Three dimensional reconstruction of energy stores for jumping in planthoppers and froghoppers from confocal laser scanning microscopy
Figures
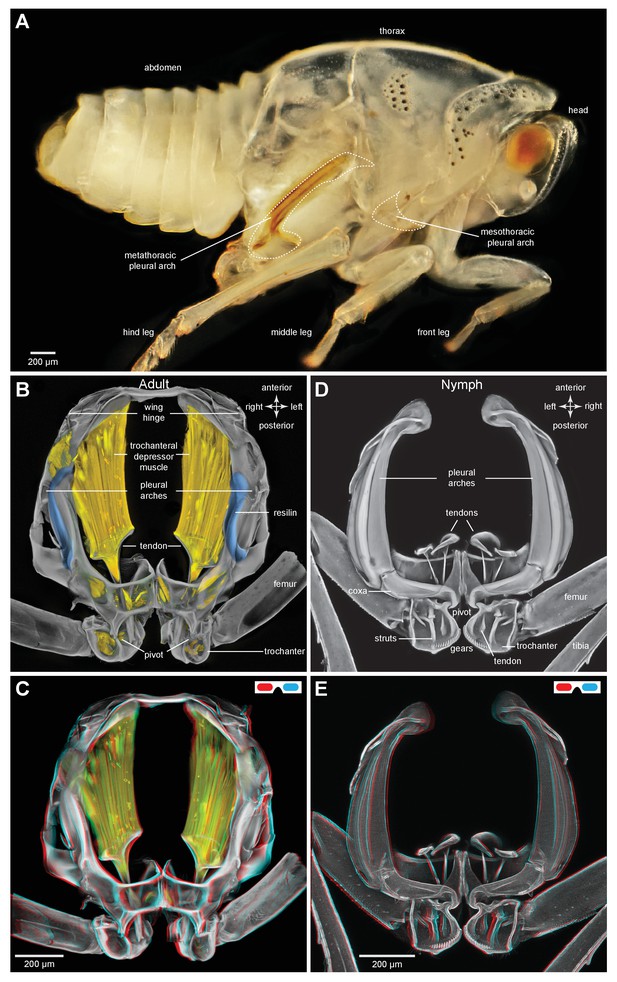
Structure of the skeletal elements in the metathorax of planthoppers associated with jumping.
(A) Side view of an intact nymph of Metcalfa pruinosa. The right pleural arch of the metathoracic segment is visible through the external cuticle. (B) Two dimensional ventral view of the thorax of an adult Delphacid planthopper, Stenocranus acutus. The large trochanteral depressor muscle (M133b/c) and parts of other muscles are shown in yellow and resilin in the pleural arches is shown in blue. All other soft tissue was removed. (C) Three dimensional rendering (indicated by the symbol of coloured glasses) of the same ventral view of the adult. Muscle M133b/c is again shown in yellow. The scale bar in (C) also refers to (B). (D) Two dimensional dorsal view of the thorax of a nymph of S. acutus. E. Three dimensional ventral view of the nymph. The scale bar in (E) also refers to (D). In (D,E) all soft tissue in the thorax was digested away to leave only the skeletal structures. Some cuticle on each coxa was also removed to reveal the articulation of a coxa with a pleural arch on each side.
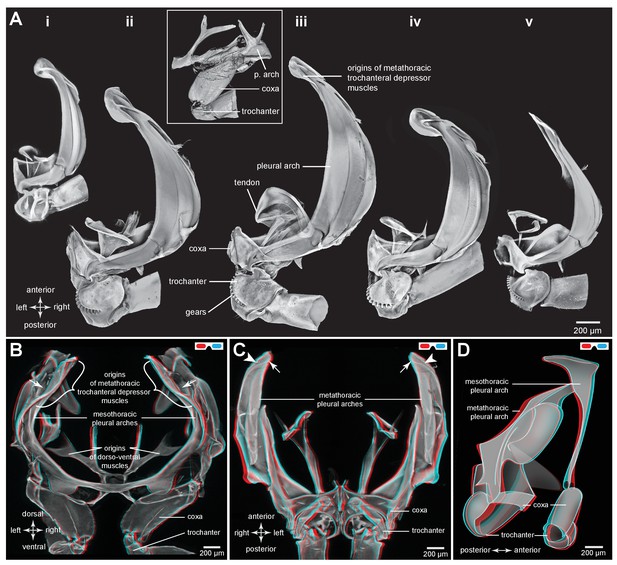
Comparison of the structure of the pleural arches in nymphs of five families of planthoppers.
(Ai) Delphacidae, Stenocranus acutus. (Aii) Issidae, Thionia bullata. (Aiii) Acanaloniidae, Acanalonia conica. (Aiv) Flatidae, Metcalfa pruinosa. (Av) Caliscelidae, Bruchomorpha oculata. In each of these nymphs the right pleural arch is viewed dorsally to show its articulation with the coxa, the tendon of the trochanteral muscle, and the gears on the trochanter. Inset: mesothoracic struts of A. conica that give rise to the mesothoracic pleural arch in the adult. (B) Mesothoracic arches of an adult Flatormenis proxima viewed dorsally, imaged using red autofluorescence (C). Metathoracic arches of F. proxima viewed ventrally. Arrows denote articulation sites of the arches; arrowheads, articulations with a wing hinge. (D) Schematic representation based on confocal data showing relative the positions of the meso- and metathoracic pleural arches viewed medially.
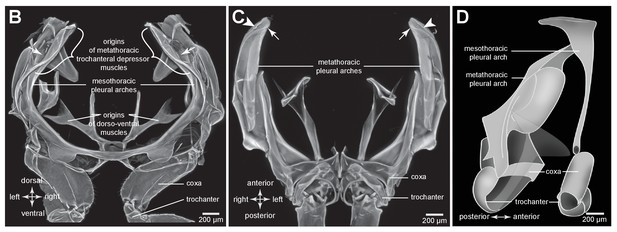
Comparison of the structure of the meso- and metathoracic pleural arches in an adult planthopper.
(B) Mesothoracic arches of an adult Flatormenis proxima viewed dorsally, imaged using red autofluorescence (C). Metathoracic arches of F. proxima viewed ventrally. Arrows denote articulation sites of the arches; arrowheads, articulations with a wing hinge. (D) Schematic representation based on confocal data showing relative the positions of the meso- and metathoracic pleural arches viewed medially.
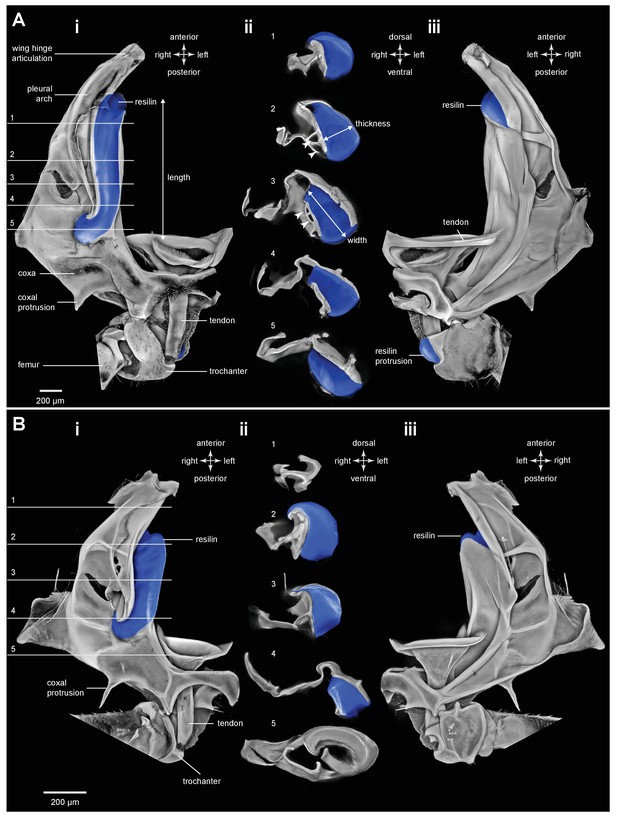
Resilin in the thorax of adult planthoppers.
(A) The skeletal elements of the right side of the metathorax of the Issid planthopper Thionia bullata after all soft tissue and soft cuticle was removed. The resilin, visualized using Acridine Orange staining, is shown in blue. (Ai) Ventral view. (Aii) Sections cut at the five levels indicated in (Ai). The white arrowheads in sections 2 and 3 indicate the holes. (Aiii) Dorsal view. (B) The same skeletal elements of the right thorax of the Derbid planthopper Apache degeeri. (Bi) Ventral view. (Bii) Sections cut at the five levels indicated in (Bi). (Biii) Dorsal view.
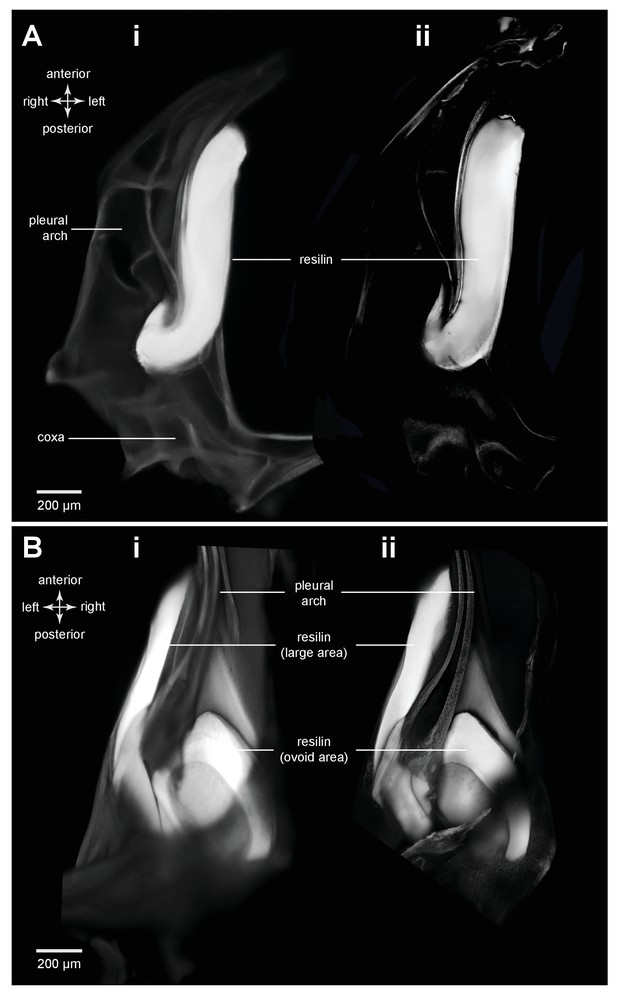
Comparison of methods for resilin visualization in planthopper and froghopper energy stores.
(A) Ventral view of the right pleural arch of planthopper Acanalonia conica. (B) Dorsal view of the right pleural arch of froghopper Lepyronia quadrangularis. (Ai and Bi) Wide-field fluorescence micrographs taken using DAPI filter cube set (excitation: 352–402 nm, emission: 414–477 nm) showing resilin autofluorescence. (Aii and Bii) CLSM maximum intensity projections showing the same distribution of resilin following staining with Acridine Orange.
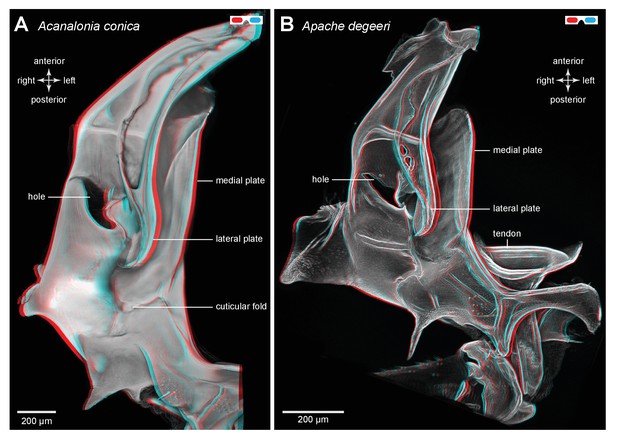
Three dimensional structure of a pleural arch.
(A) The right pleural arch of the Acanaloniid planthopper Acanalonia conica viewed ventrally. The lateral plate of the arch is reinforced by antero-posterior infoldings, and the medial plate is thin but curved. The region between these two plates is normally filled with resilin. (B) The right pleural arch of the Derbid Apache degeeri viewed ventrally and in which edges have been emphasised (using ‘find edges’ filter in ImageJ). Again the region delineated by the two plates is normally filled with resilin. The arches were imaged using red autofluorescence.
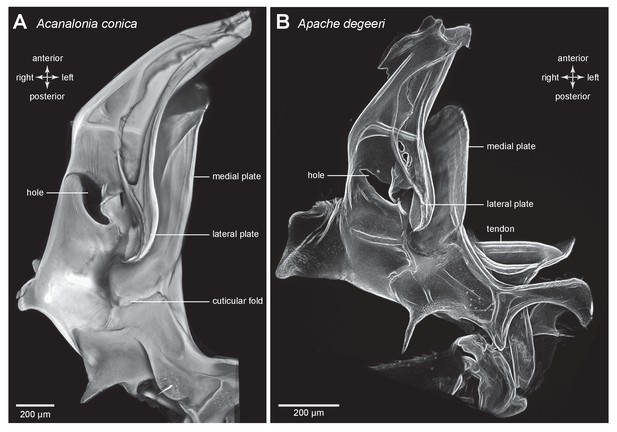
Structure of a pleural arch.
(A) The right pleural arch of the Acanaloniid planthopper Acanalonia conica viewed ventrally. The lateral plate of the arch is reinforced by antero-posterior infoldings, and the medial plate is thin but curved. The region between these two plates is normally filled with resilin. (B) The right pleural arch of the Derbid Apache degeeri viewed ventrally and in which edges have been emphasised (using ‘find edges’ filter in ImageJ). Again the region delineated by the two plates is normally filled with resilin. The arches were imaged using red autofluorescence.
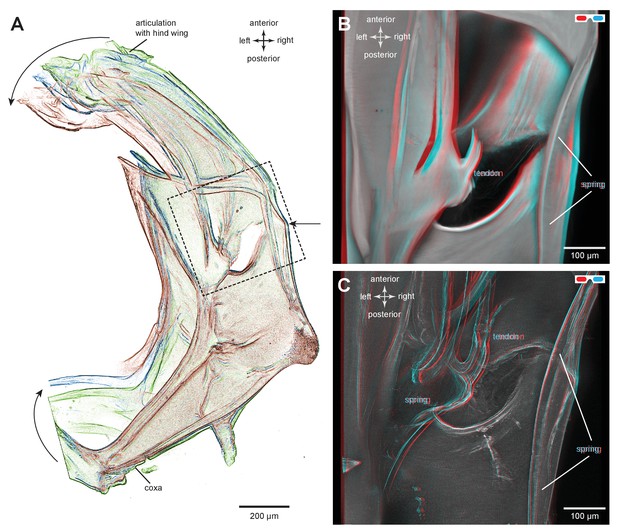
Structures associated with the bending of the pleural arches in planthoppers.
(A) Outlines of the right pleural arch viewed dorsally of the Flatid planthopper Flatormenis proxima taken from three individual frames of a movie (Supplementary material, Video 2) whilst a bending force was applied. The straight arrow points to the position of the region about which bending occurred and the curved arrows indicate the direction of bend at the anterior and posterior ends. (B) An expanded dorsal view of the area in (A) marked by the dashed box, showing the insertion of a tendon from a small muscle and the hinge region of the pleural arch. (C) The same image in which edges are emphasised to reveal a potential spring underneath the tendon insertion and a second potential curved spring on the right edge. These images can be seen in 3-D rotational views in Supplementary Material, Video 3.
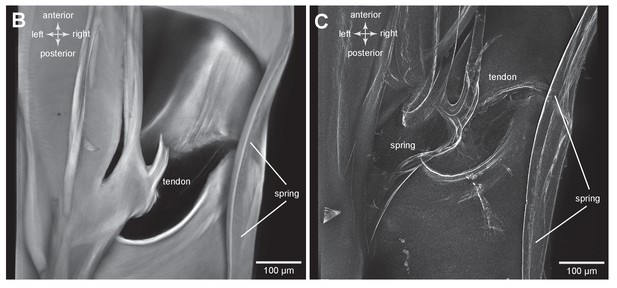
Structures associated with the bending of the pleural arches in planthoppers.
(B) An expanded dorsal view of the area in (A) marked by the dashed box, showing the insertion of a tendon from a small muscle and the hinge region of the pleural arch. (C) The same image in which edges are emphasised to reveal a potential spring underneath the tendon insertion and a second potential curved spring on the right edge. These images can be seen in 3-D rotational views in Supplementary Material, Video 3.
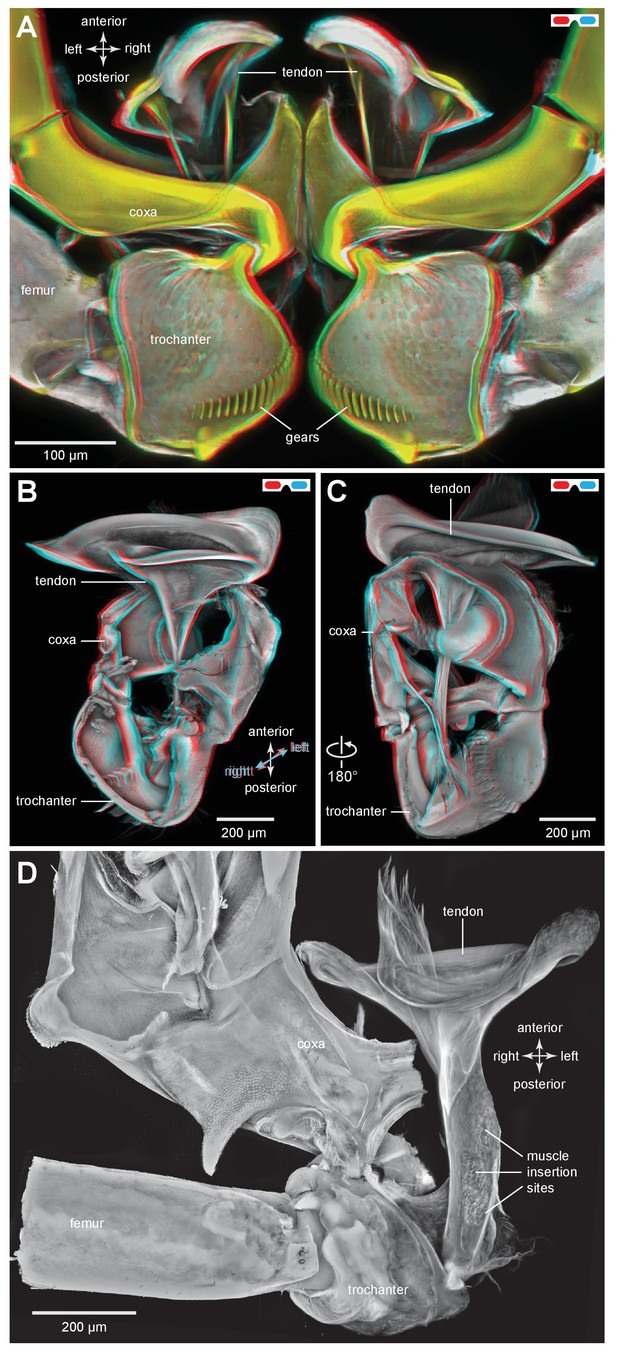
The tendon of the trochanteral depressor muscle in planthoppers.
(A) Three dimensional image, viewed dorsally, of the depressor tendons and proximal hind leg joints of a nymph of Stenocranus acutus. The helical end of the tendon on which the main body of fibres of the trochanteral depressor muscle (M133b/c) insert, protrudes in the thorax beyond the anterior edge of the coxa. The trochantera and femora of both hind legs are in their almost fully levated position so that the external coxal protrusions are close to engaging with the femora. The trochantera were displaced laterally so that their cog wheels are not engaged. (B) The tendon and proximal joints of the right side of the thorax of a nymph of Metcalfa pruinosa viewed medially. (C) The tendon and proximal joints of the right side of the thorax of a nymph of Acanalonia conica viewed ventrally. (D) The ventral wall of the right coxa of an adult Metcalfa pruinosa was removed to reveal the tendon and its insertion on the trochanter. Insertion sites of a small muscle along the straight part of the tendon are visible.
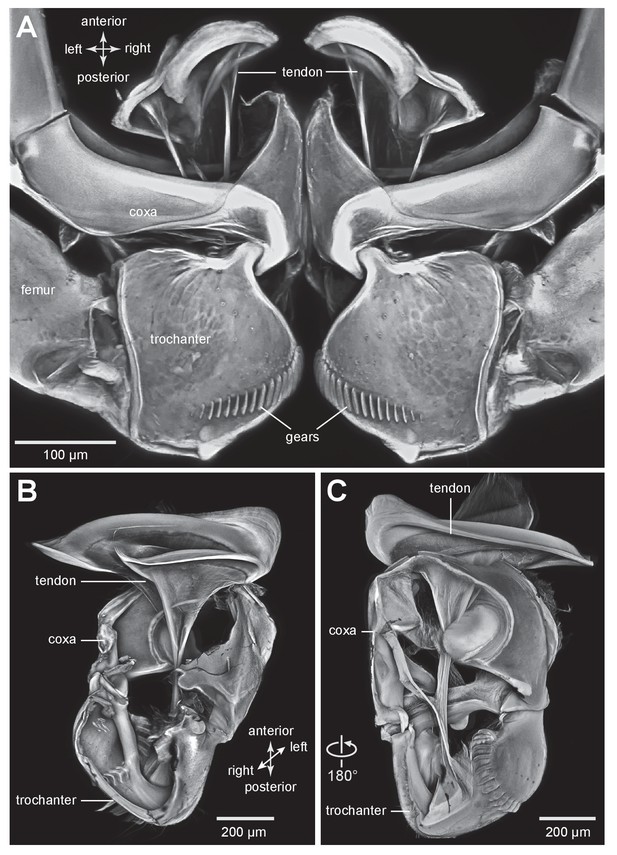
The tendon of the trochanteral depressor muscle in planthoppers.
(A) Depressor tendons and proximal hind leg joints of a nymph of Stenocranus acutus viewed dorsally. The helical end of the tendon on which the main body of fibres of the trochanteral depressor muscle (M133b/c) insert, protrudes in the thorax beyond the anterior edge of the coxa. The trochantera and femora of both hind legs are in their almost fully levated position so that the external coxal protrusions are close to engaging with the femora. The trochantera were displaced laterally so that their cog wheels are not engaged. (B) The tendon and proximal joints of the right side of the thorax of a nymph of Metcalfa pruinosa viewed medially. (C) The tendon and proximal joints of the right side of the thorax of a nymph of Acanalonia conica viewed ventrally.
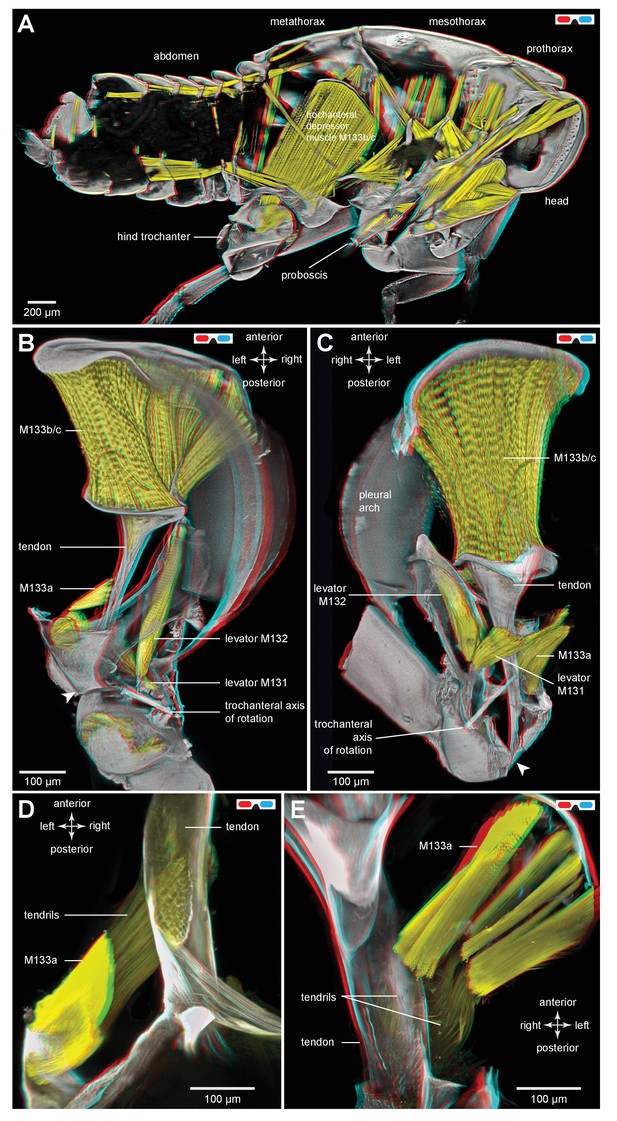
Muscles controlling movements of the hind trochantera of planthoppers.
(A) Medial view of a whole nymph of Metcalfa pruinosa. The muscles are shown in yellow. (B) Dorsal view of the right metathorax of a nymph with the right hind leg almost fully depressed, showing: the large trochanteral depressor muscle (M133b/c) with its origin in the anterior metathorax, the smaller part of this muscle (M133a) originating on the medial wall of the coxa, and a trochanteral levator muscle (M132). The two parts of the depressor muscle insert on different regions of the same tendon. The white arrowhead here (and in C) shows the insertion of the tendon on the medial wall of the trochanter. The line between the dorsal and ventral pivots of the trochanter indicates the axis of its rotation about the coxa. (C) The right half of the thorax viewed ventrally and with the hind leg fully levated. The axis of trochanteral rotation is again indicated by the line between the pivots. (D) Dorsal view of the insertions, by narrower tendrils, of fibres from the small part of the muscle (M133a) onto the main tendon in an adult. The hind leg was depressed as in (B). (E) Ventral view of these same insertions.
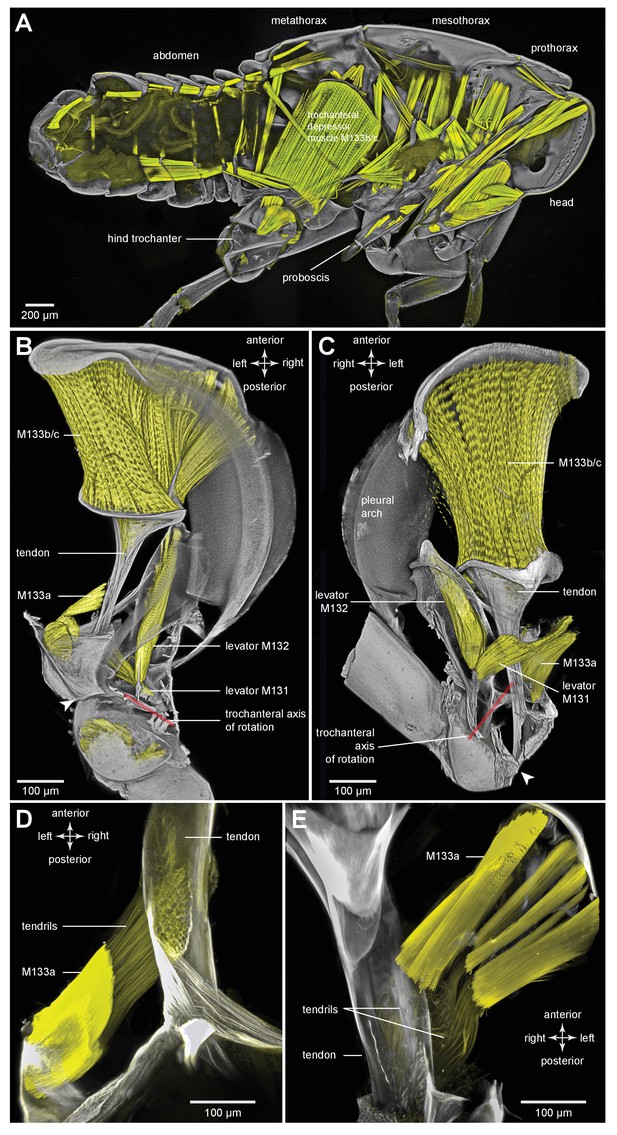
Muscles controlling movements of the hind trochantera of planthoppers.
(A) Medial view of a whole nymph of Metcalfa pruinosa. The muscles are shown in yellow. (B) Dorsal view of the right metathorax of a nymph with the right hind leg almost fully depressed, showing: the large trochanteral depressor muscle (M133b/c) with its origin in the anterior metathorax, the smaller part of this muscle (M133a) originating on the medial wall of the coxa, and a trochanteral levator muscle (M132). The two parts of the depressor muscle insert on different regions of the same tendon. The white arrowhead here (and in C) shows the insertion of the tendon on the medial wall of the trochanter. The red line between the dorsal and ventral pivots of the trochanter indicates the axis of its rotation about the coxa. (C) The right half of the thorax viewed ventrally and with the hind leg fully levated. The axis of trochanteral rotation is again indicated by the red line between the pivots. (D) Dorsal view of the insertions, by narrower tendrils, of fibres from the small part of the muscle (M133a) onto the main tendon in an adult. The hind leg was depressed as in (B). (E) Ventral view of these same insertions.
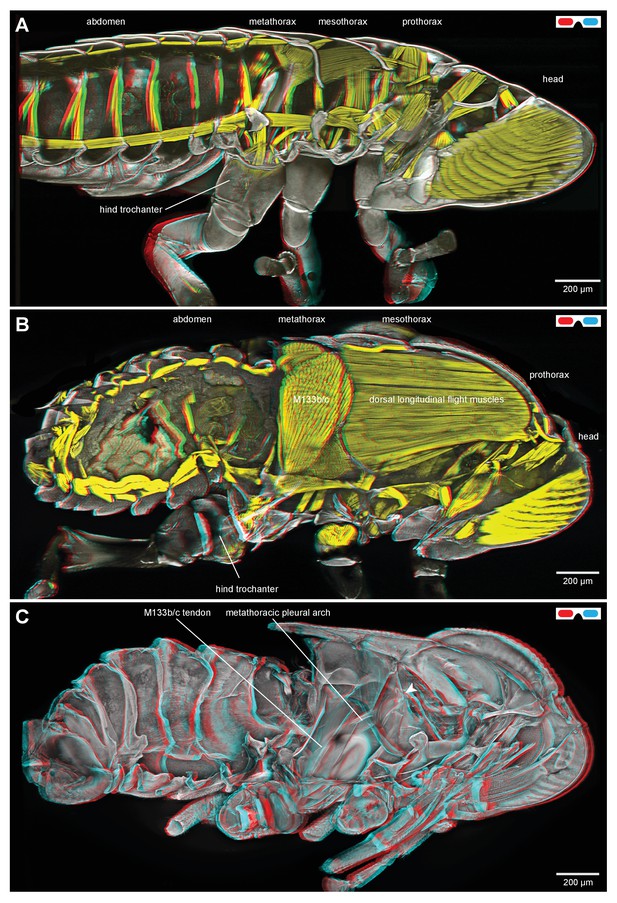
Thoracic muscles in nymphal and adult froghoppers.
(A) Medial view of a nymph of the froghopper Clastoptera obtusa. (B) Medial view of an adult of this species showing the large metathoracic trochanteral depressor muscle, and large mesothoracic flight muscles. (C) The same specimen as in (B) to show the internal skeleton of the thorax after removal of the soft tissues. The articulation of the metathoracic pleural arch with the mesothoracic pleural arch at the origin of the trochanteral depressor M133c is marked with a white arrowhead.
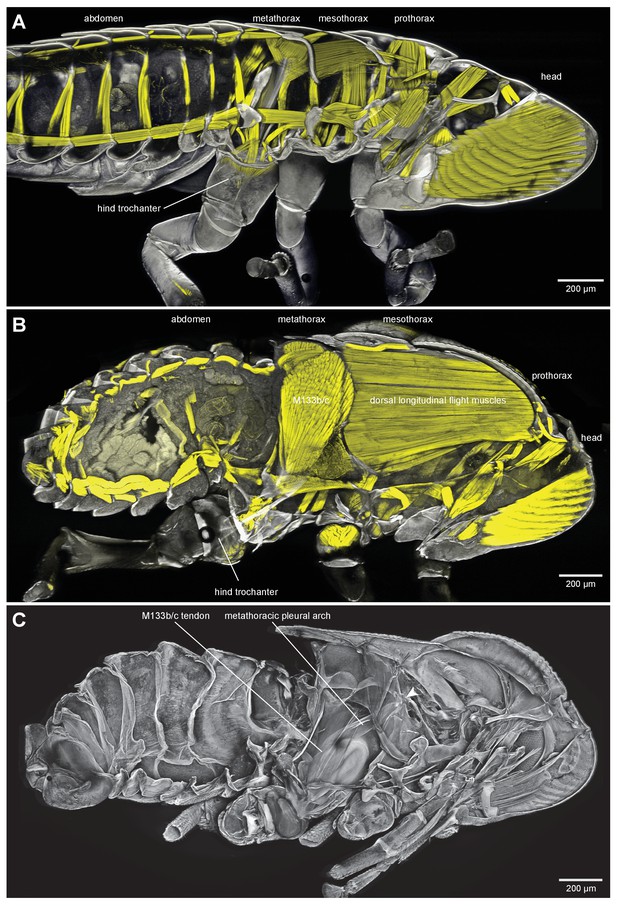
Thoracic muscles in nymphal and adult froghoppers.
(A) Medial view of a nymph of the froghopper Clastoptera obtusa. (B) Medial view of an adult of this species showing the large metathoracic trochanteral depressor muscle, and large mesothoracic flight muscles. (C) The same specimen as in (B) to show the internal skeleton of the thorax after removal of the soft tissues. The articulation of the metathoracic pleural arch with the mesothoracic pleural arch at the origin of the trochanteral depressor M133c is marked with a white arrowhead.
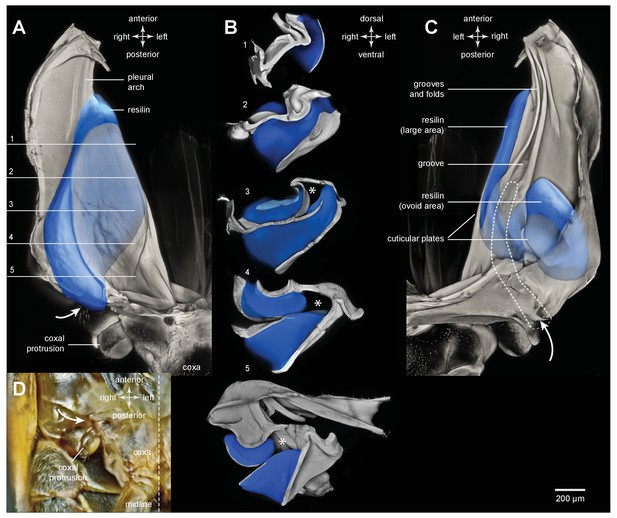
Resilin and the pleural arches of an adult froghopper.
(A) Ventral view of the right pleural arch of Lepyronia quadrangularis. The large region of resilin (blue) is sandwiched between an inner and an outer layer of cuticle. (B) Sections cut at the five levels indicated by the lines and numbers in (A). The asterisks indicate a tunnel-like space in the pleural arch. (C) Dorsal view of the right pleural arch in which two regions of resilin are visible; the first, long and medial strip is the edge of the large region visible ventrally in (A); the second, ovoid region is more lateral and posterior, and is separated from the first by a thin central layer of cuticle and is bounded by an outer layer of cuticle. The dashed outline indicates the path of the tunnel from its entrance near the coxal protrusion (curved white arrow) to its blind ending at the level where the groove forms in the medial plate of the pleural arch. (D) Photograph of the ventral surface of the right side of the metathorax to show the external entrance to the tunnel (curved white arrow) relative to the coxal protrusion and the right hind coxa.
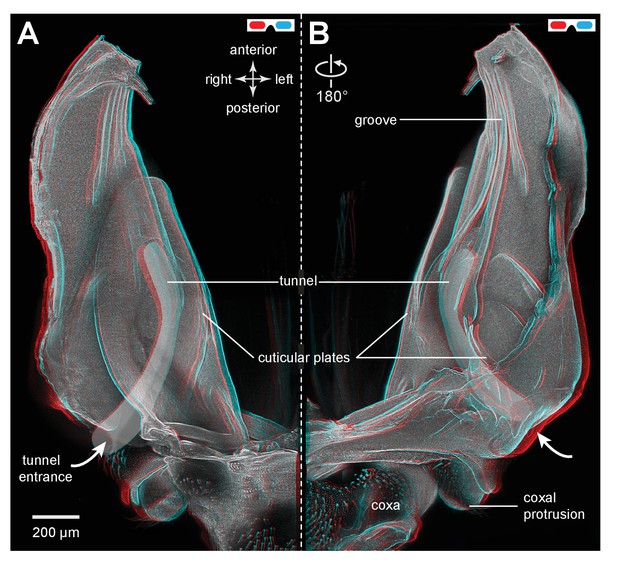
Cuticular plates of a pleural arch that surround the resilin in an adult froghopper.
(A) Three dimensional ventral view of a right pleural arch of Lepyronia quadrangularis showing the medial and lateral plates of cuticle that normally encase the large region of resilin. (B) Three dimensional dorsal view of the right pleural arch showing the more posterior plates that normally encase the smaller ovoid region of resilin. A drawing of the tunnel that connects with the outside air (curved white arrows) near the coxal protrusion is superimposed on both (A) and (B). The structures were imaged using red autofluorescence.
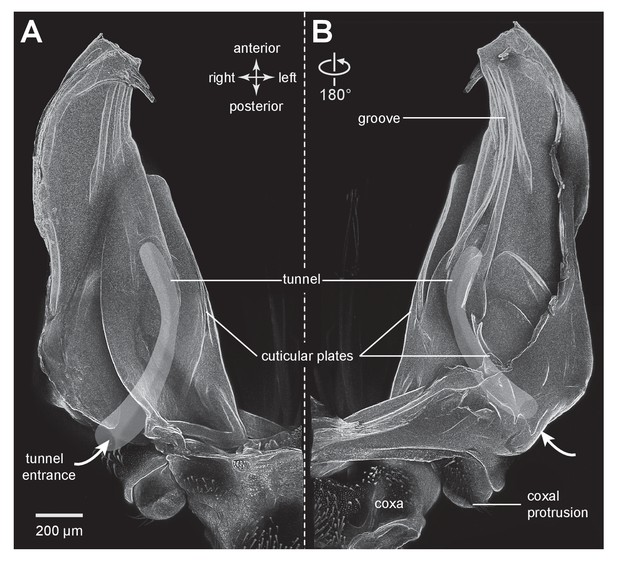
Cuticular plates of a pleural arch that surround the resilin in an adult froghopper.
(A) Ventral view of a right pleural arch of Lepyronia quadrangularis showing the medial and lateral plates of cuticle that normally encase the large region of resilin. (B) Dorsal view of the right pleural arch showing the more posterior plates that normally encase the smaller ovoid region of resilin. A drawing of the tunnel that connects with the outside air (curved white arrows) near the coxal protrusion is superimposed on both (A) and (B). The structures were imaged using red autofluorescence.
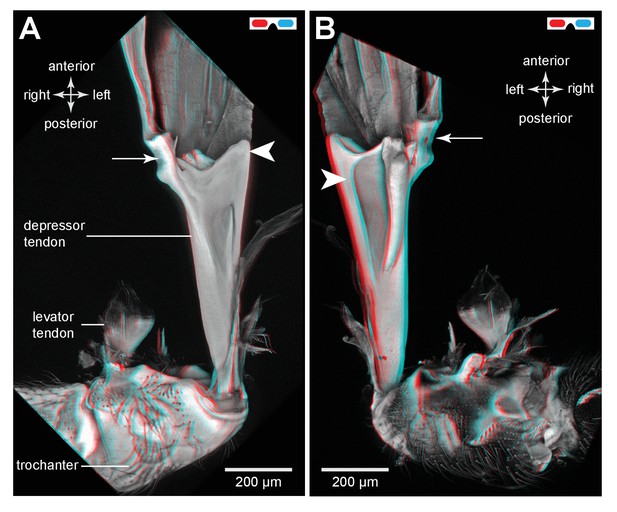
The tendon of the trochanteral depressor muscle in an adult froghopper.
(A) Three dimensional ventral view of the tendon of Lepyronia quadrangularis as it passes from the thorax, through the coxa to insert on the medial wall of the trochanter. Within the thorax the tendon bifurcates into a thick branch (arrowhead) onto which the fibres of the depressor muscle (M133c) inserts and a thinner lateral branch (arrow) on which the pinnate fibres of M133b insert. (B) Three dimensional dorsal view of the same preparation as in (A). Shorter tendons of the trochanteral levator muscles are also shown. The midline is close to the right of the image in (A) and to the left of the image in (B).
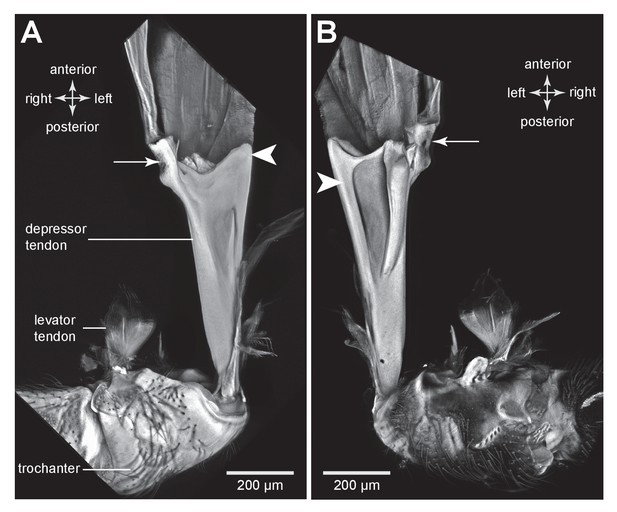
The tendon of the trochanteral depressor muscle in an adult froghopper.
(A) Ventral view of the tendon of Lepyronia quadrangularis as it passes from the thorax, through the coxa to insert on the medial wall of the trochanter. Within the thorax the tendon bifurcates into a thick branch (arrowhead) onto which the fibres of the depressor muscle (M133c) inserts and a thinner lateral branch (arrow) on which the pinnate fibres of M133b insert. (B) Dorsal view of the same preparation as in (A). Shorter tendons of the trochanteral levator muscles are also shown. The midline is close to the right of the image in (A) and to the left of the image in (B).
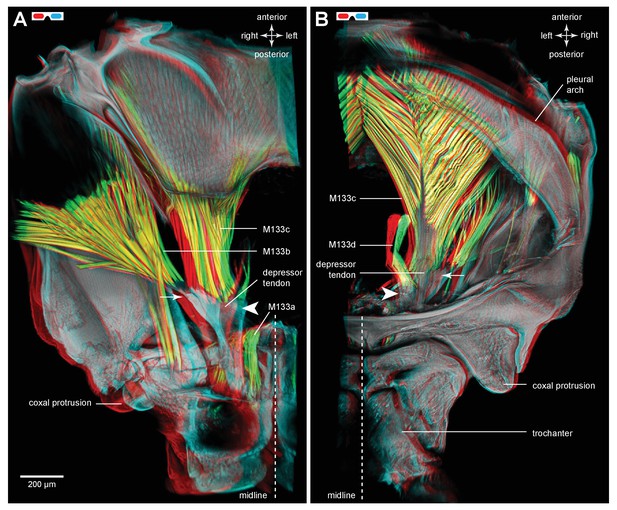
Muscles controlling movement of the hind trochantera of adult froghoppers.
(A) Ventral view of the right side of the metathorax of Clastoptera obtusa. The insertion site of muscle M133b is occluded by darkly-pigmented cuticle. (B) Dorsal view of the right side showing the insertion of M133d and the pinnate arrangement of fibres in M133b.
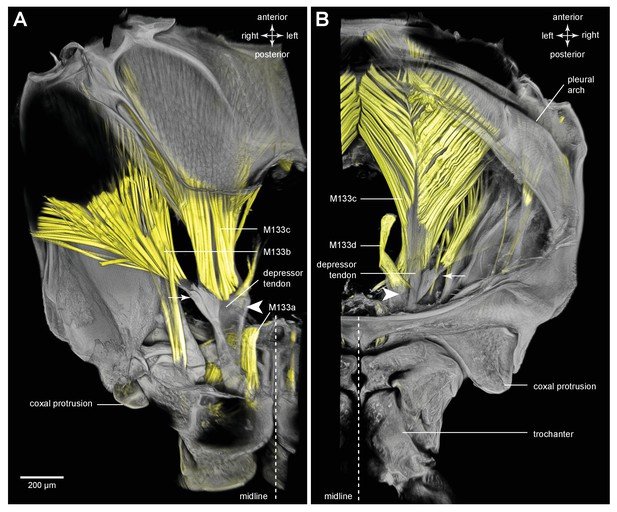
Muscles controlling movement of the hind trochantera of adult froghoppers.
(A) Ventral view of the right side of the metathorax of Clastoptera obtusa. The insertion site of muscle M133b is occluded by darkly-pigmented cuticle. (B) Dorsal view of the right side showing the insertion of M133d and the pinnate arrangement of fibres in M133b.
Videos
Bending of the pleural arches of a planthopper during natural jumping.
High speed movie taken at 30 000 frames per second and with an exposure time of 0.03 ms of the jumping movements of an adult planthopper Issus coleoptratus. Before a jump, the hind legs are moved into their fully levated position so that the coxae are located anteriorly within the metathorax. The large trochanteral depressor muscles then contract without moving the hind legs, resulting in the pleural arches being bent. As the jump is released, the pleural arches unfurl and both coxae move backwards rapidly as a unit. The more distal parts of the two hind legs also move rapidly in unison.
Imposed bending movements of a pleural arch of a planthopper.
The isolated pleural arch of the planthopper Flatormenis proxima was restrained between coverslips and a sustained force was applied to the top coverslip while a scan was made. The movie consists of three frames showing the pleural arch in a relaxed state and two compressed conformations.
Three dimensional rotational view of the jumping muscles in an adult planthopper Stenocranus acutus.
Both hind legs are in the fully levated position. The small parts of the trochanteral depressor muscles (M133a and M133d) are best viewed during the ventral portion of the rotation and the levator muscles (M131, M132) during the dorsal part.
Three dimensional rotational view of the jumping muscles in an adult froghopper Clastoptera obtusa.
Right half of the metathorax with the leg fully extended. The origins of depressor muscle (M133b) are obscured by dark, opaque cuticle.
Tables
Summary of the stains and the excitation and emission wavelengths used when imaging specimens.
| Tissue | Staining | Excitation wavelengths, nm | Emission wavelengths, nm |
|---|---|---|---|
| Chitin | Autofluorescence | 561 | 568–683 |
| Chitin and Muscles (nymphs) | Calcofluor White | 405 | 410–570 |
| Texas Red Phalloidin | 594 | 599–660 | |
| Chitin and Muscles (adults) | Autofluorescence | 405 | 410–465 |
| Texas Red Phalloidin | 594 | 599–660 | |
| Chitin and Resilin | Autofluorescence | 405 | 410–465 |
| Acridine Orange | 488 | 520–580 |
Volumes of resilin in a pleural arch, and ratios of both arches relative to body mass in the planthopper Thionia bulllata and the froghopper Lepyronia quadrangularis. See Figure 3Ai, ii for definitions of width, thickness and length and Figure 9A–C for definitions of the two resilin regions in froghoppers.
| Width, µm | Thickness, µm | Length, µm | Volume one arch, mm3 | Ratio, both arches: body mass, mm3 g−1 | |
|---|---|---|---|---|---|
| Planthopper | 500 | 200 | 1250 | 0.138 | 12.8 |
| Froghopper | |||||
| Large region | 710 | 290 | 1450 | 0.121 | |
| Ovoid region | 440 | 150 | 710 | 0.044 | |
| Total | 0.165 | 18.7 |





