
Separation of nucleosomes and hexasomes made with the Widom 601 sequence.
(A) Hexasomes but not nucleosomes migrate differently by native PAGE when flanking DNA is on the left or right of the 601 sequence. These two gels, poured from the same solution, are representative of 0-601-80 and 80-601-0 reconstitutions made using histone octamer. (B) Separation of hexasomes from nucleosomes. Shown is a representative purification over a 7% native acrylamide column using a Prep Cell apparatus. The elution fractions were analyzed by native PAGE. (C) Purified nucleosome and hexasome pools, analyzed by native PAGE. (D) As shown by SDS-PAGE, the hexasome species lack one H2A/H2B dimer. The bar graph is a quantification of gel band intensities from three different nucleosome/hexasome purifications. All histone bands were normalized to histone H4. The H2A and H2B bands often migrate close together, and therefore the relative intensities of H2A/H2B bands are shown summed together. Within each nucleosome/hexasome pair, the intensity of H2A/H2B in hexasomes was 47 ± 6% of that of nucleosomes.
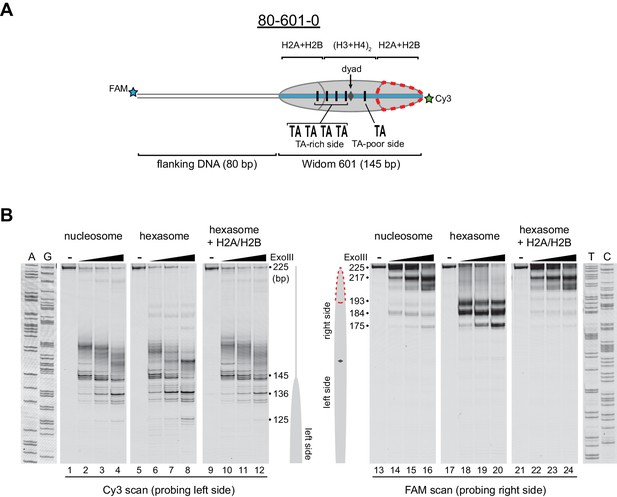
Oriented hexasomes can be generated using the Widom 601 sequence.
(A) Schematic representation of the 80-601-0 nucleosome and hexasome. With limiting amounts of H2A/H2B dimer, the side of the hexasome lacking the dimer (red dotted outline) corresponds with the TA-poor side of the Widom 601 sequence. (B) ExoIII analysis of 80-601-0 demonstrates that hexasomes specifically retain the H2A/H2B dimer on the TA-rich side of the 601 sequence. Purified nucleosomes, hexasomes, and hexasomes plus H2A/H2B were incubated with 0, 10, 40, and 160 units of ExoIII and resolved on urea denaturing gels. Lanes 9–12 and 21–24 show addition of 200 nM H2A/H2B dimer to 100 nM hexasomes, which recovered nucleosome digestion patterns. The size (bp) of major products are indicated. These gels are representative of two independent experiments. Dideoxy sequencing lanes (A, G, T, C) were run on the same gel as the samples shown. See also Figure 2—figure supplements 1 and 2.
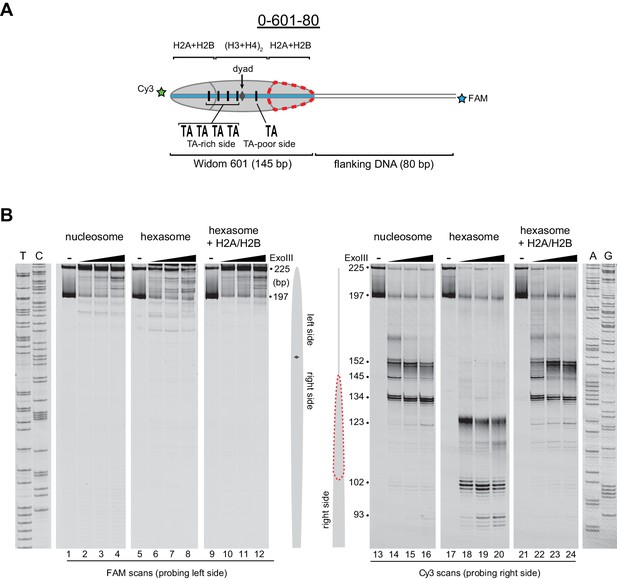
Flanking DNA does not influence the orientation of the hexasome.
(A) Schematic representation of the 0-601-80 nucleosome and hexasome. As shown for 80-601-0 in Figure 2, limiting amounts of H2A/H2B favors a hexasome that lacks the dimer on the TA-poor side of the Widom 601 sequence. (B) ExoIII digestions performed as in Figure 2 except using purified 0-601-80 nucleosomes and hexasomes, where flanking DNA was on the opposite (right) side of the 601. Comparing digestion patterns of 0-601-80 and 80-601-0 hexasomes revealed that hexasomes lacked H2A/H2B on the TA-poor side of the 601 regardless of flanking DNA. Addition of two-fold molar excess H2A/H2B to hexasomes restored nucleosome digestion patterns (lanes 21–24). ExoIII digestions of 0-601-80 and 80-601-0 hexasomes and nucleosomes were performed in parallel, run on the same gel and are each representative of two independent experiments.
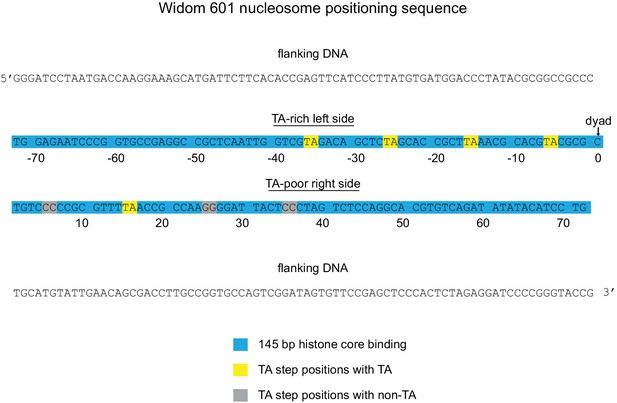
Sequence and orientation of the Widom 601 sequence used in this study.
Shown is the core 145 bp histone binding region of the 601 (blue) with flanking DNA. Numbering gives the distances from the dyad (zero).
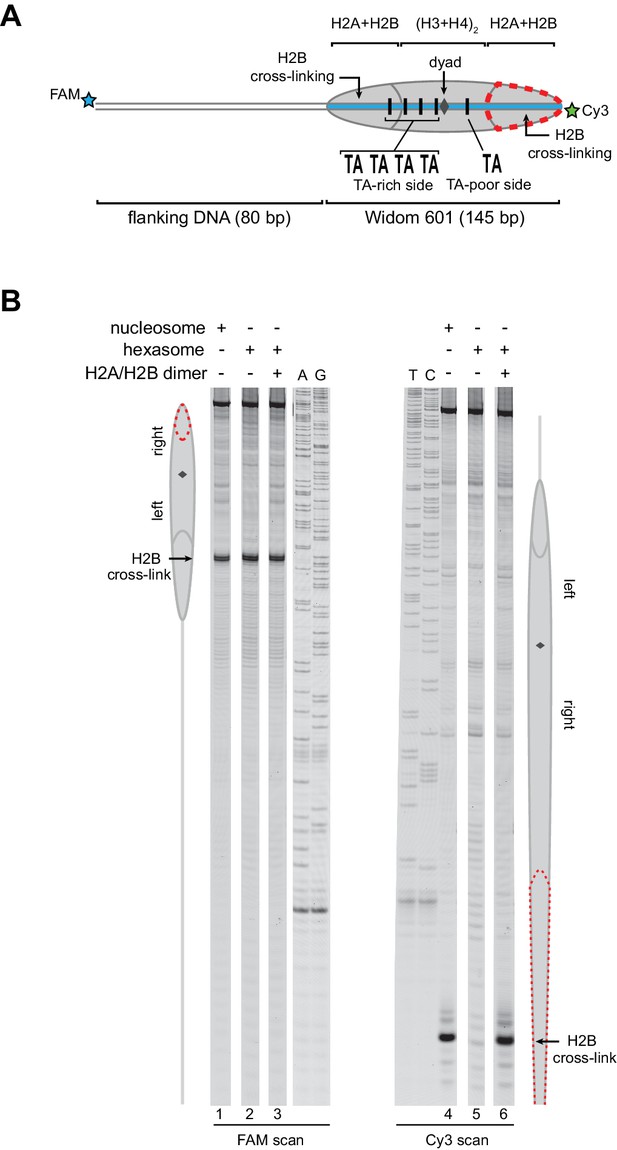
Addition of H2A/H2B dimer to hexasomes produces canonical nucleosomes.
(A) Schematic representation of the 80-601-0 nucleosome and hexasome, highlighting the locations where H2B-S53C cross-links to DNA. Due to the absence of one H2A/H2B dimer, H2B cross-linking with hexasomes is limited to the TA-rich side of the Widom 601. (B) Histone mapping demonstrates that canonical nucleosomes can be generated by addition of H2A/H2B dimer to hexasomes. For reactions containing hexasomes plus H2A/H2B, the hexasomes (10 nM) were incubated for 2–3 min with H2A/H2B (20 nM) prior to labeling with APB. Nucleosome and hexasome alone were subjected to the same brief incubation. Following UV cross-linking and DNA extraction, the DNA was cleaved at the crosslinking site and the products separated on a denaturing gel alongside a sequencing ladder to determine the cross-linking position. Results are representative of three or more independent experiments. See also Figure 3—figure supplement 1.
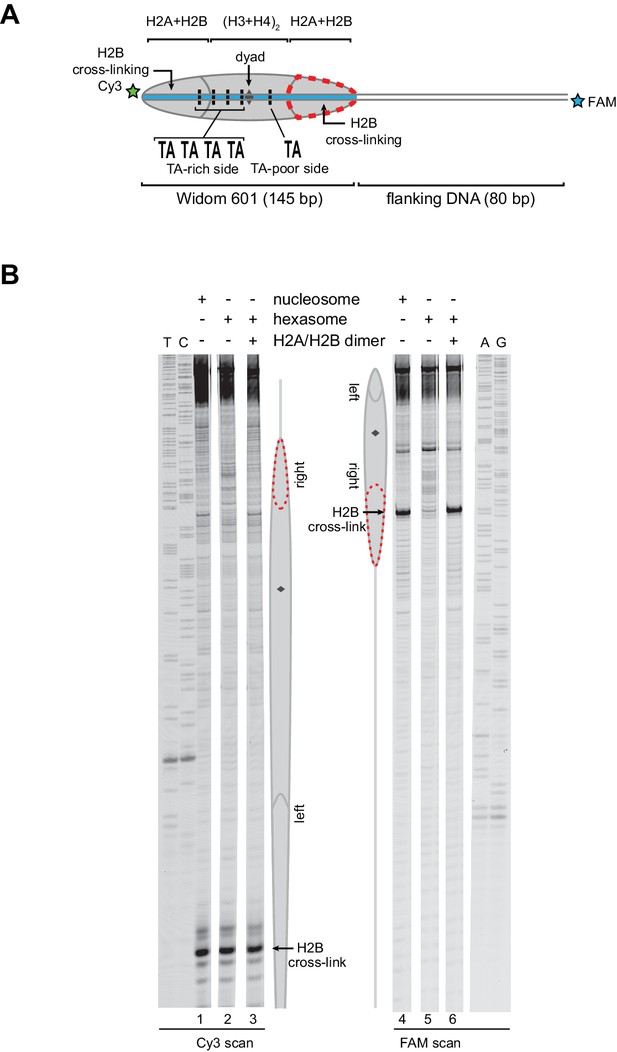
Addition of H2A/H2B dimer to hexasomes produces canonical nucleosomes, regardless of flanking DNA location.
(A) Schematic representation of the 0-601-80 nucleosome and hexasome, highlighting the location where the hexasome lacks one H2A/H2B dimer. (B) Histone mapping experiments using 0-601-80 nucleosomes, hexasomes, and hexasomes plus excess H2A/H2B dimer. Reactions were carried out as described for Figure 3. Sequencing ladder markers (T, C, A, G) were run in the same gel as the samples. Representative of three or more experiments.
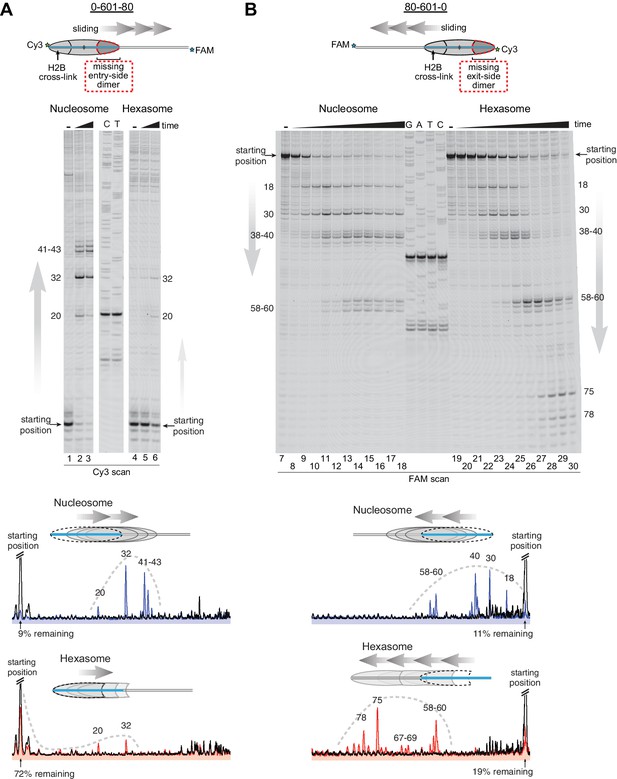
Chd1 requires entry side H2A/H2B for robustly repositioning hexasomes.
(A) Nucleosome and hexasome sliding reactions, visualized through histone mapping. For 150 nM hexasome and nucleosome 0-601-80 constructs, sliding reactions were monitored after incubation with 50 nM Chd1 and 2 mM ATP for 0, 1, and 64 min. Reactions were quenched at time points with the addition of EDTA and competitor DNA. Comparison of intensity profiles for histone mapping reactions are shown below. Samples before ATP addition (0 min) are black, nucleosome sliding reactions after 64 min are blue, and hexasome sliding reactions after 64 min are red. (B) Sliding reactions and intensity profiles carried out with 80-601-0 constructs as described for (A). Time points were 0, 0.25, 0.5, 1, 2, 4, 8, 16, 32, and 64 min. Sliding experiments for 0-601-80 and 80-601-0 were each performed six or more times with similar results. See also Figure 4—figure supplement 1.
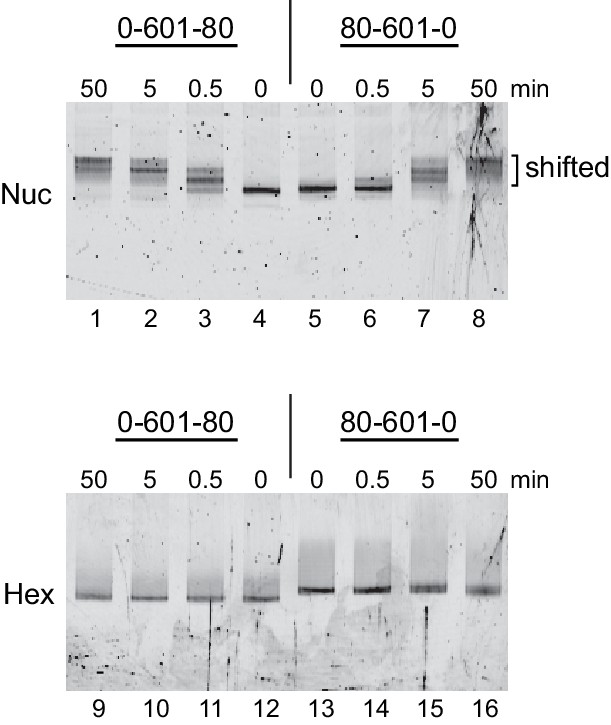
Chd1 remodeling dramatically alters nucleosome but not hexasome mobility as assessed by native PAGE.
Purified nucleosomes (top) or hexasomes (bottom), each 150 nM, were incubated with yeast Chd1 (50 nM) and 2.5 mM ATP in sliding buffer for 0, 0.5, 5, and 50 min. After quenching with EDTA and competitor DNA, reaction products were separated by native PAGE. Shown is a representative of four independent experiments.
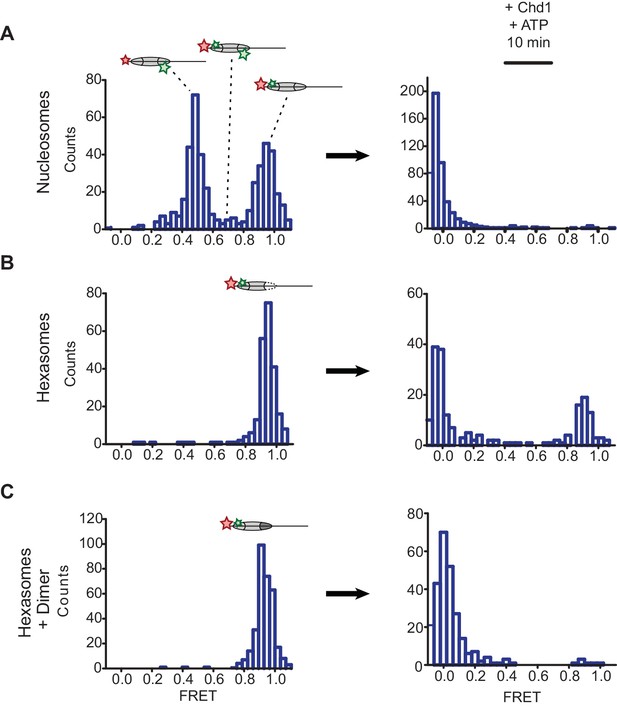
Oriented hexasomes allow targeted placement of modified H2A/H2B dimers on the nucleosome.
(A) Analysis of dual labeled 3-601-80 nucleosomes (H2A T120C-Cy3 and DNA-Cy5) by single-molecule FRET (smFRET) reveals multiple species prior to nucleosome sliding by Chd1. Nucleosomes were surface-immobilized by biotin on the 80 bp flanking DNA. Infusion of 300 nM Chd1 and ATP initiated remodeling. (B) Oriented 3-601-80 hexasomes (H2A-Cy3 and DNA-Cy5) uniformly show one dye pair that yields high FRET. Right panel shows relatively poor mobilization of hexasomes by Chd1. (C) Incubation of a two-fold molar excess of unlabeled H2A/H2B dimer with the labeled 3-601-80 hexasomes yielded asymmetric nucleosomes, only possessing the high FRET dye pair. After remodeling with Chd1 and ATP, the FRET population was similar to nucleosome.
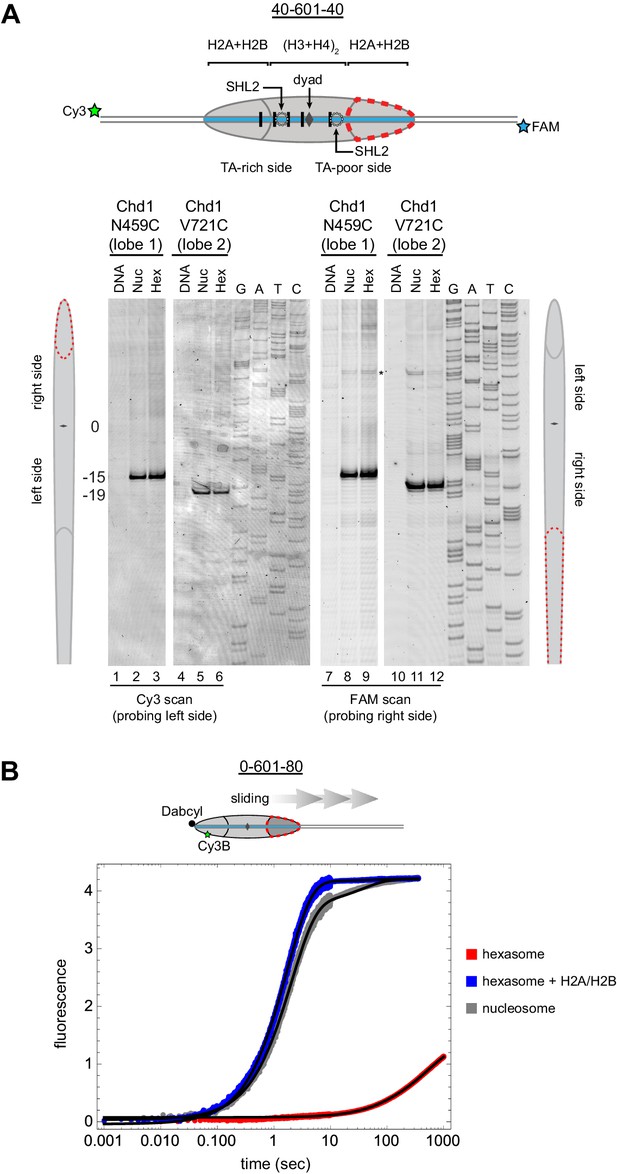
Chd1 requires entry side H2A/H2B for sliding but not binding.
(A) Chd1 cross-linking to 40-601-40 nucleosomes and hexasomes. Single cysteine variants on lobe 1 (N459C) and lobe 2 (V721C) of the Chd1 ATPase cross-linked to DNA 15 and 19 bp from the dyad, respectively, on both sides of nucleosomes and hexasomes. Chd1 was labeled with APB and incubated in a 2:1 ratio with DNA, nucleosomes, or hexasomes in the presence of ADP∙BeF3. After UV irradiation, DNA extraction and cleavage, cross-linking sites were determined by separating DNA fragments on a denaturing gel alongside a sequencing ladder. The gel shown is representative of two independent experiments. Asterisk marks cross-linking from a non-cysteine residue (Nodelman et al., 2017). (B) Stopped flow sliding reactions comparing the activity of Chd1 on 10 nM 0-601-80 nucleosomes, hexasomes, and hexasomes plus 12 nM dimer. Nucleosomes and hexasomes were labeled with Cy3B on H2A-T120C and with Dabcyl quencher on the zero end of the DNA. Reactions were initiated with the addition of saturating (600 nM) Chd1 and 1 mM ATP. Black lines represent double exponential fits of the data. Each progress curve is an average of 3–6 replicate injections, and representative of two independent experiments. See also Figure 6—figure supplement 1.
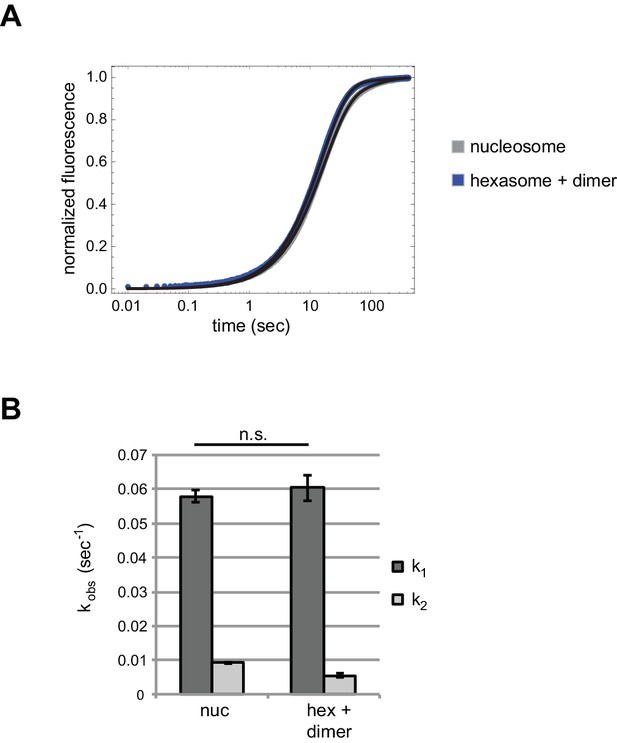
Chd1 repositions nucleosome and hexasome plus dimer at similar rates with limiting ATP.
(A) Progress curves from stopped flow experiments performed with 400 nM Chd1 on 10 nM 0-601-80 nucleosomes (gray) or 10 nM hexasomes plus 12 nM dimer (blue) in the presence of 25 μM ATP. Reactions were monitored by Cy3B-Dabcyl SQOF. Black lines depict double exponential fits of the data. Each progress curve is the average of 3–6 technical replicates and is representative of three independent experiments. (B) Comparison of sliding rates. The bar chart shows observed fast and slow rates (k1, k2) from the fits as means ± standard deviations from three independent experiments. n.s. = not significant.
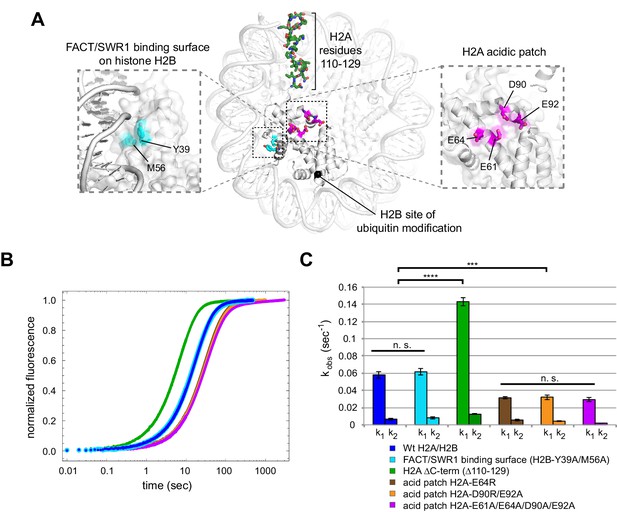
Disruptions in the nucleosome acid patch only moderately decrease sliding by Chd1.
(A) Overview of disruptions introduced on H2A/H2B. Nucleosome crystal structure shown is PDB code 1KX5 (Davey et al., 2002). (B) Stopped flow sliding reactions using asymmetric nucleosomes containing H2A or H2B disruptions on the entry side H2A or H2B. Asymmetric nucleosomes were generated by incubating 10 nM 0-601-80 hexasomes with 12 nM H2A/H2B containing one of the following sequence variants: Wt (blue); FACT/SWR1 binding surface disruption (H2B-Y39A/M56A) (cyan); H2A C-terminal tail truncation (∆110–129) (green); acid patch single mutant (H2A-E64R) (brown); acid patch double mutant (H2A-D90R/E92A) (orange); acid patch quadruple mutant (H2A-E61A/E64A/D90A/E92A) (magenta). Reactions were performed with 400 nM Chd1 and 25 μM ATP and followed by Cy3B-Dabcyl SQOF. Each progress curve is an average of 3–6 technical replicates. (C) Summary of observed rates (k1, k2) obtained from double exponential fits to stopped flow data as shown in (A). In every case the observed fast rate (k1) contributes >90% of the amplitude of the progress curve. Error bars represent standard deviation from three (six for Wt) independent experiments. Statistics compare k1 rates for indicated constructs: *** p-value <0.00001; **** p-value <0.0000001; n.s., not significant. See also Figure 7—figure supplements 1 and 2.
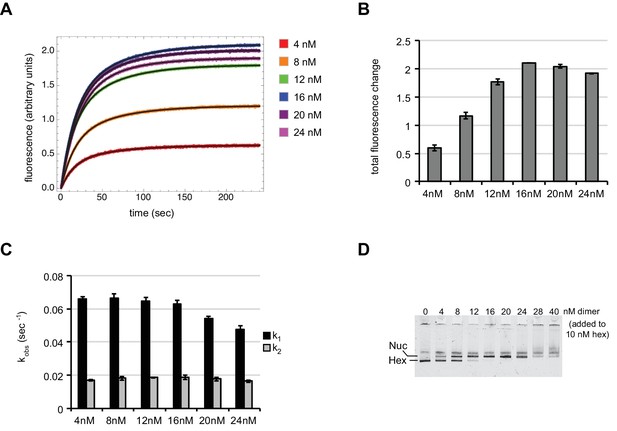
With subsaturating H2A/H2B dimer addition, rates of nucleosome sliding by Chd1 are not sensitive to nucleosome:hexasome ratios.
(A) Stopped flow nucleosome sliding reactions monitored by Cy3-Cy3 fluorescence quenching . The legend indicates the amount of H2A/H2B dimer in each reaction, which contained 10 nM 0-601-80 hexasome, 50 nM Chd1, and 25 μM ATP. Each trace is an average of 4 or more injections from the same stopped flow experiment. The black curves represent double exponential fits to the data. (B) Graph of overall intensity changes at each H2A/H2B dimer concentration added to hexasomes, with higher intensity reflecting a greater proportion of nucleosomes that were shifted. Error bars indicate the range from two independent sets of experiments. (C) Graph of sliding rates for stopped flow H2A/H2B dimer addition. The observed rates (given as k1 and k2) were determined from double exponential fits to the data. Error bars indicate the range from two independent sets of experiments. (D) Native PAGE visualization of nucleosomes generated by addition of H2A/H2B dimer to hexasomes. Shown is a representative of ten similar titrations performed using wild-type or modified H2A/H2B dimers.
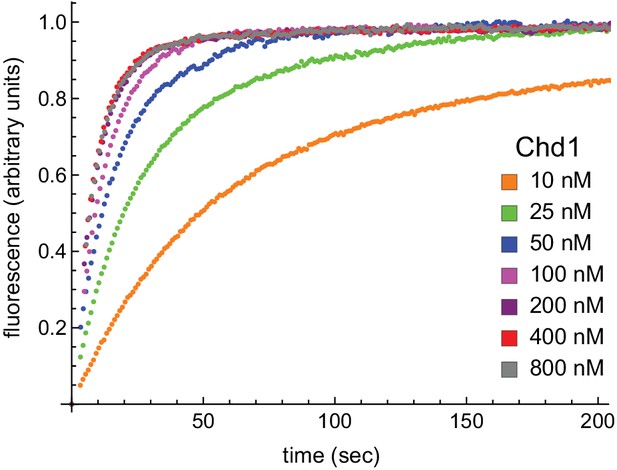
With limiting ATP, remodeling saturates at 400 nM Chd1.
Nucleosomes formed by adding 12 nM dimer to 10 nM 0-601-80 hexasomes were titrated with 10, 25, 50, 100, 200, 400, and 800 nM Chd1 and 25 μM ATP. Reactions were monitored by Cy3-Cy3 SQOF via fluorometer, and show that remodeling plateaued at 400 nM Chd1. The progress curves shown are representative of two independent Chd1 titrations using unmodified H2B (Wt-Wt). Similar results were observed for duplicate titrations using Ub-Wt, Wt-Ub, and Ub-Ub nucleosomes.
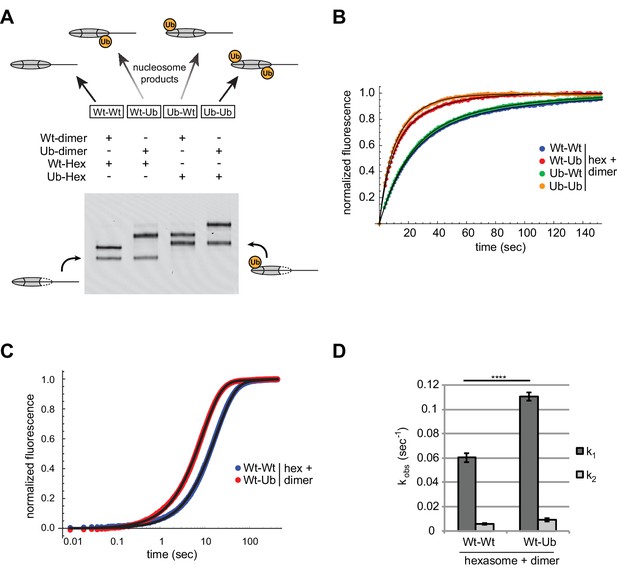
Entry-side H2B-Ubiquitin stimulates nucleosome sliding by Chd1.
(A) Generation of symmetric and asymmetric nucleosomes with site-specific placement of H2B-Ubiquitin. Nucleosomes were formed from subsaturating H2A/H2B dimer (12 nM) addition to 0-601-80 hexasomes (10 nM). Hexasomes and H2A/H2B dimer contained either unmodified (Wt) or ubiquitinated (Ub) H2B as indicated, and resulting nucleosome and hexasome species were visualized by native PAGE. Shown is a representative from six independent dimer addition experiments. (B) Comparison of remodeling reactions with subsaturating (25 nM) Chd1, using hexasomes (10 nM) and H2A/H2B dimers (12 nM) containing unmodified or Ub-conjugated H2B. Shown are progress curves for remodeling reactions monitored using a Cy3-Cy3 pair at 25 μM ATP. Black traces represent fits to the data. Progress curves are representative of two independent experiments. (C) Representative progress curves of nucleosome sliding reactions monitored by stopped flow using Cy3B-Dabcyl at 25 μM ATP and saturating (400 nM) Chd1. Each progress curve is an average of 3–6 technical replicates. Black traces represent fits to the data. (D) Comparison of observed sliding rates monitored with Cy3B-Dabcyl at 25 μM ATP and saturating Chd1 (400 nM). Error bars show standard deviations from three independent experiments. **** p-value <0.0001.
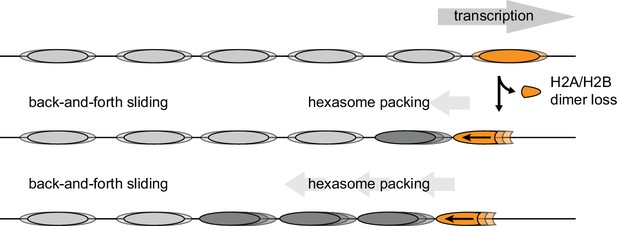
Model for nucleosome packing by oriented hexasomes.
As others have shown, transcription by Pol II through nucleosomes is facilitated by removal of the promoter-distal H2A/H2B dimer (Kulaeva et al., 2009). Our results indicate that Chd1 would slide a hexasome of this orientation upstream. We propose that one or more hexasomes would corral intervening nucleosomes toward the promoter. Alternately, if every transcribed nucleosome were briefly converted to a hexasome, unidirectionally sliding of each hexasome would maintain tight nucleosome packing.





