Cortex-wide BOLD fMRI activity reflects locally-recorded slow oscillation-associated calcium waves
Figures
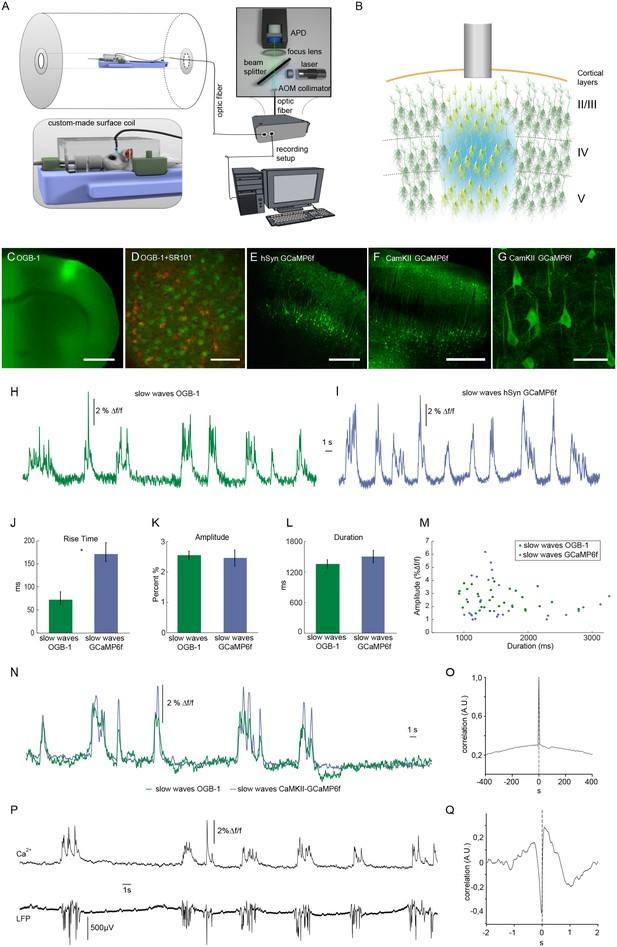
Optical recordings of slow oscillation-associated calcium waves within a small animal MR scanner in anesthetized rats.
(A) Scheme of optical recording setup comprising of a solid state laser (488 nm) coupled to a multimode optic fiber with a diameter of 200 μm. Emitted light is recorded by an avalanche photo diode (APD) and converted to a voltage signal. For simultaneous fiber-based calcium recordings and fMRI, a custom-built surface coil with a lead-through for the optic fiber is employed. (B) Upon bolus loading with OregonGreen BAPTA-1 (OGB-1) or after viral transduction with GCaMP6f, an optic fiber (grey shaded rectangle) is implanted in primary somatosensory cortex to record an integrated, yet spatially defined neural population signal. Cortical layers are indicated. (C) Fluorescence micrograph of a coronal brain slice depicting the area of OGB-1 staining at the level of primary somatosensory cortex, scale bar = 1 mm. (D) Fluorescence micrograph of co-staining with OGB-1 (green) and SR101 (red) (scale bar = 100 µm) (E) Expression of GCaMP6f four weeks after injection of AAV1.Syn.GCaMP6f (scale bar = 500 µm). (F–G) Expression of GCaMP6f four weeks after injection of AAV1.CamKII.GCaMP6f scale bar F = 500 µm G = 50 µm. (H) Calcium trace upon OGB-1 staining recorded in the MR scanner during slow wave activity. Optically recorded spontaneous stereotypical calcium waves are interrupted by periods of network quiescence. (I) Optical recordings in GCaMP6f expressing rats similarly reveal slow calcium waves. (J–M) Quantifications of slow calcium wave parameters in OGB-1 stained vs. GCaMP6f expressing rats reveal significant differences only in rise times (p=0.0001), whereas amplitude and durations are not significantly different (p=0.13; p=0.61). (N) Simultaneous optical calcium recordings outside the MR scanner using two optic fibers implanted into left hemisphere with OGB-1 staining (green trace) and with CaMKII-GCaMP6f staining. An overlay of recorded traces shows that with both indicators the occurrence of a slow wave is reflected by similar transients. (O) Cross-correlation of simultaneously recorded optical calcium signals obtained in (N) indicates high similarity between both signals. (P) Simultaneous optical calcium and LFP recordings outside of the MR scanner indicating correlation of slow calcium waves with electrically recorded slow oscillations under isoflurane anesthesia. (Q) Cross-correlation of optically and electrically recorded traces result in a prominent peak with almost zero time shift, indicating a close similarity of the two signals.
-
Figure 1—source data 1
Amplitude, duration and rise time values for quantification of OGB-1 slow calcium wave events (data shown in Figure 1J–M).
Rise time, duration and amplitude of calcium transients were assessed and statistically compared in animals measured upon OGB-1 injections (n = 8 animals). Rise time was defined as time between onset of the transients and 50% of maximum intensity peak. Duration of the transients was defined as the time between the first and the last intensity value exceeding 50% of maximum intensity peak. Amplitude was determined as the intensity difference (∆f) from the baseline and the highest intensity value of the transients.
- https://doi.org/10.7554/eLife.27602.005
-
Figure 1—source data 2
Amplitude, duration and rise time values for quantification of GCaMP6f slow calcium wave events (data shown in Figure 1J–M).
Rise time, duration and amplitude of calcium transients were assessed and statistically compared in GCaMP6f transduced rats (n = 2 animals). Rise time, duration and amplitude were defined as described for OGB-1 slow calcium wave events (Figure 1—source data 1).
- https://doi.org/10.7554/eLife.27602.006
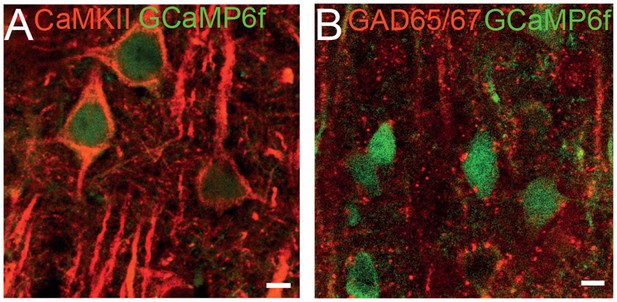
Immunofluorescent characterization of GCaMP6f expressing neurons.
(A) Confocal micrograph of GCaMP6f expression under CaMKII promoter co-stained with a CaMKII antibody. (B) Confocal micrograph of GCaMP6f expression under CaMKII promoter co-stained with a GAD 65/67 antibody. (Scale bars = 10 µm).
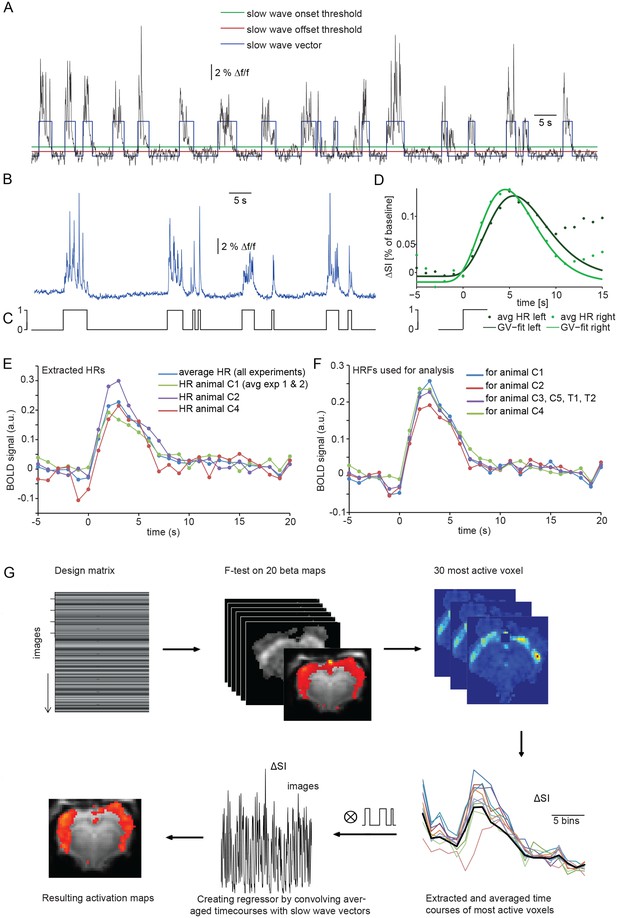
Identifying a BOLD response upon slow wave events detected in cortical calcium recordings.
(A) Onset and termination thresholds based on EMA filters detect slow wave events in a calcium trace recorded in the MR scanner and can be used to create a slow wave vector. (B,C) The binarized slow wave vector (C) containing the precise timing of the slow calcium wave events (B) can be used as an event array for subsequent fMRI analysis. (D) Averaged BOLD signal in cortical ROIs in both hemispheres of an individual experiment upon the onset of slow calcium wave events, fitted with a gamma variate (GV). Note, that due to subsequent slow calcium wave events within the time frame of the respective BOLD response (see A,B), the raw signal does not decay to baseline levels. (E) Extracted hemodynamic responses (HRs) with finite impulse response (FIR) methods. Intra- and interindividual timecourses show almost identical signal characteristics. (F) Individual HRs were averaged and used as HRF for subsequent fMRI analysis (for animal C1 HRs from animal C2 and animal C3 were averaged; for animal C2 HRs from animal C1 and animal C3; for animal C3 HRs from animal C1 and C2; for animal C4 HRs from animal C1, C2; for animal C3, C5, T1 and T2 HRs from animal C1, C2 and C4 were averaged). (G) Flow chart of fMRI analysis procedure starting with the design matrix, defined by the FIR basis function set and the spontaneous calcium wave as event.
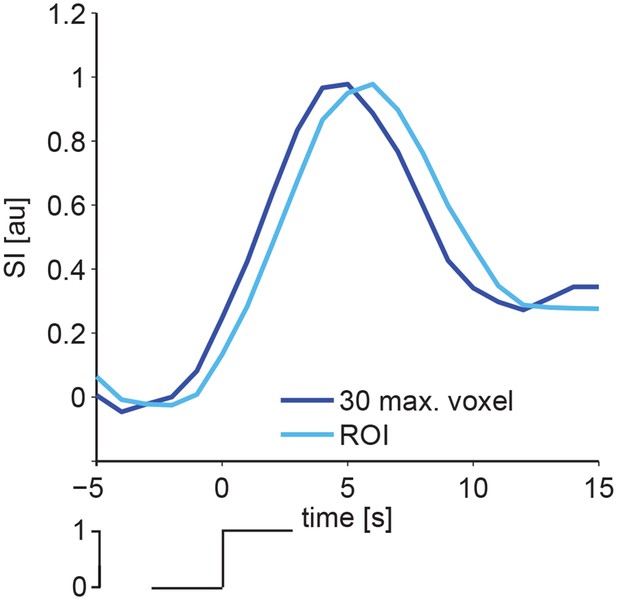
Comparison of HR extracted in the 30 most active voxel and in ROI placed in a cortical area.
Timecourses of BOLD responses upon an averaged slow wave onset reveal similar signal dynamics for a ROI defined from the 30 voxel of highest intensity in the previously calculated F-maps (dark blue curve) or a ROI placed in a cortical area showing BOLD activation (light blue curve).
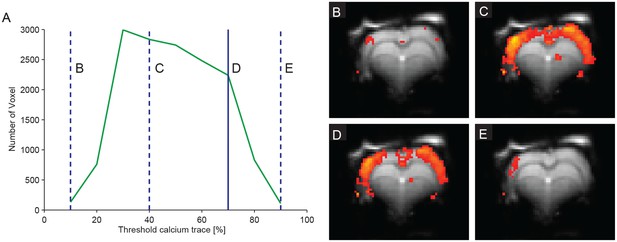
Number of active voxel depends on the threshold for calcium wave detection.
(A) Graph depicting the relation of active voxel revealed by a SPM fMRI analysis and varying thresholds for the detection of the calcium waves. (B–E) Activation maps at 10%, 40%, 70% and 90% threshold in the same animal (C2).
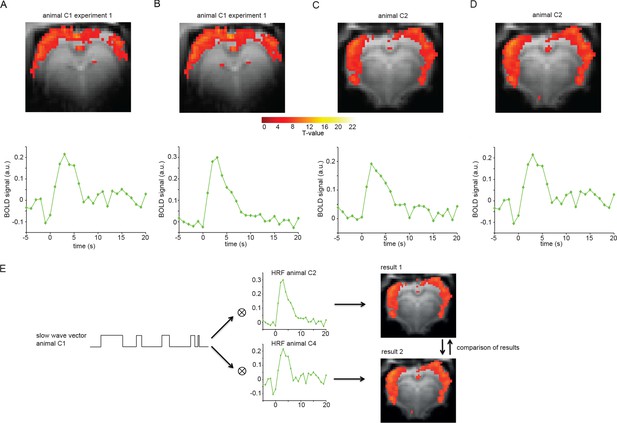
Cross-validation procedure to test for impact of respective dataset used for HRF extraction.
(A) Animal C1 experiment 1 analyzed with HRF extracted from animal C4; cluster size = 810, T-value (max.)=10.86. (B) Same animal as in (A) analyzed with HRF extracted from animal C2; cluster size = 641, T-value (max.)=11.36. (C) Animal 2 analyzed with HRF extracted from animal C1 (average of both experiments); cluster size = 1492, T-value (max.)=13.16. (D) Same animal as in (C) analyzed with HRF extracted from animal C4; cluster size = 1912, T-value (max.)=13.95. (E) Flow chart depicting cross-validation procedure.
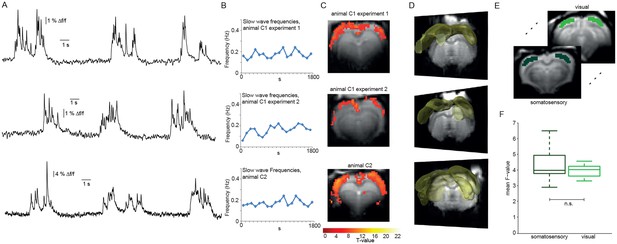
Relating neurophysiologically defined slow calcium wave events to fMRI BOLD signal reveals cortex-wide activation.
(A) Excerpts of calcium traces acquired during fMRI measurements in three experiments depicting slow calcium waves. (B) Frequencies of slow calcium waves within an imaging experiment. (C) BOLD correlate upon FIR-based event-related fMRI analysis in the cortex. (D) 3D renderings depict cortex-wide expansion of activity shown in (B) (light-yellow shading shows above threshold activity). (E,F) Quantitative comparison of BOLD activation in somatosensory (10 ROIs, 4 animals, 5 experiments) and visual cortex (6 ROIs, 4 animals, 5 experiments). No significant difference in the mean F-values could be detected (mean F-values in somatosensory cortex: 4.3 ± 0.3, in visual cortex: 4.0 ± 0.1, p=0.41).
-
Figure 3—source data 1
Cluster sizes and T-values for experimental HRF versus canonical HRF for all experiments (Table for data shown in Figure 3—figure supplement 2).
Mean T-value over all ROIs for canonical HRF: 3.95 ± 0.53, mean T-value for FIR-extracted HRF: 3.91 ± 0.32. Means do not differ significantly between groups (Wilcoxon rank-sum test).
- https://doi.org/10.7554/eLife.27602.015
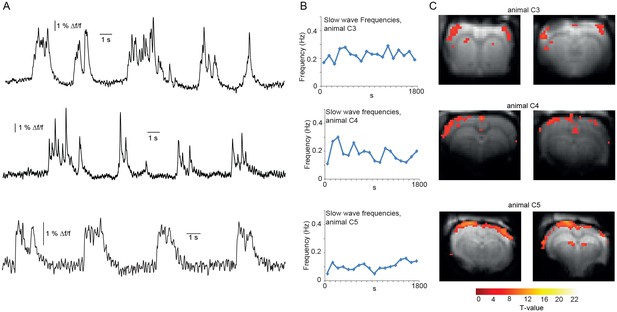
BOLD correlate upon FIR-based event-related fMRI analysis of additional datasets.
(A) Excerpts of calcium traces acquired during fMRI measurements depicting slow calcium waves. (B) Frequencies of slow calcium waves within an imaging experiment. (C) Corresponding BOLD correlates upon slow calcium wave events.
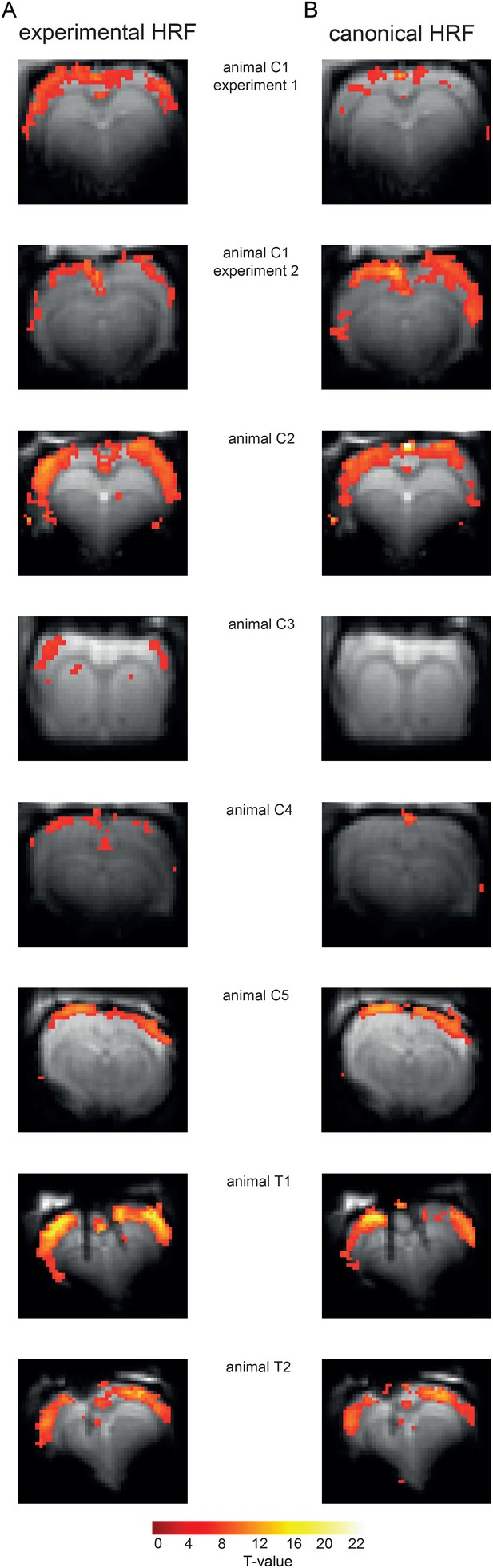
Similar activity patterns upon employing HRFs extracted from datasets based on FIR approach and classical canonical HRF model.
All activation maps were scaled to the overall maximum detected T-value (note that this maximum T-value in animal C2 (B) is only achieved because of spurious activity in the sinus vein detected by the canonical HRF approach). (A) Activation maps resulting from FIR-based event-related fMRI analysis of datasets shown in Figure 3, Figure 3—figure supplement 1 and Figure 4. (B) Activation maps resulting from event-related fMRI analysis of the same datasets while using the standard canonical HRF implemented in SPM, which is modelled by two gamma functions.
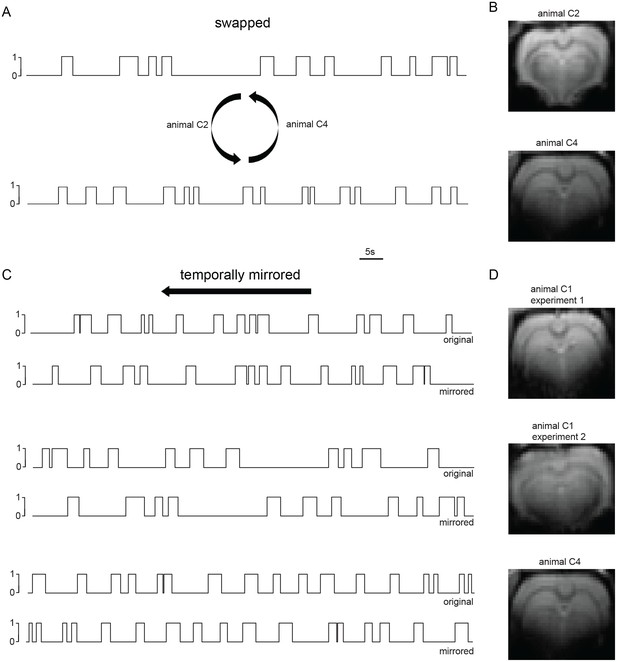
Slow wave BOLD correlates depend on precise slow calcium wave dynamics detected in individual experiments.
(A, B) Swapping of slow wave vectors between two animals (A) reveals no BOLD activity upon FIR-based event-related fMRI analysis (B). (C, D) Temporally mirroring slow wave vectors within the same experiment (C) again reveals no activation (D).
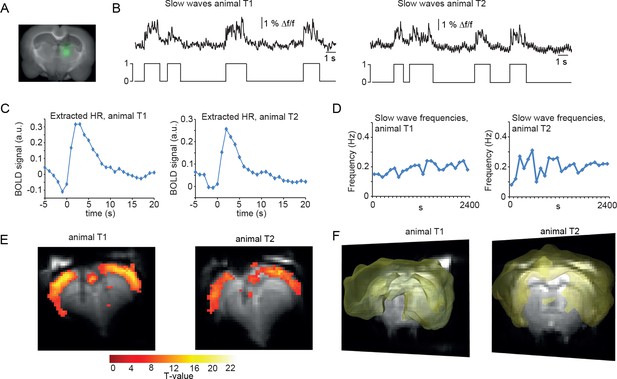
BOLD correlate upon FIR-based event-related fMRI analysis using slow wave vectors acquired in the thalamus.
(A) Micrograph of a coronal brain slice depicting the area of OGB-1 staining at the level of thalamus (POm). (B) Excerpt of calcium trace acquired in thalamus during fMRI measurements depicting slow calcium waves and the binarized slow wave vector derived from these calcium waves. (C) Extracted hemodynamic responses (HRs) with FIR methods. The time course shows almost identical signal characteristics as in previous datasets using cortical calcium recordings. (D) Frequencies of slow calcium waves within an imaging experiment. (E) BOLD correlate upon FIR-based event-related fMRI analysis in the cortex of animal T1 and T2. (F) 3D rendering depicts cortex-wide expansion of activity shown in (E) (light-yellow shading shows above threshold activity).
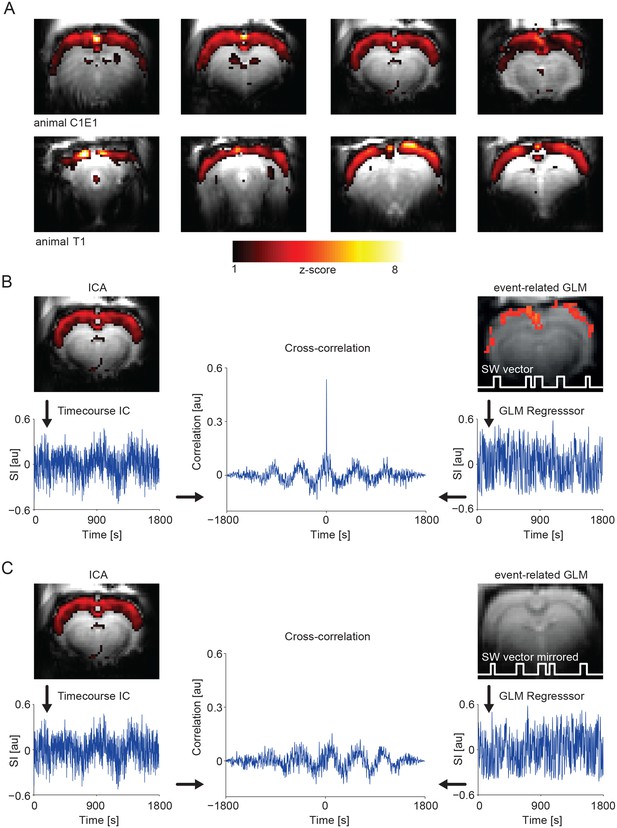
IC analysis in slow activity results in a component showing pan-cortical activation specifically correlating to the calcium slow wave vector.
(A) Pan-cortical component in slow wave activity (isoflurane anesthesia). Spatial correlate of two exemplary ICs showing consistent high values covering the entire cortex. Two representative animals and four imaging slices each are depicted. (B) The timecourse of the pan-cortical activation component is highly correlated with the regressor extracted from the slow wave vector (SW vector) obtained from the calcium recordings (r = 0.535 and r = 0.628) for upper and lower ICs shown in (A), when this SW vector is mirrored in time, this correlation completely disappears (C). (B) and (C) both shown for animal C1E1.

Complete component set of IC analysis in slow wave activity.
(A) ICs derived from group ICA (B) corresponding single subject ICs sorted by their mean power in the range up to 0.1 Hz (higher to lower). ICs extracted based on minimum description length criterion (MDL) originally revealed 37 components, after visual inspection all components showing only activity outside the brain were discarded, resulting in a total of 23 components shown on 21 slices with a distance of 0.6 mm between the slices and an isovoxel resolution of 0.3 mm³ after DARTEL normalization and reconstruction. Components #3 and #6 show pancortical activation (outlined in red), components related to typical default mode activity networks including auditory (IC #1) and visual (IC #8) cortex, striatum (IC #7) and hippocampus. (IC #20) were also identified (outlined in green).
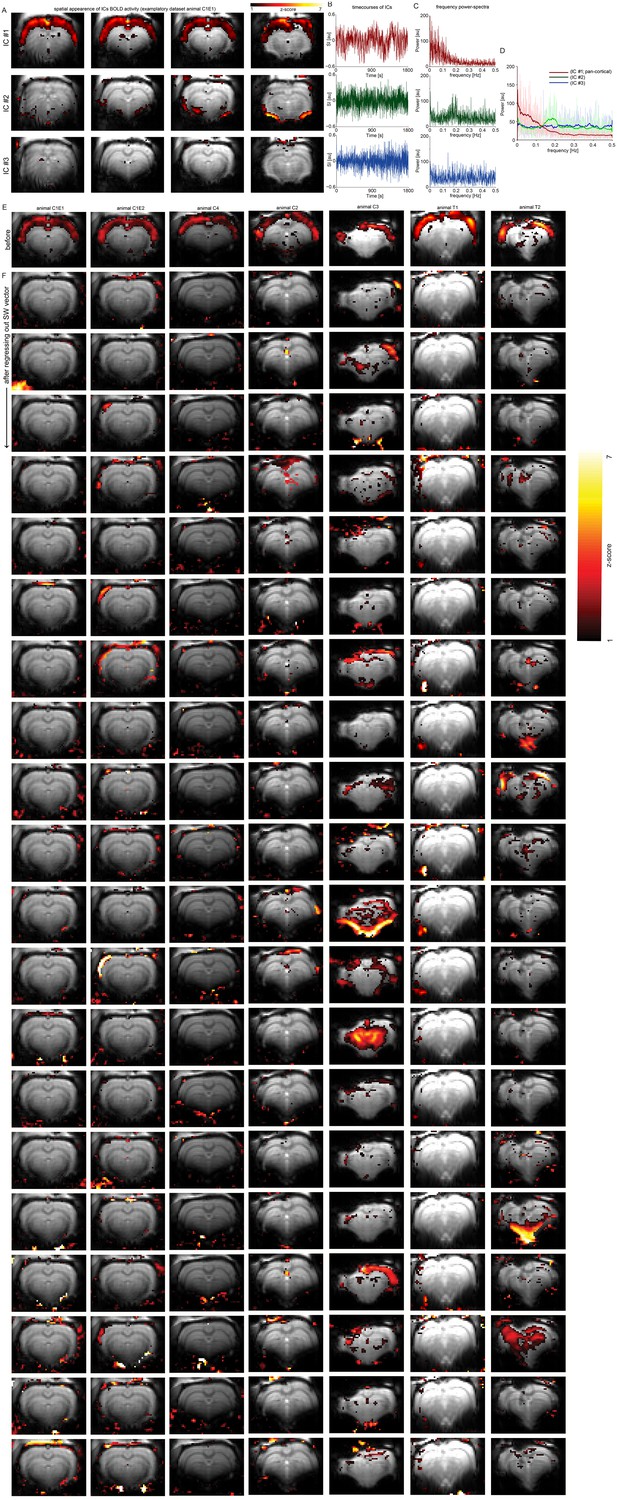
Pan-cortical BOLD appears only in ICs with higher power in lower frequencies of their time courses’ power spectra.
Regressing out the slow calcium wave regressor before ICA leads to absence of pan-cortical activity ICs. (A) ICs of pancortical activation (1), non-cortical activity (2) and activity ourtside of the brain (3). (B) Timecourses of these three ICs. (C) Power spectra derived by Fast Fourier transform of the IC time courses. (D) The three independent components are related to differences in the frequency power-spectra of their timecourses. The component revealing pan-cortical activity (1) shows a timecourse which has higher power in frequencies in the range up to 0.1 Hz, the components which are related to other types of activity show power mostly in higher frequency ranges, i.e. unspecific non-cortical activity (2) shows a peak in frequencies around 0.18–0.2 Hz, or a component showing mostly activity outside of the brain and is likely related to unspecific noise (3) does not show specific frequency peaks which could be attributed to any type of oscillatory sources. (E) ICA revealed component showing pan-cortical activation prior to regressing out the timecourse derived by the slow calcium wave vector (SW vector) of the datasets (one examplatory slice is shown each). (F) The previously present pancortical component (E) is absent in all of the 20 components in all datasets after regressing out the timecourse derived by the slow calcium wave vector previously to performing ICA.
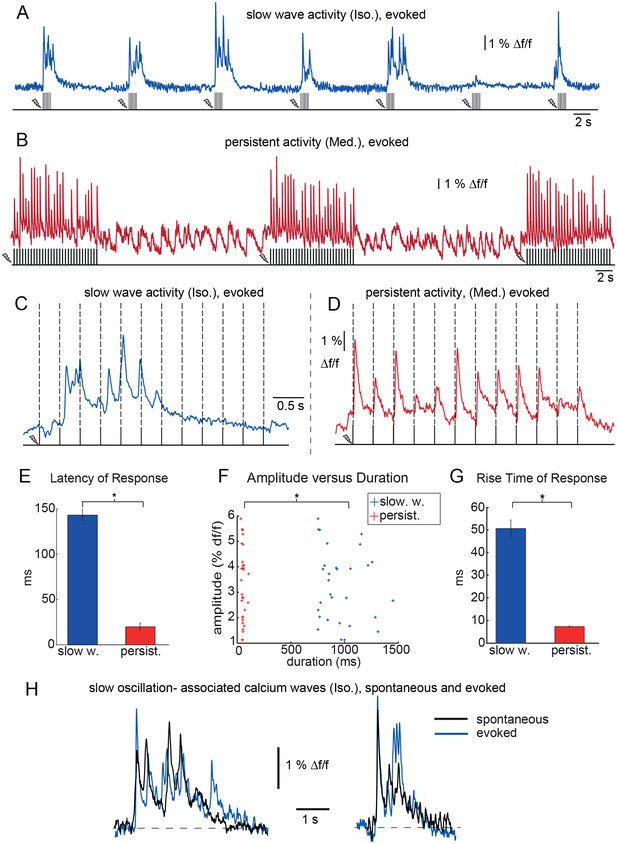
Neuronal response patterns upon sensory stimulation for slow wave and persistent activity.
(A) Calcium trace recorded during slow wave activity (blue). Electric forepaw stimulation at 9 Hz for 1 s every 10 s. Calcium waves were evoked in an all-or-none manner. The last pulse train started during the occurrence of a spontaneous wave. (B) Calcium trace recorded during persistent activity (red). Electric forepaw stimulation at 3 Hz for 10 s every 30 s resulted in short-latency calcium transients upon each pulse. (C) Individual sensory evoked slow calcium wave, note that wave is locked to the onset of the first stimulus with a latency of 140 ms (stimulation paradigm: 3 Hz, 4 s). (D) Sensory-evoked calcium responses during persistent activity (stimulation paradigm: 3 Hz, 4 s). Each pulse evoked an individual response. (E) Latencies of evoked calcium waves during slow wave activity (blue) vs. responses during persistent activity (red). Latencies differ significantly by more than 100 ms. (F) Scatter plot displaying mean response amplitudes versus durations measured at 50% of maximum peak in the two conditions. Duration of responses (slow wave vs. persistent response) differs significantly by more than 900 ms. (G) Mean rise times of responses in the the two conditions differ significantly. Asterisks indicate significant difference (rank sum test, p<0.0001, 30 events, three animals). (H) Slow oscillation-associated calcium waves evoked by sensory stimulation are indistinguishable from spontaneously occurring waves. For slow wave activity, we observed electric forepaw stimulation to reliably evoke slow waves locked to stimulus onset (A) with a mean latency of 143 ms ± 7 ms (30 events, three animals, (E), in reasonable agreement with studies in mice using visual or auditory stimulation (Grienberger et al., 2012; Stroh et al., 2013). Regarding signal characteristics, stimulus-evoked calcium waves were not distinguishable from spontaneous waves (H), again in agreement with previous studies (Stroh et al., 2013). Stimulating with multiple pulses with a frequency of 3 Hz and pulse train duration of 4 s led to calcium waves with latencies similar to those waves induced by single pulses (C). Subsequent pulses within a given pulse train did not evoke additional waves, suggesting refractory behavior of those waves, as previously shown in mice. Waves followed all-or-none characteristics (A), with a probability of induction of 72%±8% (five animals, 12 traces). In the persistent activity condition, we detected neuronal calcium responses upon sensory stimuli with a significantly shorter latency of 20 ms ± 4 ms and a significantly shorter duration of 55 ms ± 3 ms, in agreement with previous studies (Schmid et al., 2016) versus 959 ms ± 36 ms in slow wave state (30 events, three animals, (E, F). The amplitudes of persistent activity responses slow waves did not differ significantly (F), but varied between individual animals due to different staining efficiencies. It has to be noted that amplitudes of calcium transients can only be meaningfully compared within one experiment, as the fluorescence amplitudes depend on many local parameters, such as the concentration of the calcium indicator in the region and the position of the optic fiber. Responses during persistent activity exhibited significantly shorter rise times compared to slow waves (G), suggesting a different mechanism of activity initiation.
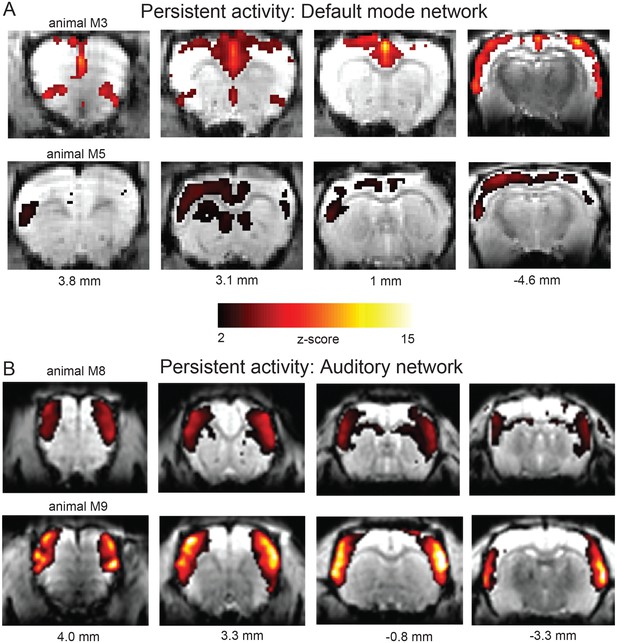
IC Analysis showing typical resting state networks during persistent activity.
(A) ICA component showing canonical default mode network activation. The activation maps of two representative animals (out of 4) are depicted (labelled with AP coordinates in mm relative to bregma): At 3.8 mm activation of orbital and prelimbic cortex can be observed, at 3.0 and 1.0 mm cingulate cortex and at 4.6 mm temporal association cortex, posterior parietal cortex and retroslenial cortex. (B) Auditory cortical network component from the same animals as in (A). This component resembles activation of an auditory network involving sensorimotor (medial superior) and auditory (inferior parietal) cortices.
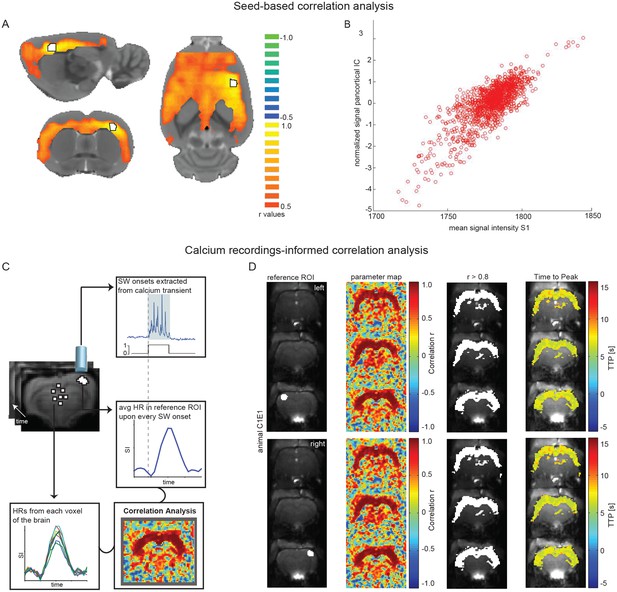
Seed-based and calcium recordings-informed correlation analyses reveal pan-cortical BOLD activation during slow wave activity.
(A) Signal correlation between primary somatosensory cortex ROI (S1; white with black outline) and the rest of the cortex in a representative animal during slow wave activity. (B) Correlation between the average signal in S1 and the pan-cortical independent component (IC) from this representative animal’s ICA (pearson correlation coefficient, r = 0.799, p<0.001). (C) Flow-chart of correlation analysis procedure employing slow wave onsets from calcium transients. (D) ROIs on both hemispheres (ipsi- and contralateral sites of the fiber recordings), parameter maps showing r-values and regions of highest correlation (r > 0.8; white). The mean HR was calculated from these thresholded regions and time to peak of voxel-by-voxel HR was determined.
-
Figure 6—source data 1
Values for seed-based correlation between S1 and cortex during slow wave and persistent activity (n = 4 animals; table for data shown in Figure 6 and Figure 6—figure supplement 1).
Number of clusters (>300 voxel) differ significantly between conditions (two-sample t-test (6)=−2.5538, p=0.0433).
- https://doi.org/10.7554/eLife.27602.026
-
Figure 6—source data 2
Values for seed-based correlation between hippocampal formation (hf) and cortex during slow wave and persistent activity (n = 4 animals; table for data shown in Figure 6—figure supplement 2).
Number of clusters (>300 voxel) differ significantly between conditions(two-sample t-test (6)= −3.6667, p=0.0105).
- https://doi.org/10.7554/eLife.27602.027
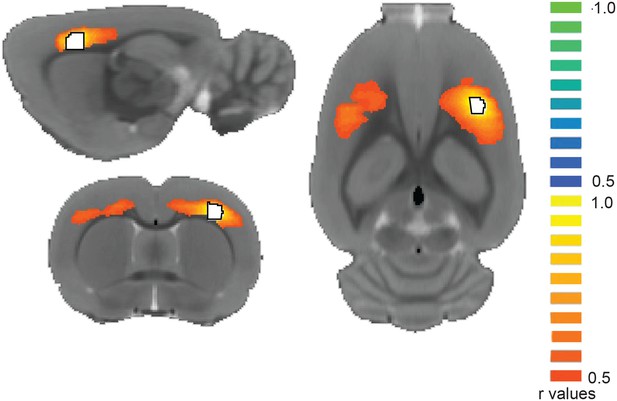
Seed-based correlation analysis during persistent activity.
During persistent activity signal correlation between primary somatosensory cortex ROI (white with black outline) and the rest of the cortex does not reveal pancortical BOLD activation but shows bilateral somatosensory activity (same animal as in main Figure 6A).
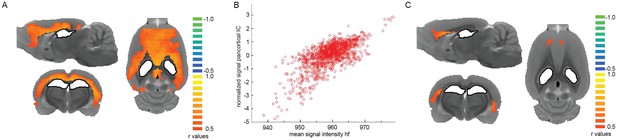
Seed-based correlation analysis originating from hippocampal formation (hf).
(A) Signal correlation between hippocampal ROI (white with black outline) and cortex in a representative animal during slow wave activity (isoflurane anesthesia). (B) Correlation between average signal in hf and the pan-cortical independent component (IC) from this animal’s ICA (pearson correlation coefficient, r = 0.701, p<0.001). (C) Signal correlation between hippocampal ROI and cortex in the same animal during persistent activity (medetomidine sedation) reveals activation in the prefrontal cortex to be correlated with the hippocampal seed.
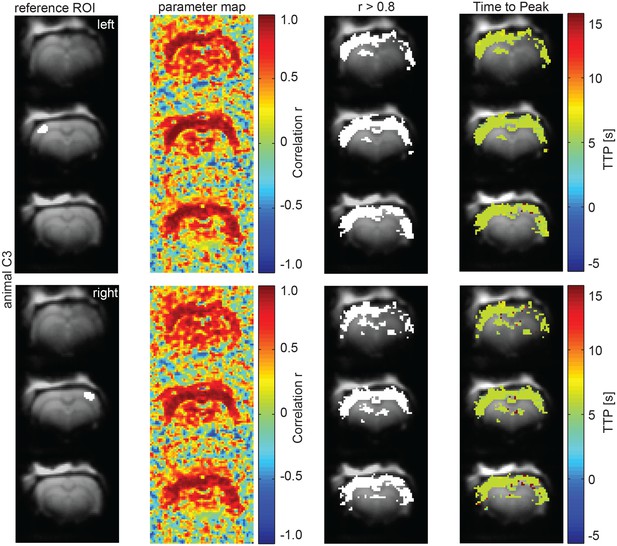
Calcium recordings-informed correlation analysis reveals pan-cortical BOLD activation during slow wave activity.
ROIs on both hemispheres (ipsi- and contralateral sites of the fiber recordings), parameter maps showing r-values and regions of highest correlation (r > 0.8; white). The mean HR was calculated from these thresholded regions and time to peak of voxel-by-voxel HR was determined (same analysis as in main Figure 6C,D in different animal).
Additional files
-
Transparent reporting form
- https://doi.org/10.7554/eLife.27602.028





