Activation of the same mGluR5 receptors in the amygdala causes divergent effects on specific versus indiscriminate fear
Figures
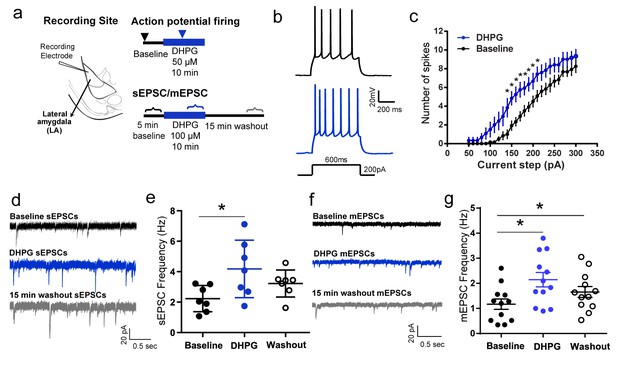
Pharmacological activation of mGluR5 using DHPG enhances intrinsic and synaptic excitability in LA principal neurons in vitro.
(a) Placement of recording electrode in a coronal brain slice of the lateral amygdala (LA) (left). Schematic of experimental protocols (right). Current-clamp recordings of action potential firing (right, top) was compared using a range of current injections (600 ms, 50–300 pA, 10 pA steps) between baseline firing before DHPG application (▾) and during a 10 min bath application of 50 µM DHPG ( ). Voltage-clamp recordings of spontaneous EPSCs and miniature EPSCs were made (right, bottom) before (
). Voltage-clamp recordings of spontaneous EPSCs and miniature EPSCs were made (right, bottom) before ( ), during (
), during ( ) and 15 min after (
) and 15 min after ( ) bath application of 100 µM DHPG for 5 min each in the same neuron. (b) Representative traces of accommodating action potential firing from a LA principal neuron in response to depolarizing current injections (200 pA) before (top, black) and after DHPG application (bottom, blue). (c) Averaged number of spikes fired shows an increase in excitability after DHPG application across a range of current injections (n = 6 neurons, p<0.05). (d) Representative traces showing voltage clamp recordings (VHOLD = −70 mV) of sEPSCs from LA neurons before (black), during (blue) and 15 min after washout of DHPG (grey). (e) DHPG causes a significant increase in the mean frequency of sEPSCs that returns to baseline levels after washout (n = 7 neurons, p<0.05). (f) Representative traces showing voltage clamp recordings (VHOLD = −70mV) of mEPSCs from LA neurons before (black), during (blue) and 15 min after washout of DHPG (grey). (g) DHPG causes a significant increase in the mean frequency of mEPSCs that remains elevated even after 15 min of washout (n = 12 neurons, p<0.05). *p<0.05 in all the graphs.
) bath application of 100 µM DHPG for 5 min each in the same neuron. (b) Representative traces of accommodating action potential firing from a LA principal neuron in response to depolarizing current injections (200 pA) before (top, black) and after DHPG application (bottom, blue). (c) Averaged number of spikes fired shows an increase in excitability after DHPG application across a range of current injections (n = 6 neurons, p<0.05). (d) Representative traces showing voltage clamp recordings (VHOLD = −70 mV) of sEPSCs from LA neurons before (black), during (blue) and 15 min after washout of DHPG (grey). (e) DHPG causes a significant increase in the mean frequency of sEPSCs that returns to baseline levels after washout (n = 7 neurons, p<0.05). (f) Representative traces showing voltage clamp recordings (VHOLD = −70mV) of mEPSCs from LA neurons before (black), during (blue) and 15 min after washout of DHPG (grey). (g) DHPG causes a significant increase in the mean frequency of mEPSCs that remains elevated even after 15 min of washout (n = 12 neurons, p<0.05). *p<0.05 in all the graphs.
-
Figure 1—source data 1
Data for individual cells representing the number of spikes generated in response to current injection (Figure 1c), frequency of sEPSCs (Figure 1e) and frequency of mEPSCs (Figure 1g) during baseline recordings, in the presence of DHPG and after washout of DHPG.
- https://doi.org/10.7554/eLife.25665.003
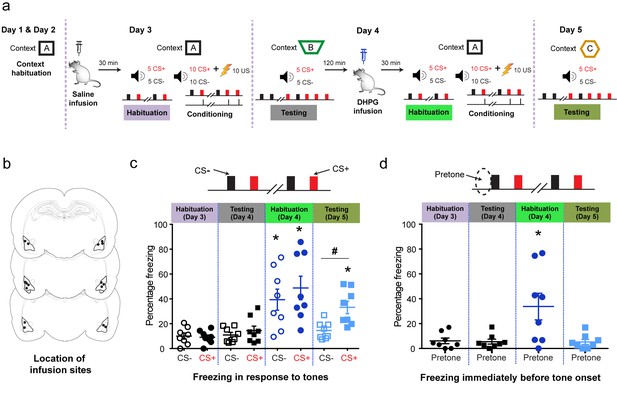
Targeted in vivo activation of mGluR5 in the BLA by itself initially causes indiscriminate fear, but it eventually leads to selective strengthening of cue-specific fear when combined with conditioning.
(a) Experimental protocol for discriminative auditory fear conditioning (10 CS+-US pairings were interleaved with 10 CS− presentations during conditioning) using a weak US (0.4 mA) combined with in vivo infusion (1.0 μl per side) of saline (0.9% NaCl) followed by DHPG (50 µM of DHPG) into the BLA of the same animal. (b) Schematic coronal sections depicting cannula infusion sites for saline/DHPG. (c) Mean freezing levels 30 min after saline infusions on Day 3 showed no difference between the CS+ and CS− during habituation (N = 8 rats, p>0.05). During testing 1 d after weak conditioning (Day 4), the CS+ did not evoke higher freezing relative to either the CS− (p>0.05), or habituation (p>0.05). However, 30 min after DHPG infusion, the same animals exhibited significantly higher freezing to both CS+ and CS− (*p<0.05), and the freezing levels were indistinguishable between CS+ and CS− (Habituation, Day 4: p>0.05). Strikingly, subsequent conditioning in the presence of DHPG using the same weak US (Day 4, 0.4 mA) strengthened cue-specific fear. Thus, during testing 1 d after weak conditioning, the CS+ evoked higher freezing relative to the CS− (Testing, Day 5: #, p<0.05), as well as CS+-evoked freezing 1 d after weak conditioning in saline (Testing, Day 4: *, p<0.05). (d) Mean freezing levels in the different spatial contexts immediately before the presentation of tones (pretone) during the same tests described in (c). Enhanced pretone freezing in the context was observed only immediately after DHPG infusion (Habituation, Day 4: *p<0.05). *p<0.05 between sessions in all graphs; #p<0.05 between CS+ and CS− within sessions in all graphs.
-
Figure 2—source data 1
Data for individual animals representing freezing response to CS+, CS- and pretone during the different phases of behaviour (Figure 2c and d).
- https://doi.org/10.7554/eLife.25665.005
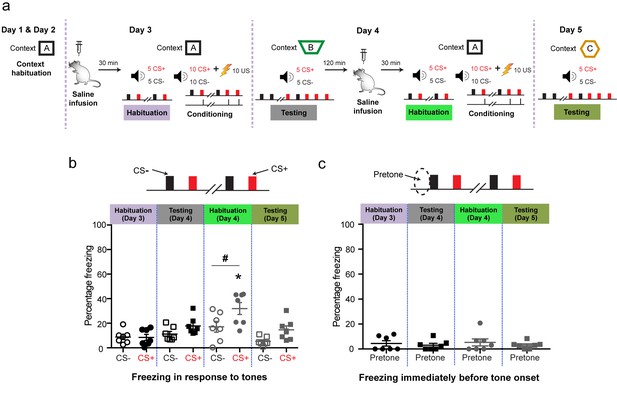
Control experiments using the same discriminative auditory fear conditioning with a weak US combined with in vivo infusion (1.0 µl per side) of saline (0.9% NaCl) followed by a second episode of the same saline infusion into the BLA of the same animal did not cause the increase in indiscriminate fear and cue-specific fear seen with DHPG infusion (as reported in Figure 2).
‘Mean freezing levels 30 min after saline infusions on Day 3 showed no difference between the CS+ and CS− during habituation (N = 7 rats, p>0.05). During testing 1 d after weak conditioning (Day 4), the CS+ did not evoke higher freezing relative to either the CS− (p>0.05), or habituation (p>0.05). However, 30 min after the second saline infusion, the same animals exhibited significantly higher freezing specifically for CS+ and not for CS− (*p<0.05) when habituated in the same conditioning context. The freezing levels were specific to CS+ (Habituation, Day 4: #p<0.05). Subsequently, a second episode of conditioning in the same animals in the presence of saline using the same weak US (Day 4, 0.4 mA) did not affect fear memory. Thus, during testing 1 d after weak conditioning, the CS+ did not evoke higher freezing relative to the CS− (Testing, Day 5: #p<0.05), as well as CS+-evoked freezing 1 d after first weak conditioning in saline (Testing, Day 4: *p<0.05). (c) Mean freezing levels in the different spatial contexts immediately before the presentation of tones (pretone) during the same tests described in (b). There was no difference in pretone freezing to the context during any of the sessions (p>0.05). *p<0.05 between sessions in all graphs; #p<0.05 between CS+ and CS− within sessions in all graphs.’.
-
Figure 2—figure supplement 1—source data 1
Data for individual animals representing freezing response to CS+, CS- and pretone during the different phases of behaviour (Figure 2—figure supplement 1b and c).
- https://doi.org/10.7554/eLife.25665.007
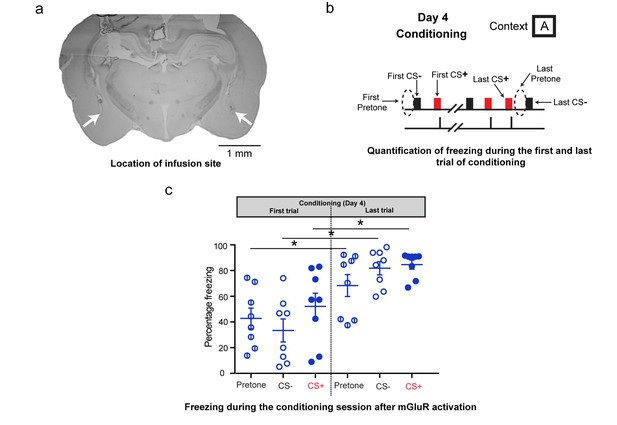
Additional analysis of freezing behavior during differential conditioning after DHPG infusion on Day 4.
(a) A sample image showing the location of the infusion cannula. The white arrows denote the tip of the infusion cannula. (b) To assess the effect of mGluR activation on the freezing response during fear conditioning, the freezing response to the first CS+, CS- and the first pretone were analysed. These were compared to the freezing response to the last CS+, CS- and last pretone of the conditioning session. (c) The high level of indiscriminate freezing to CS+/CS-/pretone is visible not only at the beginning of the conditioning, but also at the end. Moreover, the levels of freezing at the end of conditioning show an overall increase for pretone as well as CS+/CS- (*p<0.05). Thus, although the conditioning session leads to enhanced freezing, this freezing continues to be indiscriminate.
-
Figure 2—figure supplement 2—source data 1
Data for individual animals representing freezing response to CS+, CS- and pretone during the first and last trial of conditioning after DHPG infusion on day 4 (Figure 2—figure supplement 2c).
- https://doi.org/10.7554/eLife.25665.009
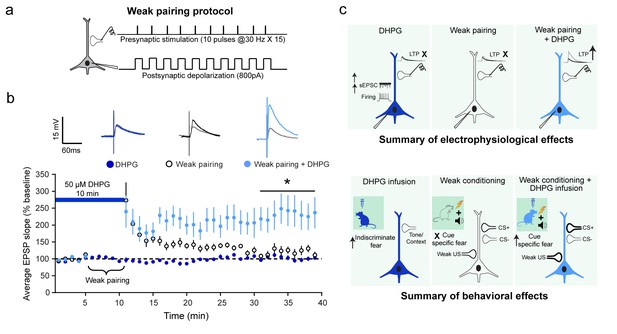
Although DHPG and a weak associative pairing protocol on their own did not elicit LTP, the two together caused a robust facilitation of LTP.
(a) Schematic of the weak associative pairing protocol that involved pairing EPSPs evoked by presynaptic stimulation of thalamic afferents (trains of 10 pulses at 30 Hz, repeated 15 times) with weak postsynaptic depolarization of LA neurons (somatic injection of 800 pA current) timed to coincide with the peak of EPSPs evoked by presynaptic stimuli (see Materials and methods). (b) 10 min bath application of 50 µM DHPG (blue bar) alone did not induce LTP ( , n = 11 neurons, p>0.05). The weak pairing protocol ( ⏟ ) caused only a transient enhancement in the EPSP slope, but no LTP (○, n = 11 neurons, p>0.05). However, in the presence of DHPG, the same weak pairing protocol induced robust LTP (
, n = 11 neurons, p>0.05). The weak pairing protocol ( ⏟ ) caused only a transient enhancement in the EPSP slope, but no LTP (○, n = 11 neurons, p>0.05). However, in the presence of DHPG, the same weak pairing protocol induced robust LTP ( , n = 12 neurons, *p<0.05). Superimposed representative EPSP traces before (grey) and30 minutes after the three manipulations – DHPG alone (dark blue), weak pairing alone (black), and weak pairing + DHPG (light blue). (c) Summary of the electrophysiological effects of in vitro activation of mGluR5 in LA slices and how they relate to the behavioral effects of in vivo activation of mGluR5, as described in the text.
, n = 12 neurons, *p<0.05). Superimposed representative EPSP traces before (grey) and30 minutes after the three manipulations – DHPG alone (dark blue), weak pairing alone (black), and weak pairing + DHPG (light blue). (c) Summary of the electrophysiological effects of in vitro activation of mGluR5 in LA slices and how they relate to the behavioral effects of in vivo activation of mGluR5, as described in the text.
-
Figure 3—source data 1
Data for individual cells representing the EPSP slope, normalized to baseline with DHPG treatment, weak pairing and weak pairing in the presence of DHPG (Figure 3b).
- https://doi.org/10.7554/eLife.25665.011





