Inhibition of Transient Receptor Potential Melastatin 3 ion channels by G-protein βγ subunits
Figures
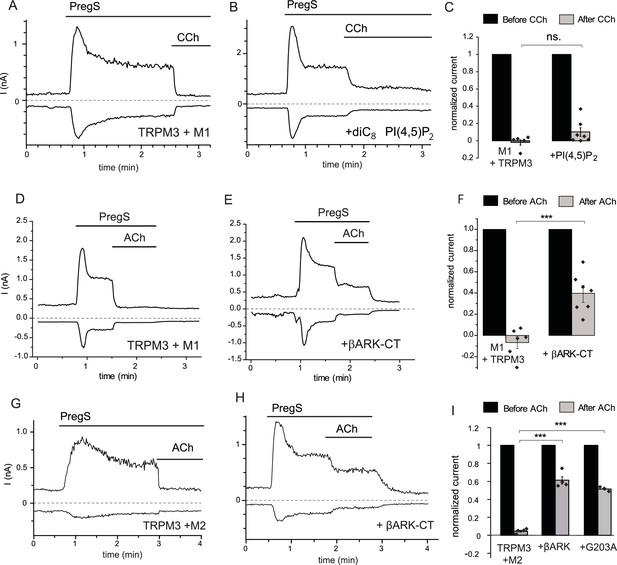
Inhibition of TRPM3 by Gq-coupled M1 and Gi-coupled M2 muscarinic receptors via Gβγ.
Whole-cell patch clamp experiments on HEK cells expressing mTRPM3α2 and Gq-coupled M1 or Gi-coupled M2 muscarinic receptors were performed as described in Materials and methods. TRPM3 currents were evoked by 50 μM PregS, currents are plotted at −100 and 100 mV (lower and upper traces), dashed lines show zero current. (A–B) Representative traces for inhibition by 100 μM carbachol (CCh), without (A) or with 100 μM diC8 PI(4,5)P2 (B) in the whole-cell patch pipette in cells expressing M1 muscarinic receptors. (C) Summary of the data (n = 5 for control and n = 7 for PI(4,5)P2, ns: p=0.103, two sample t-test). (D) Representative trace showing inhibition by 5 μM ACh, in a cell expressing M1 muscarinic receptors (E) similar experiment in a cell co-expressing the C-terminus of βARK which binds to Gβγ. (F) Summary data (n = 6 for control and n = 7 for βARK-CT, ***p=0.00032, two sample t-test). (G) Representative trace showing inhibition by 5 μM ACh in a cell expressing the Gi-coupled M2 muscarinic receptors and mTRPM3α2, (H) similar experiment in a cell co-expressing the C-terminus of βARK. (I) Summary data, (n = 4 for control, n = 4 for βARK-CT, n = 3 for G203A). ***p=0.000003 and p=0.000022, one-way analysis of variance with Bonferroni post hoc comparison.
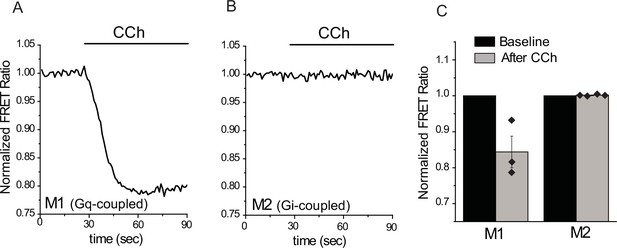
Activation of M1, but not M2 muscarinic receptors induces PI(4,5)P2 hydrolysis.
FRET-based PI(4,5)P2 measurements were performed as described in the Materials and methods section, downward deflection of the FRET ratio curve indicates a decrease in plasma membrane PI(4,5)P2 levels. (A–B) Representative FRET measurements in HEK cells expressing the CFP- and YFP-tagged tubby domain PI(4,5)P2 sensors in HEK cells co-transfected with M1 (A) and M2 (B) muscarinic receptors. (C) Summary data for the FRET measurements (n = 3 for M1 and n = 4 for M2).
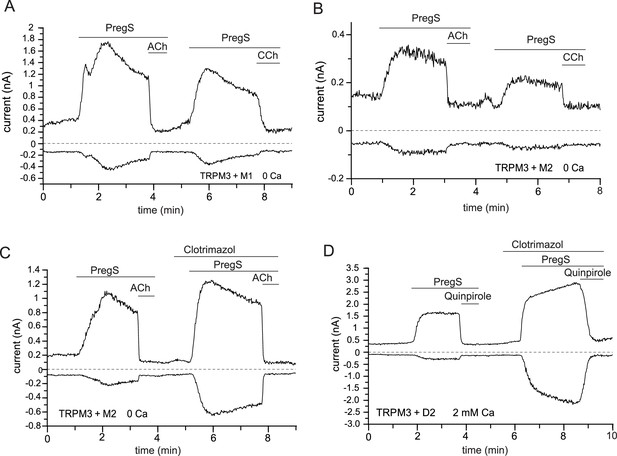
Activation of GPCRs inhibit TRPM3 currents in various conditions.
Whole-cell patch clamp experiments in HEK cells transfected with mTRPM3α2 were performed as described in Materials and methods; TRPM3 currents were evoked by 50 μM PregS, currents are plotted at −100 and 100 mV (lower and upper traces). Additional GPCR constructs cotransfected with TRPM3 are indicated at the individual panels. Current inhibition was essentially complete in all experiments, thus only representative traces are shown. (A) Cell transfected with M1 muscarinic receptors, the applications of 5 μM ACh and 100 μM CCh are indicated by the horizontal lines. The extracellular solution contained no added Ca2+ and 1 mM EGTA (n = 3). (B) Similar experiment to that shown in panel (A) but in cells transfected with M2 receptors (n = 3). (C) Similar experiment to that shown in B, but 10 μM clotrimazole was co-applied with PregS to stimulate the alternative conduction pathway of TRPM3 (n = 2). (D) Experiment in cells also transfected with D2 dopaminergic receptors; the applications of 200 nM Quinpirole and 10 μM Clotrimazole are shown by the horizontal lines, the experiment was performed in the presence of 2 mM extracellular Ca2+ (n = 5).
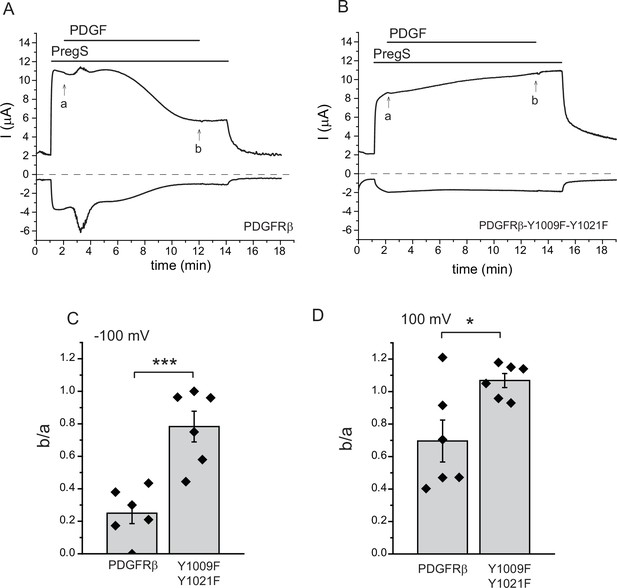
PLCγ activation by the PDGFRβ inhibits TRPM3 activity.
TEVC measurements were performed as described in Materials and methods, in Xenopus oocytes injected with cRNA encoding hTRMP3 and either the wild-type PDGFRβ (A) or the Y1009F-Y1021F mutant (B), which does not activate PLCγ. Currents are plotted at 100 mV (upper trace) and −100 mV (lower trace); the applications of 50 ng/ml PDGF and 50 μM PregS are indicated by the horizontal lines. Note the development of the transient Ca2+-activated Cl- current indicating PLC activation in oocytes expressing the wild-type PDGFRβ, and the lack of it in the experiments with the Y1009F-Y1021F mutant. (C) Summary of the inhibition at −100 mV. (D) Summary of the inhibition at 100 mV (n = 6 for both groups); b/a refers to the ratio between the current amplitudes at time points indicated by b and a in panels A and B. Statistical analysis was performed with two sample t-test *p=0.02, ***p=0.0008.
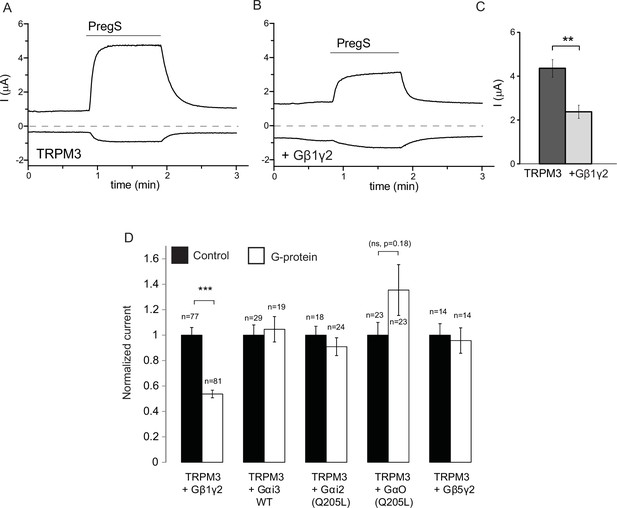
Co-expressed Gβ1γ2, but not Gαi or Gαo inhibits TRPM3 currents.
TEVC measurements in Xenopus oocytes expressing hTRPM3 were performed as described in Materials and methods; currents are plotted at 100 mV (upper traces) and −100 mV (lower trace). Currents were evoked by 50 μM PregS in control oocytes (A) and in oocytes expressing Gβ1γ2 (B). (C) Summary data for current amplitudes at 100 mV (n = 17 for each groups from one representative experimental day) (D) Normalized PregS-induced current amplitudes in oocytes co-expressing hTRPM3 and different G-protein constructs at 100 mV. Black bars are normalized current levels for control hTRPM3 expressing oocytes (see Materials and methods for details), empty bars are normalized current levels for oocytes also expressing the various G-protein subunits. The number of measurements on individual oocytes are indicated for each group. Statistical analysis was performed with two sample t-test ***p<0.005, corrected for multiple comparisons.
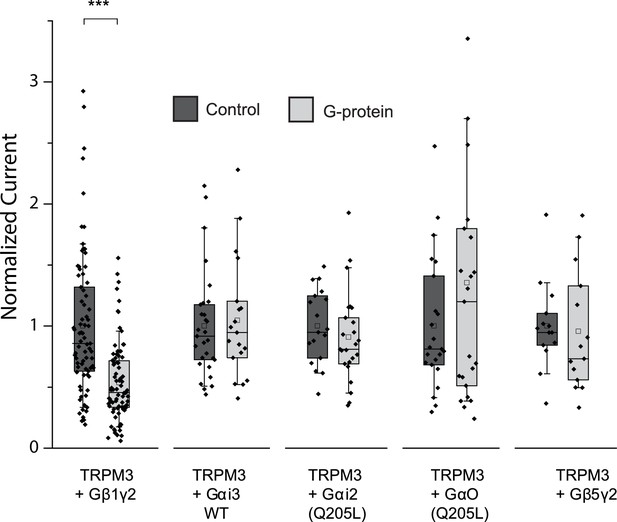
Co-expressed Gβ1γ2, but not Gαi or Gαo inhibits hTRPM3 currents; box and scatter plots.
Box and scatter plot of data from Figure 2D. Boxes show 25th and 75th percentiles, whiskers 90 and 10 percentiles.
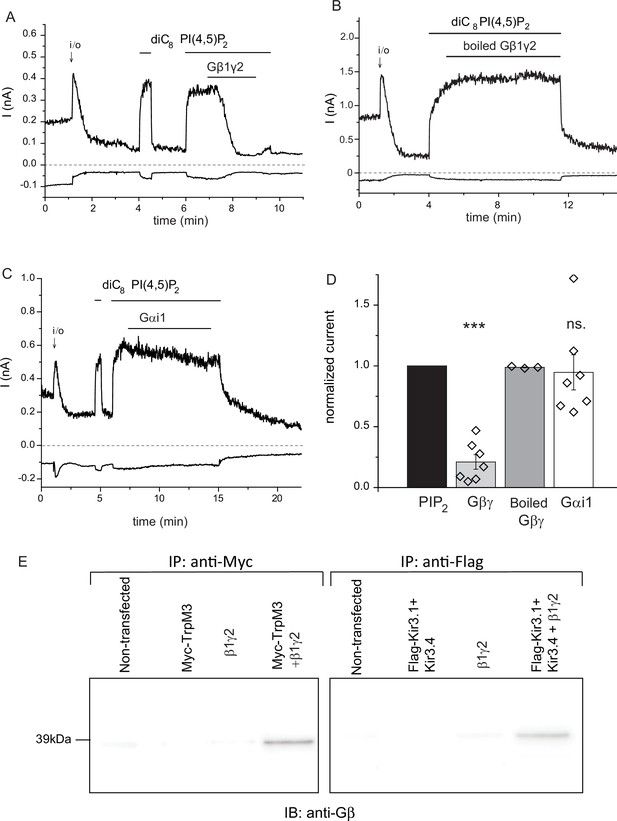
Purified recombinant Gβ1γ2 inhibits TRPM3 currents in excised patches.
(A–C) Excised inside-out patch clamp experiments were performed in Xenopus oocytes expressing hTRPM3, with 100 μM PregS in the patch pipette, as described in Materials and methods, currents at −100 mV (lower traces) and 100 mV (upper traces) are shown. The establishment of the inside-out (i/o) configuration is marked with the arrow, the application of 25 μM diC8 PI(4,5)P2 is shown with the horizontal line. (A) the effect of intact Gβ1γ2 (50 ng/ml), (B) the effect of Gβ1γ2 boiled for 15 min before the experiment. (C) The effect of 50 ng/ml Gαi1 (D) Summary of the data, the effects of the G-proteins were normalized to the currents induced by PI(4,5)P2 before the application the G-protein (n = 3 for boiled Gβγ, n = 7 for Gβγ and for Gαi1). (E) Co-immunoprecipitation of myc-TRPM3 (left panel) and flag-Kir3.4 was performed as described in the materials and methods section. HEK cells were transfected with the constructs indicated, immunoprecipitated using an anti-myc (left) or anti-flag antibody, and immunoblotted with an anti-Gβ antibody. Blots are representatives for four independent experiments, from four different transfections. Statistical analysis for the electrophysiological experiments was performed with one sample t-test ***p<0.00001, ns: p=0.72.
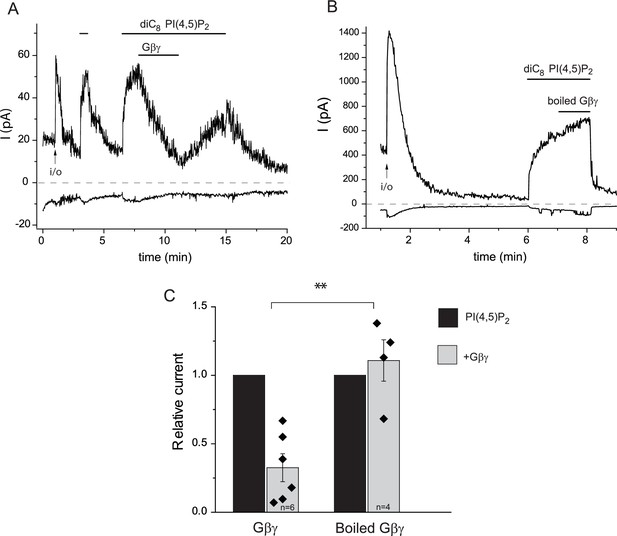
Inhibition TRPM3 in excised patches by Gβγ purified from bovine brain.
Excised inside-out patch clamp experiments were performed in Xenopus oocytes expressing hTRPM3, with 100 μM PregS in the patch pipette, as described in Materials and methods and as shown in Figure 3. The establishment of the inside-out (i/o) configuration is marked with the arrow, the application of 25 μM diC8 PI(4,5)P2 is shown by the horizontal line. (A) the effect of intact Gβ1γ2 (50 ng/ml), (B) the effect of Gβ1γ2 boiled for 15 min before the experiment. (C) Summary of the data. Statistical analysis was performed with two sample t-test **p=0.002.
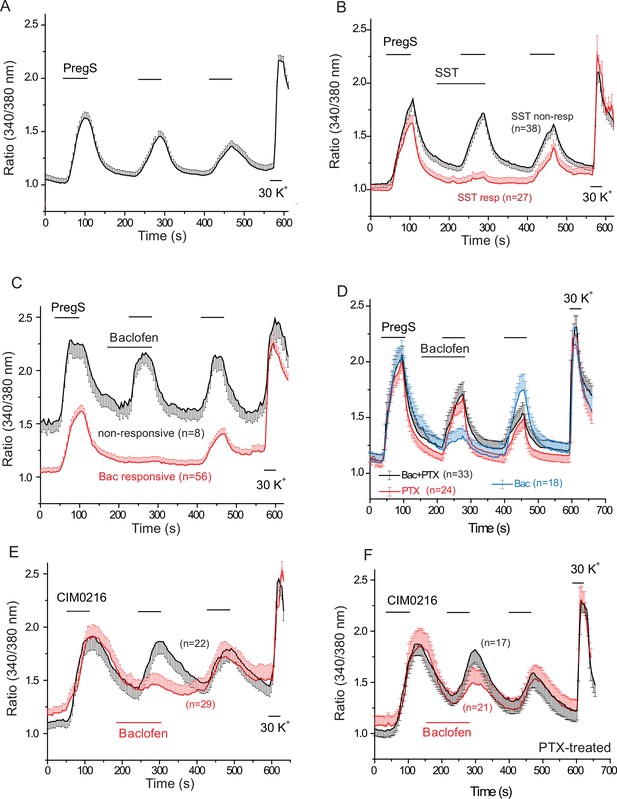
PregS-induced Ca2+ signals are inhibited by agonists of Gi-coupled receptors in DRG neurons.
Ca2+ imaging experiments in DRG neurons were performed as described in Materials and methods. (A) Average trace ± SEM showing the effect of three consecutive applications of 12.5 μM PregS from neurons responsive to this compound; 30 mM KCl was applied at the end of the experiment. In (B) 1 μM somatostatin (SST) was applied before the second application of PregS, the two traces show the average ratios ± SEM in cells that responded to somatostatin (red) and in cells that did not (black). (C) Shows a similar measurement with 25 μM baclofen. (D) DRG neurons were treated overnight with 300 ng/ml PTX, the effects of 25 μM baclofen are compared in PTX treated (black) and non-treated (blue) cells. The red trace shows PTX treated cells without the application of baclofen. For these experiments, we pooled baclofen responsive and non-responsive cells, as cells not responding to baclofen would have been difficult to identify in the PTX treated group. (E) Measurements similar to panel C using the synthetic TRPM3 agonist CIM0216 (1 μM). Black trace is control cells not treated with baclofen, red trace represents baclofen treated cells. (F) Similar measurements to panel E in cells pretreated overnight with 300 ng/ml PTX; red trace represents baclofen treated cells, black shows control cells.
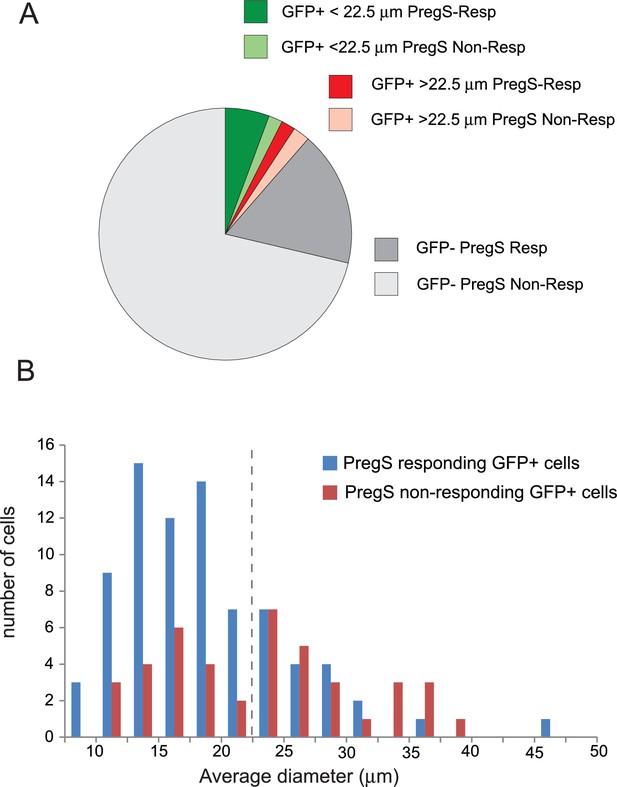
Distribution of PregS responsive and non-responsive DRG neurons of TRPM8-GFP reporter mice.
(A) Pie chart of PregS-responsive and non-responsive neurons based on analysis of Ca2+ responses to 12.5 μM PregS of 1063 KCl responsive cells. The GFP-positive population was divided into smaller cells (<22.5 μm) and larger cells (>22.5 μm); average diameter of the cells was determined by the Image Master five software. (B) Size distribution of PregS responsive and non-responsive GFP-positive cells; dashed line shows the 22.5 μm cutoff for smaller and larger cells.
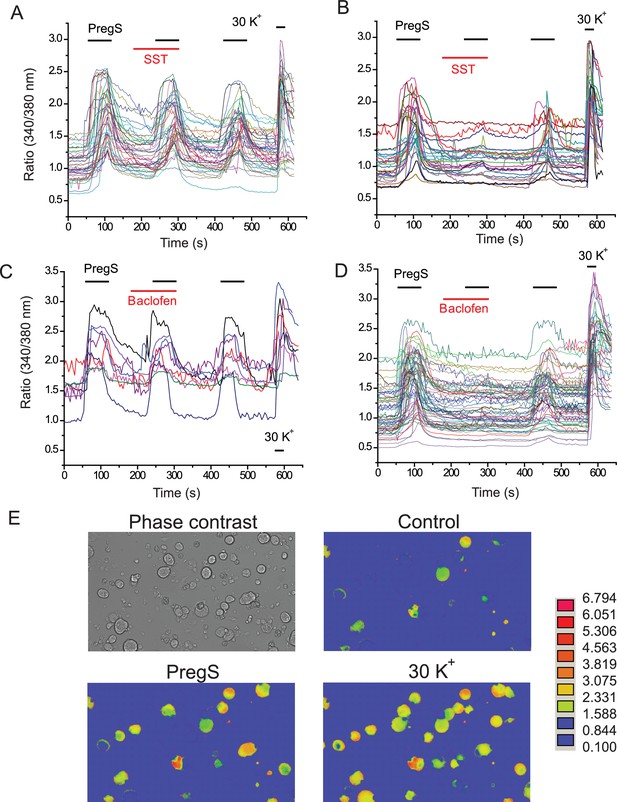
Individual traces and representative images for Ca2+ imaging experiments.
(A, B) Individual 340/380 nm ratios traces for Fura2-loaded DRG neurons; application of 12.5 μM PregS and 1 μM somatostatin (SST) are indicated by the horizontal lines. (A) SST non-responsive, (B) SST responsive cells. (C, D). Individual 340/380 nm ratio traces; application of 12.5 μM PregS and 25 μM baclofen are indicated by the horizontal lines, (C) baclofen non-responsive, (D) baclofen responsive cells. E representative images for responses to PregS and 30 mM KCl.
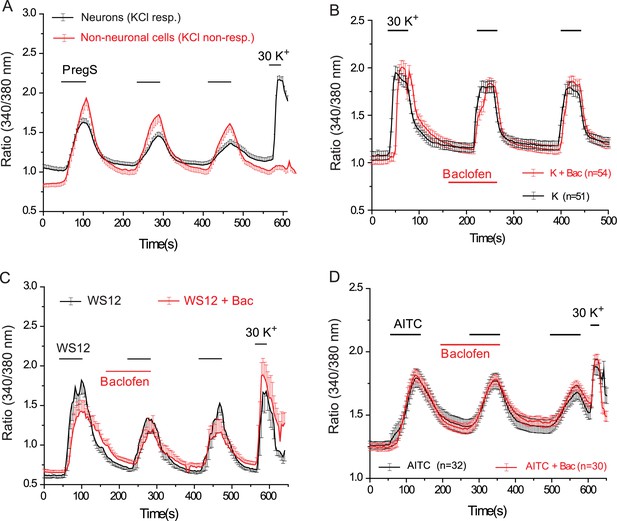
Baclofen does not inhibit PregS-induced Ca2+ signals in non-neuronal cells, and Ca2+ signals in DRG neurons evoked by KCl, the TRPM8 agonist WS12, or the TRPA1 agonist AITC.
(A) Ca2+ signals (mean ± SEM of 340/380 nm ratios) in Fura2-loaded KCl-responsive and KCl non-responsive cells induced by three consecutive applications of 12.5 μM PregS. The Ca2+ traces for non-neuronal (KCl-non-responsive) cells is an average of control and baclofen (25 μM) treated cells; the data were pooled, because the traces were indistinguishable; control trace for neurons without the application of baclofen is shown for reference (B) Lack of effect of baclofen on Ca2+ signals induced by 30 mM KCl (C) Ca2+ signals induced by three consecutive applications of 1 μM WS12 in control cells (black) and in cells where the second WS12 application was preceded by 25 μM baclofen, as indicated by the horizontal lines, n = 6 for both control and baclofen treated cells. (D) Ca2+ signals induced by three consecutive applications of 25 μM allyl isothyaocyanate (AITC) in control cells (black) and in cells where AITC application was preceded by 25 μM baclofen.
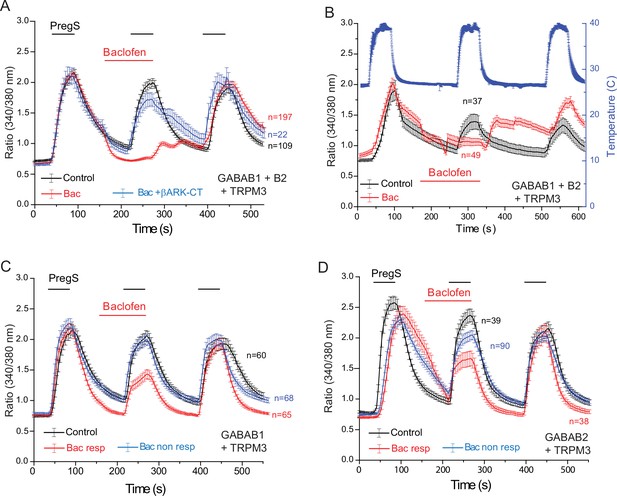
Baclofen inhibits PregS-induced Ca2+ signals in HEK cells expressing the GABAB1 and GABAB2 receptors in a Gβγ-dependent manner.
Ca2+ imaging experiments in HEK cells were performed as described in Materials and methods. Average traces ± SEM showing the effect of three consecutive applications of 12.5 μM PregS and the effect of 25 μM baclofen. The cells were transfected with mTRPM3 plus (A, B) GABAB1 + GABAB2 receptor, and in a subset of cells the Gβγ sink βARK-CT (blue trace in panel A), (C) GABAB1 receptor, (D) GABAB2 receptor. In panel A, note the almost complete inhibition of PregS-induced Ca2+ signal by baclofen, and the increase of Ca2+ after washout of baclofen (‘off’ effect). In panel B, Ca2+ responses to three consecutive heat pulses are shown (temperature: blue curve), note the marked off-response after washout of baclofen. In panels C and D the baclofen treated cells were subdivided into cells showing no response to baclofen (Bac non-resp), and cells in which baclofen induced a partial reduction of the PregS-induced Ca2+ signals (Bac resp).
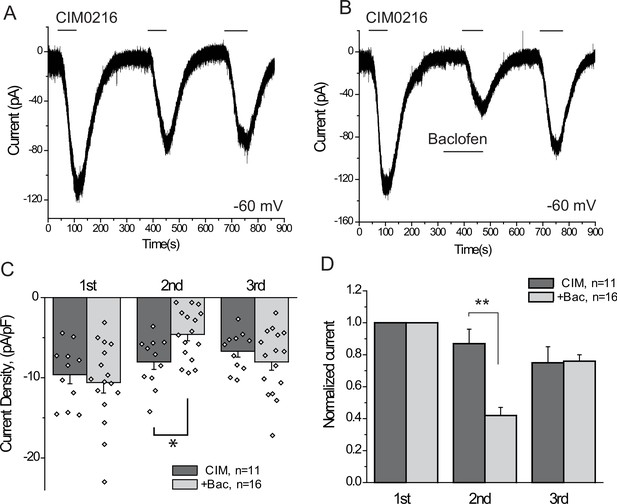
The GABAB receptor agonist baclofen inhibits inward currents induced by the TRPM3 channel agonist CIM0216.
(A–B) Whole-cell patch clamp measurements in small GFP-positive DRG neurons were performed as described in Materials and methods at −60 mV holding potential in nominally Ca2+ free solution. The applications of 5 μM CIM0216 and 25 μM baclofen are indicated by the horizontal lines. (C) Summary of current densities, (D) Summary of data normalized to the amplitude of the first peak current. Statistical analysis was performed with two sample t-test *p<0.05, **p<0.01.
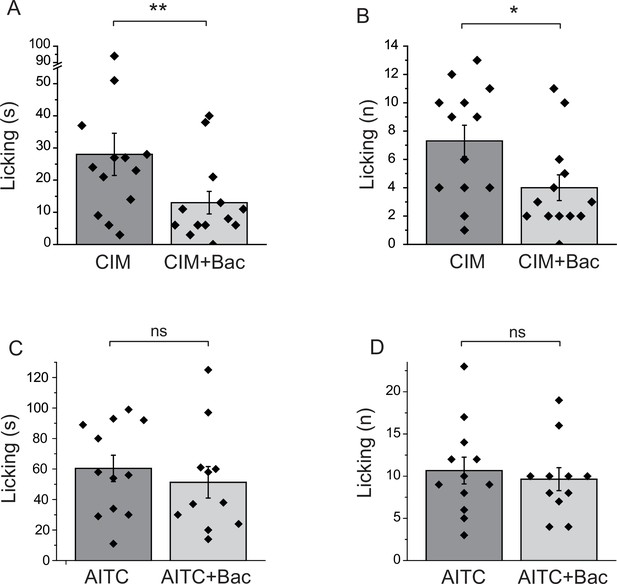
Baclofen inhibits nocifensive behavioral responses induced by the TRPM3 channel agonist CIM0216, but not responses to the TRPA1 agonist AITC.
(A–B) Nocifensive responses to the injection of CIM0216 (50 nmol/paw) were recorded as described in Materials and methods in control animals, and in animals where 12.5 nmol/paw baclofen was also injected in the same hind paw. (A) Duration of licking, (B) number of licking (n = 13 for both groups). (C, D) Nocifensive responses to hind paw injection of 100 nmol/paw AITC were recorded as described in Materials and methods in control animals, and in animals where 12.5 nmol/paw baclofen was co-injected. (C) Duration of licking, (D) number of licking (n = 12 for AITC and n = 11 for AITC + baclofen). Statistical analysis was performed with two sample t-test *p<0.05, **p<0.01, ns: p=0.5 (C) and p=0.63 (D).





