Chronic pain induces generalized enhancement of aversion
Figures
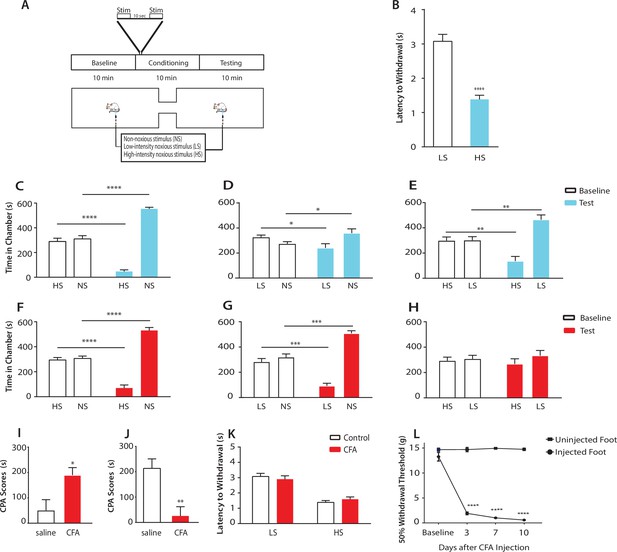
Chronic pain enhances the aversive response to acute pain at anatomically unrelated sites.
(A) Schematic for the conditioned place aversion (CPA) test. Each episode of peripheral stimulation lasted until paw withdrawal or in cases of no withdrawal (non-noxious stimulus or NS) a total of 5 s. (B) Latency to paw withdrawal was shorter with higher intensity pain stimulus (HS) than lower intensity stimulus (LS). n = 12; p<0.0001, Student’s t test. (C) During conditioning, rats received HS in one chamber and NS in the other chamber. After conditioning, rats spent less time in the chamber paired with HS during the test phase than at baseline, and more time in the chamber paired with NS. n = 14; p<0.0001, paired Student’s t test. (D) After conditioning with LS and NS in separate chambers, rats spent less time in the chamber paired with LS during the test phase than at baseline, and more time in the chamber paired with NS. n = 14; p=0.0131. (E) After conditioning with HS and LS, rats spent less time in the chamber paired with HS during the test phase than at baseline, and more time in the chamber paired with LS. n = 10; p=0.0012. (F) 10 days after CFA treatment, rats underwent conditioning by receiving HS stimulation of the uninjected paw in one chamber and NS stimulation of the same paw in the other chamber. During the test phase, rats spent less time in the chamber paired with HS than at baseline, and more time in the chamber paired with NS. n = 9; p<0.0001; paired Student’s t test. (G) After CFA treatment, after conditioning with LS and NS in separate chambers, rats spent less time in the chamber paired with LS stimulation of the uninjected paw during the test phase than at baseline, and more time in the chamber paired with NS. n = 9; p=0.0002. (H) After CFA treatment, rats could not differentiate between HS and LS treatments of the uninjected paws on the CPA test. n = 13; p=0.4923. (I) CFA treatment resulted in increased aversive response to LS. Rats were conditioned with LS and NS, and the CPA or aversion score was calculated by subtracting the amount of time rats spent during the test phase from baseline in the LS-paired chamber (see Materials and methods section). CPA scores for CFA-treated rats were significantly higher than saline control. n = 9–10; p=0.0179, unpaired Student’s t test. (J) After CFA treatment, rats could not distinguish between HS and LS. Rats were conditioned with HS in one chamber and LS in the other chamber. The CPA score for HS was calculated by subtracting the amount of time spent in the HS-paired chamber during the test phase from baseline. CFA-treated rats demonstrated a significant decrease in the CPA score, indicating an inability to distinguish between the aversive values of HS and LS. n = 8–13; p=0.0029, unpaired Student’s t test. (K) CFA treatment did not alter paw withdrawal latency in the uninjected paw. n = 12; p=0.8381, two-way ANOVA with repeated measures and post-hoc Bonferroni test. (L) CFA treatment caused mechanical allodynia in the injected but not uninjected paw. n = 12; p<0.0001. two-way ANOVA with repeated measures and post-hoc Bonferroni test.
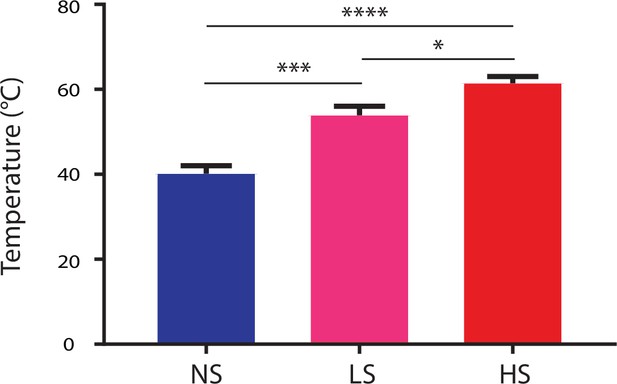
HS, LS and NS trigger high-intensity, low-intensity noxious and non-noxious stimulus respectively.
Temperature measurements were conducted with a temperature sensor (Extech Instruments, 65EA, Waltham, MA) after 5 s in the case of NS, 3 s in the case of LS, and 1.5 s in the case of HS. In the cases of LS and HS, the timing of temperature measurements correlated with the averaged time of paw withdrawals. Averaged temperature at the time of recordings was 40.2 ± 1.9, 53.1 ± 2.1, and 61.4 ± 1.6 for NS, LS and HS, respectively. p=0.0003 for comparison between NS and LS, <0.0001 for comparison between NS and HS, and 0.0123 for comparison between LS and HS. n = 10–24 recording sessions. One-way ANOVA with post-hoc Bonferroni’s multiple comparison tests.
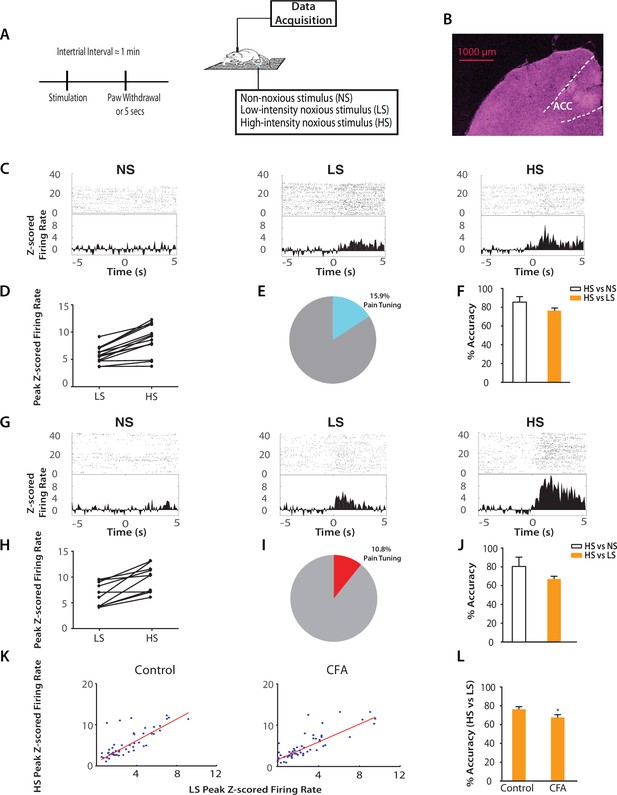
Chronic pain increases the responsiveness of ACC neurons to acute pain signals.
(A) Timeline and schematic for electrophysiological recordings in freely moving rats. Each trial of peripheral stimulation lasted until paw withdrawal or in cases of no withdrawal (non-noxious stimulus or NS) a total of 5 s. (B) Histology showing the location of ACC tetrode recordings. (C) Raster plots and peristimulus time histograms (PSTHs) before and after NS, LS and HS stimulations. Time zero denotes the onset of stimulus. Y-axis shows z-scored firing rates. To calculate the z-scored firing rate, we used the following equation: (FR – mean of FRb) / SD of FRb, where FR indicates firing rate and FRb indicates baseline firing rate prior to NS, LS or HS (see Materials and methods section). (D, E) A subset of neurons was identified among pain responsive neurons that showed higher firing rates after HS stimulation compared with LS (see Materials and methods for definition of pain responsive neurons). n = 14 out of a total of 88 neurons; for D, p<0.0001, paired Student’s t test. (F) Population-decoding analysis using a SVM classifier demonstrated decoding accuracy to distinguish between HS and NS (85%) or HS and LS (76%), n = 9. See Materials and methods. (G) Raster plots and PSTHs before and after NS, LS and HS stimulations in rats 10 days after CFA injections in the opposite paws. (H, I) A subset of neurons was found among pain responsive neurons that showed higher firing rates after HS stimulation compared with LS in the chronic pain condition. n = 10 out of a total of 93 neurons; for H, p<0.0001, paired Student’s t test. (J) Population-decoding analysis demonstrated decoding accuracy to distinguish between HS and NS (81%) or HS and LS (67%) in rats after chronic pain, n = 15. (K) A robust linear regression model was used to fit the peak z-scored firing rates and to calculate slope of the fit for all ACC neurons that demonstrated higher firing rates at HS compared with LS. Slope = 1.31 ± 0.09, R2 = 0.5937, n = 50 neurons. After chronic pain, neurons with higher firing rates at HS than LS showed a flatter tuning curve between LS and HS responses (see Materials and methods). Slope = 1.07 ± 0.07, R2 = 0.6904, n = 53 neurons. The two slope parameters are statistically different (p<0.05, unpaired Student’s t-test). (L) Decoding analysis showed that after chronic pain, there was a decrease in decoding accuracy to distinguish between HS and LS, n = 9 for pre-CFA, 15 for post-CFA; p<0.05, unpaired Student’s t test.
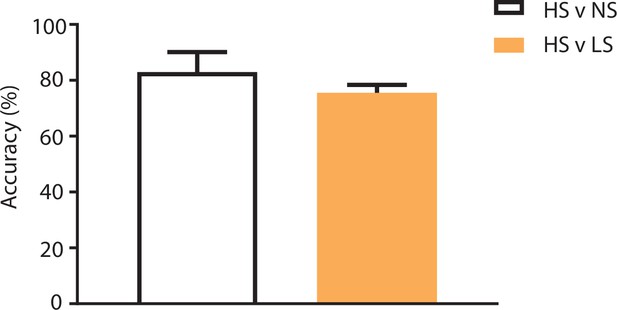
Population-decoding analysis using a shorter 3 s time window.
SVM analysis using a shorter 3 s time window to distinguish between HS and NS (83%) or HS and LS (76%) yielded similar results to analysis using a longer 5 s time window (Figure 2F). n = 9.
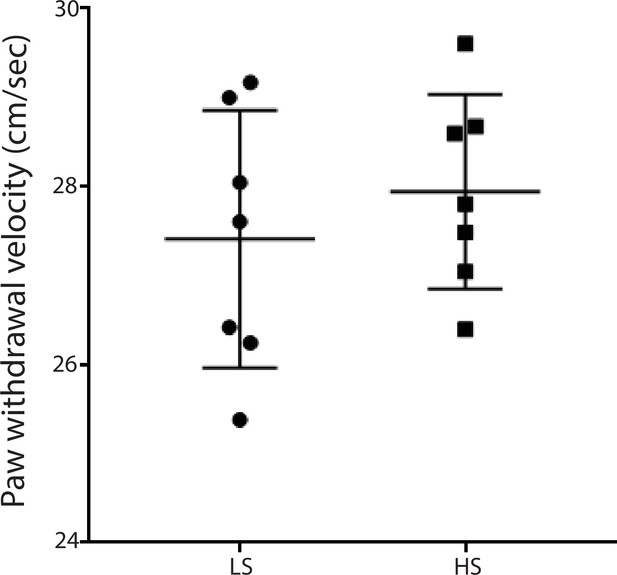
LS and HS trigger similar paw withdrawal movements.
Paw withdrawal velocity was measured using a high-speed camera. We measured the paw withdrawal velocity by dividing the highest point each paw reached by the time it took to reach this point (see Materials and methods). There was not a statistically significant difference in the average velocity of withdrawal between the responses to LS and HS. n = 7 rats for each stimulus. p=0.4503, unpaired Student’s t test.
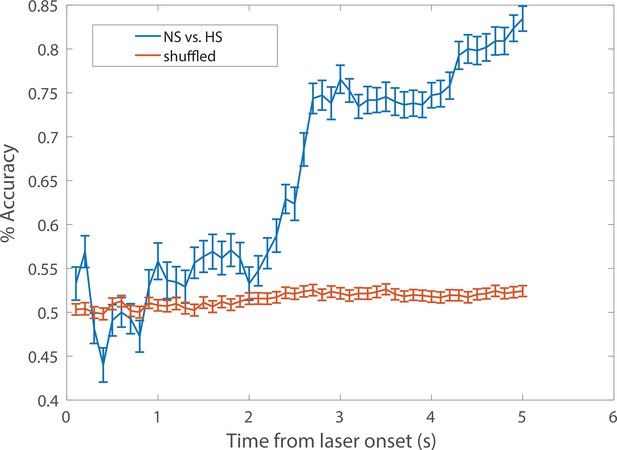
A representative example of cumulative decoding accuracy curves using the SVM classifier.
Time zero denotes the laser onset. The blue curve denotes the decoding accuracy (n1 = 24 trials for NS, n2 = 32 trials for HS; C = 7 ACC neurons) derived from the data with true labels; the error bar denotes the SEM from 100 Monte Carlo simulations based on 5-fold cross-validation; the maximum decoding accuracy was 0.83. The red curve denotes the decoding accuracy derived from the shuffled data with randomly permuted labels; the error bar denotes the SEM from 500 random permutations; the maximum decoding accuracy was 0.53. The chance level 53% (instead of 50%) was partially due to the imbalance of the sample size (n1≠ n2). See Materials and methods for details.
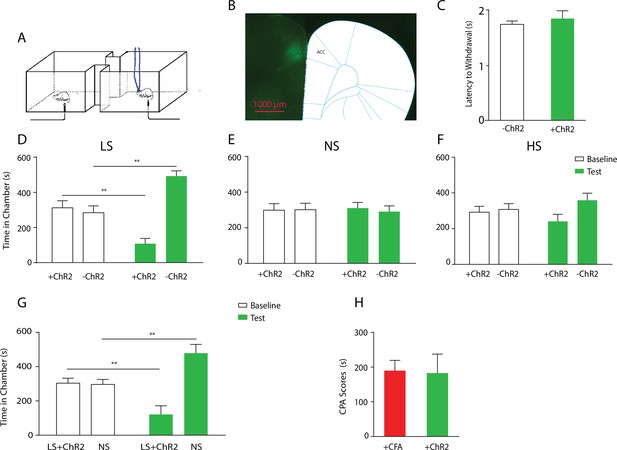
Optogenetic activation of the ACC has similar effects as chronic pain in enhancing the aversive response to acute pain.
(A) Schematic for a CPA test during optogenetic activation of the ACC. Light activation of the ACC was temporally coupled with peripheral stimulation to the paw. (B) Histologic expression of ChR2 in the ACC. (C) Light activation of the ACC did not alter paw withdrawal latency to noxious stimuli. n = 5; p=0.4654, paired Student’s t test. (D) ACC activation increased the aversive response to LS. One of the chambers was paired with optogenetic stimulation of the ACC and LS; the other chamber was paired with LS without ACC activation. Rats spent less time during the test phase than at baseline in the chamber paired with LS coupled with ACC activation and more time in the chamber paired with LS alone. n = 8; p=0.0026. (E) ACC activation did not elicit an aversive response to NS. One of the chambers was paired with ACC activation and NS; the other chamber was paired with NS alone. n = 9; p=0.7514. (F) ACC activation did not increase the aversive response to HS in a statistically significant manner. One of the chambers was paired with ACC activation and HS; the other chamber was paired with HS alone. n = 10; p=0.1584. (G) Coupling with ACC activation increased the aversive response of LS compared with NS. One of the chambers was paired with optogenetic stimulation of the ACC and LS; the other chamber was paired with NS alone. Rats spent significantly less time during the test phase than at baseline in the chamber paired with ACC activation and LS. n = 11; p=0.0075. (H) ACC activation caused a similar increase in the aversive response to LS as chronic pain. For comparison, rats were conditioned with LS and NS in separate chambers in both experiments. In one experiment (3G), LS was coupled with ACC activation, and the CPA score was calculated by subtracting the amount of time spent during the test phase from baseline in the chamber paired with simultaneous ACC activation and LS. In the second experiment, conditioning was performed in CFA-treated rats without ACC activation. A CPA score was calculated by subtracting the amount of time spent during the test phase from baseline in the chamber paired with LS in CFA-treated rats. The CPA scores from these two different experiments were similar. n = 9–11; p=0.9161, unpaired Student’s t test.
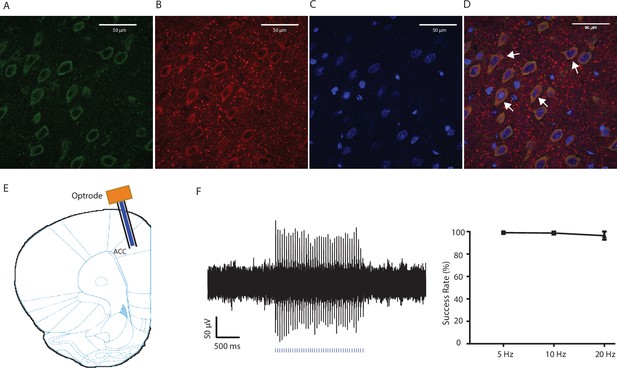
Expression of YFP-ChR2 in the ACC.
(A–D) High-magnification (63x) view of the expression of YFP-ChR2 in the ACC demonstrated the co-staining of excitatory neuronal markers (VGLUTs) with YFP. (A) YFP-ChR2 staining; (B) VGLUTs 1/2 staining; (C) DAPI staining; (D) merged images of A–C). Arrows point to examples of co-stained neurons. The extent of YFP-ChR2 expression in excitatory neurons was calculated, and 95.8 ± 1.4% of the YFP-positive cells were also VGLUTs-positive, suggesting that ChR2 was expressed almost exclusively in excitatory neurons. n = 8 sections from three rats. (E) Schematic for in vivo optrode recordings. (F) In vivo recording at the virus injection site demonstrated that laser pulses reliably triggered spikes in ACC neurons. Left: a representative trace showing increased neuronal firing in response to optogenetic activation. Right: success rate of optogenetic activation (defined by the rate of spiking induced by each laser pulse). n = 6 recording sessions from two rats.
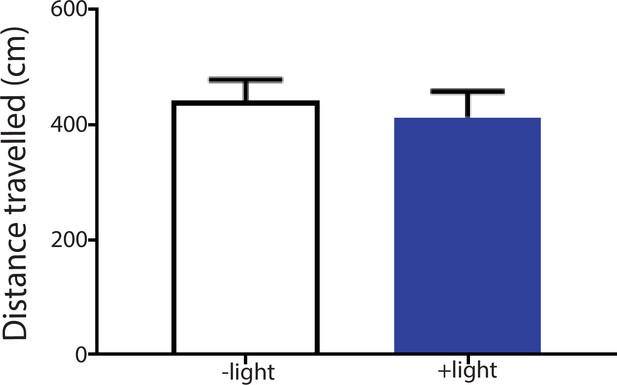
Activation of the ACC did not alter locomotion.
We measured locomotion over 10 min for rats that received optogenetic activation of the ACC. Light was turned on for 3 s every 10 s during the locomotion test. Light treatment did not change locomotor activities during the test. n = 8; p=0.6517, paired Student’s t test.
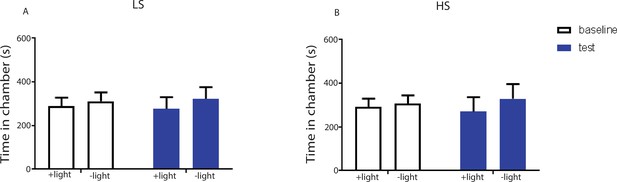
Light treatment of the ACC injected with an YFP-only vector did not impact conditioned place aversion.
We injected a viral vector that expressed only YFP without any opsins into the ACC. We applied laser light to the ACC in rats injected with this virus during the CPA tests. (A) During the conditioning phase of the CPA assay, we paired one chamber with light treatment and LS, and the opposite chamber with LS alone. We did not observe any preference or aversion for the chamber paired with light treatment. n = 7; p=0.6875, paired Student’s t test. (B) During the conditioning phase of the CPA assay, we paired one chamber with light treatment and HS, and the opposite chamber with HS alone. We did not find any preference or aversion for the chamber paired with light treatment. n = 7; p=0.6149, paired Student’s t test.
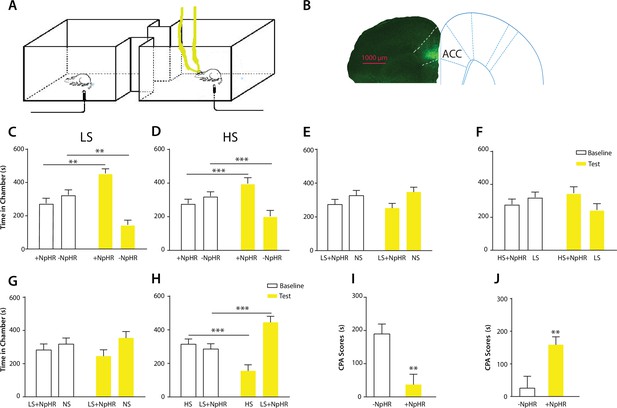
Optogenetic inhibition of the ACC diminishes the effect of chronic pain on the aversive response to acute pain.
(A) Schematic for a CPA test during optogenetic inhibition of the ACC. Optogenetic inhibition of the ACC was temporally coupled with peripheral stimulation to the paw. (B) Histologic expression of NpHR in the ACC. (C) ACC inhibition decreased the aversive response to LS. One of the chambers was paired with optogenetic inhibition of the ACC and LS; the other chamber was paired with LS alone. Rats spent more time during the test phase than at baseline in the chamber paired with light inhibition of the ACC coupled with LS and less time in the LS-alone chamber. n = 10; p=0.0043, paired Student’s t test. (D) ACC inhibition decreased the aversive response to HS. One of the chambers was paired with ACC inhibition and HS; the other chamber was paired with HS alone. Rats spent more time during the test phase than at baseline in the chamber paired with ACC inhibition coupled with HS and less time in the HS-alone chamber. n = 11; p=0.0006. (E) ACC inhibition abolished the aversive value of LS. One chamber was paired with ACC inhibition coupled with LS; the other chamber was paired with NS without ACC modulation. There was no statistically significant difference between test and baseline preference for either chamber. n = 9; p=0.5797. (F) ACC inhibition abolished the difference in aversive valuation between HS and LS. One chamber was paired with ACC inhibition coupled with HS; the other chamber was paired with LS. There was no statistically significant difference between test and baseline preference for either chamber. n = 12; p=0.1617. (G) ACC inhibition abolished the aversive value of LS even in CFA-treated rats. One chamber was paired with ACC inhibition coupled with LS; the other chamber was paired with NS. There was no statistically significant difference between test and baseline preference for either chamber. n = 10; p=0.2638. (H) ACC inhibition during the presentation of LS restored the normal differentiation between the aversive values of HS and LS in CFA-treated rats. One chamber was paired with ACC inhibition coupled with LS; the other chamber was paired with HS. Rats spent more time during the test phase than baseline in the chamber paired with ACC inhibition and LS, and less time in the HS-paired chamber. n = 10; p=0.0001. (I) ACC inhibition decreased the aversive value of LS in CFA-treated rats. CPA scores were compared in CFA-treated rats. In the control group (− NpHR), LS was not coupled with ACC inhibition during conditioning. In the test group (+ NpHR), LS was coupled with optogenetic inhibition of the ACC. In both groups, LS was conditioned against NS. CPA scores were calculated by subtracting the amount of time spent in the chamber paired with LS during the test phase from baseline. ACC inhibition reduced the CPA score for LS. n = 9–10; p=0.0026, unpaired Student’s t test. (J) ACC inhibition restored the normal difference in the aversive response to HS vs LS even after CFA treatment. In the control group (− NpHR), LS was not coupled with ACC inhibition during conditioning. In the test group (+ NpHR), LS was coupled with optogenetic inhibition of the ACC. In both groups, LS was conditioned against HS. CPA scores were calculated by subtracting the amount of time spent during the test phase in the chamber paired with HS from baseline. ACC inhibition increased the CPA score for HS. n = 10–13; p=0.0097.
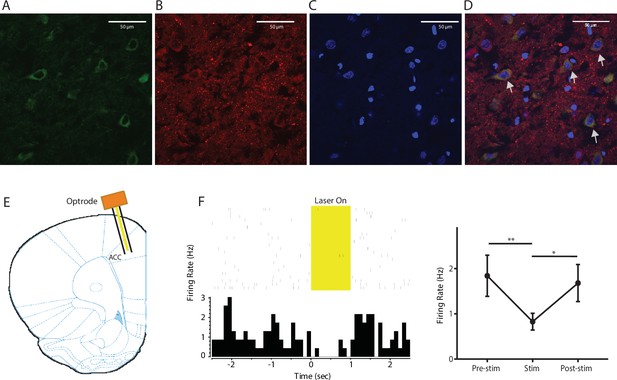
Expression of YFP-NpHR in the ACC.
(A–D) High-magnification (63x) view of the expression of YFP-NpHR in the ACC demonstrated the co-staining of excitatory neuronal markers (VGLUTs) with YFP. (A) YFP-NpHR staining; (B) VGLUTs 1/2 staining; (C) DAPI staining; (D) merged images of A–C). Arrows point to examples of co-stained neurons. The extent of YFP-NpHR expression in excitatory neurons was calculated, and 92.0 ± 3.1% of the YFP-positive neurons were also VGLUTs-positive, suggesting that NpHR was expressed almost exclusively in excitatory neurons. n = 7 sections from three rats. (E) Schematic for in vivo optrode recordings. (F) In vivo recording at the virus injection site demonstrated that light inhibited ACC firing. Left: a representative PSTH showing a neuron in response to optogenetic inhibition. Right: neurons demonstrated decreased firing rate in response to optogenetic inhibition. n = 7 neurons from two rats, p=0.0099 (comparing pre-stimulation with stimulation), p=0.0226 (comparing stimulation with post-stimulation), and p>0.9999 (comparing pre- with post-stimulation), one-way ANOVA with repeated measures and post-hoc Dunn’s tests.
Inhibition of the ACC did not alter locomotion.
We measured locomotion over 10 min for rats that received optogenetic inhibition of the ACC. Light was turned on for 3 s every 10 s during the locomotion test. n = 6; p=0.9415, paired Student’s t test.
Coupling of light inhibition of the ACC with non-noxious stimulation (NS) did not result in aversion.
One of the chambers was paired with light inhibition of the ACC and NS; the other chamber was paired with NS alone. n = 11; p=0.5792, paired Student’s t test.





