Transient external force induces phenotypic reversion of malignant epithelial structures via nitric oxide signaling
Figures
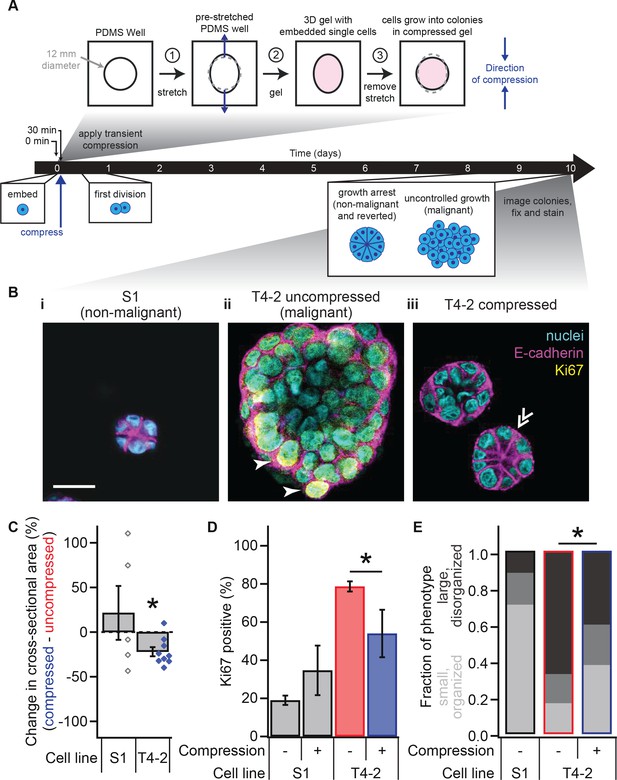
Transient compression of breast epithelial cells grown in laminin-rich ECM reverts the malignant phenotype.
(A) Cells were compressed after 30 min of lrECM polymerization at 37°C, before first division (day 1) or formation of growth-arrested colonies or large disorganized colonies (day 10). Compressive strain dissipated on a minutes time scale (Figure 1—figure supplement 1) and affected cells grown in a modified culture geometry (Figure 1—figure supplement 2). (B) Representative confocal immunofluorescence images of uncompressed S1 (i) and T4-2 colonies (ii), and compressed T4-2 colonies (iii). Solid arrowheads indicate cells that stained positive for Ki67. Double arrowheads indicate a T4-2 colony with its E-cadherin arranged in a ‘star pattern’ through its central section. Scale bar 25 μm. Compressed T4-2 colonies resemble T4-2 cells treated with chemical reverting agents (Figure 1—figure supplement 3). (C) 23% compression of single malignant cells led to reduction in colony size (N = 5, 9 pairs of gels, bars are mean ± SEM, points are means of individual experiments, with 90 ± 44 colonies per sample [mean ± SD]). *Difference is significant at p<0.05 in paired t-test, comparing compressed and uncompressed samples in same chamber preparation (see also Figure 1—figure supplement 2). Analogous experiments using MCF10A cancer progression series cells gave similar results (Figure 1—figure supplement 4). (D) Colonies grown from compressed single cells exhibited enhanced growth arrest (N = 4, 4, 14, and 6 gels, mean ± SEM, with 33 ± 12 colonies per sample [mean ± SD]), determined by the absence of Ki67 stain. *Difference is statistically significant at p<0.02 in two-sample t-test. (E) Bar graphs displaying qualitative analysis of phenotype of uncompressed S1, uncompressed T4-2, and compressed T4-2 colonies after 10 days of embedded culture in lrECM. Confocal immunofluorescence images were scored in a blinded fashion. Colonies were categorized as having a ‘small, organized’ phenotype (characteristic of normal development or reversion) if they contained at least three of the following four features: (1) a small size, (2) a round shape, (3) the absence of Ki67, and (4) well-organized E-cadherin cell-cell junctions (appearing as a star pattern in cross section) or a cleared lumen. Colonies with one or none of these four features were categorized as ‘large, disorganized’ (characteristic of malignant behavior). The increase in the proportion of colonies with an organized phenotype and the decrease in proportion of a disorganized phenotype in demonstrate that a fraction of the population of malignant T4-2 cells were phenotypically reverted with compression. This difference is statistically significant (*) between the T4-2 uncompressed and compressed (p<0.05, two-sample t-test, N = 3, 3 experiments). Each bar represents the average fraction of each phenotype category across 2, 3, and 3 gels, respectively, with 34 ± 13 colonies per gel (mean ± SD).
-
Figure 1—source data 1
Colony size and proliferation status in compressed and uncompressed T4-2 and S1 cells.
- https://doi.org/10.7554/eLife.26161.007
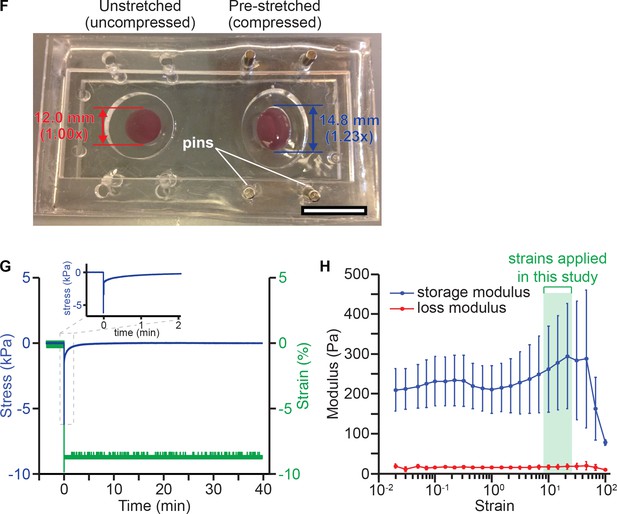
Application of transient compression to 3D cultured breast epithelial cells.
(F) Photograph of PDMS chamber for applying compression to 3D cultured cells, demonstrating the unstretched and pre-stretched configurations. In an experiment, one well was pre-stretched and held in place in an acrylic frame with steel pins. Then, lrECM was polymerized and cells were seeded in both wells. To apply compression, the steel pins were removed from the pre-stretched well, allowing the chamber to relax to the same size and shape as the unstretched well. To describe the amount of strain, we compared the lengths of the stretched and unstretched wells in the primary direction of the stretch, as shown here for 0% strain (1.00x, unstretched) and 23% compressive strain (1.23x, pre-stretched). Scale bar, 20 mm. (G) Representative stress relaxation of lrECM gel under compression. Stress in gels relaxed to zero with a characteristic time constant on the scale of a minute (relaxation time 66 ± 16 s, mean ± SEM, N = 4 gels). Inset, detail of the first 2 min of the stress relaxation trace. (H) Comparison of moduli at 0.01% and 21.5% strain showed that lrECM gels did not significantly stress stiffen or soften under strains applied in this study (two-sided t-test, p=0.579, 0.699; N = 5 gels, mean ± SEM).
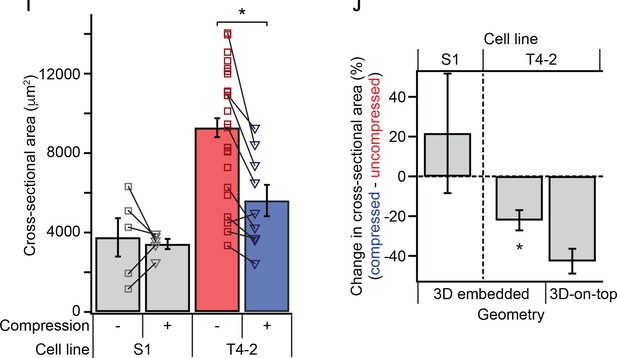
Mechanical reversion by transient compression occurs in both 3D embedded and 3D-on-top culture geometries.
(I) 23% compression of single malignant cells led to reduction in colony size. *Difference is significant at p<0.05 in one-way analysis of variance with Tukey-Kramer multiple comparison test. Each point represent mean colony size within an individual experiments, with paired compressed (triangles) and uncompressed (squares) and 99 ± 25 colonies per condition (mean ± SD). Lines indicate paired gels. This is an alternative representation of the data in Figure 1C with additional unpaired colony size measurements. (J) Percent change in cross-sectional area of colonies in 3D embedded and 3D-on-top geometries after 10 days of culture. Malignant cells demonstrated a reduction in colony size with compression in either geometry. Each bar represents aggregate of paired samples from the same gel preparation, compressed and uncompressed (N = 5, 9, and 2 gels, mean ± SEM, with 147 ± 27 colonies per sample [mean ± SD]). *Difference is statistically significant at p<0.02 in paired t-test.
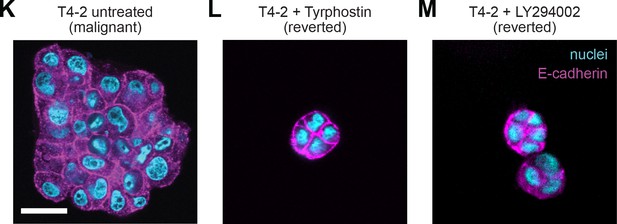
Chemical agents revert the malignant phenotype.
Representative confocal immunofluorescence images of T4-2 colonies after 10 days of embedded culture in lrECM with (K) no drug treatment, (L) the EGFR inhibitor Tyrphostin AG 1478 (70 nM) or (M) with PI3K inhibitor LY294002 (4 μM). Scale bar 25 μm.
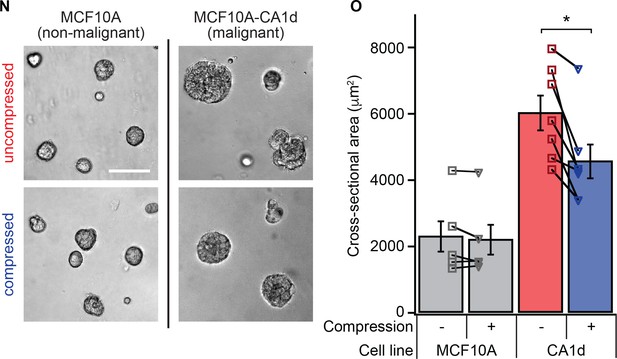
Transient compression reduces growth of in cultured malignant cells of the MCF10A series.
(N) Brightfield images of compressed and uncompressed cells of the MCF10A breast cancer progression series. Non-malignant MCF10A cells and malignant MCF10A-CA1d cells were seeded and compressed in the 3D-on-top culture geometry as was done with the HMT3522 series cell lines. (O) After 5 days of culture, colony size in malignant cells was reduced by 24 ± 5% (mean ± SEM; p=0.007 by paired t-test, N = 7 pairs of gels) in malignant cells with no reduction in colony size in non-malignant cells (p=0.29 by paired t-test, N = 5 pairs of gels). Bars represent mean colony cross-sectional area across experiments. Each point represents the mean colony size within a condition in an individual experiment with 104 ± 21 colonies per condition (mean ± SD). Lines indicate paired gels within an experiment.
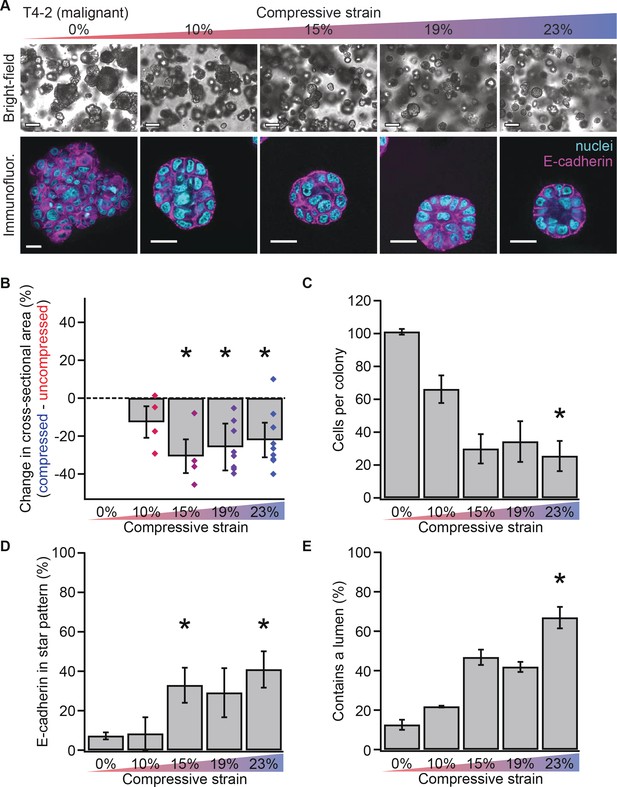
Mechanical reversion occurs above a threshold strain.
(A) Bright-field and confocal immunofluorescence images of malignant T4-2 cell colonies grown for 10 days in compressed gels. Scale bars 100 μm bright-field and 25 μm confocal. (B) Colonies grown in compressed matrices were smaller (N = 4, 4, 7, and 9 gels, each with paired uncompressed control, bars mean ± SEM, points are means of individual experiments, with 121 ± 62 colonies per sample [mean ± SD]). *Differences are statistically significant at p<0.05 compared to control samples in paired t-test with a gel made from the same preparation. (C) Compressed colonies contained fewer cells as measured by counting nuclei (N = 11, 3, 3, 2, and 4 gels, mean ± SEM, with 40 ± 18 colonies per sample [mean ± SD]). *Difference is statistically significant at p<0.02 compared to control samples in paired t-test with a gel made from the same preparation. (D) Additionally, colonies grown in compressed matrices had multicellular architectures resembling non-malignant colonies (N = 15, 3, 3, 2, and 6 gels, mean ± SEM, with 39 ± 17 colonies per sample [mean ± SD]). *Differences are statistically significant at p<0.001 in analysis of variance. (E) Compression increased the frequency of lumen formation (N = 11, 3, 3, 2, and 4 gels, mean ± SEM, with 40 ± 18 colonies per sample [mean ± SD]). *Difference is statistically significant at p<0.001 in analysis of variance.
-
Figure 2—source data 1
T4-2 colony size and architecture by compressive strain.
- https://doi.org/10.7554/eLife.26161.009
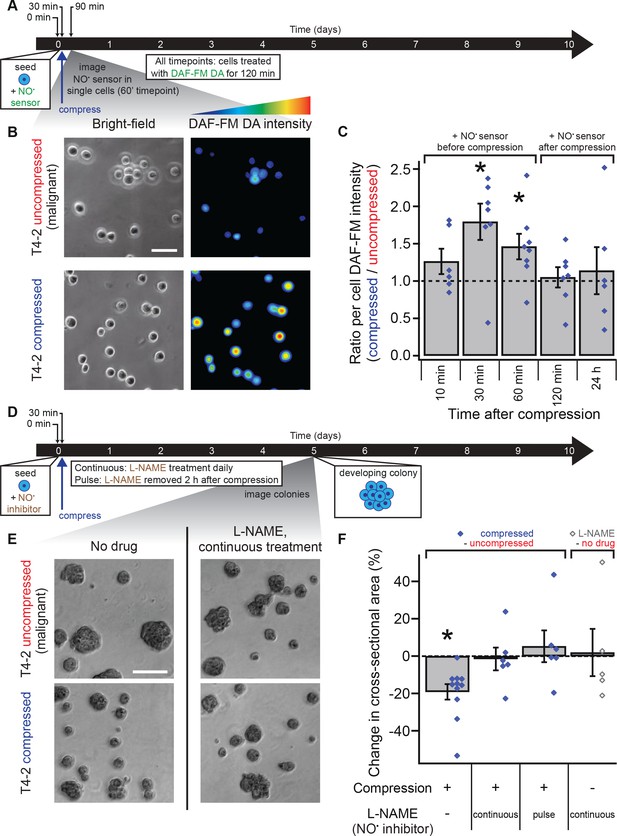
Mechanical reversion occurs via nitric oxide signaling.
(A) Malignant T4-2 cells were compressed 30 min after seeding and coating with 5% lrECM drip in the presence of the nitric oxide (NO) sensor dye DAF-FM DA. At the indicated times after compression, cells were washed with sensor-free media and imaged in wide field fluorescence. For all time points, cells were treated with DAF-FM DA for 2 hr before being washed and imaged. For 10, 30, and 60 min time points treatment with DAF-FM DA began before compression. For 120 min time point, treatment began immediately after compression. For 24 hr time point, treatment did not begin until 22 hr after compression. (B) Bright-field and fluorescence images of compressed and uncompressed malignant T4-2 cells. Scale bar 50 μm. The fluorescent images were pseudocolored from the raw images in ImageJ using one of the default colorscales (blue-to-red). (C) Compression induced NO production in T4-2 cells, as measured by DAF-FM DA fluorescence (N = 6, 7, 8, 7, 6 gels, each with paired uncompressed control, mean ± SEM, with 21 ± 9 cells per sample [mean ± SD]). *Difference is statistically significant at p≤0.03 compared to control samples in paired t-test. (D) T4-2 cells were compressed 30 min after seeding and coating with 5% lrECM in the presence of the NO inhibitor L-NAME. The media was replaced with L-NAME free media after two hours (pulse treatment) or with fresh media containing L-NAME daily (continuous treatment). Five days after compression, cells were imaged. Scale bar 200 μm. (D) Bright-field images of T4-2 colonies, compressed at the single-cell stage and treated continuously with the 0.5 mM L-NAME. Scale bar 200 μm. (F) Treatment of T4-2 cells with the NO inhibitor L-NAME blocked growth sensitivity to compression while L-NAME treatment alone did not alter colony size (N = 11, 6, 6, and 5 gels, points are means of individual experiments, with 176 ± 30 colonies per sample [mean ± SD]). *Difference is statistically significant at p<0.02 compared to uncompressed control samples in paired t-test with a gel made from the same preparation. Treatment of T4-2 cells with NO donor molecules in the absence of compression also yielded smaller colonies (Figure 3—figure supplement 1).
-
Figure 3—source data 1
Nitric oxide sensor in T4-2 cells and nitric oxide synthase inhibitor-treated T4-2 colonites.
- https://doi.org/10.7554/eLife.26161.012
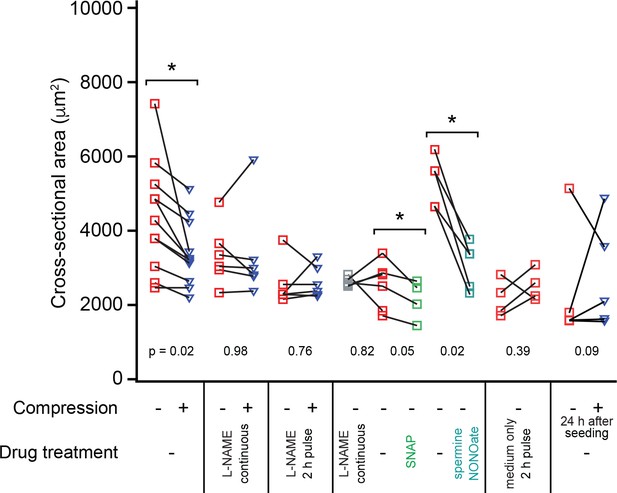
Pairwise comparison of colony sizes while manipulating nitric oxide conditions and mechanical stimulation.
Cross-sectional area of T4-2 colonies after 5 days of culture under a given pair of condition across individual experiments. Each point is the mean colony size of 159 ± 42 colonies (mean ± SD; range 69–331 colonies) of a given condition. Squares represent uncompressed colonies; triangles are compressed colonies. Only compression alone or addition of the nitric oxide donor molecules SNAP or spermine NONOate produced statistically significant reduction in colony sizes as compared with matched controls (p<0.05 by paired t-test). From left to right, pairs of conditions are: compressed vs. uncompressed, compressed vs. uncompressed with 0.5 mM L-NAME supplemented daily, compressed vs. uncompressed with 0.5 mM L-NAME at the time of compression and washed out after 2 hr, 0.5 mM L-NAME vs. untreated vs. 10 μM SNAP (each supplemented daily), untreated vs. 5 μM spermine NONOate (only initial treatment), untreated (left) vs. medium exchange 2 hr after seeding (right), compressed vs. uncompressed where compression was applied 24 hr after seeding. The first four pairs of conditions are an alternative presentation of the data in Figure 3F. Several of the control conditions for NO inhibitor and donor treatments were shared in PDMS chambers containing more than two wells (shared on plot for comparison), and several of the control conditions for medium exchange experiments were also used as controls for L-NAME and SNAP treatments (duplicated on plot for clarity).
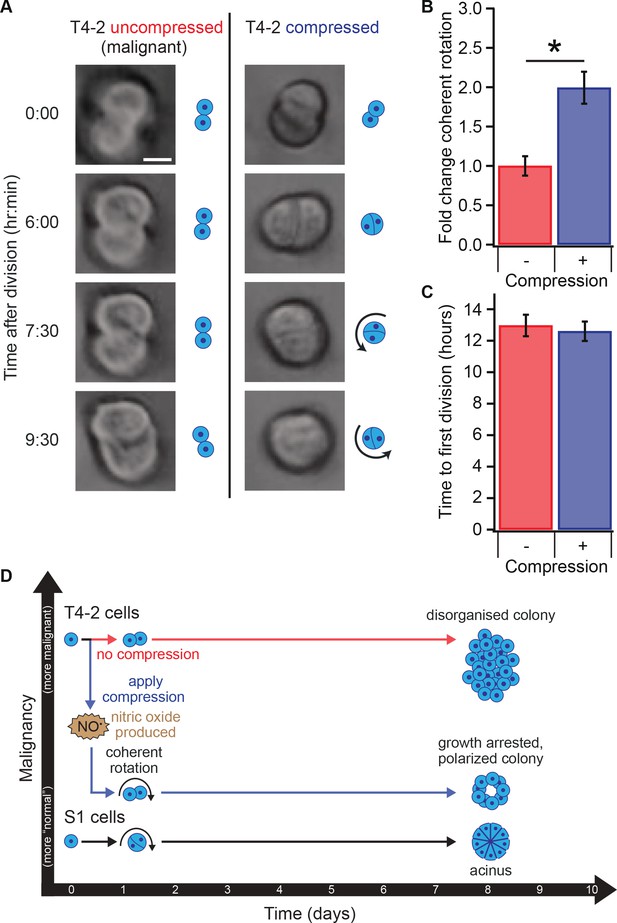
Compression allows restoration of coherent rotation after division.
(A) Time series of malignant T4-2 cells growing in uncompressed and compressed gels. Scale bar 10 μm. Time series were taken from Figure 4—Video 1 and Figure 4—Video 1. (B) Cells at the two-cell stage rotated coherently more often in compressed matrices. Fold change coherent rotation is the ratio of the fraction of cell doublets that exhibited rotation with compression to the fraction that exhibited rotation without compression (Fold change = 1, for uncompressed gels). *Fold change is statistically significant with p<0.01 in a paired t-test with control samples made from the same preparation and imaged simultaneously. Coherent rotation behavior depends on proper cell-cell contacts, and treatment of cells with function-blocking E-cadherin antibodies disrupted sensitivity to compression (Figure 4—figure supplement 1). (C) Time to first division of T4-2 cells was not sensitive to compression. For B and C, N = 4 gels, each with paired control, mean ± SEM, with 49 ± 29 cell doublets per sample (mean ± SD). *Fraction of cell doublets that underwent coherent rotation is statistically significant with p<0.02 in a paired t-test with control samples made from the same preparation and imaged simultaneously. (D) Compression induces nitric oxide production in single breast epithelial cells, which activates ‘normal’ signaling and development and diminishes the malignant character of colonies days after the compression occurred.
-
Figure 4—source data 1
- https://doi.org/10.7554/eLife.26161.015
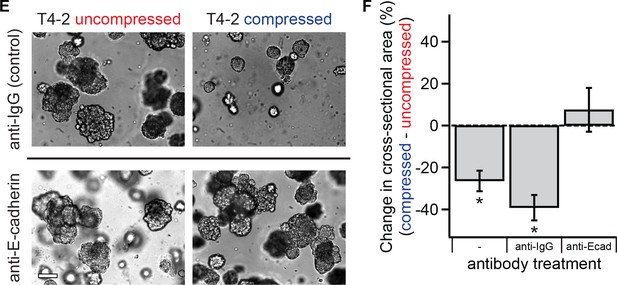
E-cadherin function is required for mechanical reversion.
Single malignant T4-2 cells were treated with function-blocking E-cadherin antibody before compression was applied (E) Bright-field images of colonies treated with function-blocking E-cadherin antibodies or control. Scale bar 100 μm. (F) Blocking E-cadherin prevented compression-induced reduction in colony size (N = 6, 3, 3 gels, each with paired control, mean ± SEM, with 174 ± 111 colonies per sample [mean ± SD]). Once E-cadherin function was blocked, the resulting cells and colonies did not exhibit a compression response (1-sided paired t-test, p=0.80). *Significant at p<0.05 in a paired t-test with control samples made from the same preparation.
Uncompressed malignant T4-2 doublets undergo incoherent movement.
Time-lapse of uncompressed malignant T4-2 breast epithelial cells. Scale bar 25 μm, time stamp hours:minutes.
Compression restores coherent rotation of malignant T4-2 doublets.
Time-lapse of compressed malignant T4-2 breast epithelial cells. Scale bar 25 μm, time stamp hours:minutes.
Additional files
-
Transparent reporting form
- https://doi.org/10.7554/eLife.26161.018





