Structural basis for the hijacking of endosomal sorting nexin proteins by Chlamydia trachomatis
Figures
SNX5, SNX6 and SNX32 tare recruited to C. trachomatis inclusions.
(A) HeLa cells stably expressing the mCherry-Rab25 inclusion membrane marker (red) were infected with C. trachomatis serovar L2 (24 hr) and transfected with myc-tagged SNX expression constructs. The samples were fixed and immunolabeled with anti-myc (green) and anti-chlamydial HtrA antibodies (white) and counterstained with DAPI (blue). Similar experiments using GFP-tagged proteins are shown in Figure 1—figure supplement 1A.
SNX5, SNX32 and SNX1 are recruited to C. trachomatis inclusions and membrane tubules.
(A) Hela cells were transiently transfected with GFP-tagged SNX and mCherry-Rab25 proteins as indicated, and infected with C. trachomatis serovar L2. Cells were imaged by confocal fluorescence microscopy for GFP-tagged proteins (green), endogenous SNX1 (blue), mCherry-Rab25 (red) and DAPI-stained nuclear material (white). Both GFP-SNX5 and GFP-SNX32 are recruited to inclusion membranes, but the distantly related SNX-BAR protein SNX8 is not. The images are maximum projections. (B) An example of SNX1-decorated tubules (green) often observed emanating from inclusion membranes (mCherry-Rab25 in red; DAPI staining in blue). The image is a maximum projection.
Recruitment of SNX1, SNX2 and SNX5 to inclusions is not dependent on 3-phosphoinositides.
HeLa cells stably expressing mCherry-Rab25 were infected with C. trachomatis serovar L2 (MOI ~0.5) for 24 hr and imaged by immunofluorescence microscopy using antibodies to SNX1, SNX2 and SNX5. mCherry-Rab25 provides marker for the inclusion membrane. The upper panels show control infections and lower panels show cells treated with wortmannin or Vps34-IN1 with concomitant loss of SNX association with endosomal compartments while inclusion localisation is unaffected. The images are maximum projections. Endosomal compartments are labeled with antibodies to endogenous Rab5 or Vps35 and Pearson’s correlation coefficients used to quantify loss of endosomal recruitment (100 cells per group; error bars, S.D). A movie showing the effect of wortmannin on GFP-SNX5 is shown in Video 1.
IncE from C. trachomatis binds the PX domains of SNX5, SNX6 and SNX32.
(A) Binding affinity between IncE peptide (residues 107–132) and SNX PX domains by ITC. Top panels show raw data and lower panels show normalised integrated data. See Table 1 for the calculated binding parameters. Truncation analyses of the IncE peptide by ITC are shown in Figure 3, Table 2. (B) Sequence alignment of human SNX1, SNX5, SNX6 and SNX32 PX domains. Conserved residues are indicated in red. Side-chains that directly contact IncE in the crystal structure are indicated with black circles. Mutations that block IncE binding are highlighted with red triangles, and mutations that do not affect binding indicated with green circles. Secondary structure elements derived from the SNX5 crystal structure are indicated above. (C) Sequence alignment of IncE from C. trachomatis and putative homologues from C. muridarum and C. suis. IncE side-chains that directly contact SNX5 in the crystal structure are indicated with black circles. Mutations that block SNX5 binding are highlighted with red triangles, and mutations that do not affect binding indicated with green circles. Predicted transmembrane regions are indicated and C-terminal IncE sequences that form β-strands in complex with SNX5 are shown.
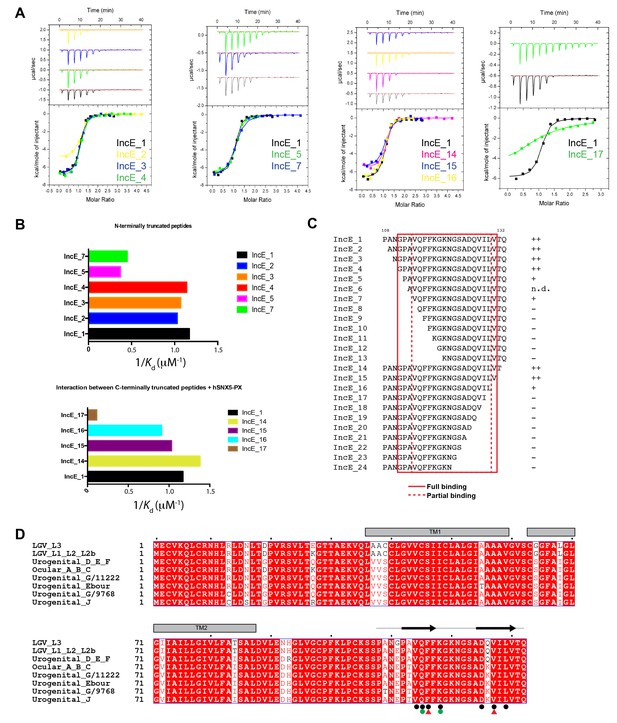
IncE residues 110–131 are sufficient for full recognition of the SNX5 PX domain.
(A) Representative ITC experiments for truncated IncE peptides. These experiments were conducted using a single batch of SNX5 PX domain to minimize batch-to-batch protein variation. (B) Plots of the affinity constants for selected peptides to highlight the progressive loss of binding with N and C-terminal truncations. (C) Sequences of the truncated IncE peptides are given, with a qualitative indication of binding strength relative to the IncE_1 peptide containing residues 107–132. Full binding is indicated by ‘++’ reduced binding by ‘+’ and lack of binding by ‘−‘. All sequence information and their Kd values are given in Table 2. When compared to the reference ITC experiment the binding affinity of peptides was unaffected when the first three N-terminal residues were removed (IncE_2, IncE_3 and IncE_4) and gradually became weaker until IncE_7, after which binding was abolished. Results from IncE_6 are inconclusive due to the difficulty in successfully dissolving the peptides in buffer (n.d.). C-terminal truncations showed that IncE_14 and IncE_15 had similar high binding affinities to the reference, while the binding of IncE_16 and IncE_17 became progressively weaker and peptides shorter than IncE_17 showed no binding. This data indicates that the minimal IncE binding sequence retaining full SNX5 binding is GPAVQFFKGKNGSADQVILVT, and a shorter fragment VQFFKGKNGSADQVIL can bind to SNX5, albeit with a slightly reduced affinity. (D) Sequence alignment of IncE from different C. trachomatis serovars.
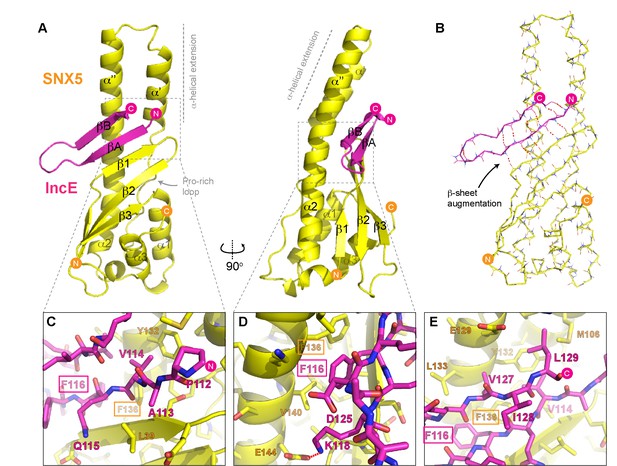
Structure of the SNX5 PX domain in complex with the IncE C-terminal domain.
(A) Crystal structure of the SNX5 PX domain (yellow) in complex with IncE residues 107–132 (magenta) shown in cartoon representation. (B) Backbone atoms of the SNX5 and IncE proteins are shown to highlight the prominent β-sheet augmentation mediating the association between the two molecules. (C) Close up view of the SNX5-IncE interface highlighting specific contact areas at the N-terminus of the IncE peptide. (D) Close up of the SNX5-IncE interface highlighting specific contact areas at the hairpin loop of the IncE peptide shown at 90° to Figure 4C. (E). Close up of the SNX5-IncE interface highlighting contact areas at the C-terminus of the IncE peptide in approximately the same orientation as Figure 4C. Residues in SNX5 (Phe136) and IncE (Phe116) that are critical for binding based on mutagenesis are boxed.
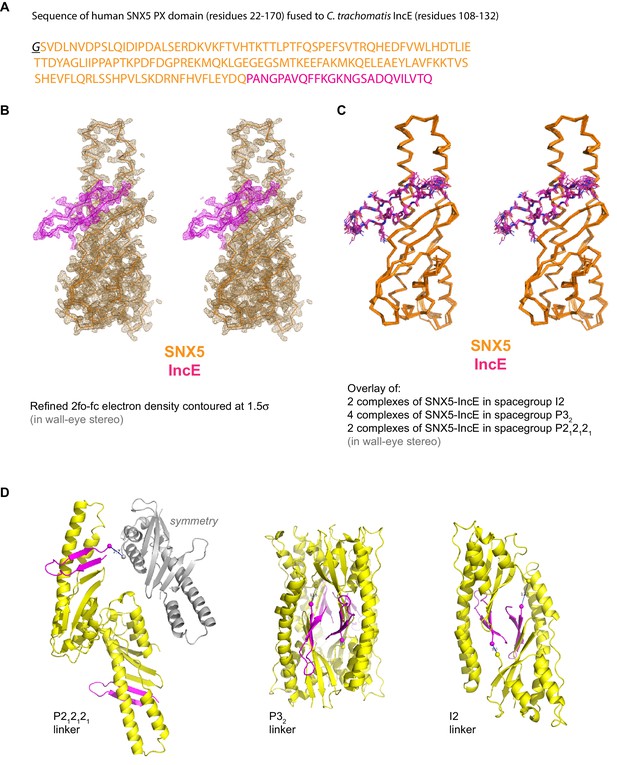
Supplementary images for SNX5-IncE crystal structures.
(A) Sequence of the SNX5 PX domain fusion protein with the IncE C-terminal peptide. (B) Refined 2fo-fc electron density contoured at 1.5σ for the SNX5-IncE structure in spacegroup P212121. (C) Overlay of each independent SNX5-IncE complex observed in the three crystal forms. (D) Ribbon structures indicating the locations of the linker regions in each crystal form. The C-terminal SNX5 residues and the N-terminal IncE residues are shown by spheres with distances indicated.
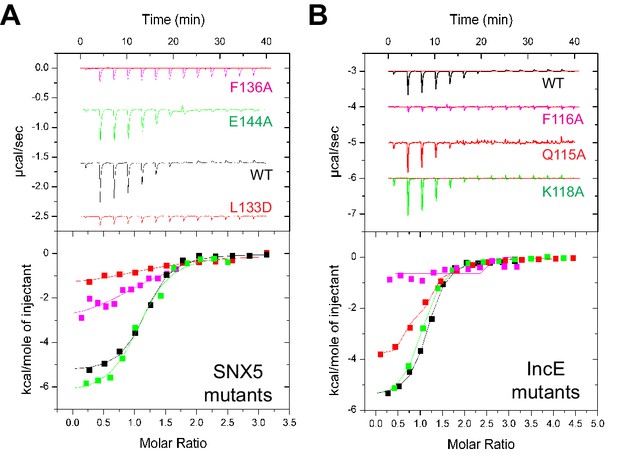
Mutations in the SNX5 and IncE proteins prevent complex formation in vitro.
(A) ITC experiments testing the effect of SNX5 mutations on IncE binding. Both L133D and F136A mutations prevented IncE binding, but the A144A mutation had little effect. (B) ITC experiments testing the effect of IncE mutations on SNX5 binding. Both F116A and V127D blocked SNX5 interaction, while Q115A had a partial effect and K118A had no effect on association.
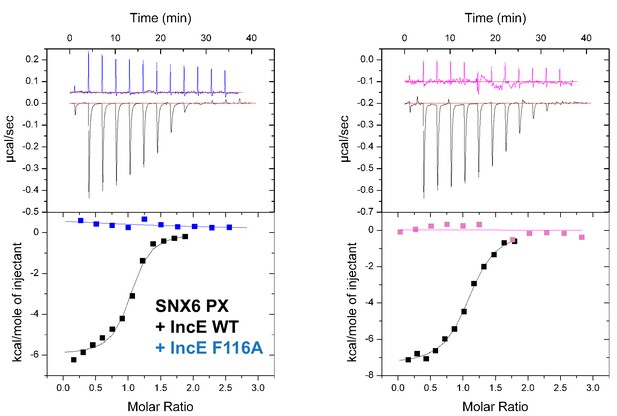
SNX6 and SNX32 PX domains bind IncE at the same site as SNX5.
ITC experiments testing the effect of IncE peptide mutations on binding to SNX6 and SNX32 PX domains. The IncE F116A mutation blocks interaction with both PX domains similarly to SNX5 (Figure 5B).
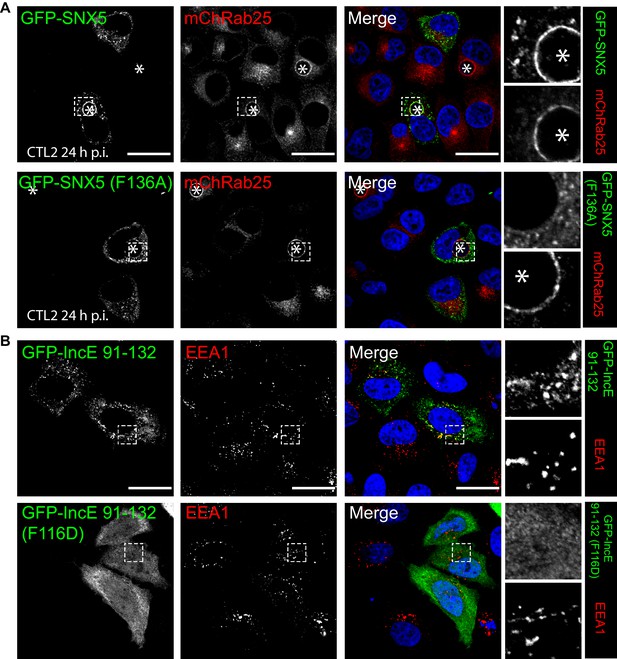
Mutations in the SNX5 and IncE proteins prevents their interaction in cells.
(A) Single amino-acid mutation in the PX domain of the SNX5 (F136A) abolishes recruitment to the chlamydial inclusion. HeLa cells stably expressing mCherry-Rab25 (red) were transfected transiently with GFP-SNX5 or GFP-SNX5 (F136A) (green) and infected with Chlamydia trachomatis L2 for 18–24 hr. The cells were fixed and the nucleic materials were counter-stained with DAPI (blue). (B) HeLa cells were transfected transiently with GFP-IncE(91-132) or GFP-IncE(91-132)(F116D) (green) and co-labelled for the early endosomal marker EEA1 (red). Mutation in the SNX5 binding IncE peptide (F116D) abolishes recruitment to endosomal structures. *Represents the inclusion. Scale bar 20 μm.
GFP-IncE C-terminal domain is localised to endosomes but not inclusions.
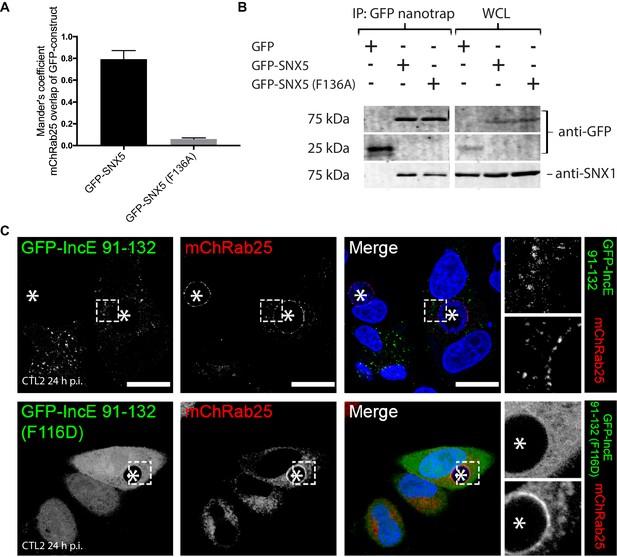
(A) Quantitation of the degree of overlap between the GFP-SNX5 constructs and the mCherry-Rab25 inclusion membrane marker from Figure 6A. Mander’s correlation coefficient of mChRab25 signal over GFP-SNX5 signals (10 cells per group; error bars, S.D). (B) Co-immunoprecipitation of GFP-SNX5 from HeLa cells shows that both the wild-type and mutant protein (F136A) interact equally with endogenous SNX1. This indicates that both proteins are correctly folded and otherwise functional. (C) HeLa cells stably expressing mCherry-Rab25 (red) were transfected transiently with GFP-IncE(91-132) or GFP-IncE(91-132)(F116D) (green) and infected with C. trachomatis L2 for 18–24 hr. Mutation in the SNX5 binding IncE peptide (F116D) abolishes recruitment to endosomal structures. Neither construct is recruited to inclusions, which is consistent with the lack of transmembrane regions. *Represents the inclusion. Scale bar 20 μm.

Conformational changes in SNX5 and a model for SNX-BAR recruitment to inclusion membranes.
(A) Comparison of the SNX5-IncE complex (yellow-magenta) with the previously reported apo- SNX5 PX domain crystal structure (blue) (PDB ID 3HPB)(Koharudin et al., 2009). The α-helical extension undergoes a significant displacement in the bound state. The enlarged panels to the right show several close-up views of the binding pocket highlighting conformational changes that are required to accommodate IncE. (B) A model for the SNX5-SNX1 PX-BAR heterodimer and its interaction with IncE at the inclusion membrane. The PX-BAR structure was modeled in silico (see methods). The left panel shows cartoon representations of the structure, viewed from the side and from the membrane surface. Middle panels show the same structures in electrostatic surface representation (red, negative; blue positive). The right panels show close ups of the putative PtdIns3P-binding pocket in SNX1 and SNX5, with a PtdIns3P head-group (shown in spheres) docked by aligning the previous SNX9 crystal structure (Pylypenko et al., 2007). SNX1 has a canonical PtdIns3P pocket, while SNX5 lacks a clear site for lipid head-group binding. (C) Sequence conservation of SNX5-related proteins was calculated and plotted using CONSURF. The surface representation indicates exposed side-chains that are evolutionarily conserved in green. The IncE peptide binds to a highly conserved surface groove, while the putative phosphoinositide binding region (Koharudin et al., 2009) on the opposite face is neither highly conserved nor poised to allow docking. (D) Cartoon model depicting the recruitment of SNX5 and related proteins to the inclusion membrane. Heterodimers with SNX1 or SNX2 will be recruited via IncE in infected cells, and this recruitment will be in competition with the binding of SNX1 and SNX2 to PtdIns3P for normal endosomal association, as well as interactions with other proteins including retromer and unidentified molecules that potentially bind to the conserved groove of the SNX5 PX domain.
Videos
Movie showing that wortmannin disrupts SNX5 recruitment to endosomes but not the chlamydial inclusion.
HeLa cells stably expressing mCherry-Rab25 (red) were transfected transiently with GFP-SNX5 (green) and infected with Chlamydia trachomatis L2 for 24 hr. Time-lapse videomicroscopy was performed using an interval of 1 min on an inverted Nikon Ti-E deconvolution microscope with environmental control at 40 x magnification. 10 min into recording 100 nM wortmannin was added.
Animation highlighting the mechanism of interaction between SNX5 and IncE.
The SNX5 PX domain is shown in yellow ribbons and the IncE peptide is shown in magenta.
Tables
Thermodynamic parameters of IncE binding to SNX PX domains*.
| Sample cell | Titrant | Kd (µM) | △H (kcal/mol) | T△S (kcal/mol) | △G (kcal/mol) | N |
|---|---|---|---|---|---|---|
| SNX5 PX | IncE peptide† | 0.95 ± 0.07 | −6.9 ± 0.3 | −1.9 ± 0.05 | −8.2 ± 0.01 | 1.01 ± 0.01 |
| SNX6 PX | IncE peptide | 1.13 ± 0.08 | −5.0 ± 0.9 | −3.0 ± 1 | −8.0 ± 0.07 | 1.01 ± 0.08 |
| SNX32 PX | IncE peptide | 1.15 ± 0.07 | −6.9 ± 0.4 | −1.3 ± 0.8 | −8.2 ± 0.4 | 1.06 ± 0.005 |
| SNX1 PX | IncE peptide | No binding |
-
*Values are the mean from three experiments ±SEM.
-
b.†IncE synthetic peptide sequence PANGPAVQFFKGKNGSADQVILVTQ.
ITC data for SNX5 PX domain binding to truncated and mutated IncE peptides*.
| Protein | Peptide | Sequence | Kd (µM) | △H (kcal/mol) | T△S (kcal/mol) | △G (kcal/mol) | N |
|---|---|---|---|---|---|---|---|
| SNX5 PX | IncE_1 | PANGPAVQFFKGKNGSADQVILVTQ | 0.95 ± 0.07 | −6.9 ± 0.3 | −1.9 ± 0.05 | −8.2 ± 0.01 | 1.01 ± 0.01 |
| IncE_2 | ANGPAVQFFKGKNGSADQVILVTQ | 1 | −5.0 | −2.6 | −8.1 | 0.98 | |
| IncE_3 | NGPAVQFFKGKNGSADQVILVTQ | 0.93 | −6.7 | −1.4 | −8.1 | 1.03 | |
| IncE_4 | GPAVQFFKGKNGSADQVILVTQ | 0.87 | −6.8 | −1.2 | −8.2 | 1.03 | |
| IncE_5 | PAVQFFKGKNGSADQVILVTQ | 2 | −5.9 | −1.2 | −8.3 | 0.99 | |
| IncE_6 | AVQFFKGKNGSADQVILVTQ | / | / | / | / | / | |
| IncE_7 | VQFFKGKNGSADQVILVTQ | 2.2 | −6.9 | −1.1 | −7.7 | 0.99 | |
| IncE_8 | QFFKGKNGSADQVILVTQ | No binding | / | / | / | / | |
| IncE_9 | FFKGKNGSADQVILVTQ | No binding | / | / | / | / | |
| IncE_10 | FKGKNGSADQVILVTQ | No binding | / | / | / | / | |
| IncE_11 | KGKNGSADQVILVTQ | No binding | / | / | / | / | |
| IncE_12 | GKNGSADQVILVTQ | No binding | / | / | / | / | |
| IncE_13 | KNGSADQVILVTQ | No binding | / | / | / | / | |
| IncE_14 | PANGPAVQFFKGKNGSADQVILVT | 0.72 | −5.1 | −1.6 | −8.4 | 1 | |
| IncE_15 | PANGPAVQFFKGKNGSADQVILV | 0.97 | −6.5 | −1.3 | −8.2 | 0.98 | |
| IncE_16 | PANGPAVQFFKGKNGSADQVIL | 1.1 | −5.6 | −1.4 | −8.12 | 0.99 | |
| IncE_17 | PANGPAVQFFKGKNGSADQVI | 8.7 | −2.7 | −2.5 | −6.9 | 0.99 | |
| IncE_18 | PANGPAVQFFKGKNGSADQV | No binding | / | / | / | / | |
| IncE_19 | PANGPAVQFFKGKNGSADQ | No binding | / | / | / | / | |
| IncE_20 | PANGPAVQFFKGKNGSAD | No binding | / | / | / | / | |
| IncE_21 | PANGPAVQFFKGKNGSA | No binding | / | / | / | / | |
| IncE_22 | PANGPAVQFFKGKNGS | No binding | / | / | / | / | |
| IncE_23 | PANGPAVQFFKGKNG | No binding | / | / | / | / | |
| IncE_24 | PANGPAVQFFKGKN | No binding | / | / | / | / | |
| IncE Q115A | PANGPAVAFFKGKNGSADQVILVTQ | 6.3 | −5.3 | −1.6 | −6.9 | 0.90 | |
| IncE F116D | PANGPAVQAFKGKNGSADQVILVTQ | No binding | |||||
| IncE K118A | PANGPAVQFFAGKNGSADQVILVTQ | 2.8 | −6.0 | −1.5 | −7.5 | 0.91 | |
| IncE V127D | PANGPAVQFFKGKNGSADQDILVTQ | No binding | |||||
| SNX5 PX L133D | IncE_1 | PANGPAVQFFKGKNGSADQVILVTQ | No binding | ||||
| SNX5 PX F136A | IncE_1 | PANGPAVQFFKGKNGSADQVILVTQ | No binding | ||||
| SNX5 PX E144A | IncE_1 | PANGPAVQFFKGKNGSADQVILVTQ | 15 | −9.9 | −3.1 | −13 | 0.99 |
-
*Except for IncE_1 all other peptide-binding experiments were performed only once.
Summary of crystallographic structure determination statistics*.
| Crystal | SNX5 PX-IncE Form 1 | SNX5 PX-IncE Form 2 | SNX5 PX-IncE Form 3 |
|---|---|---|---|
| PDB ID | 5TGI | 5TGJ | 5TGH |
| Data collection | |||
| Wavelength (Å) | 0.95370 | 0.95370 | 0.95370 |
| Space group | P212121 | I2 | P32 |
| Cell dimensions | |||
| a, b, c (Å) | 60.7, 67.5, 88.2 | 58.4, 80.3, 94.6 | 100.6, 100.6, 71.7 |
| α, β, γ (°) | 90, 90, 90 | 90, 97.2, 90 | 90, 90, 120 |
| Resolution (Å) | 60.7–1.98 (2.03–1.98) | 31.9–2.6 (2.72–2.60) | 50.3–2.80 (2.95–2.80) |
| Rmerge | 0.104 (0.525) | 0.153 (0.659) | 0.101 (0.713) |
| Rmeas | 0.112 (0.572) | 0.18 (0.777) | 0.124 (0.873) |
| Rpim | 0.042 (0.225) | 0.096 (0.408) | 0.051 (0.363) |
| <I> / σI | 12.4 (3.4) | 39.6 (3.2) | 11.7 (2.3) |
| Total number reflections | 178868 (11000) | 46691 (5757) | 115149 (16861) |
| Total unique reflections | 26075 (1805) | 13432 (1632) | 20001 (2923) |
| Completeness (%) | 100 (100) | 99.9 (100.0) | 100 (100) |
| Multiplicity | 6.9 (6.1) | 3.5 (3.5) | 5.8 (5.8) |
| Half-set correlation (CC(1/2)) | 0.997 (0.868) | 0.986 (0.55) | 0.997 (0.683) |
| Refinement | |||
| Resolution (Å) | 45.1–1.98 (2.02–1.98) | 31.9–2.6 (2.69–2.60) | 41.2–2.8 (2.87–2.80) |
| No. reflections/No. Rfree | 26021/2000 | 13421/1342 (1208/134) | 19975/1972 (1301/144) |
| Rwork/Rfree | 0.192/0.214 (0.221/0.246) | 0.199/0.242 (0.276/0.332) | 0.236/0.254 (0.329/0.372) |
| No. atoms | |||
| Protein | 2579 | 2619 | 5189 |
| Solvent | 281 | 69 | 0 |
| Average B-factor (Å2) | 31.8 | 42.5 | 56.0 |
| R.m.s deviations | |||
| Bond lengths (Å) | 0.012 | 0.011 | 0.015 |
| Bond angles (°) | 1.27 | 1.15 | 1.27 |
-
*Highest resolution shell is shown in parentheses.





