Restraint of presynaptic protein levels by Wnd/DLK signaling mediates synaptic defects associated with the kinesin-3 motor Unc-104
Figures
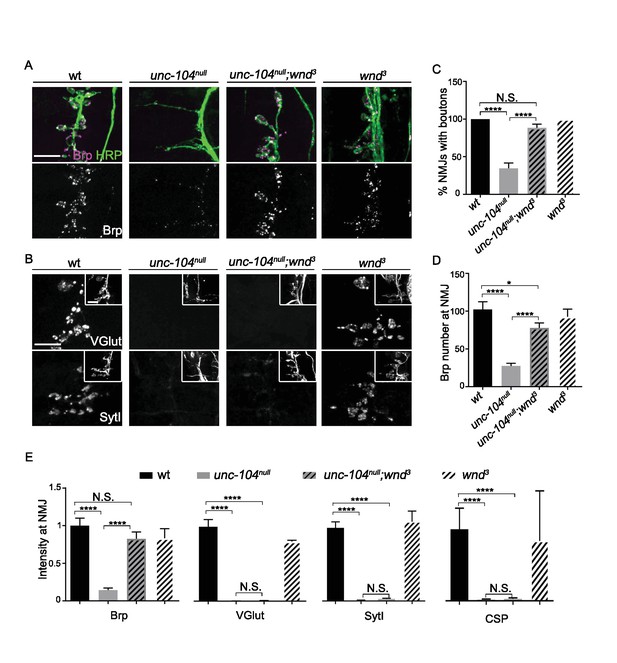
AZ transport and Synaptic bouton formation but not SVP transport defects in unc-104-null mutants are rescued by mutations in wnd.
Representative images of ISNb neuromuscular junction (NMJ) terminals at muscles 6, 7, 12 and 13 at embryonic stage 17 (20–21 hr AEL). We observed similar results with multiple independently generated unc-104-null alleles including unc-104P350, unc-104170and unc-10452. Representative images with unc-104P350 are shown here. (A) Images of ISNb NMJ terminals immunostained for a neuronal membrane marker to show axons and terminal boutons (HRP, green in upper panel) and the AZ component Brp (magenta in upper panel, white in lower panel). Boutons failed to form and AZs failed to localize to NMJ in unc-104null mutants. Both defects were restored in unc-104null(P350);wnd3 mutants. (B) Images of ISNb NMJ terminals immunostained for presynaptic vesicle proteins (VGlut, upper panels; SytI, lower panel). The insets show HRP staining of the nerve terminals. Note VGlut and SytI failed to localize to NMJ in both unc-104null(P350) and unc-104null(P350); wnd3 mutants. (C) The bouton formation defect was quantified as the percentage of ISNb NMJ terminals that contained at least one presynaptic varicosity, identified by HRP staining (and scored while blind to genotype). This method over-estimates the actual number of boutons in unc-104null mutants, since any varicosity within any of the ISNb terminals (on muscles 6, 7, 12 or 13) was counted. (D) The number of AZs (identified as Brp punctae) formed at ISNb NMJ terminals (on muscles 6, 7, 12 and 13). (E) The total (sum) intensity of Brp, VGlut, SytI and CSP measured across the ISNb NMJ terminals. All data are represented as mean ±SEM; At least 9 animals and 20 ISNb NMJ terminals were examined per genotype; N.S., not significant; ****p<0.0001, *p<0.05; Tukey test for multiple comparison; Scale bar, 10 μm.
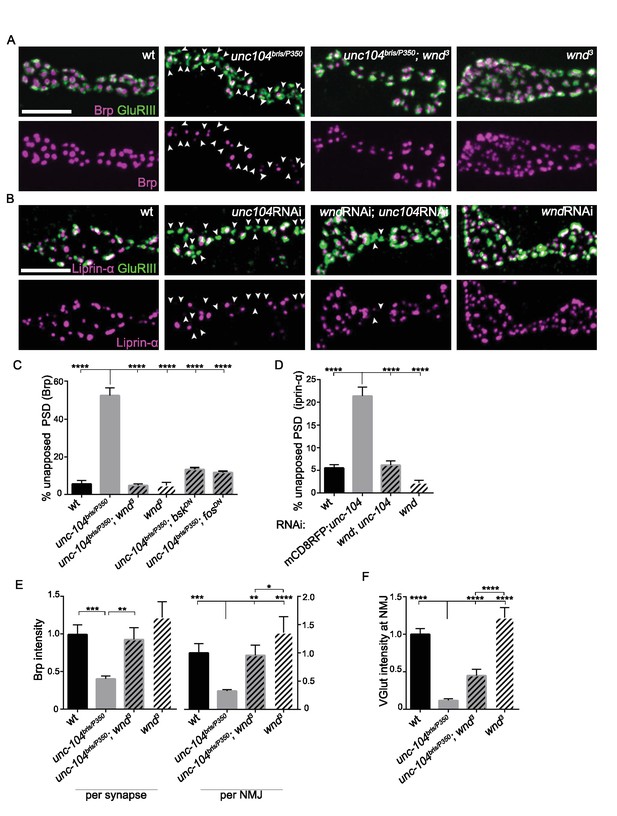
Wnd signaling pathway is required for the presynaptic defects in unc-104-hypomorph mutants.
(A–B) Representative confocal images of third instar larval neuromuscular junctions (NMJ) at muscle 4. Postsynaptic densities (PSDs) identified by GluRIII staining (Green) that lacked apposing AZ components Brp (magenta in A), or Liprin-α (magenta in B) are highlighted by arrowheads. Note that when determining the absence of AZ components, the pixel saturation threshold was reduced to ensure detection ofweak signals (this resulted in saturation of certain pixels in wt controls. For more details, see Experimental Procedures.). (A) Alignment of postsynaptic GluRIII (green) with presynaptic AZ component Brp (magenta) in Canton-S (wt), unc-104bris/P350, unc-104bris/P350;wnd3 and wnd3. Unc-104bris is a partial loss of function mutation (Kern et al., 2013), while unc-104P350 is a null allele (Barkus et al., 2008). wnd3 is a presumptive null mutation in wnd (Collins et al., 2006). (B) Alignment of postsynaptic GluRIII (green) with presynaptic AZ component Liprin-α (magenta). Liprin-α-GFP was driven by rab7 promoter and UAS-unc-104 RNAi and UAS-wnd RNAi were driven by neuronal elav-Gal4. UAS-mCD8-RFP was used as a control for UAS dosage. (C–D) The percentage of PSDs which lack any trace of apposing presynaptic AZ protein, based on (C) Brp or (D) Liprin-α-GFP. (E) Signal intensity for Brp staining at individual synapses or summed across the entire NMJ terminal (at muscle 4), normalized to that in wt (Canton S) animals. (F) The total intensity of Vglut protein measured across the entire synaptic NMJ terminal at the muscle 4, normalized to that in wt (Canton S). All data are represented as mean ±SEM; At least 6 animals and 12 muscle 4 NMJ terminals were examined per genotype; ****p<0.0001, ***p<0.001, **p<0.01, *p<0.05. Scale bar, 5 μm. For additional data, see Figure 2—figure supplements 1–2.
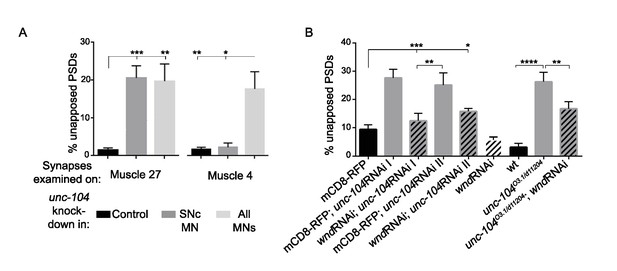
Wnd acts in a neuron-specific and cell-autonomous manner to mediate presynaptic defects in unc-104-hypomorph mutants, (related to Figure 2).
(A) The percentage of GluRIII-labeled PSDs which lack apposing AZ components. Two different Gal4 drivers were used to express unc-104 RNAi (vdrc 23465), either specifically in SNc neurons, which innervate muscle 26, 27 and 29 (m12-Gal4) or all motoneurons (OK6-Gal4). Knockdown in SNc neurons caused synaptic assembly defects on innervated muscles (quantified for muscle 27) but not on other muscles (quantified for muscle 4). Control animals share UAS-unc-104 RNAi but lack Gal4. (B) The percentage of unapposed PSDs, defined (as in A and in Figure 2) by the absence of Brp at synapses labeled by GluRIII, was rescued when wnd was neuronally knocked down by RNAi together with unc-104 (2 independent RNAi lines: vdrc 23465, I and TRiP BL43264, II) or in unc-104-hypomorph (O3.1/d11204) mutants. All UAS-RNAi were driven by BG380-Gal4. UAS-Dicer was co-expressed to enhance RNAi efficiency with unc-104RNAi II. UAS-mCD8-RFP was used as a control for dosage of UAS lines. All data are represented as mean ±SEM; N.S., not significant; ****p<0.0001, ***p<0.001, **p<0.01,*p<0.05; Tukey test for multiple comparison.
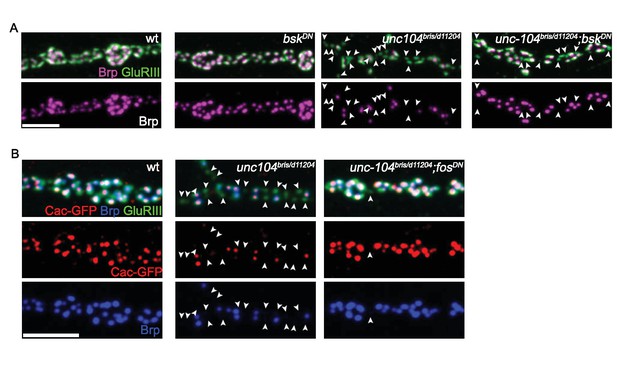
Wnd signaling components JNK/Bsk and Fos promotes presynaptic defects in the localization of AZs and release machinery in unc-104-hypomorph mutants, (related to Figure 2).
(A–B) Representative confocal images of NMJ synapses labeled with (A) Brp and (B) the L-type calcium channel Cacophony (cac)-GFP. Inhibition of (A) jnk/bsk or (B) fos via expression of a Dominant Negative (DN) transgene rescued the synaptic apposition defect of unc-104 mutants. (A): Control (OK319-Gal4), bskDN (OK319-Gal4; UAS-bskDN), unc-104bris (OK319-Gal4, unc104bris/d11204) and unc-104bris; bskDN (OK319-Gal4, unc104bris/d11204; UAS-bskDN). (B): Control (OK319-Gal4; UAS-cac-GFP), unc-104bris (OK319-Gal4, unc104bris/d11204; UAS-cac-GFP) and unc-104bris; fosDN (OK319-Gal4, unc104bris/d11204; UAS-bskDN, UAS-cac-GFP). GluRIII-labeled PSDs that lack apposing AZs (Cac-GFP or Brp) are highlighted by arrowheads. unc104d11204 is a null allele. Scale bar, 5 μm.
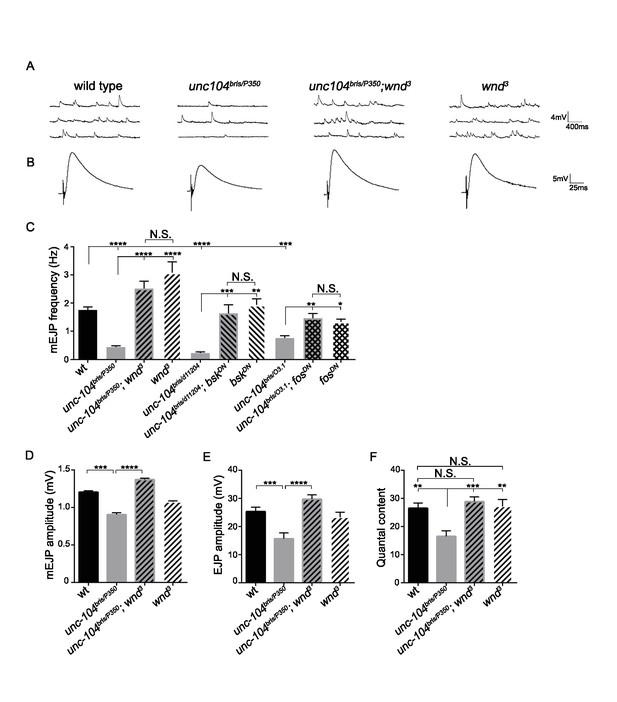
The synaptic transmission defect in unc-104 mutants is suppressed by wnd mutations.
(A–B) Representative electrophysiological traces of (A) miniature Excitatory Junctional Potentials (mEJP) and (B) Evoked Excitatory Junctional Potentials (EJP) recorded from muscle 6 of third instar larvae. (C) Quantification of impaired mEJP frequency for unc-104-hypomorph mutants and their rescue by wnd mutations or expression of dominant negative (DN) bsk or fos (driven by elav-Gal4 and BG380-Gal4, respectively). Representative traces for bskDN and fosDN are shown in Figure 3—figure supplement 1. (D–F) Quantification of (D) mEJP amplitude, (E) EJP amplitude and (F) quantal content (corrected for nonlinear summation). See also Figure 3—figure supplement 1 for additional quantification of bskDN and fosDN data. Unc-10403.1 and unc-104bris are hypomorphic alleles, while unc-104P350 and unc-104d11204 are null alleles. wnd3 is a presumptive null mutation in wnd (Collins et al., 2006). wt animals are Canton S. All data are represented as mean ±SEM; N.S., not significant; ****p<0.0001, ***p<0.001, **p<0.01, *p<0.05. Tukey test for multiple comparison. For additional data, see Figure 3—figure supplements 1–4.
-
Figure 3—source data 1
Measurements of EJP and mEJP.
The file contains data from 3 independent experiments and 6 source data sheets: EJP amplitude, corrected EJP amplitude and Quantal content, mEJP amplitude and frequency of CS, unc-104, unc-104;wnd and wnd. This is corresponding to Figure 3.
- https://doi.org/10.7554/eLife.24271.012
-
Figure 3—source data 2
Measurements of EJP and mEJP.
The file contains EJP amplitude, corrected EJP amplitude and Quantal content, mEJP amplitude and frequency of CS, unc-104bris/d11204, unc-104bris/d11204;bskDN and bskDN. This is corresponding to Figure 3C and Figure 3—figure supplement 1.
- https://doi.org/10.7554/eLife.24271.013
-
Figure 3—source data 3
Measurements of EJP and mEJP.
The file contains EJP amplitude, corrected EJP amplitude and Quantal content, mEJP amplitude and frequency of CS, unc-104bris/O3.1, unc-104bris/O3.1;fosDN and fosDN. This is corresponding to Figure 3C and Figure 3—figure supplement 1.
- https://doi.org/10.7554/eLife.24271.014
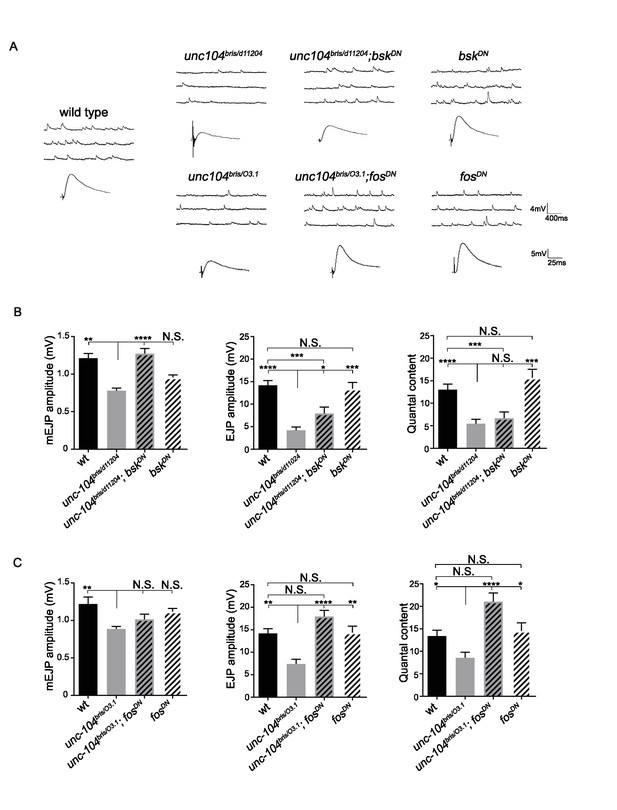
Wnd signaling components JNK/Bsk and Fos promotes synaptic transmission defects in unc-104-hypomorph mutants, (related to Figure 3).
(A) Representative electrophysiological traces of mEJP and EJP recorded from muscle 6 of third instar female larvae. The genotypes are: wt (Canton-S), unc-104bris/d11204, bskDN (elav-Gal4;; UAS-bskDN) and unc-104bris/d11204; bskDN (elav-Gal4; unc104bris/d11204; UAS-bskDN), unc-104bris/O3.1, unc-104bris/O3.1; fosDN (BG380-Gal4; unc104bris/d11204; UAS-fosDN) and fosDN (BG380-Gal4;; UAS-fosDN). (B–C) Quantification of mEJP amplitude, EJP amplitude and quantal content (corrected for nonlinear summation). All data are represented as mean ±SEM; N.S., not significant; ****p<0.0001, ***p<0.001, **p<0.01, *p<0.05. Tukey test for multiple comparison.
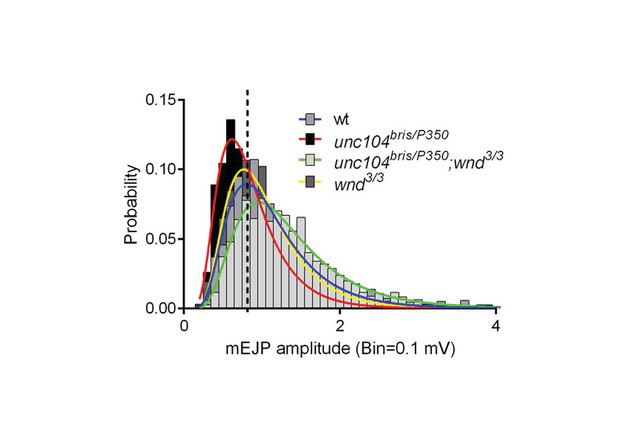
mEJP amplitude is reduced in unc-104 mutants and restored in unc-104; wnd double mutants, (related to Figure 3).
Distribution of mEJP amplitudes fit with log Gaussian. The center amplitude was determined by the peak of curve: wt (Canton S) wild type (0.80 mV), unc104bris/P350 (0.61 mV), unc104bris/P350; wnd3/3 (0.95mV) and wnd3/3 (0.76 mV). Note that both the center amplitude and the amplitude distribution were restored in double mutants.
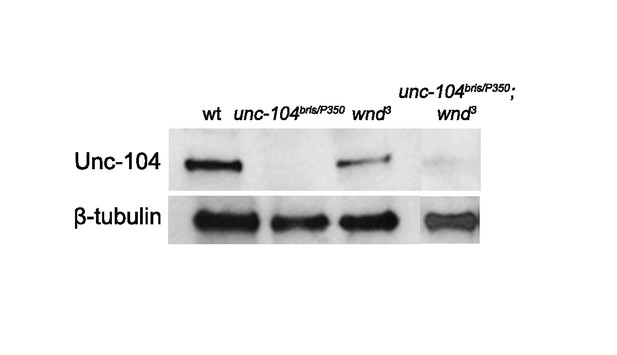
The expression of Unc-104 is barely detectable in unc-104-hypomorph mutants, (related to Figure 3).
Western blot of larva brains for Unc-104 and β-tubulin for wt (Canton S), unc-104bris/P350, wnd3 and unc-104bris/P350;wnd3.
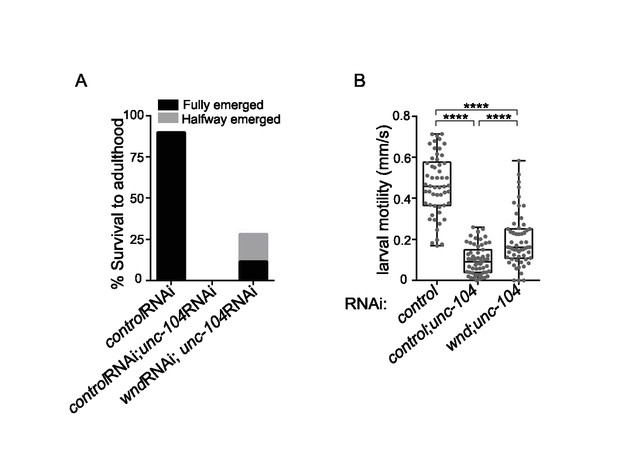
Defects in larval motility and lethality of unc-104-hypomorph mutants were only partially suppressed by wnd mutations, (related to Figure 3).
(A) Percentage pupae that survive to adulthood. 60 offspring third instar larvae of indicated genotype were transferred to grape plates and raised at 29°C to increase RNAi knock-down efficiency. The number of adults emerging from pupae were counted over the next 14 days. Adults with anterior the half of their body (head, thorax and foreleg) out of pupae, but with the posterior half body (abdomen and posterior 4 legs) stuck in the pupal case were scored as 'halfway' emerging adults. UAS-RNAi lines were driven by OK371-Gal4. UAS-Octβ2R RNAi and UAS-moody RNAi were used as control RNAi lines for UAS dosage, since no phenotypes were observed for either RNAi line when expressed in in neurons. The percentage survival was measured as the number of emerged or halfway emerged adults divided by the total number of larvae. (B) Motility of third instar larvae was quantified from 2 min videos (30 frames per second). After selection of each genotype animals put on grape plates for 1 hr to adapt before recording. For each larva, the distance and time traveled after it was released in a new plate and before it reached the plate walls was measured with the MB-Ruler (Markus Bader). A marker point in the middle of movement was only applied if the larva moved 45 degree away from the current direction and 2–5 marker points were set to determine the path. A total of 60 larvae were recorded and analyzed for each genotype. UAS-RNAi lines were driven by OK371-Gal4, as in (A). All data are represented as mean ±SEM; N.S., not significant; ****p<0.0001, ***p<0.001, **p<0.01,*p<0.05; Tukey test for multiple comparison.
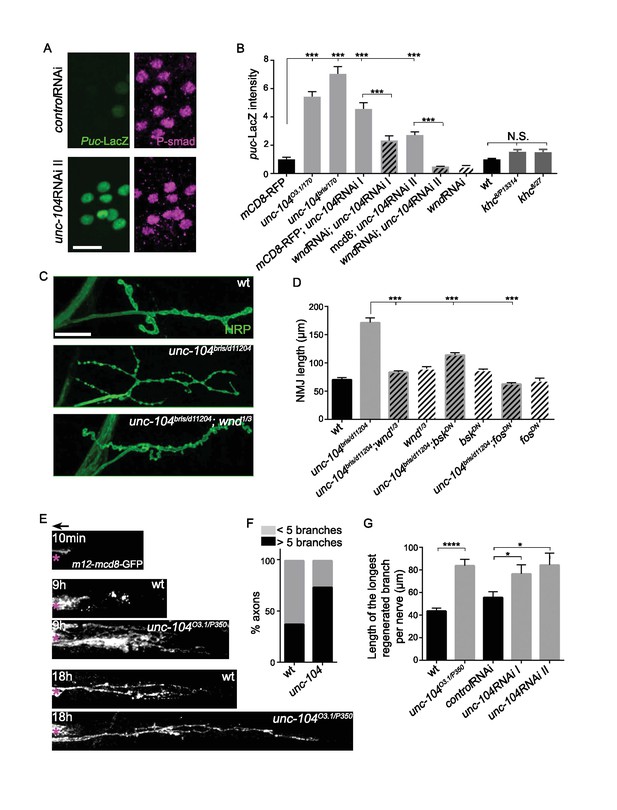
The Wnd signaling pathway is activated in unc-104 mutants.
(A) Images of motoneuron cell bodies near the midline of larval ventral nerve cord, immunostained for motoneuron nuclear marker phospho-SMAD (magenta) and the Wnd/JNK nuclear reporter puc-LacZ (green). UAS-RNAi lines (including UAS-moody RNAi as a control) were driven by pan-neuronal BG380-Gal4. At least 3 dorsal abdominal clusters of motoneurons per VNC from 6 animals per genotype were examined. (B) Quantification of puc-lacZ expression in multiple unc-104 mutant backgrounds. Unc-10403.1 and unc-104bris are hypomorphic alleles, while unc-104170 is a null allele. In addition we tested two independent unc-104 RNAi lines (vdrc 23465, I and TRiP BL43264, II). Unc-104 RNAi II was accompanied with Dicer2 expression to facilitate the knock-down. Co-expression of wnd-RNAi, compared to UAS-mCD8-ChRFP reduced the effect of unc-104 knockdown. The ‘control’ genotype has UAS-mCD8-ChRFP, (which serves as a control for dosage of UAS lines). Quantification methods are described in Experimental Procedures. (C) Presynaptic bouton morphology and branching at the NMJ, viewed via immunostaining for HRP (membrane marker) at muscle 4. The presynaptic arbor was over-branched in unc-104 bris mutants compared to wt (Canton S) control animals, and this was rescued in unc-104bris; wnd1/3 double mutants. (D) Quantification of presynaptic overgrowth was estimated by measuring the total NMJ length (from the most proximal to the most distal bouton of the presynaptic nerve terminal at muscle 4, labeled via anti-HRP staining). The data shown used unc-104bris and wnd1/3 mutations, while UAS-bskDN and UAS-fosDN were driven by elav-Gal4. wt control animals were Canton S. (E) Regenerative axonal sprouting of m12-Gal4, UAS-mcd8-GFP labeled axons 10 min, 9 hr or 18 hr after nerve crush from wt compared to unc-104O3.1(hypomorph)/P350(null() mutant animals. Asterisk (*) indicates the injury site and arrow indicates the direction of the cell body. By 9 hr after nerve crush injury, axons in wild type animal initiate new growth via short filopodia-like branches from the proximal axonal stump. At 18 hr, a few branches are stabilized and grow either towards the distal axon or the cell body. In comparison, unc-104 mutants showed a marked increase in new axonal branches at 9 hr, and at 18 hr these new axonal branches showed similar stabilization but extended nearly twice as far as wild type axons. (F) 9 hr after injury, the percentage of axons which contain more than 5 identifiable branches per axon. (G) The length of the longest branch per nerve at 18 hr after injury. unc-104O3.1/P350 mutant and 2 independent unc-104 RNAi lines and control RNAi (moody-RNAi) (driven by m12-Gal4) were examined. All data are represented as mean ±SEM; N.S., not significant; ****p<0.0001, ***p<0.001,*p<0.05, Tukey test for multiple comparison; Scale bar, 20 μm.
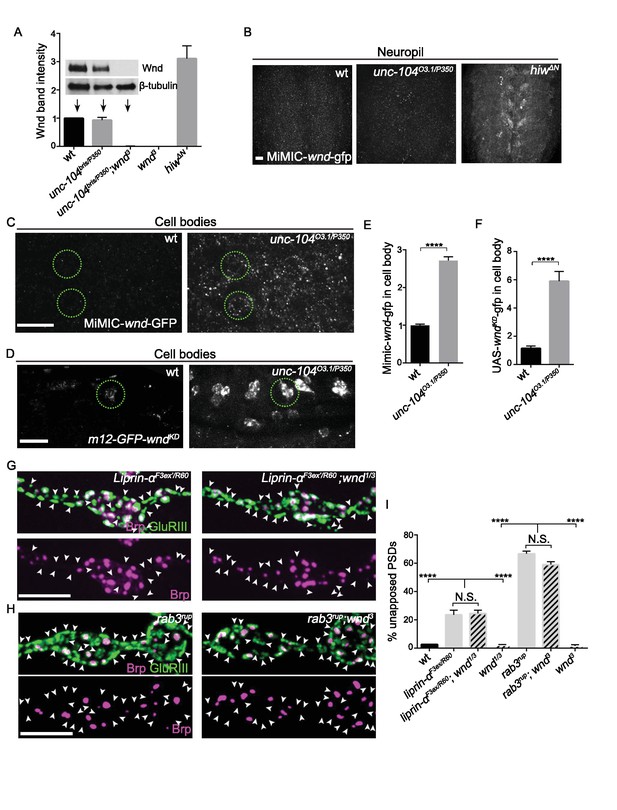
Wnd signaling is activated via an unconventional mechanism in unc-104 mutants.
(A) Representative Western blot of larval whole brain extracts for endogenous Wnd and β-tubulin, and quantification of Wnd levels normalized to β-tubulin band intensity (n ≥ 3). Mutants examined include unc-104bris(hypomoprh)/P350(null), wnd3 and hiwΔN. (B) Images of larval ventral nerve cords from MiMIC-wnd-GFP animals immunostained for GFP. Note the increased neuropil signal in hiw mutants, but not control or unc-104 mutants. This also indicates that the MiMIC-wnd-GFP is, like endogenous Wnd (Collins et al., 2006), subject to regulation by Hiw. (C) Images of motoneuron cell bodies in MiMIC-wnd-GFP animals immunostained with antibody against GFP. Compared to (B), these images are collected in a higher magnification and focal plane to view the motoneuron cell bodies. Two representative cell bodies are marked by green circles. At least 6 animals were examined per genotype. (D) Images of SNc motoneuron cell bodies via live-imaging in larvae expressing UAS-GFP-wndkd (which is kinase dead to avoid pathway activation) driven by m12-Gal4. A representative cell body is marked by a green circle. At least 6 animals were examined per genotype. (E–F) Quantification of GFP signal intensity from (C–D) in cell bodies, normalized to wt (control) animals. Note that ectopically expressed GFP-wnd kinase dead protein has a higher basal expression level which allows for increased sensitivity in detecting changes to Wnd protein. (G–H) Representative images of presynaptic Brp and postsynaptic GluRIII from (G) liprin-αF3ex/R60 and liprin-α F3ex/R60;wnd1/3 and (H) rab3rup and rab3rup;wnd3. Unapposed GluRIII-labeled PSDs are highlighted by arrowheads. Wt is Canton S.. (I) Percentage of unapposed GluRIII-labeled PSDs from (G) and (H). All data are represented as mean ±SEM; N.S., not significant, ****p<0.0001; Tukey test for multiple comparison; Scale bar (B–D) 20 μm and (G–H) 5 μm. For additional data, see Figure 5—figure supplements 1–3.
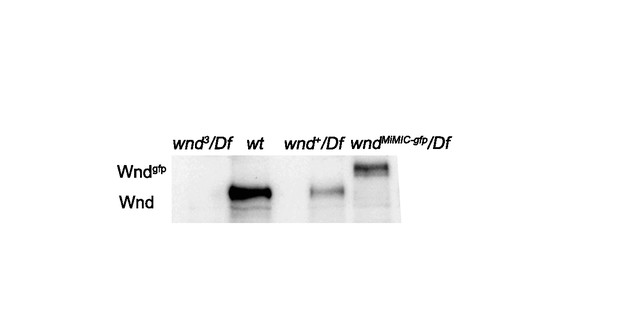
MiMIC-Wnd-GFP is a fusion protein of Wnd and GFP, (related to Figure 5).
Western blot with anti-Wnd antibody for larval brain extracts from wnd3/Df, wt, wnd+/Df and wndMiMIC-GFP/Df animals. Endogenous Wnd protein runs at approximately 130 kDa, while the MiMIC-Wnd-GFP fusion protein can be detected at an appropriately larger molecular weight (160 kDa) for the GFP-fusion, confirming the expression and stability of the fusion protein. 20 adult brains were processed for each sample.
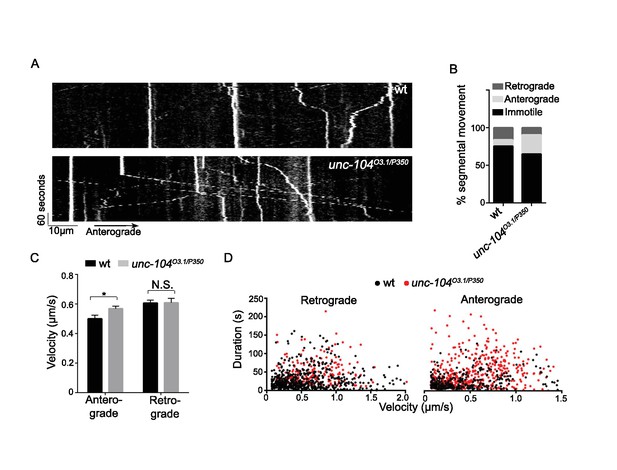
Wnd transport was not impaired in unc-104 mutants, (related to Figure 5).
(A) Kymograph of GFP-wndKD particle movement in SNc motoneuron axons (using the m12-Gal4 driver). Axons were imaged 900 μm distal to cell bodies at 0.3 Hz for 5 min in wt (m12-Gal4, UAS-GFP-wndKD) and unc-104O3.1/P350 mutant animals. Anterograde particles (which were more abundant in unc-104 mutants) moved from left to right. (B–D) Measurement of GFP-wndKD particle movement by (B) the distribution of particles across retrograde, anterograde and immotile categories, (C) velocity, and (D) scatter plot showing both duration (y-axis) and velocity (x-axis) of all anterograde and retrograde particles. In unc-104 mutants anterogradely-moving particles are more abundant and move in longer duration at higher velocity. All data are represented as mean ±SEM; N.S., not significant, ***p<0.001, **p<0.01, *p<0.05, Tukey test for multiple comparison.
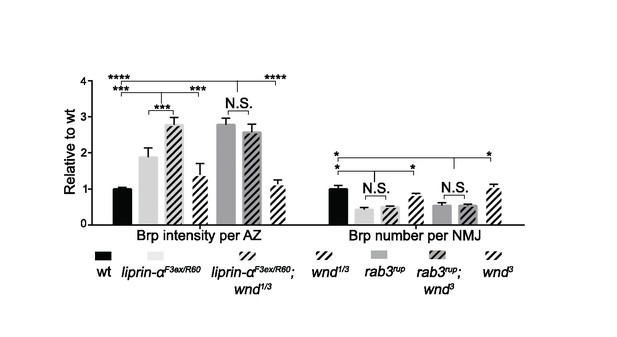
Liprin-α and Rab3 control presynaptic assembly independently of Wnd, (related to Figure 5).
Quantification of Brp intensity at individual AZs (intensity per AZ) and the number of Brp puncta per NMJ. Both liprin-α and rab3 mutants showed increased Brp intensity at individual AZs and a reduction in total number of Brp puncta. These phenotypes were not suppressed by wnd mutations. All quantification is normalized to wt (Canton S). All data are represented as mean ±SEM; N.S., not significant; ****p<0.0001, ***p<0.001,*p<0.05, Tukey test for multiple comparison.
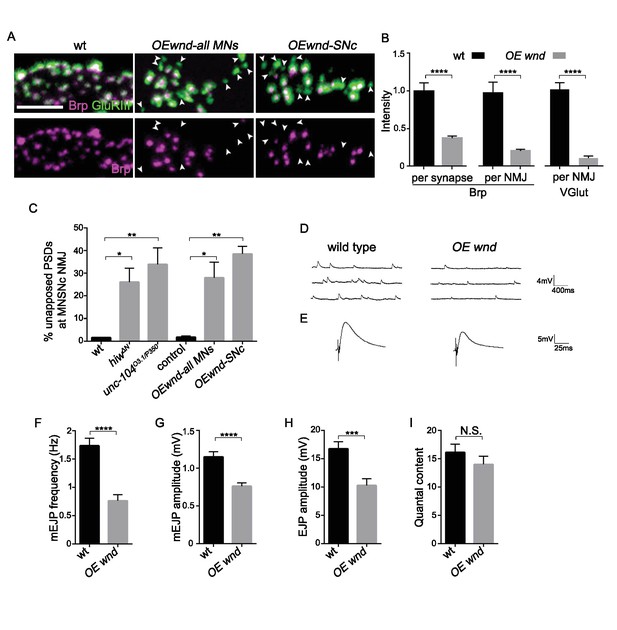
Activation of the Wnd signaling pathway in neurons is sufficient to impair presynaptic structure and synaptic transmission.
(A) Representative images of SNc synapses at muscle 26, 27 and 29 with Brp and GluRIII staining. GluRIII-labeled PSDs that lack apposed AZs (highlighted by arrowheads) increased when Wnd was over-expressed. Similar defects were observed using a pan-motoneuron driver (OK6-Gal4), and in driver line specific to the SNc motoneuron (m12-Gal4), indicating the cell autonomy of Wnd’s effect upon synapse assembly. (B) Quantification of Brp intensity at individual synapses or across entire NMJ terminals, and, similarly, total VGlut intensity across entire NMJ terminals (at muscle 4). Expression of UAS-wnd was driven with ok6-Gal4, and normalized to wt (Canton S). (C) The percentage of GluRIII-labeled PSDs which are unapposed by AZ component BRP. Quantification was carried out at SNc NMJ terminals in hiwΔN, unc-104O3.1/P350 and when UAS-wnd was over-expressed using either pan-motoneuron driver (OK6-Gal4) or a driver specific to SNc motoneurons (m12-Gal4). (D–E) Representative electrophysiological traces of (D) mEJP and (E) EJP on muscle 6 of third instar larvae in wt (Canton S) and OE wnd (expressed using the ok6-Gal4 driver). (F–I) Quantification of the (F) mEJP frequency, (G) mEJP amplitude, (H) EJP amplitude and (I) quantal content (corrected for nonlinear summation). All data are represented as mean ±SEM; N.S., not significant, ****p<0.0001, ***p<0.001, **p<0.01, *p<0.05, Tukey test for multiple comparison; Scale bar, 2 μm. For additional data, see Figure 6—figure supplements 1–2.
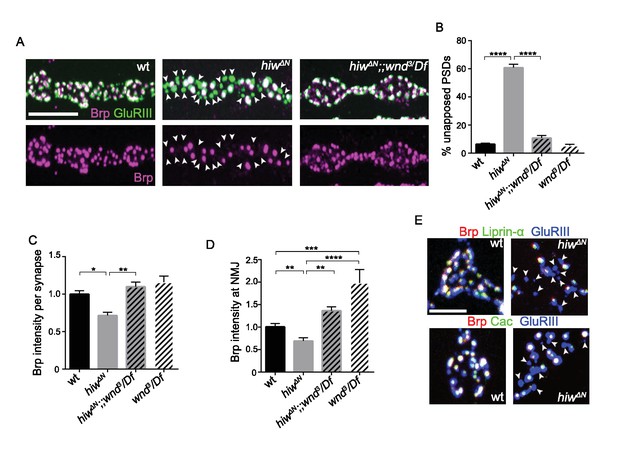
Wnd activation in hiw mutants inhibits presynaptic assembly, (related to Figure 6).
(A) Representative images of presynaptic Brp (magenta) and postsynaptic GluRIII (green) from wt (Canton S), hiwΔN(null) and hiwΔN(null);;wnd3/Df. GluRIII-labeled PSDs that lack apposed AZs (identified by Brp) are highlighted by arrowheads. (B) Quantification of the percentage of unapposed GluRIII-labeled PSDs which lack AZs from (A), normalized to wt (Canton S). (C–D) The total intensity of Brp measured at (C) individual synapses and (D) across NMJ terminals was reduced in hiw mutants, and restored in hiw;wnd double mutants. (E) Triple labeling of Brp (red), GluRIII (blue) and either Liprin-α-GFP or Cacophony-GFP (green) from wild type and hiwΔN. The co-localization of three markers resulted in white while unapposed ‘half synapses’ resulted in blue only, highlighted with arrowheads. Note that the arrowheads lack all 3 AZ markers. All data are represented as mean ±SEM; N.S., not significant, ****p<0.0001, ***p<0.001, **p<0.01, *p<0.05; Tukey test for multiple comparison, Scale bar, (A) 5 μm and (E) 2 μm.
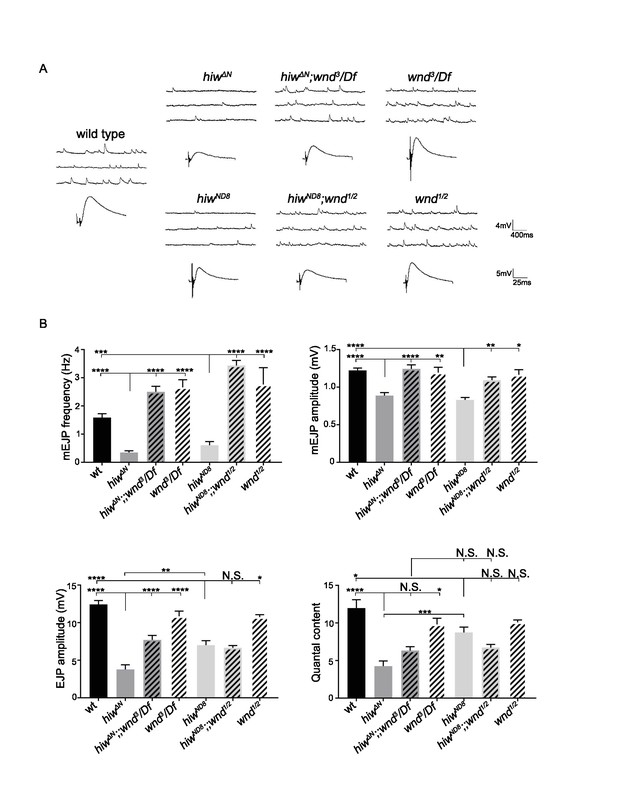
Wnd activation in hiw null mutants inhibits synaptic transmission, (related to Figure 6).
(A) Representative electrophysiological traces of mEJP and EJP on muscle 6 of third instar male larvae. The genotypes are wild type (Canton-S), hiwΔN, hiwΔN;;wnd3/Df, wnd3/Df, hiwND8, hiwND8;;wnd1/wnd2, wnd1/wnd2. hiwΔN is likely a null allele of highwire, while hiwND8 is likely a hypomorphic allele (Wu et al., 2005). (B) Quantification of the mEJP frequency, mEJP amplitude, EJP amplitude and quantal content. Note that hiwΔN exhibited a slightly stronger defect in EJP amplitude and quantal content than hiwND8. For both hiw alleles, mutations in wnd rescued the strongly reduced mEJP frequency, as well as the reduced mEJP amplitude. However, consistent with previous characterization(Collins et al., 2006) the quantal content defect (in both hiwΔN and hiwND8 mutants) was not significantly rescued by mutations in wnd. All data are represented as mean ±SEM; N.S., not significant, ****p<0.0001, ***p<0.001, **p<0.01, *p<0.05; Tukey test for multiple comparison.
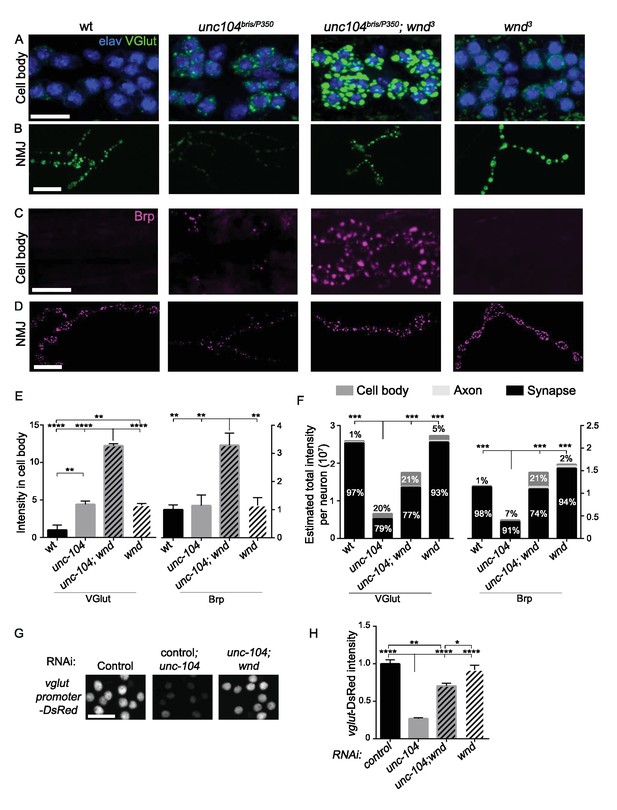
Wnd restricts the expression level of presynaptic components Brp and VGlut in unc-104 mutants.
(A–D) Immunostaining for VGlut (green, (A and B) and Brp (magenta, (C and D) in motoneuron cell bodies (A and C) and NMJ terminals (B and D). In (A) motoneuron nuclei are indicated by Elav staining (blue). Note the increased intensity of VGlut and Brp in unc-104bris/P350; wnd3 double mutants. (E) Quantification of VGlut and Brp intensity in cell bodies of motoneurons, corresponding to images in A and C. Note that total intensity measured at NMJ terminals is shown in Figure 2E–F. (F) Estimates generated for the total levels of VGlut and Brp proteins within motoneurons, accounting for the summed intensities measured in cell body, axonal, and synaptic compartments. Methods and assumptions used to calculate these estimates are described in Experimental Procedures. These proteins predominantly localize to the NMJ synaptic terminals (the proportion of the total protein localized to synapses is indicated with black shading). In unc-104-hypomorph mutants, a larger percentage is detected in cell bodies (medium gray shading), and the total summed intensity is reduced. In unc-104; wnd double mutants, the intensity increases in all of the compartments, cell body, axon (light gray shading) and synapse, compared to unc-104 mutants alone. Wt animals are Canton S. (G) Images of motoneuron nuclei, immunostained for DsRed in vglut promoter-DsRed reporter lines. (H) Quantification of vglut-DsRed intensity in (G), normalized to controlRNAi (moody RNAi). UAS-RNAi lines were driven by OK6-Gal4. For cell body analysis, at least 2 dorsal abdominal clusters of motoneurons per animal from at least 6 animals per genotype were examined. All data are represented as mean ±SEM; ****p<0.0001, ***p<0.001, **p<0.01, *p<0.05, Tukey test for multiple comparison; Scale bar, 20 μm. For additional data, see Figure 7—figure supplements 1–3.
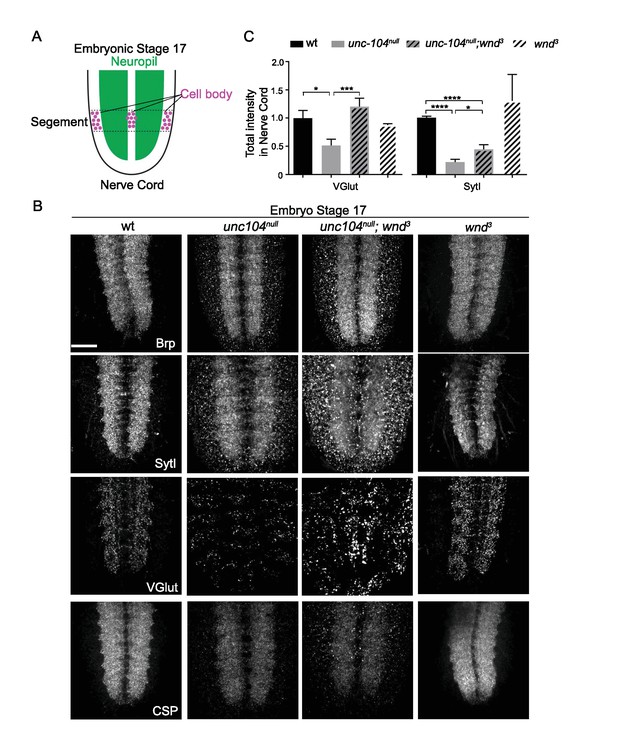
In unc-104-null mutants, multiple synaptic proteins were down-regulated by Wnd, (related to Figure 7).
(A) Schematic cartoon showing the locations of motoneuron cell bodies (within one segment, denoted by dotted lines) and Neuropil (green) in the nerve cord at embryonic stage 17. (B) Brp, SytI, VGlut and CSP immunostaining in stage 17 embryonic nerve cord (20–21 AEL). Increased staining in unc-104null;wnd3 mutants is noted in the cell body region. (C) Quantification of total intensity of SytI and VGlut from (B). At least 6 animals were examined per genotype. All data are represented as mean ±SEM; N.S., not significant, ****p<0.0001, ***p<0.001, *p<0.05, Tukey test for multiple comparison; Scale bar, 20 μm.
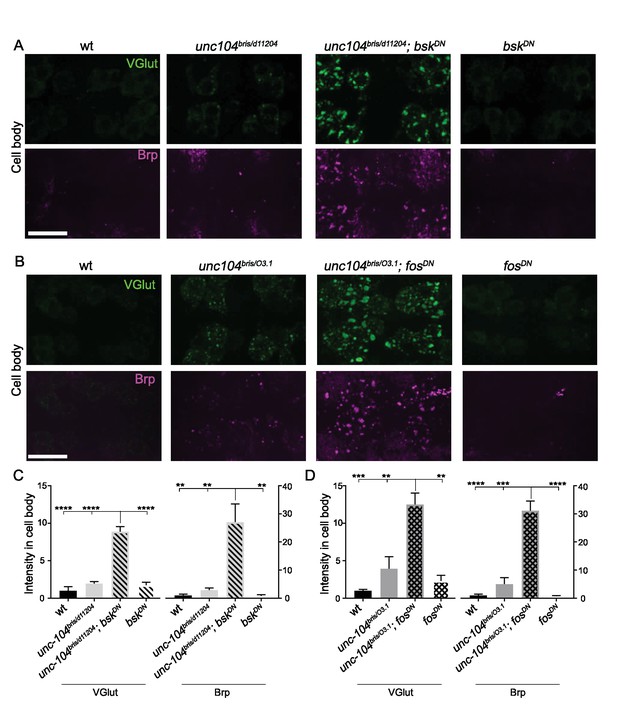
Wnd signaling components JNK/Bsk and Fos restrict the expression level of presynaptic components Brp and VGlut in cell bodies of unc-104 mutants, (related to Figure 7).
(A–B) Representative confocal images of motoneuron cell bodies immunostained for VGlut (Green) and Brp (magenta). Similarly to mutations in wnd (in Figure 7) inhibition of (A) bsk(JNK) or (B) fos in the unc-104-hypomorph mutant background via expression of dominant negative isoforms caused a dramatic enhancement of VGlut and Brp acccumulations in cell bodies. Two groups of motoneuron cell bodies are shown in each image. The genotypes are: wt (Canton-S), unc-104bris/d11204, bskDN (elav-Gal4;; UAS-bskDN), unc-104bris/d11204; bskDN (elav-Gal4; unc104bris/d11204; UAS-bskDN), unc-104bris/O3.1, unc-104bris/O3.1; fosDN (BG380-Gal4; unc104bris/d11204; UAS-bskDN) and fosDN (BG380-Gal4;; UAS-fosDN). Scale bar, 20 μm. (C–D) Quantification of VGlut and Brp from (A–B). At least 6 animals were examined per genotype.
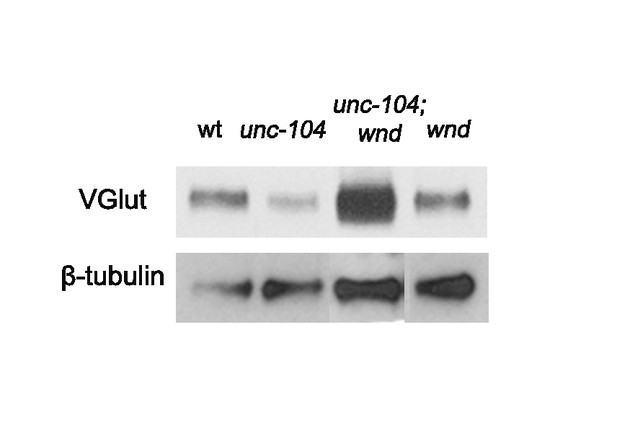
Total VGlut protein levels are reduced in unc-104-hypomorph mutants, in a Wnd-dependent manner (related to Figure 7).
Representative Western blot of 30 larval brains for VGlut and β-tubulin from wt (Canton S), unc-104bris/P350, wnd3 and unc-104bris/P350;wnd3.
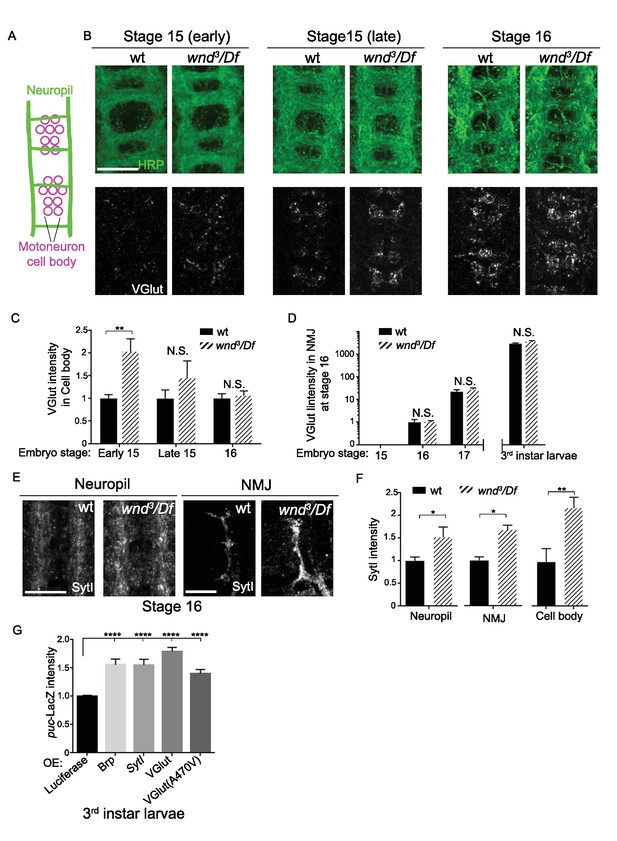
Wnd delays the expression of SV proteins during early stages of synapse development.
(A) Schematic cartoon of the embryonic nerve cord showing the neuropil (the location of neurites and developing synapses in the CNS) and motoneuron cell bodies in 2 segments at late embryonic stages (15 to 16). (B) Representative images of VGlut (white, bottom row) expression in motoneuron cell bodies of wt (w118) and wnd-null mutants (wnd3/Df) at embryonic stage early 15, late 15 and 16. Analogous segments are identified by neuropil HRP staining (top row). (C–D) Quantification of VGlut intensity in (C) motoneuron cell bodies and (D) NMJ presynaptic terminals for wt and wnd-null mutants at different embryonic stages and in third instar larvae. VGlut expression first appears in cell bodies at embryonic stage 15 (C), corresponding with the onset of NMJ synaptogenesis, but does not appear at NMJ terminals until stage 16 (D). As the NMJ matures and expands throughout development, VGlut intensity, which is predominantly localized to NMJ terminals, continues to increase (note the logarithmic scale). Quantification in both (C) and (D) is normalized to intensity for wt (w118) animals at stage 16. (E) Representative images of SytI immunostaining in CNS neuropil and NMJs. SytI intensity is elevated in wnd3/Df mutants at embryonic stage 16. (F) Quantification of SytI intensity from (E), normalized to intensity for wt (w118) animals. (G) Quantification of puc-lacZ expression in third instar larvae that ectopically express luciferase or presynaptic components (Brp, SytI, VGlut and VGlutA470V) via the Gal4/UAS system. Pan neuronal Gal4 (BG380) was used to drive expression of UAS-lines. All data were represented as mean ±SEM; ****p<0.0001, **p<0.01, *p<0.05, Tukey test for multiple comparison; Scale bar, 20 μm. For additional data, see Figure 8—figure supplement 1.
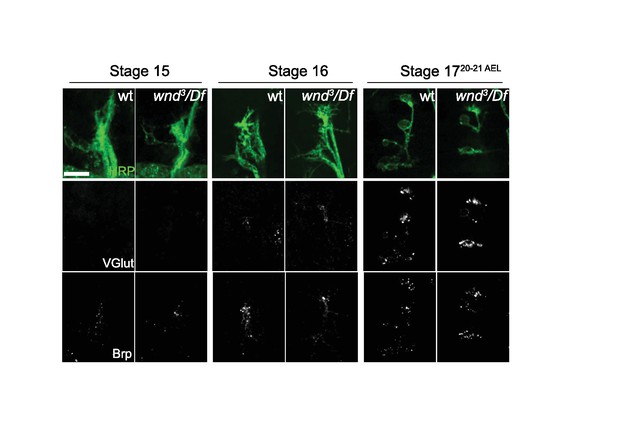
Brp and VGlut localization to NMJ terminals through early stages of synaptic development are not altered in wnd null mutants, (related to Figure 8).
VGlut (second row) and Brp (third row) immunostaining at ISNb NMJ terminals for wt (w118) and wnd3/Df null mutants at embryonic stage 15, 16 and 17. The top row shows the NMJ morphology at each stage based on staining with HRP which reveals the axonal and nerve terminal membrane. Scale bar, 10 μm.
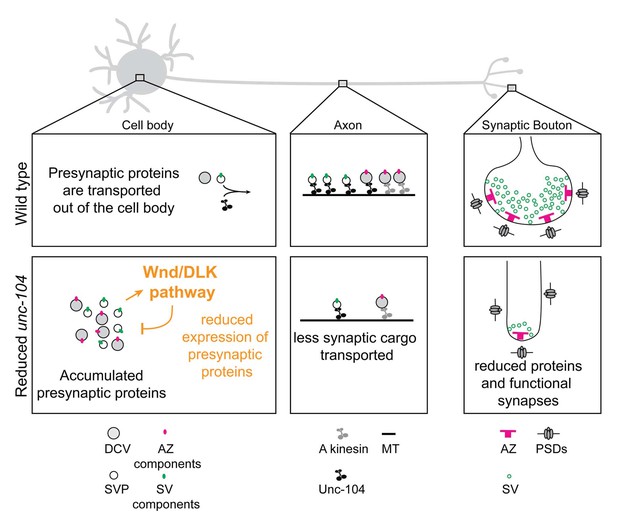
Model: Wnd/DLK signaling promotes synaptic defects by restraining total levels of presynaptic proteins when Unc-104’s function is reduced.
The formation and maintenance of synapses require the synthesis of presynaptic components (including AZ components (magenta dots) and SV components (green dots), which ultimately assemble into mature AZs (magenta ‘T’ bars) and SVs (green circles) at synapses. SV components are transported in the form of SV precursors (SVPs), while AZ components are thought to be transported via association with dense core vesicles (DCVs) (Shapira et al., 2003). While much remains to be learned about the mechanism by which individual synaptic components are transported, we can infer from this study that the Unc-104 kinesin (black) plays an essential role in the transport of SV components but not AZ components (which may be carried by an additional kinesin, indicated in gray). When Unc-104’s function is impaired/reduced, presynaptic components accumulate in cell bodies. However Wnd signaling activation reduces the accumulations by reducing total expression levels of presynaptic components. This reduction eventually manifests as a reduction in synapse number and mismatched pre- and postsynaptic structures.
Additional files
-
Transparent reporting form
- https://doi.org/10.7554/eLife.24271.030





