Mating and male pheromone kill Caenorhabditis males through distinct mechanisms
Figures
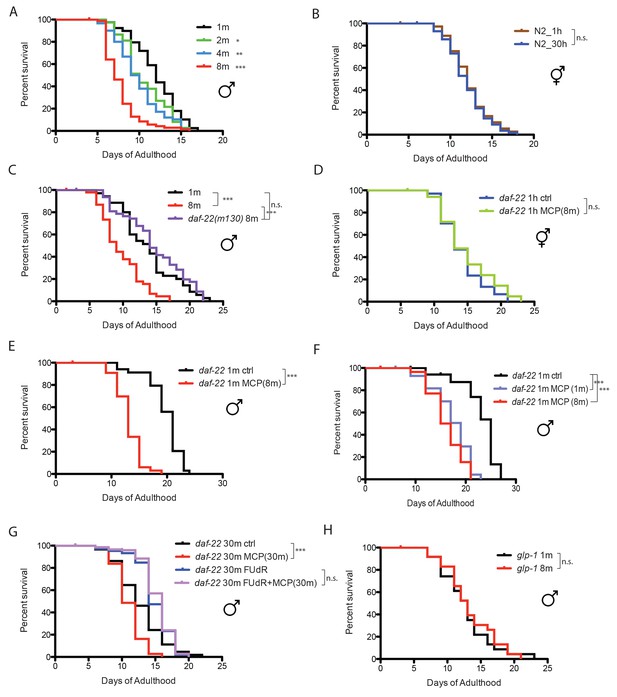
Male pheromone leads to early death in C. elegans males.
(A) Lifespans of grouped fog-2(q71) males. We used fog-2(q71) mutants, as fog-2 males are equivalent to wild-type (N2) males (Schedl and Kimble, 1988). Solitary males: 12.0 ± 0.4 days, n = 40; two males: 10.6 ± 0.4 days, n = 40, p=0.0397; four males: 9.9 ± 0.4 days, n = 60, p=0.0012; eight males: 7.7 ± 0.2 days, n = 80, p<0.0001. For all the lifespan assays performed in this study, Kaplan-Meier analysis with log-rank (Mantel-Cox) test was used to determine statistical significance. All the lifespan results are included in Supplementary file 1. (B) Lifespans of solitary N2 hermaphrodites: 12.3 ± 0.4 days, n = 40; grouped N2 hermaphrodites: 12.0 ± 0.3 days, n = 58, p=0.6436. (C) Grouped daf-22(m130) males have a similar lifespan to solitary wild-type fog-2 males. (daf-22(m130) mutants are ascaroside pheromone-production deficient.) Solitary fog-2 males: 13.8 ± 0.7 days, n = 35; eight fog-2 males: 9.8 ± 0.5 days, n = 48, p<0.0001; eight daf-22(m130) males: 14.7 ± 0.7 days, n = 48, p=0.4039. Details about male-conditioned plates lifespan assays are included in Materials and methods and Figure 1—figure supplement 1B. (D) Lifespans of solitary C. elegans daf-22(m130) hermaphrodites on plates conditioned by eight fog-2 males. Solitary daf-22 hermaphrodites on control plates: 14.2 ± 0.6 days, n = 35; solitary daf-22 hermaphrodites on male-conditioned plates: 14.8 ± 0.8 days, n = 35, p=0.4356. (E) Lifespans of solitary C. elegans daf-22(m130) males on plates conditioned by eight fog-2 males. daf-22(m130) mutants are ascaroside pheromone-production deficient. Therefore, the effect of male pheromone is due to the male pheromone secreted by wild-type males when conditioning the plates. Solitary daf-22 males on control plates: 19.7 ± 0.5 days, n = 34; solitary daf-22 males on male-conditioned plates: 13.1 ± 0.4 days, n = 33, p<0.0001. (F) daf-22(m130) male lifespans on plates conditioned by wild-type fog-2 males. Solitary daf-22(m130): 23.0 ± 0.9 days, n = 30; daf-22(m130) on plates conditioned by one fog-2 male: 17.3 ± 0.7 days, n = 29, p<0.0001; daf-22(m130) on plates conditioned by eight fog-2 male: 16.1 ± 0.6 days, n = 30, p<0.0001. (G) Male pheromone-induced shorter lifespan of grouped daf-22(m130) males is inhibited in the presence of 50 µM FUdR. Grouped daf-22 males on NGM: 12.7 ± 0.3 days, n = 150; grouped daf-22 males on male-conditioned plates (MCP): 11.0 ± 0.2 days, n = 150, p<0.0001; grouped daf-22 males on NGM with FUdR: 14.9 ± 0.2 days, n = 150, grouped daf-22 males on MCP with FUdR: 15.3 ± 0.2 days, n = 150 p=0.2964. (H) Lifespans of grouped glp-1(e2141) males. Solitary males: 12.7 ± 0.8 days, n = 44; eight males: 13.3 ± 0.8 days, n = 56, p=0.699.
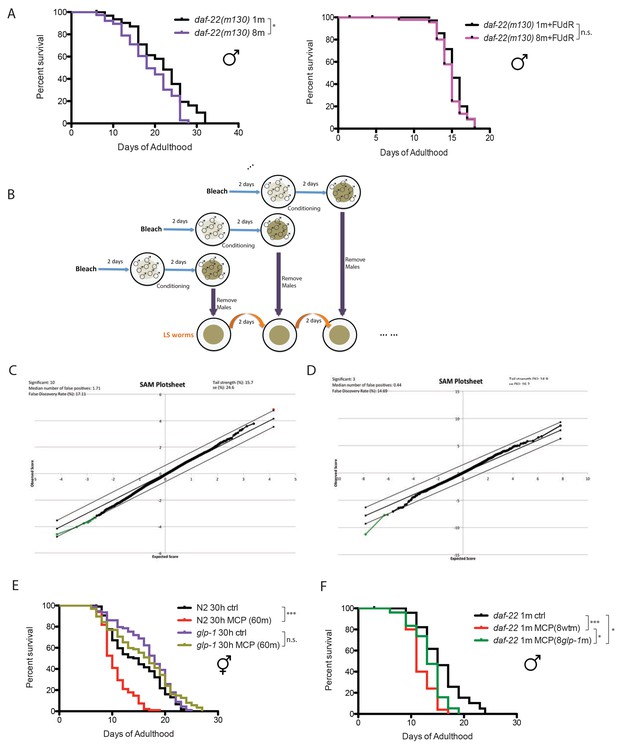
Male pheromone-mediated toxicity requires germline.
(A) Grouped daf-22(m130) males live slightly shorter than the solitary control (left). Solitary daf-22 males: 21.7 ± 1.2 days, n = 32; eight daf-22 males: 18.8 ± 1.0 days, n = 38, p=0.0394. Lifespans are not different between solitary and grouped daf-22(m130) in the presence of FUdR (right). Solitary daf-22(m130): 15.3 ± 0.3 days, n = 35; eight daf-22(m130): 14.7 ± 0.3 days, n = 48, p=0.2117. (B) Schematic illustration of how lifespan assays on male-conditioned plates were performed. Detailed description is included in Materials and methods. (C) SAM plot of daf-22(m130) males ± MCP(30m) for 6 days in the presence of 50 µM FUdR. Only 10 genes’ expression significantly changed, even at high FDR (17%). (D) SAM plot of glp-1(e2141) hermaphrodites ± MCP(60m) for 6 days in the presence of 50 µM FUdR. Only three genes had significant changes in expression, even at high FDR (14%). (E) Lifespans of grouped N2 and glp-1 hermaphrodites on plates conditioned by 60 wild-type fog-2 males. Grouped N2 control: 14.7 ± 0.5 days, n = 107; grouped N2 hermaphrodites on MCP: 10.6 ± 0.3 days, n = 98, p<0.0001; Grouped glp-1(e2141) control: 16.9 ± 0.5 days, n = 96; grouped glp-1(e2141) on MCP: 15.9 ± 0.6 days, n = 97, p=0.8933. Lifespan assay of glp-1 hermaphrodites was performed at 25°C in the first week of adulthood and at room temperature afterwards. (F) Germline also affects the production of male pheromone. Lifespans of solitary daf-22(m130) males control: 15.8 ± 0.9 days, n = 25; solitary daf-22(m130) males on plates conditioned by eight wild-type males: 11.8 ± 0.5 days, n = 25, p=0.0002; solitary daf-22(m130) males on plates conditioned by eight germline-less glp-1 males: 13.4 ± 0.6 days, n = 25, p=0.0339 (compared to the control), p=0.0472 (compared to MCP(8 wt m)).
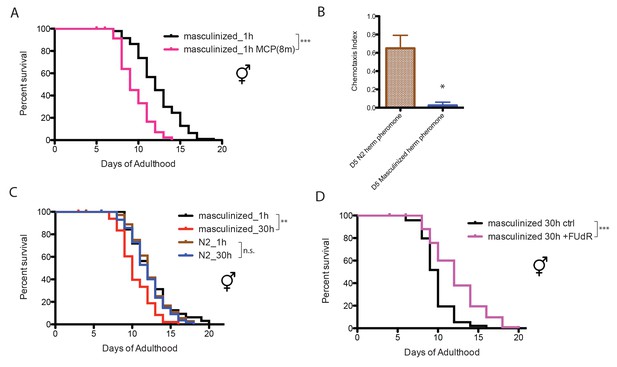
Neuronal masculinization of C. elegans hermaphrodites.
(A) Neuronal masculinization of C. elegans hermaphrodites increases their sensitivity to male pheromone toxicity. Lifespans of solitary masculinized hermaphrodites: 12.3 ± 0.3 days, n = 96; solitary masculinized hermaphrodites on plates conditioned by eight males: 9.6 ± 0.3 days, n = 56, p<0.0001. (B) Supernatant solutions from Day 5 C. elegans hermaphrodites and masculinized hermaphrodites and Day one fog-2 males were used to do the chemotaxis assay. See Materials and methods for detailed description. Chemotaxis Index (C.I.) of wild-type hermaphrodites’ supernatant: 0.65 ± 0.10, C.I. of masculinized hermaphrodites’ supernatant: 0.03 ± 0.03; p=0.0261, unpaired t-test. (C) Neuronal masculinization is sufficient to induce male-like population-density-dependent lifespan decrease in hermaphrodites. Lifespans of solitary N2 hermaphrodites: 12.3 ± 0.4 days, n = 40; grouped N2 hermaphrodites (about 30 worms per 35 mm plate): 12.0 ± 0.3 days, n = 58, p=0.6436. Solitary masculinized hermaphrodites: 12.4 ± 0.5 days, n = 40; grouped masculinized hermaphrodites (30 per plate): 10.4 ± 0.3 days, n = 60, p=0.0015. (D) FUdR rescue the lifespans of grouped masculinized hermaphrodites. Lifespans of grouped masculinized hermaphrodites: 9.8 ± 0.2 days, n = 111, grouped masculinized hermaphrodites (30 per plate) in the presence of 50 µM FUdR: 12.2 ± 0.3 days, n = 119, p<0.0001.
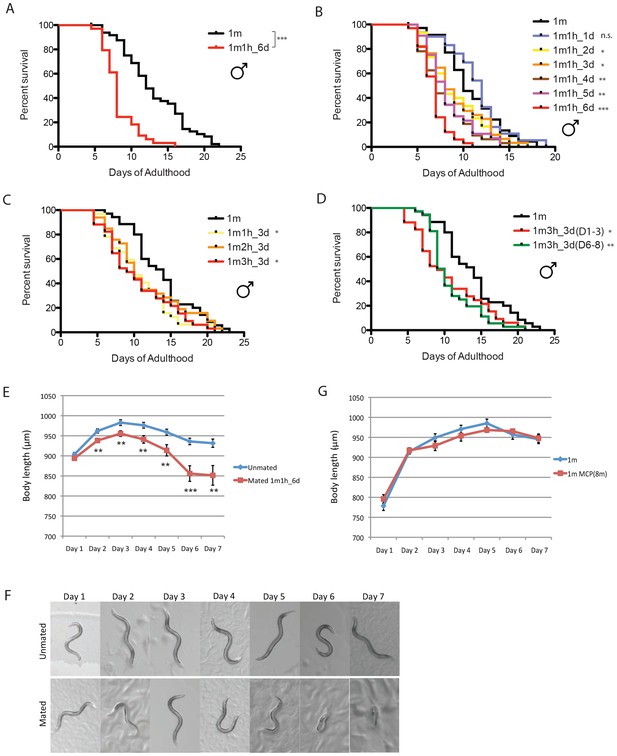
C. elegans males shrink and die early after mating.
(A) Lifespans of unmated solitary and mated fog-2(q71) males. Solitary males: 13.1 ± 0.6 days, n = 50; mated males: 8.3 ± 0.4 days, n = 34, p<0.0001. Each male was paired with a fog-2(q71) hermaphrodite on a single 35 mm plate during Day 1–6 of adulthood. Unless noted, all the hermaphrodites used are fog-2(q71). (B) Male post-mating lifespan decrease is mating duration-dependent: Unmated solitary males: 10.9 ± 0.6 days, n = 35; one male and one hermaphrodite mating on Day 1 of adulthood: 11.4 ± 0.6 days, n = 31, p=0.3697; mating from Day 1–2: 9.0 ± 0.6 days, n = 30, p=0.0325; mating from Day 1–3: 9.1 ± 0.6 days, n = 34, p=0.0452; mating from Day 1–4: 7.9 ± 0.5 days, n = 32, p=0.0002; mating from Day 1–5: 8.3 ± 0.4 days, n = 34, p=0.0006; mating from Day 1–6: 6.8 ± 0.3 days, n = 33, p<0.0001. (C) Lifespans of one male paired with different number of hermaphrodites during Day 1–3 of adulthood: solitary unmated males: 13.8 ± 0.7 days, n = 35; one male with one hermaphrodite: 10.8 ± 0.6 days, n = 32, p=0.0175; one male with two hermaphrodites: 11.6 ± 0.9 days, n = 33, p=0.1435; one male with three hermaphrodites: 10.6 ± 0.8 days, n = 34, p=0.0147. (D) Lifespans of one male paired with three hermaphrodites for 3 days but at different time of adulthood. Solitary unmated males: 13.8 ± 0.7 days, n = 35; mating during Day 1–3 of adulthood: 10.6 ± 0.8 days, n = 34, p=0.0147; mating during Day 6–8 of adulthood: 10.8 ± 0.6 days, n = 37, p=0.0022. (E) Length of unmated and mated fog-2 males: t-test, **p<0.01, ***p<0.001. (F) Representative pictures of the same unmated solitary male and male paired with one hermaphrodite from Day 1-Day 6 of adulthood. (G) Male pheromone does not induce body shrinking. Length of solitary fog-2 males on plates conditioned by eight wild-type males.
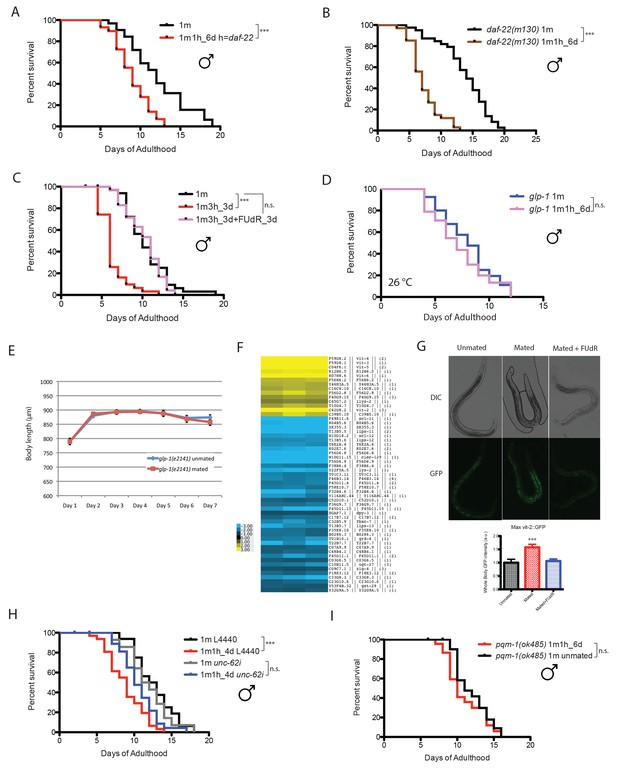
Mating-induced death is germline-dependent.
(A) Lifespans of fog-2 males mated with daf-22(m130) hermaphrodites. Unmated solitary fog-2 males: 12.1 ± 0.6 days, n = 32; mated males: 9.0 ± 0.4 days, n = 29, p=0.0001. In the mated group, one fog-2(q71) male was paired with one daf-22(m130) hermaphrodite from Day 1- Day 6 of adulthood. (B) Lifespans of unmated and mated daf-22(m130) males. Unmated solitary daf-22(m130) males: 13.8 ± 0.6 days, n = 40; mated daf-22(m130) males: 7.4 ± 0.4 days, n = 34, p<0.0001. In the mated group, one daf-22(m130) male was paired with one fog-2(q71) hermaphrodite from Day 1- Day 6 of adulthood. (C) FUdR can rescue male post-mating early death. Unmated solitary males: 10.5 ± 0.5 days, n = 35; one male with three hermaphrodites for three days: 6.4 ± 0.3 days, n = 31, p<0.0001; one male with three hermaphrodites for three days but in the presence of 50 µM FUdR during the three days’ mating: 10.2 ± 0.4 days, n = 36, p=0.7086 (compared with unmated solitary group). (D) Lifespans of unmated and mated glp-1(e2141) males: unmated solitary glp-1 males: 8.0 ± 0.4 days, n = 40; mated glp-1 males: 7.2 ± 0.4 days, n = 40, p=0.3178. The assay was performed at 26°C, in mated group, one glp-1 male was paired with one fog-2 hermaphrodite from Day 1–6. (E) Length of mated and unmated glp-1(e2141) males. (The same population as in Figure 4D.). (F) Expression heatmap of genes whose expression is significantly changed in mated males based on SAM analysis. (G) Ectopic expression of VIT-2::GFP in mated males is germline-dependent. 5 days’ mating, pictures were taken on Day 6 of adulthood. Representative images are shown above the quantification of VIT-2::GFP expression [maximum ± SE (error bars)], a.u., arbitrary units. ***p<0.001, t-test. (H) unc-62 RNAi suppresses male post-mating early death. Unmated solitary male on L4440: 12.6 ± 0.7 days, n = 25; mated males on L4440: 8.8 ± 0.5 days, n = 33, p=0.0001. Unmated males on unc-62 RNAi: 11.9 ± 0.8 days, n = 25; mated males on unc-62 RNAi: 10.6 ± 0.5 days, n = 34, p=0.1249 (compared to unmated males on unc-62 RNAi). (I) pqm-1(ok485) mated males have similar lifespans as unmated controls. Unmated solitary pqm-1(ok485) males: 11.9 ± 0.5 days, n = 25; mated pqm-1(ok485) males: 11.0 ± 0.6 days, n = 29, p=0.2782. In the mated group, one pqm-1(ok485) male was paired with one fog-2(q71) hermaphrodite from Day 1- Day 6 of adulthood.
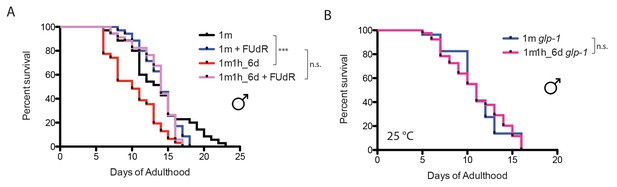
Mating-induced lifespan decrease is germline-dependent.
(A) Germline proliferation blocking with FUdR can rescue male post-mating early death. Unmated solitary males: 13.8 ± 0.7 days, n = 35; one male with one hermaphrodite for six days: 10.3 ± 0.6 days, n = 31, p=0.0006; solitary male in the presence of 50 µM FUdR: 13.9 ± 0.4 days, n = 35, p=0.4079 (compared to unmated solitary group). One male mated with one hermaphrodites for 6 days in the presence of FUdR: 13.6 ± 0.5 days, n = 34, p=0.3992 compared to unmated solitary group. (B) Lifespans of unmated and mated glp-1(e2141) males: unmated solitary glp-1 males: 11.1 ± 1.0 days, n = 27; mated glp-1 males: 11.1 ± 0.5 days, n = 43, p=0.9149. The assay was performed at 25°C; in mated group, one glp-1 male was paired with one fog-2 hermaphrodite from Day 1–6.
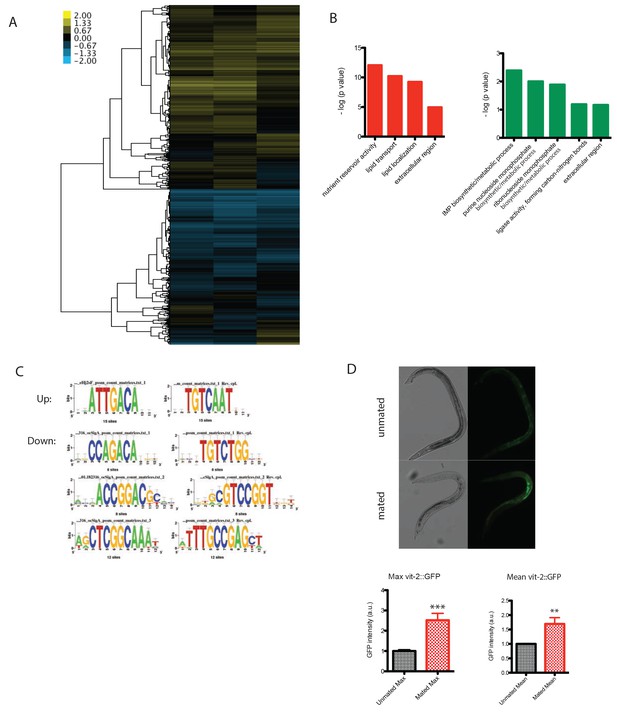
Microarray analysis of mated males.
(A) Clustered heat map of whole transcriptome expression comparison of mated vs unmated males. Individual males were paired with one hermaphrodite for 3.5 days and collected on Day four for microarrays. (B) Enriched GO terms for significantly up- and down-regulated genes in mated males. (C) Enriched motifs in promoter region (1 kb upstream of TSS) of significantly up- and down-regulated genes using (RSAT) Regulatory Sequence Analysis Tools (www.rsat.eu). (D) VIT-2::GFP expression in males increases significantly after mating. Upper: DIC and GFP images; Lower: GFP intensity quantitation, left: max ± SE (error bars); right: mean ± SE (error bars), a.u., arbitrary units. **p<0.01, t-test.
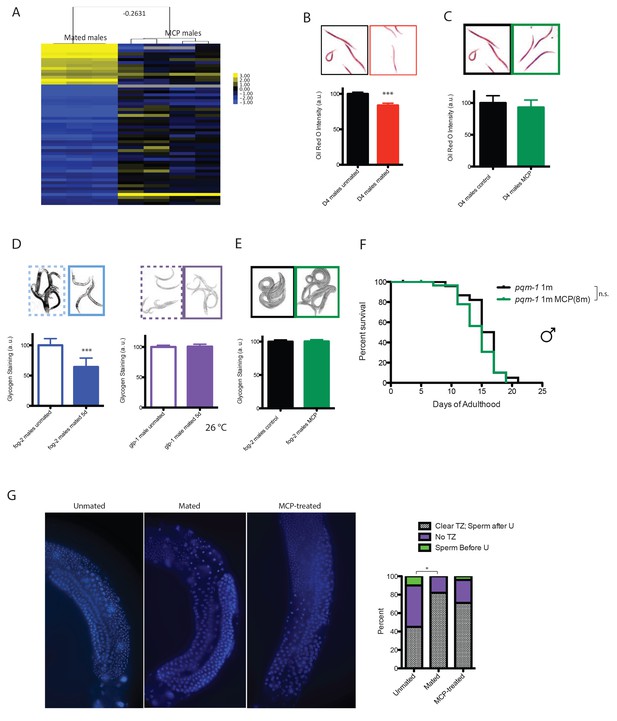
Mating-induced and male pheromone-induced death are distinct.
(A) Transcriptional profiles of mated males and MCP-treated males are different. Heatmap cluster of mated males (left) and MCP-treated vs untreated grouped daf-22 males (right); Pearson correlation = −0.27. The cluster only contains genes with significant changes in mated males by SAM, 0% FDR. (B) Mating induces significant fat loss in males. Representative pictures of Oil red O staining are shown above the quantitation. Males lost about 20% of their fat after mating on Day 4, p<0.001. Error bars represent SE. (C) Male pheromone exposure fails to induce fat loss in males. Four days’ MCP treatment. Representative pictures of Oil red O staining are shown above the quantitation. Unconditioned control males are framed by black lines, and MCP-treated males are framed by green lines. (D) Glycogen staining of mated and unmated males. Left: mated fog-2 (wt) males lost over 30% glycogen after 5 days’ mating; ***p<0.001. Right: mated glp-1 males did not lose glycogen after mating. The staining intensity was normalized to unmated males of each genotype. Representative pictures are shown above the quantitation. Unmated males are framed by dashed lines, and mated males are framed by solid lines. (E) No glycogen loss after male pheromone exposure. Four days’ MCP treatment. Representative pictures of iodine staining are shown above the quantitation. Unconditioned control males are framed by black lines, and MCP-treated males are framed by green lines. (In D–E, error bars represent SD.). (F) Loss of the DAE-dependent transcription factor PQM-1 suppresses male pheromone-induced death. Lifespans of control solitary pqm-1(ok485) males: 15.6 ± 0.6 days, n = 25; solitary pqm-1(ok485) males on plates conditioned by eight males: 14.4 ± 0.6 days, n = 25, p=0.1627. (G) DAPI staining of Day six males’ germlines (left). Right: categorical quantification of germline morphology: mating causes more obvious change in male germline morphology than male pheromone does. TZ: transition zone; U: U-shaped turn of male germline. See Figure 5—figure supplement 4 for details.
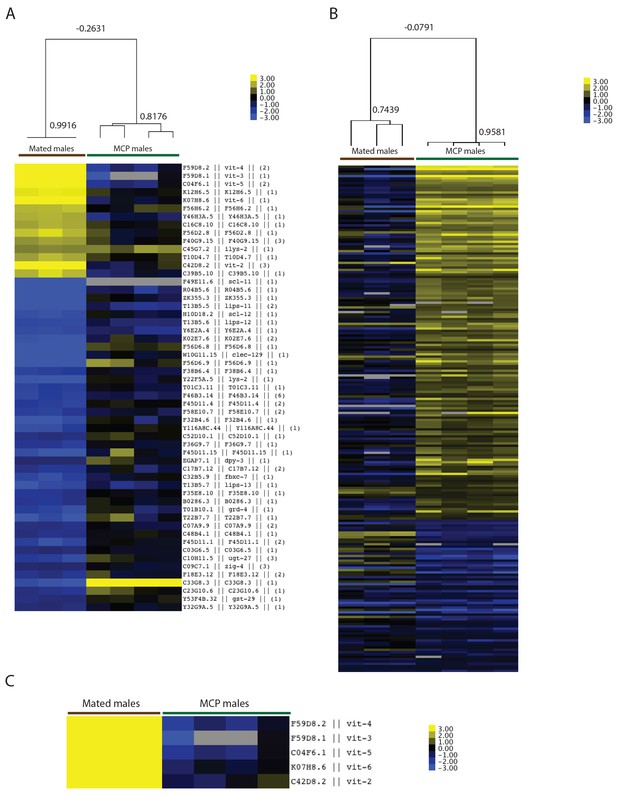
Transcriptional profiles of mated and male pheromone-induced males are distinct.
(A) Heatmap of (1) mated males (genes with significant changes by SAM, 0% FDR) and (2) MCP treated vs untreated grouped daf-22 males. Top: Pearson correlation. (B) Heatmap of (1) mated males and (2) MCP treated vs untreated grouped daf-22 males (genes with significant changes by SAM, 1% FDR). Top: Pearson correlation. (C) Vit genes are not up-regulated in males exposed to male pheromone.
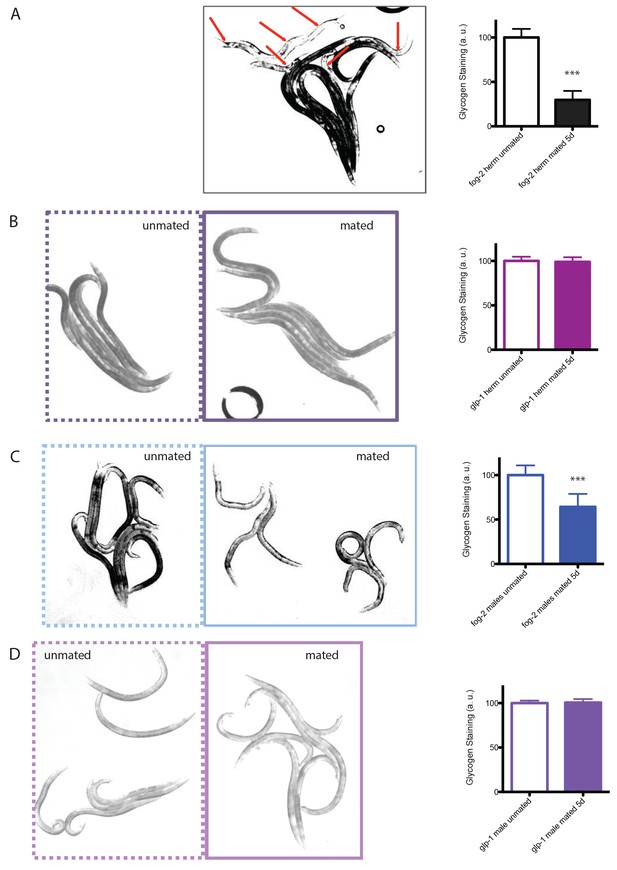
Glycogen staining of mated vs unmated hermaphrodites and males.
Left: representative pictures of iodine staining of worms. Unmated worms are framed by dashed lines, whereas mated worms are framed by solid lines. In the first picture, mated and unmated fog-2 hermaphrodites were mixed together, with red arrows pointing to mated fog-2 hermaphrodites. Worms were mated from Day 1 – Day 5 and were imaged on Day 5. Right: quantitation of iodine staining. The intensity of mated worms was normalized to unmated control of the same genotype. Mated fog-2 hermaphrodites have only 30% of the glycogen levels of unmated fog-2 hermaphrodites of the same age (p<0.0001). Mated glp-1 hermaphrodites have 99% glycogen compared to unmated glp-1 hermaphrodites control (p=0.6070). Mated fog-2 males have 64% of the glycogen level of unmated fog-2 males of the same age (p<0.0001). Mated glp-1 males have 101% glycogen compared to unmated glp-1 males control (p=0.7107). Error bars represent SD. ***p<0.0001, t-test.
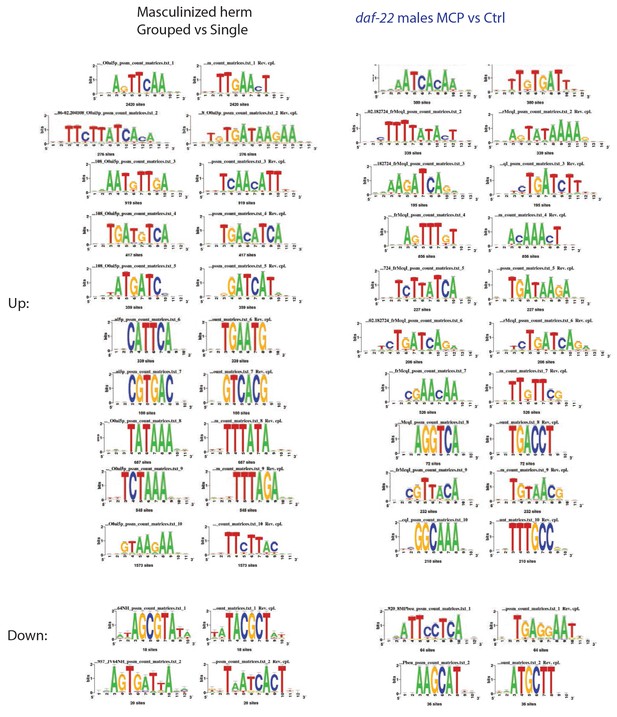
Enriched motifs of male pheromone-induced transcriptional changes.
Enriched motifs associated with significantly up- and down-regulated genes predicted by RSAT (Regulatory Sequence Analysis Tools) in grouped vs. single neuronal masculinized hermaphrodites (left) and MCP treated vs untreated grouped daf-22 males.
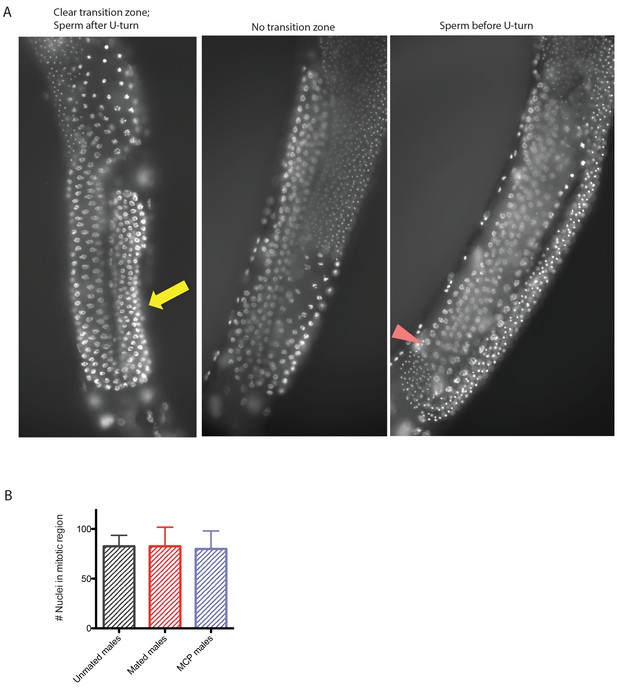
Germline of mated and MCP-treated males.
(A) Representative micrographs of each category: left: male germline with clear transition zone (yellow arrow indicates transition zone marked by crescent shaped nuclei); middle: male germline with no apparent transition zone; right: male germline with mature sperm appearing before U shaped turn. (B) Mating (one male one hermaphrodites for 4 days) or treating solitary males with male pheromone (4 days on MCP(8m)) does not change the number of proliferating nuclei in the male germline. The number of nuclei in mitotic proliferating zone (only counting germlines with clear transition zone) is similar in all three conditions.
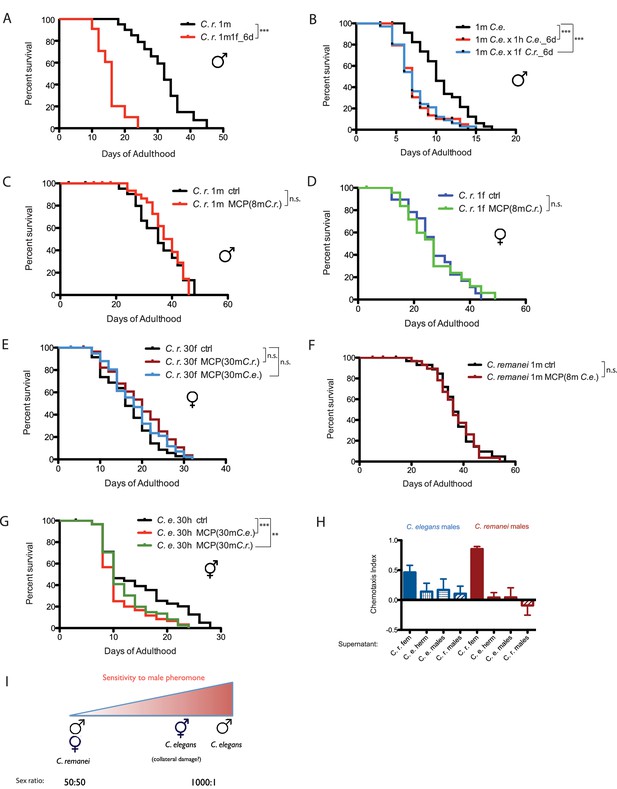
Mating-induced death is evolutionarily conserved, whereas male pheromone-induced death is not.
(A) Mated C. remanei males also live shorter. Unmated solitary C. remanei males: 31.4 ± 1.7 days, n = 72; mated C. remanei males: 15.7 ± 1.2 days, n = 28, p<0.0001. In mated group: one C. remanei male was paired with one C. remanei female from Day 1-Day 6 of adulthood. (B) Lifespans of C. elegans males mated with C. elegans hermaphrodites and C. remanei females. Unmated solitary C. elegans males: 10.2 ± 0.6 days, n = 35; C. elegans males mated with C. elegans hermaphrodites: 7.4 ± 0.4 days, n = 35, p=0.0001; C. elegans males mated with C. remanei females: 7.4 ± 0.4 days, n = 35, p=0.0003. In mated groups, one C. elegans male was paired with either one C. elegans hermaphrodite or one C. remanei female from Day 1–6 of adulthood. (C) Lifespans of solitary C. remanei males on plates conditioned by eight C. remanei males. Solitary C. remanei males on control plates: 35.8 ± 2.0 days, n = 34; solitary C. remanei males on male-conditioned plates: 37.8 ± 1.2 days, n = 34, p=0.8501. (D) Lifespans of solitary C. remanei females on plates conditioned by eight C. remanei males. Solitary C. remanei females on control plates: 27.6 ± 2.2 days, n = 24; solitary C. remanei females on male-conditioned plates: 27.0 ± 2.5 days, n = 30, p=0.8306. (E) Lifespans of grouped C. remanei females on plates conditioned by 30 males. C. remanei females on control plates: 15.8 ± 0.9 days, n = 60; C. remanei females on plates conditioned by C. remanei males: 19.5 ± 1.3 days, n = 30, p=0.0636; C. remanei females on plates conditioned by C. elegans fog-2 males: 18.5 ± 0.9 days, n = 60, p=0.1770. (F) Lifespans of solitary C. remanei males on plates conditioned by eight C. elegans males. Solitary C. remanei males on control plates: 37.2 ± 1.7 days, n = 40; solitary C. remanei males on C. elegans male-conditioned plates: 36.7 ± 1.4 days, n = 38, p=0.7774. (G) Lifespans of grouped C. elegans fog-2 hermaphrodites on plates conditioned with 30 males. fog-2 hermaphrodites control: 14.4 ± 0.8 days, n = 90. fog-2 hermaphrodites on plates conditioned by fog-2 males: 10.9 ± 0.6 days, n = 60, p=0.0004; fog-2 hermaphrodites on plates conditioned by C. remanei males: 11.9 ± 0.5 days, n = 90, p=0.0042. (H) Chemotaxis of C. elegans (left, blue) and C. remanei (right, red) to supernatants from C. elegans males, C. remanei males, C. elegans N2 hermaphrodites, and C. remanei females. See Materials and methods for detailed description. C. e. males to supernatant of C. r. females: Chemotaxis Index (CI) is 0.46 ± 0.11 (mean ± SEM, n = 12 [plates]); C. e. males to supernatant of C. e. hermaphrodites: CI = 0.14 ± 0.13 (n = 10); C. e. males to supernatant of C. e. males: CI = 0.17 ± 0.17 (n = 12); C. e. males to supernatant of C. r. males: CI = 0.11 ± 0.12 (n = 11); C. r. males to supernatant of C. r. females: CI = 0.85 ± 0.04 (n = 12); C. r. males to supernatant of C. e. hermaphrodites: CI = 0.04 ± 0.08 (n = 12); C. r. males to supernatant of C. e. males: CI = 0.04 ± 0.15 (n = 12); C. r. males to supernatant of C. r. males: CI = −0.09 ± 0.16 (n = 12). (I) Sensitivity to male pheromone-induced lifespan reduction. C. elegans males are the most sensitive to male pheromone-induced killing, whereas both C. remanei sexes are immune to this effect.
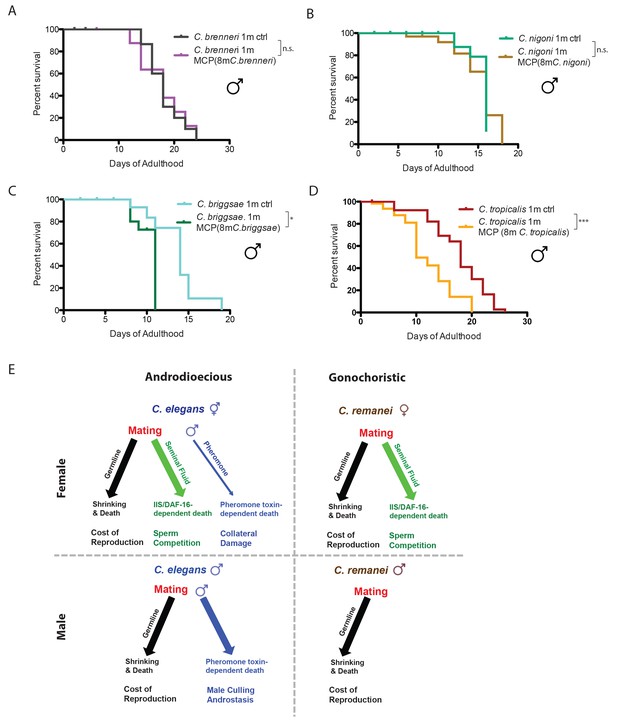
Gonochoristic species are immune to male pheromone killing; androdioecious species are susceptible.
(A) Lifespans of solitary C. brenneri males on plates conditioned by 8 C. brenneri males. Solitary C. brenneri males control: 18.1 ± 0.8 days, n = 33; solitary C. brenneri males on MCP: 17.8 ± 1.1 days, n = 32, p=0.9915. (B) Lifespans of solitary C. nigoni males on plates conditioned by 8 C. nigoni males. Solitary C. nigoni males control: 15.3 ± 0.4 days, n = 32; solitary C. nigoni males on MCP: 15.2 ± 0.6 days, n = 40, p=0.7443. (C) Lifespans of solitary C. briggsae males on plates conditioned by 8 C. briggsae males. Solitary C. briggsae males control: 13.7 ± 0.8 days, n = 38; solitary C. briggsae males on MCP: 10.3 ± 0.3 days, n = 54, p=0.0192. (D) Lifespans of solitary C. tropicalis males on plates conditioned by 8 C. tropicalis males. Solitary C. tropicalis males control: 17.7 ± 0.8 days, n = 40; solitary C. tropicalis males on MCP: 12.2 ± 1.0 days, n = 60, p=0.0002. (E) Model of the effects of mating and male pheromone on androdioecious and gonochoristic female and males. C. elegans hermaphrodites (upper left); C. remanei females (upper right); C. elegans males (lower left); C. remanei males (lower right).
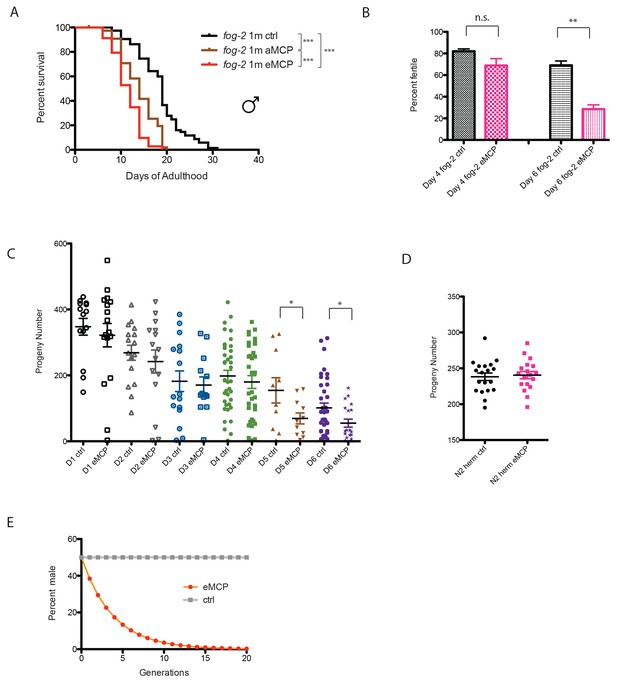
Male pheromone reduces male offspring.
(A) Male pheromone conditioning from egg onward causes a more severe lifespan shortening. (Pooled results from two independent assays. See Figure 8—figure supplement 2 for results of separate lifespan assays, which were also significant.) Solitary C. elegans fog-2 males control: 18.5 ± 0.6 days, n = 75; solitary C. elegans fog-2 males on MCP from early adulthood onward (aMCP): 13.9 ± 0.4 days, n = 79, p<0.0001 (compared to the control); solitary C. elegans fog-2 males on MCP from egg onward (eMCP): 11.4 ± 0.4 days, n = 80, p<0.0001 (compared to the control), p<0.0001 (compared to aMCP). (B) Male pheromone decreases fog-2 male fertility on Day 6. Male treatment started from egg onward. Each male was paired with one virgin fog-2 hermaphrodite at indicated time for 24 hr. On Day 4 of adulthood, the percent of males who were able to fertilize Day 1 virgin fog-2 hermaphrodites: three biological replicates: ctrl: 82 ± 2%; MCP: 69 ± 6%, p=0.12, unpaired t-test. By Day 6 of adulthood, the percent of males who were able to fertilize Day one virgin fog-2 hermaphrodites (three biological replicates) had significantly decreased: ctrl: 69 ± 4%; MCP: 29 ± 4%, p=0.0019. (C) Male pheromone treatment decreases the number of progeny produced by those animals who do successfully mate. The difference appears by Day 5. See Figure 8—figure supplement 3 for detailed numbers. *p<0.05, unpaired t-test. (D) Brood size of self-fertilized N2 hermaphrodites is not affected by male pheromone. Ctrl: 238.2 ± 5, n = 19; MCP-treated: 240.5 ± 5, n = 18, p=0.7571, unpaired t-test. (E) Theoretical calculation of male pheromone’s effect on male population control (not considering any other contributing factors). See Discussion and Figure 8—figure supplement 3 for more details.
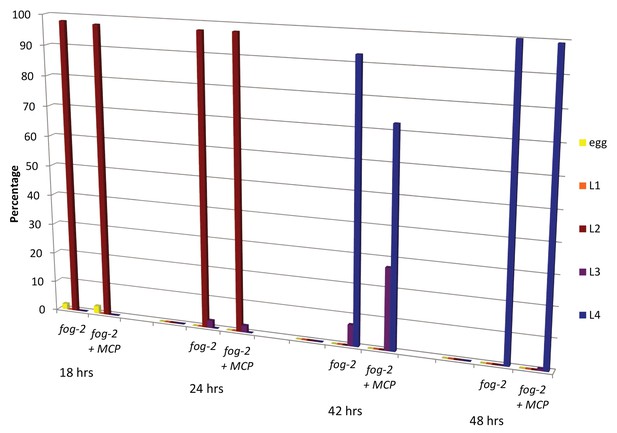
Male pheromone does not affect developmental rates.
Eggs from fog-2 worms were bleached onto control (n = 359) or MCP (n = 257) plates. The number of eggs, L1, L2, L3, and L4 worms was counted at 18, 24, 42, and 48 hr post-bleach. We observed a slight deceleration in development at L3 and no difference by L4, resulting in no significant change in total developmental rate upon treatment with MCP.
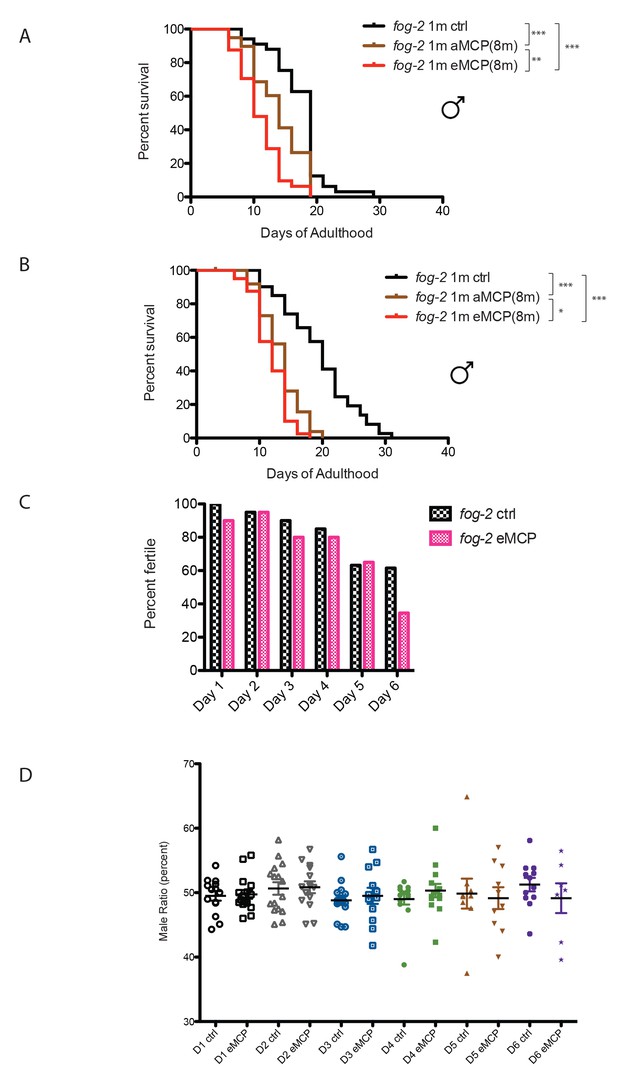
Male pheromone treatment from egg onward severely affects both lifespan and cross-offspring production.
(A) Male pheromone conditioning from egg onward causes a severe lifespan shortening (replicate 1). Solitary C. elegans fog-2 males control: 17.4 ± 0.7 days, n = 34; solitary C. elegans fog-2 males on MCP from early adulthood onward (aMCP): 13.9 ± 0.7 days, n = 39, p<0.0001 (compared to the control); solitary C. elegans fog-2 males on MCP from egg onward (eMCP): 11.1 ± 0.6 days, n = 40, p<0.0001 (compared to the control), p=0.0021 (compared to aMCP). (B) Male pheromone conditioning from egg onward causes a severe lifespan shortening (replicate 2). Solitary C. elegans fog-2 males control: 19.5 ± 0.9 days, n = 41; solitary C. elegans fog-2 males on MCP from early adulthood onward (aMCP): 13.4 ± 0.5 days, n = 40, p<0.0001 (compared to the control); solitary C. elegans fog-2 males on MCP from egg onward (eMCP): 11.8 ± 0.4 days, n = 40, p<0.0001 (compared to the control), p=0.024 (compared to aMCP). (C) Percentages of fog-2 males who are able to successfully fertilize virgin fog-2 hermaphrodites in the presence and absence of MCP treatment. See Figure 8—figure supplement 3 for details. (D) Male pheromone treatment does not selectively kill one sex in offspring. The male ratio is always ~50% (checked from the progeny produced by fog-2 hermaphrodites successfully fertilized by males in C).
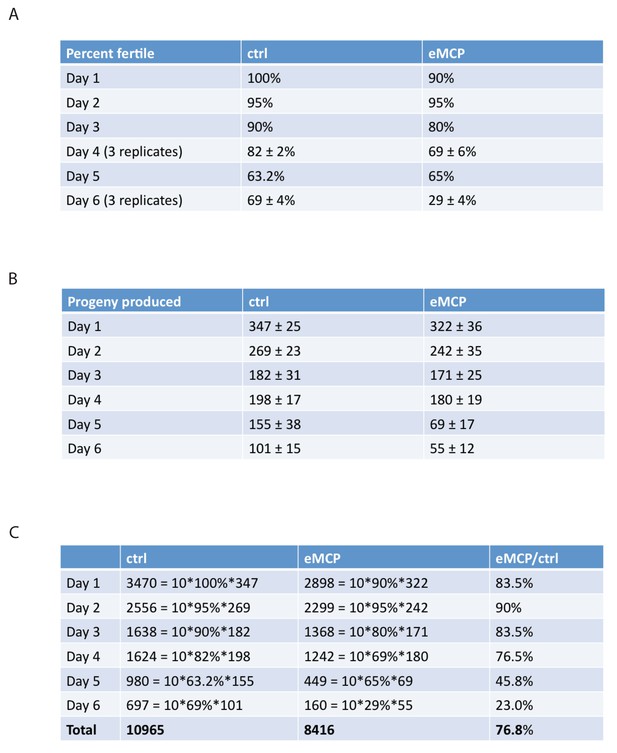
Male pheromone treatment from egg onward reduces offspring and might be a novel mechanism to cull male population in hermaphroditic species.
(A) Percentage of fog-2 males who were able to successfully fertilize virgin fog-2 hermaphrodites in the presence and absence of MCP treatment. (B) Progeny produced by those animals who do successfully mate. (C) Example of theoretical calculation of how male pheromone affects cross progeny production in the next generation (simplified modeling, not considering any other factors). In both control and eMCP conditions, the population starts with 60 males. Assuming they have equal chance of mating from Day 1 – Day 6 of adulthood, the data of percent fertile and number of progeny produced in A and B were used at each time points, in the end, the males in eMCP group produce about 76.8% of the total progeny produced by males in control group. This number was used when plotting the curve in Figure 8E, for generation X, percent males of control group is 50%, percent males of eMCP group is 50%*(76.8%)X.
Additional files
-
Supplementary file 1
Summary of all lifespan assays performed in this study.
- https://doi.org/10.7554/eLife.23493.021
-
Supplementary file 2
Summary of body size measurements.
- https://doi.org/10.7554/eLife.23493.022
-
Supplementary file 3
Significantly up- and down-regulated genes in mated males identified by Significance Analysis of Microarrays (FDR = 0%).
- https://doi.org/10.7554/eLife.23493.023
-
Supplementary file 4
Significantly up- and down-regulated genes under male pheromone-induced conditions identified by Significance Analysis of Microarrays (FDR = 1%).
Sheet 1: grouped vs. solitary neuronally-masculinized hermaphrodites. Sheet 2: grouped daf-22 males on plates conditioned by wild-type males vs. grouped daf-22 males on control plates. Sheet 3: overlap between the two conditions.
- https://doi.org/10.7554/eLife.23493.024
-
Supplementary file 5
Comparison of the list of genes significantly up- and down-regulated upon MCP treatment of daf-22 males to previously published arrays of male pheromone-treated hermaphrodites (Maures et al., 2014).
Sheet 1: comparison of up-regulated genes. Sheet 2: comparison of down-regulated genes.
- https://doi.org/10.7554/eLife.23493.025





