Histone gene replacement reveals a post-transcriptional role for H3K36 in maintaining metazoan transcriptome fidelity
Figures
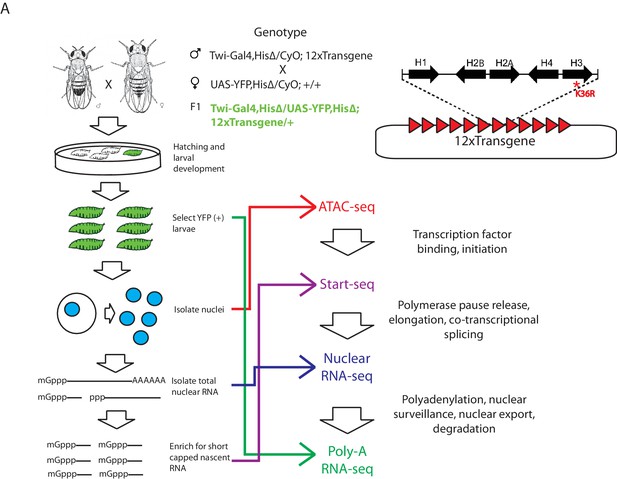
Strategy for interrogating the transcriptomic life cycle of H3K36R animals.
(A) Schematic of experimental high-throughput sequencing methods applied to H3K36R animals. Twelve tandem copies of the histone repeat unit were cloned into a custom BAC vector and site-specifically integrated into the D. melanogaster genome as described in McKay et al. (2015). Poly-A-selected RNA was sequenced from whole third instar larvae, ATAC-seq and rRNA-depleted nuclear RNA-seq were carried out from nuclei isolated from third instar larvae, and short, nascent, capped RNAs were selected from nuclei and subjected to ‘Start-seq’ (Henriques et al., 2013).
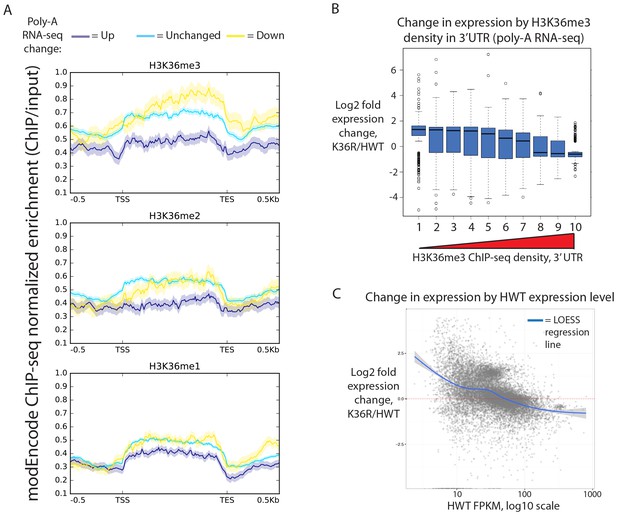
Transcriptome dystregulation in H3K36R mutants is correlated with H3K36me3 ChIP-seq.
(A) Metagene plot describing the density of H3K36me3 (top), H3K36me2 (middle), and H3K36me1 (bottom) ChIP-seq across genes that are upregulated (purple), unchanged (blue), or downregulated (yellow) in H3K36R mutants as compared with HWT controls. (B) Boxplot of differential expression of gene cohorts stratified by density of H3K36me3 signal in the 3’ UTR (1=lowest density decile, 10=highest decile). (C) MA plot with accompanying LOESS regression line plotting log2 fold change (y-axis) vs. HWT FPKM (x-axis) interpreted from poly-A RNA-seq data.
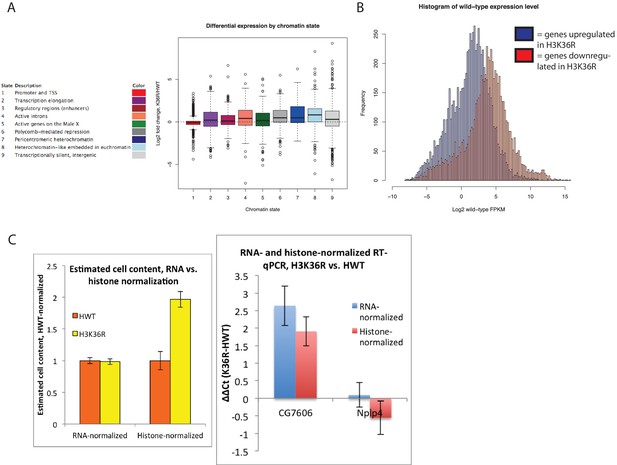
Gene expression changes in H3K36R mutants.
(A) Boxplot depicting log2 fold expression change for genes whose start sites reside in each of the nine different chromatin states defined in Kharchenko et al., 2011. (B) Histogram of mean FPKM for genes significantly upregulated (blue) or downregulated (red) in K36R animals (p<0.05). (C) Left: Boxplot of comparison of estimated cell content from five third instar larvae between HWT and K36R, based on normalizing by either quantitated RNA (left) or band intensity from histone H3 western blot signal (right). Given that K36R larvae are expected to have higher cell content based on histone normalization, total RNA per cell is likely overestimated in K36R, and therefore log2 fold change gene expression values are not expected to be overestimated based on bias from higher RNA per cell in K36R. Right: This interpretation is confirmed by lower log2 fold expression change in histone- vs. RNA-normalized RT-qPCR for select differentially expressed genes.
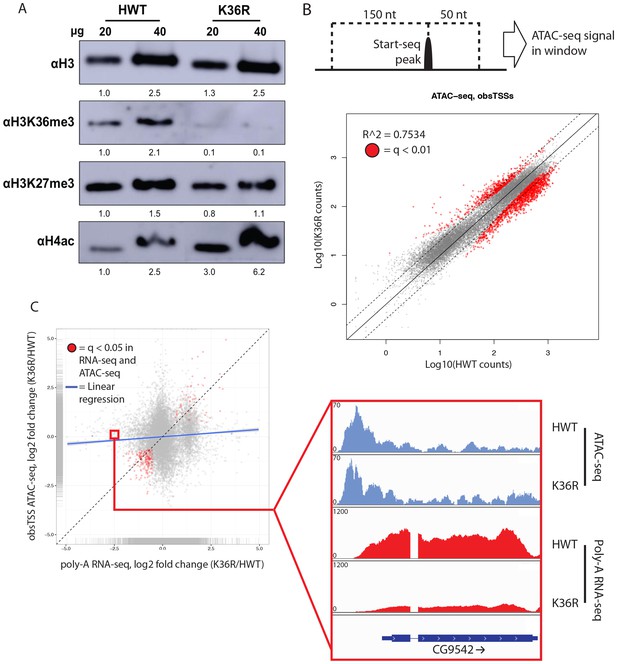
H4 acetylation enrichment in mutants does not result in open-chromatin-dependent changes in gene expression.
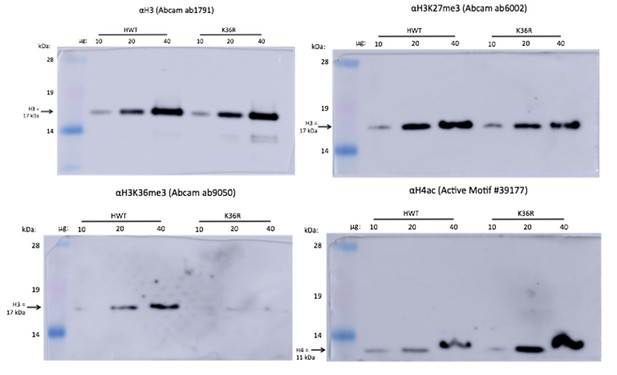
(A) Western blots measuring enrichment of histone H3, H3K36me3, H3K27me3, and pan H4 acetylation (H4ac) in H3K36R mutants and HWT controls. Signal relative to first lane is denoted below each band. (B) Scatterplot of ATAC-seq signal mapping in a 200 nt window (as denoted at top) around obsTSSs, with R2 value indicated. (C) Scatterplot of log2 fold change of poly-A RNA-seq (x-axis) vs. that of ATAC-seq (y-axis) signal in a window around the corresponding gene’s transcription start site (as identified by start-seq). Genes with codirectional, statistically significant changes in both RNA-seq and ATAC-seq are indicated in red. Example browser shot of a gene differentially expressed in mutants in the absence of changes in chromatin accessibility at its start site is shown at right.
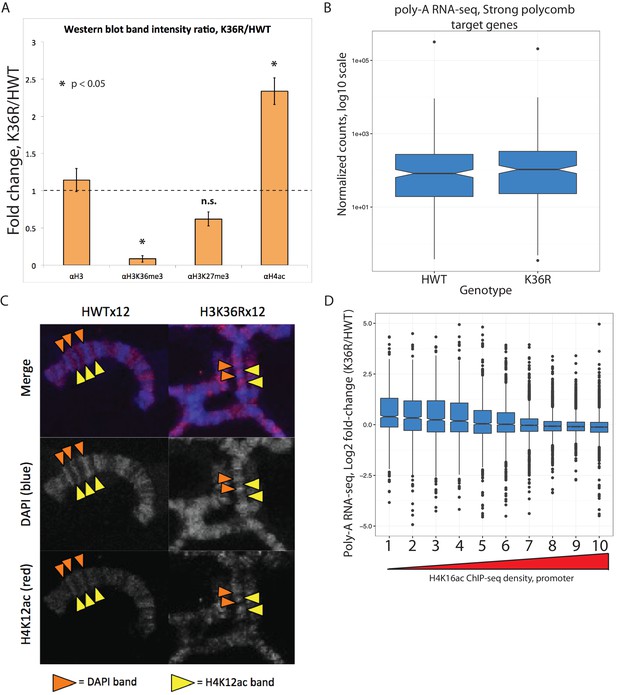
Histone crosstalk and gene expression changes in H3K36R animals.
(A) Barplot displaying the fold change in western blot signal intensity quantified from two biological replicates of whole larval nuclear lysate for the antibodies indicated. Asterisk indicates T-test p-value between H3 and indicated PTM is <0.05. (B) Boxplot of normalized poly-A RNA-seq counts mapping to genes that co-occur with a strong polycomb regulatory region (Schwartz et al., 2006) in HWT and K36R. (C) Polytene chromosome salivary gland squash and immunofluorescent stain for H4K12ac from HWT and K36R third instar larvae. H4K12ac-bright (yellow arrowheads), and transcriptionally silent DAPI bright (orange arrowheads) regions, are anticorrelated in both genotypes, suggesting H4K12ac accumulates to transcriptionally active regions in K36R mutants D) Boxplot of differential expression of gene cohorts stratified by density of H4K16ac ChIP-seq signal in a 400 nt window surrounding the annotated gene start site (1=lowest density decile, 10=highest decile).
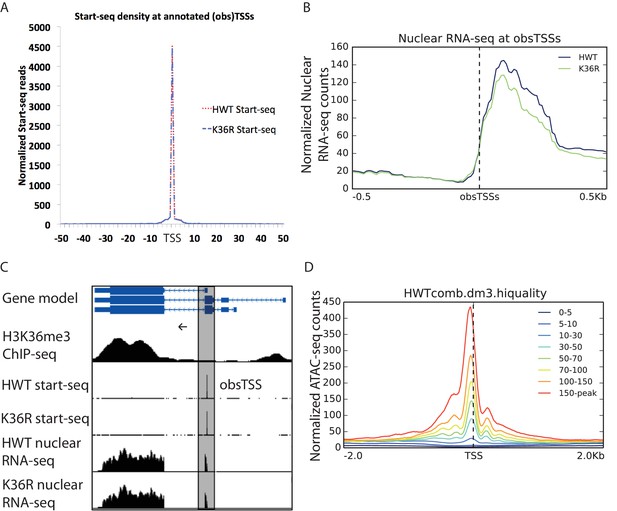
Metagene analysis of Start-seq reads at previously annotated (observed) transcription start sites, obsTSSs.
(A) Metaplot of Start-seq signal aligned in a 100 nt window around all annotated TSSs in the dm3 reference gene model. (B) Metaplot of HWT and K36R nuclear RNA-seq signal aligned in a 1 kb window around all obsTSSs identified in Start-seq data. (C) Representative browser shot of Start-seq signal pileup at annotated gene promoter (obsTSS). Direction of transcription denoted by arrow. (D) Metaplot of ATAC-seq signal in a 4 kb window surrounding obsTSSs identified by Start-seq. obsTSSs are binned by the average normalized signal across the window (denoted in the legend).
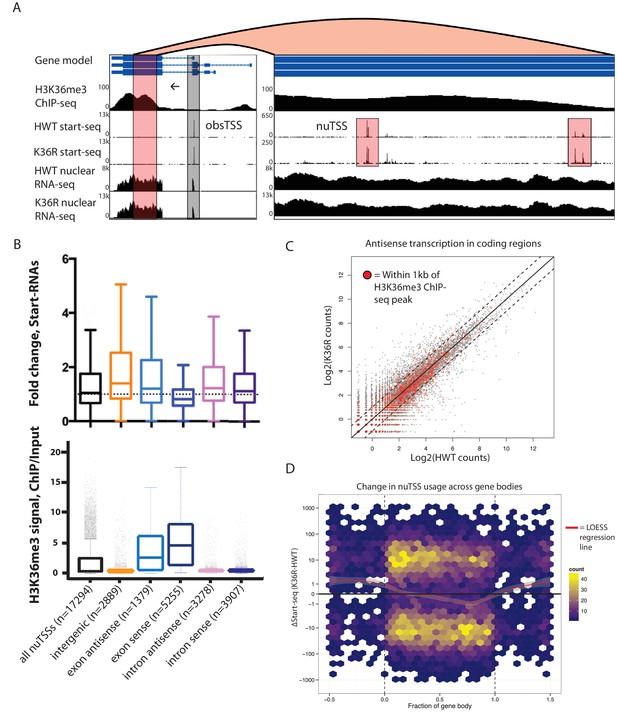
H3K36 modification does not suppress cryptic transcription initiation in coding regions.
(A) Representative browser shot of gene containing novel unannotated transcription start sites (nuTSSs, highlighted in red). Direction of transcription denoted by arrow, and read counts denoted on Y-axis. (B) Boxplot describing the fold change in Start-seq signal for nuTSSs classified by their genomic localization and strand of origin relative to the resident gene if applicable. Lower boxplot describes H3K36me3 ChIP-seq signal (ChIP/input) for the same gene cohorts. (C) Scatterplot of normalized nuclear RNA-seq reads mapping antisense to genes in the dm3 reference gene model in HWT (x-axis) or K36R (y-axis). Genes containing or within 1 kb of a local H3K36me3-ChIP-seq peak are denoted by red dots. (D) Hex-plot heatmap plotting nuTSSs by their location relative to the gene boundaries of the nearest gene, and the absolute change in their Start-seq signal (K36R – HWT).
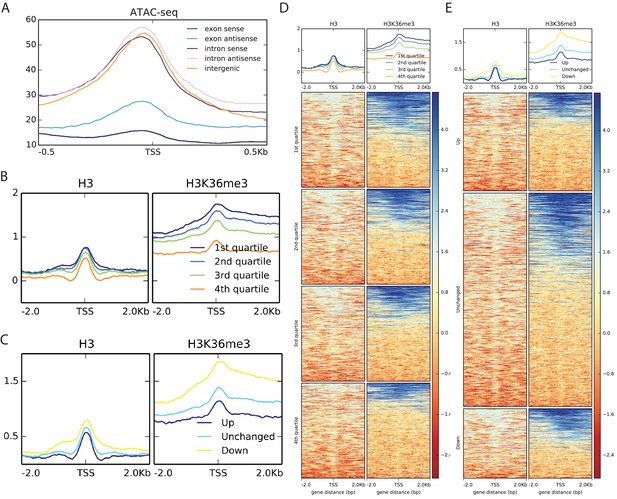
Metagene analysis of Start-seq reads at novel, unannotated (nu)TSSs in comparison to open chromatin, nucleosome positioning and H3K36 trimethylation.
(A) Metaplot of ATAC-seq signal mapping in a 1 kb window around nuTSSs, classified as in Figure 4B. (B) Metaplots of H3 (left) and H3K36me3 (right) ChIP-seq signal mapping to a 4 kb window around nuTSSs, separated by quartiles of absolute Start-seq signal change between K36R and HWT. (C) Metaplots of HWT ATAC-seq signal mapping to a 4 kb window around nuTSSs, separated by log2 fold change in Start-seq signal. ‘Up’ denoted increased by more than two fold in K36R, ‘Down’ denotes decreased by more than two fold in K36R, and ‘Unchanged’ denotes all other nuTSSs. (D) Heatmaps displaying H3 (left) and H3K36me3 (right) ChIP-seq signal mapping to a 4 kb window around nuTSSs representing the catergories listed in B. (E) Heatmaps displaying H3 (left) and H3K36me3 (right) ChIP-seq signal mapping to a 4 kb window around nuTSSs representing the catergories listed in C.
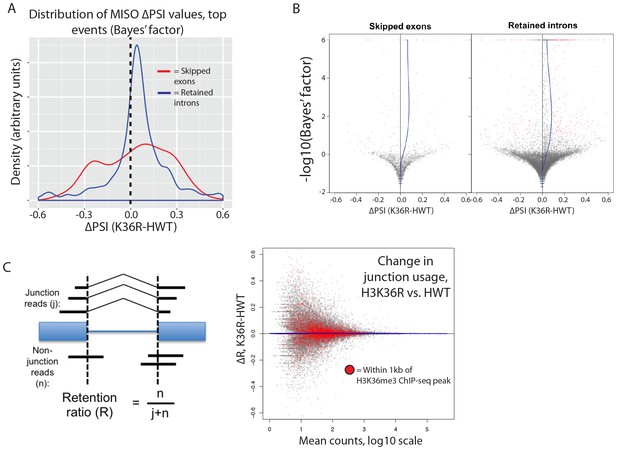
H3K36 modification does not regulate alternative splicing.
(A) Density plots reflecting the distributions of change in percent spliced in (∆PSI) values for skipped exon (red) or retained intron (blue) alternative splicing events manually classified as significant based on MISO parameters (see Materials and methods). (B) Volcano plots for skipped exon (left) and retained intron (right) events, with a local regression line (blue line) reflecting the skew in ∆PSI values (x-axis) based on Bayes factor (y-axis). (C) Global analysis of splice junction usage, where R denotes the ‘retention ratio’ in one condition, and ∆R denotes the difference in R between K36R and HWT.
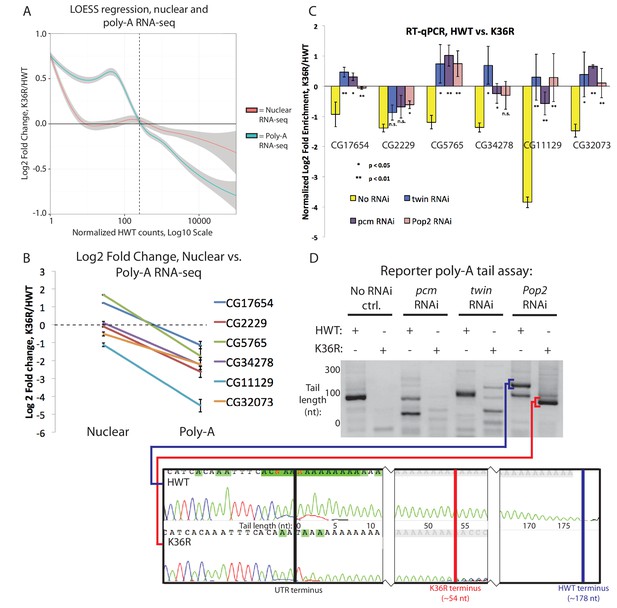
A class of highly expressed genes is subject to exonuclease degradation and inefficient post-transcriptional processing in H3K36R mutants.
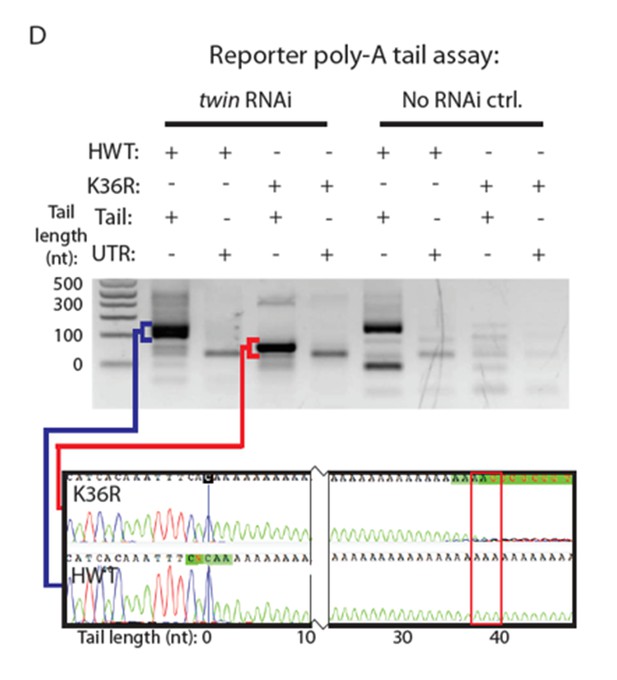
(A) LOESS regression lines generated from MA plots of either nuclear or poly-A RNA-seq, plotting gene log2 fold change (y-axis) vs. normalized read counts in HWT (x-axis). (B) Log2 fold change values between K36R and HWT in nuclear (left) and poly-A (right) RNA-seq, plotted for genes selected for further RT-PCR analysis. (C) RT-qPCR quantification of differential expression between HWT and K36R for select genes in a no RNAi, pacman RNAi, twin RNAi, or Pop2 RNAi background, using the -∆∆Ct method. (D) LM-PAT assay results for the YFP transcript in HWT and K36R, in a no RNAi, pcm RNAi, twin RNAi, or Pop2 RNAi background. Sanger sequencing trace confirming the poly-A site (leftmost panel) and differential tail lengths (right two panels) is shown below.
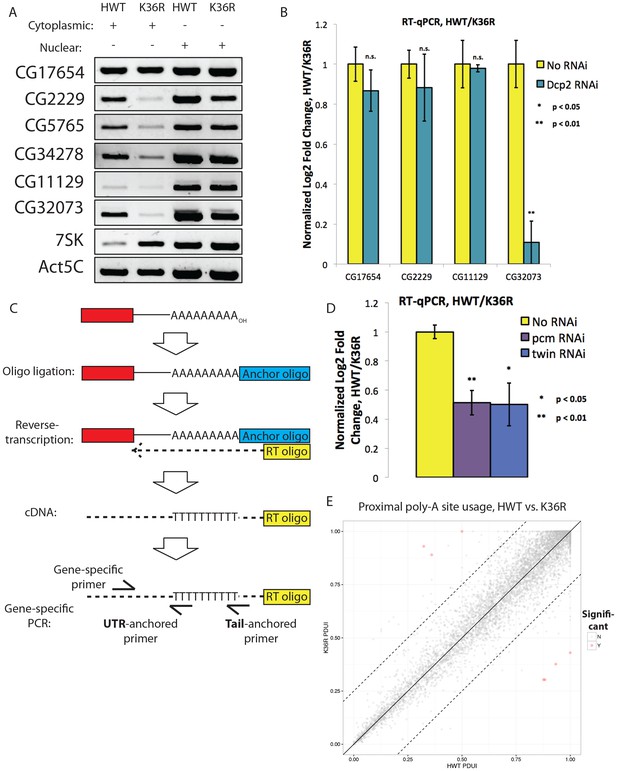
RT-PCR controls, alternative polyadenylation analysis and schematic of assay for gene-specific poly A tail length assay (LM-PAT) showing relative positions of primers.
(A) RT-PCR for select genes from cytoplasmic (lanes 1 and 2) or nuclear (lanes 3 and 4) RNA from HWT (lanes 1 and 3) or K36R (lanes 2 and 4) animals. 7SK RNA is a control for nuclear enrichment. (B) RT-qPCR for select genes from Figure 6B measuring normalized Log2 fold change (HWT/K36R) in no RNAi or Dcp2 RNAi background. P-value obtained via t-test. (C) Schematic of modified LM-PAT assay, in which an adenylated oligonucleotide anchor is ligated to the 3’ end of total RNA, cDNA is generated using an anchor-specific RT primer, and genes of interest are amplified using a gene-specific forward primer and an anchor specific reverse primer that contains either an oligo-T sequence at its 3’ end (tail-anchored) so as to extend from the ends of poly-A tails, or an oligo-T-N sequence (UTR-anchored) in order to extend from the terminus of the 3’ UTR. (D) RT-qPCR for YFP measuring normalized Log2 fold change (HWT/K36R) in no RNAi, pcm RNAi, or twin RNAi backgrounds. P-value obtained via t-test. (E) Genome wide analysis of alternative polyadenylation using DaPars (Xia et al., 2014), with percentage of distal poly-A site usage (PDUI) for each gene in HWT and K36R plotted on the x- and y-axes, respectively.
Additional files
-
Source code 1
Custom perl script used to extract base-specific TSS counts from a Start-seq read SAM file.
Briefly, an input SAM file is parsed for strand orientation and mate pair status, with second mates thrown out, and the genomic coordinates of the first base in the first mate hashed with a running count of reads corresponding to that entry. An optional input removes TSSs whose accumulate counts fail to reach a user-defined threshold.
- https://doi.org/10.7554/eLife.23249.014
-
Source code 2
Custom perl script used to detect splice junctions de novo from an input RNA-seq SAM file and quantify junction and non-junction read counts for each entry.
Briefly, for each read spanning a splice junction (i.e. containing an ‘N’ SAM flag), intron coordinates are defined and hashed with a running count of reads corresponding to those coordinates, which are deposited in a BED file. Non-junction reads are subsequently determined by intersecting output BED file with the original SAM file using bedtools (Quinlan and Hall, 2010).
- https://doi.org/10.7554/eLife.23249.015
-
Supplementary file 1
Excel file summarizing DESeq2 (Love et al., 2014) results from comparing K36R to HWT from total nuclear or poly-A RNA-seq.
For each gene, and each experiment (nuclear and poly-A), there are listed values (from left to right) for mean counts, log2 fold change (K36R/HWT), log2 fold change standard error, test statistic, p-value, and adjusted p-value.
- https://doi.org/10.7554/eLife.23249.016
-
Supplementary file 2
List of primers used for qPCR and LM-PAT assays (see methods).
- https://doi.org/10.7554/eLife.23249.017





