Organization of the Drosophila larval visual circuit
Figures
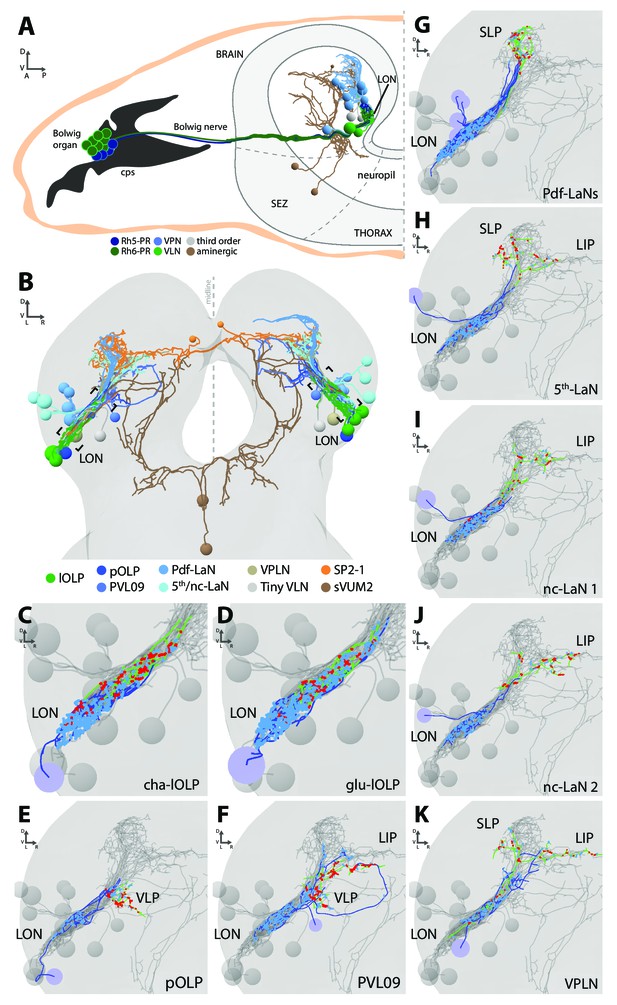
Overview of the larval optic neuropil.
(A) Schematic of the larval visual system with EM-reconstructed skeletons of all larval optic neuropil (LON) neurons. The Rh5-PRs (dark blue) and Rh6-PRs (dark green) cell bodies form the Bolwig organ sitting in the cephalopharyngeal skeleton (cps). They extend their axons to the brain via the Bolwig nerve. In the brain, neurons cell bodies are in the outer layer (gray) and project neurits into the neuropil. We can distinguish four main classes of neurons: visual projection interneurons (VPN, blue), visual local interneurons (VLN, green), third-order interneurons (gray) and aminergic modulatory neurons (brown). Octopaminergic/tyraminergic modulatory neurons cell bodies sit in the subesophageal zone (SEZ). (B) 3D reconstruction of all LON-associated neurons from the ssTEM dataset in both hemispheres (except Bolwig nerves): VLN in green: local optic lobe pioneer neurons (lOLPs); VPN in shades of blue: the projection OLP (pOLP), a novel neuron which is located in the posterior ventral lateral cortex (PVL09), the Pdf-lateral neurons (Pdf-LaNs), the 5th-LaN and the non-clock-LaNs (nc-LaNs); third-order neurons: VPLN in light brown and Tiny VLN in gray; aminergic modulatory neurons: serotonergic neuron (SP2-1, orange) and SEZ-ventral-unpaired-medial-2 octopaminergic/tyraminergic neurons (sVUM2, brown). Posterior view. (C–K) 3D reconstructions from ssTEM dataset, posterior view, dendrites in blue, axons in green, presynaptic sites in red, postsynaptic sites in cyan, other LON neurons in gray. VLP: ventral lateral protocerebrum. SLP: superior lateral protocerebrum. LIP: lateral inferior protocerebrum. Both lOLPs (cha-lOLP (C) and glu-lOLP (D)) have dense arborizations within the LON. (E) The pOLP cell body is situated with the lOLP and projects to the VLP. (F) PVL09 cell body is situated postero-ventro-laterally to the LON and has an axon with a characteristic loop shape, extending first towards the ventro-medial protocerebrum, then towards the LIP before curving down back to the VLP, where it forms most of its synaptic output. (G) The four Pdf-LaNs project to the SLP. (H) The 5th-LaN projects both to the SLP and the LIP region, whereas nc-LaN 1 and 2 (I and J) mainly project to the LIP. (K) The third-order neuron VPLN cell body is situated anteriorly to the LON and it has an axon coming back in the LON in top of its projections within both SLP and LIP regions.
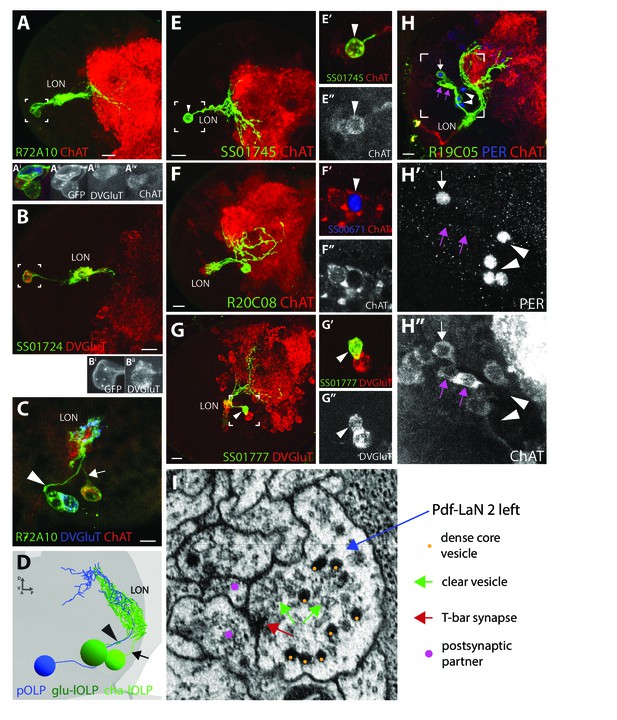
Neurotransmitter expression in the LON.
(A-C), (E-H) Confocal z-projections, dorsal view. (A) R72A10 > UAS-myr::GFP (green) showing the three OLPs (neuropil marker: ChAT, red) and close-up on the cells bodies (box) showing that one OLP is glutamatergic (Aiii) and at least another one is clearly cholinergic (Aiv). B: SS01724 >UAS-myr::GFP (green) showing a lOLP with dense arborization within the LON, reduced projections, and which is glutamatergic (DVGluT in red) (Bi), (Bii) close-up of the cell body (box)). (C) R72A10 > UAS-myr::GFP (green) close up on the three OLPs cell bodies where we can observe the strongly cholinergic cell (ChAT, red) sending its axon towards the LON via a different path (arrow) than the glutamatergic cell (DVGluT, blue) and the third cell (arrowhead). (D) 3D reconstruction of the three OLPs in the left hemisphere of the ssTEM dataset where we could observe two cells sending their axons together to the LON (arrowhead), whereas the third one takes a separate path (arrow). Comparing (C) and (D) and based on their anatomy, the three OLPs can be distinguished in the ssTEM dataset (D): the projection OLP (pOLP, blue) and the glutamatergic lOLP (glu-lOLP, green) axons fasciculate together (arrowhead) but not with the cholinergic lOLP (cha-lOLP, light green, arrow). (E) SS01745 >UAS-myr::GFP (green) showing the pOLP anatomy with weak arborization within the LON and deep projection into the neuropil. (E’, E’’) Close up on pOLP cell body showing a weak but clear cholinergic cell (arrowhead, ChAT in red/white). (F) R20C08 > UAS-myr::GFP (green) showing a single looping neuron with a strong overlap in the LON and having its cell body situated postero-ventro-laterally, corresponding to PVL09 (neuropil marker ChAT in red). (F’), (F’’) Close up on a PVL09 cell body (SS0671 >UAS-H2B-RFP, RFP in blue) that is cholinergic (ChAT, red, arrowhead). Single section. (G) SS01777 >UAS-myr::GFP (green) showing a stochastic single clone of the third-order neuron VPLN that appears glutamatergic (arrowhead, DVGluT in red/white, (G’), (G’’) close up). (H) R19C05 > UAS-myr::GFP (green) showing three cells among which only one is PER-positive (5th-LaN, white arrow) and two are PER-negatives (nc-LaNs, magenta arrow) (H’), PER in blue/white). All three cells are cholinergic (H’’), ChAT in red/white). Four additional cells weakly covered by the Gal4 line and expressing PER correspond to the Pdf-LaNs (H’), arrowheads). Scale bars: A, B, E, F, G, H: 10 μm. C: 5 μm. (I) Electron microscopy view of a bouton rich in dense-core vesicles and clear vesicles in the Pdf-LaN numbered 2 of the left hemisphere.
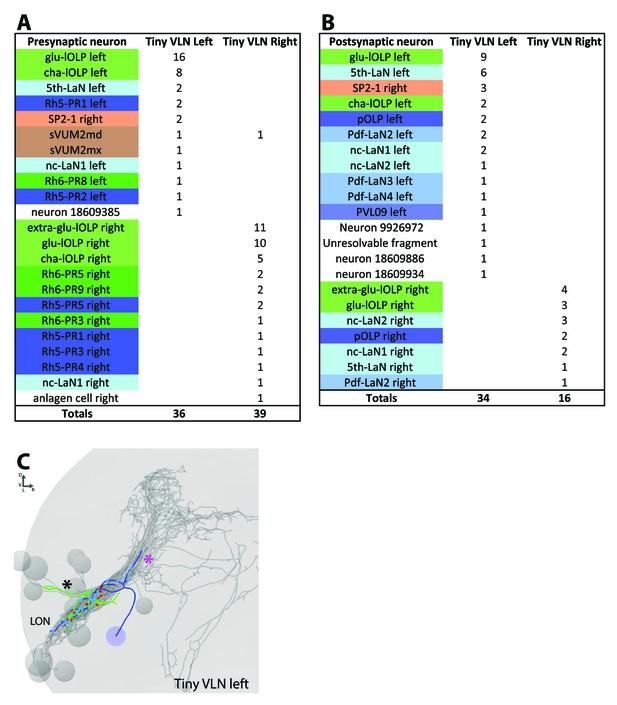
Connections and anatomy of the small third-order neuron Tiny VLN.
(A) The main inputs of the Tiny VLN in both hemispheres come from the lOLPs in particular the glu-lOLP (and extra-glu-lOLP in the right hemisphere). (B) Tiny VLNs have few outputs, especially in the right hemisphere, but it seems that their main targets are back to the glu-lOLPs. (C) 3D reconstruction of the left Tiny VLN with a medial-situated cell body, sparse connections in the LON and neurites entering different primary tracts (BLAd tract: black star; central optic tract: magenta star). Posterior view, dendrites in blue, axon in green, presynaptic sites in red, postsynaptic sites in cyan, other LON neurons in gray.
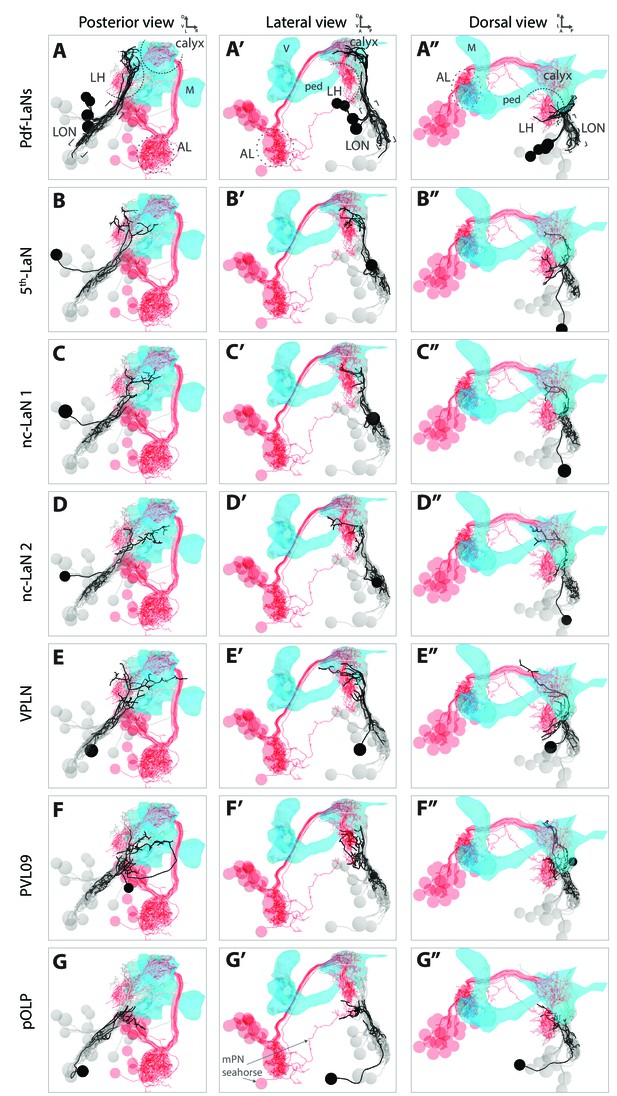
: 3D reconstructions of each VPN relative to the lateral horn and to the mushroom body.
3D reconstructions from different views (posterior, lateral, dorsal) relative to the lateral horn (LH) shown by displaying olfactory projection interneurons (in red, AL: antennal lobe), and to the mushroom body (MB, blue mesh, V: vertical lobe, M: medial lobe, ped: peduncle). VPN of interest in black, other LON neurons in gray. (A–A’’) The Pdf-LaNs project above the LH. The 5th-LaN (B–B’’), the nc-LaN 1 (C–C’’), the nc-LaN 2 (D–D’’) and the third-order neuron VPLN (E–E’’) project to the LH and MB calyx. (F–F’’) PVL09 projects to the LH and, after a long looping branch below the MB, project to the same region as pOLP. (G–G’’) pOLP projects like the multiglomerular olfactory projection neurons (mPN) Seahorse in the lower LH (two Pdf-LaNs were remove for these panels to unmask the lower LH). Also see Figure 1—video 2 for 3D animation movies.
Animation movie of the 3D reconstruction of the larval visual system with all 60 LON neurons. Color code as in Figure 1B.
https://doi.org/10.7554/eLife.28387.0063D animations of the rotating brain with all interneurons of the LON in the left hemisphere, first together and then individually, relative to the lateral horn shown by displaying olfactory projection interneurons in red and to the mushroom body as a blue mesh.
https://doi.org/10.7554/eLife.28387.007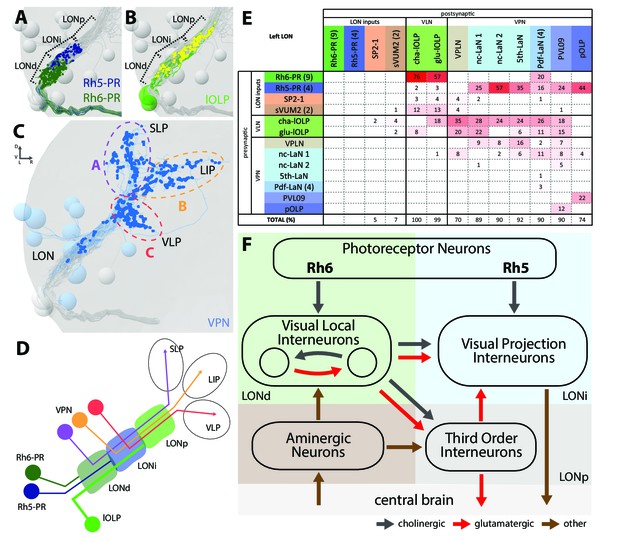
The structural organization of the larval optic neuropil.
(A-D) 3D representations of the presynaptic sites of LON neurons in the left lobe, posterior view. (A) Rh6-PRs presynaptic terminals (dark green) define a distal LON layer (LONd) while Rh5-PRs presynaptic connections (dark blue) define an intermediate LON layer (LONi). A third layer of the LON, more proximal (LONp) is devoid of PR terminals. Other LON neurons in gray. (B) All LON layers, including the LONp, contain presynaptic sites from the lOLPs (skeletons in green, synapses in yellow). VPNs, Tiny VLN and Bolwig nerve in gray. (C) VPNs (blue) make synaptic connections in three main regions outside the LON. VPNs projections define three domains: dorsal domain (A, violet) defined by projections in the superior lateral protocerebrum (SLP), lateral domain (B, orange) in the lateral inferior protocerebrum (LIP), ventral domain (C, red) in the ventral lateral protocerebrum (VLP). VLNs and Bolwig nerve in gray. (D) Schematic of the LON three layers: LONd innervated by Rh6-PRs (dark green), LONi innervated by Rh5-PRs (dark blue) and LONp innervated by lOLPs (green); and of the three domains outside the LON were different VPNs subtypes project to (violet, orange and red empty circles). lOLPs also make presynaptic connections in the LONd and LONi (thick line). (E) Connectivity table of the left LON with the percentage of postsynaptic sites of a neuron in a column from a neuron in a row. Neurons of same type are grouped, in brackets number of neurons in the group. Same colors as in Figure 1B. Only connections with at least two synapses found in both hemispheres were used. (F) Simplified diagram of the larval visual system. PRs inputs are cholinergic and define two pathways. Rh5-PRs target VPNs (blue area, LONi) while Rh6-PRs target the two main larval VLNs (green area, LONd). Between these two VLNs, one is cholinergic while the other one is glutamatergic and they both inputs onto VPNs. These VLNs also integrate aminergic modulatory inputs (brown area) that potentially bring information from the central brain. In the LONp, the two third-order neurons receive from the lOLPs. The third-order neuron VPLN, which is glutamatergic, additionally receives from the serotonergic neuron, connects onto the VPNs and also projects towards the central brain (gray area) like other VPNs. Black arrows: cholinergic connections. Red arrows: glutamatergic connections. Brown arrows: connections with other neurotransmitters.
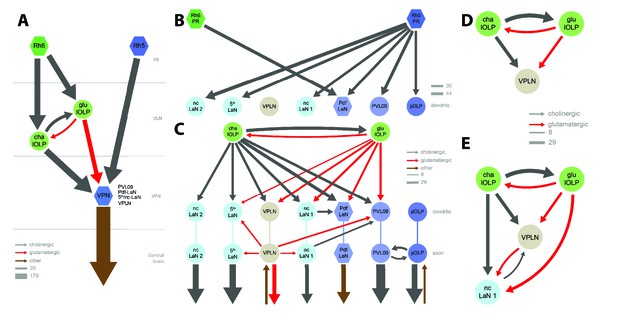
Connectivity and circuit motifs of the larval visual system.
Left hemisphere, hexagons represent group of cells, circles represent single cell, arrow thickness weighted by the square root of the number of synapses, arrow thickness scale shows minimum and median. Black arrows: cholinergic connections. Red arrows: glutamatergic connections. Brown arrows: connections with other neurotransmitters. VPNs in shades of blue for Rh5-PRs targets plus the VPLN in light brown. (A) Wiring diagram of both lOLPs (light green circles). The two lOLPs receives from Rh6-PRs (dark green) and are reciprocally connected. They also share the same type of targets: VPNs (blue) including PVL09, all LaNs and the VPLN, that are direct targets of Rh5-PRs (dark blue) (except the VPLN) and are outputs of the LON towards the central brain. (B) The four Pdf-LaNs are the only VPNs that receive from both Rh6-PRs and Rh5-PRs. The VPLN is a third-order neuron that does not receive any inputs from PRs. All other VPNs receive visual inputs uniquely from Rh5-PRs. All inputs from PRs onto VPNs are situated on the target dendrites. (C) VPNs, except pOLP, are targets of the two lOLPs and these connections are situated on the VPNs dendrites. Additionally, PVL09 receives inputs from both the VPLN and nc-LaN one while the Pdf-LaNs receive only from the nc-LaN 1, and the 5th-LaN receive only from the VPLN. PVL09 and pOLP are reciprocally connected on their axons. All VPNs transfer light information to neurons deeper in the brain. The VPLN and pOLP additionally receive on their axons some inputs from other neuronal circuits. (D) Circuit motif of the VPLN receiving from both lOLPs. E: Circuit motif of the nc-LaN one that is under regulations from cha-lOLP, glu-lOLP and the VPLN. Moreover, nc-LaN one connects back to the VPLN. Similar motifs can be described for other VPNs (Figure 3—figure supplement 1).
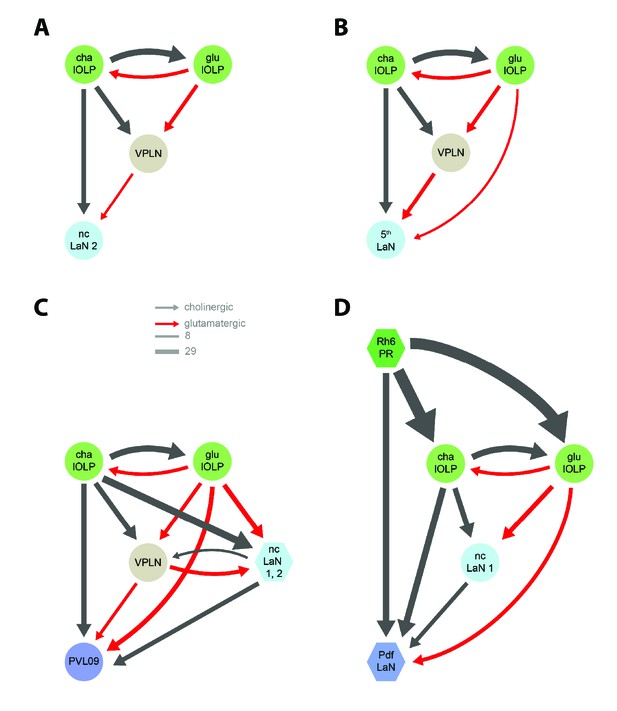
Network motifs of the nc-LaN 2, the 5th-LaN, PVL09 and the Pdf-LaNs.
Black arrows: cholinergic connections. Red arrows: glutamatergic connections. The nc-LaN 2 (A) and the 5th-LaN (B) are controlled in similar fashion by the two lOLPs and the third-order neuron VPLN. (C) PVL09 activity is shaped by the similar motifs as the nc-LaN 1 and the 5th-LaN with connections from both the lOLPs and the VPLN, and is additionally controlled by a second level of coherent feedforward loops (FFL) from the two nc-LaNs. (D) Unlike other VPNs, the Pdf-LaNs receive direct inputs from Rh6-PRs. The Pdf-LaNs are downstream of two interlocked coherent FFLs from the Rh6-PRs via cha-lOLP and nc-LaN 1. Left hemisphere, hexagons represent group of cells, circles represent single cell, arrow thickness weighted by the square root of the number of synapses, arrow thickness scale shows minimum and median.
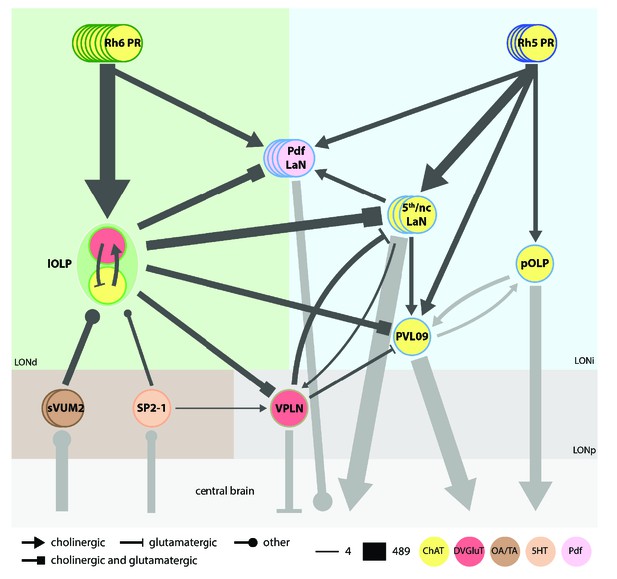
Model of the complete larval visual neural network.
Cell circle outline color defines the neuron identity. Cell filled color represents the neurotransmitter/neuropeptide expression: yellow for cholinergic cells, red for glutamatergic cells, brown for octopaminergic/tyraminergic cells, light orange for serotonergic cell, pink for Pdf neuropeptide. Each PRs subtypes have specific targets. Rh6-PRs (dark green) mainly contact the two lOLPs while Rh5-PRs (dark blue) contact VPNs. Only the Pdf-LaNs receive inputs from both PRs and therefore could be placed at the limit between both Rh6-PRs and Rh5-PRs pathways (green and blue areas, also corresponding to LONdistal (LONd) and LONintermediate (LONi) layers). All PRs neurons are cholinergic (Yasuyama et al., 1995; Keene et al., 2011). sVUM2 neurons (brown) are octopaminergic/tyraminergic. SP2-1 (orange) is serotonergic. One lOLP (light green) is cholinergic while the other one is glutamatergic. VPNs (shades of blue): pOLP and PVL09 are cholinergic; Pdf-LaNs express the Pdf neuropeptide and may co-express glycine (Frenkel et al., 2017); both nc-LaNs and the 5th-LaN are cholinergic. The third-order neuron VPLN (light brown) is glutamatergic and receives its inputs in the LONproximal layer (LONp, stronger gray area). For simplicity, the 5th-LaN and the two nc-LaNs are grouped together. Both lOLPs form a reciprocally connected pair that modulates almost all other VPNs. The VPLN also modulates the 5th/nc-LaNs group as well as PVL09. nc-LaN1 additionally connects onto the Pdf-LaNs, and both nc-LaNs connect onto the VPLN. pOLP is the only VPN that is not modulated by other visual interneurons, except for its strong reciprocal connections with PVL09 at their axonal level. The three aminergic neurons (brown area) target mainly the two lOLPs and SP2-1 further connect onto the VPLN. Black arrows represent connections within LON neurons while gray arrows represent connections beyond. Additional external inputs onto some VPNs are not represented here. Arrow thickness weighted by the square root of the number of synapses, arrow thickness scale shows minimum and maximum.
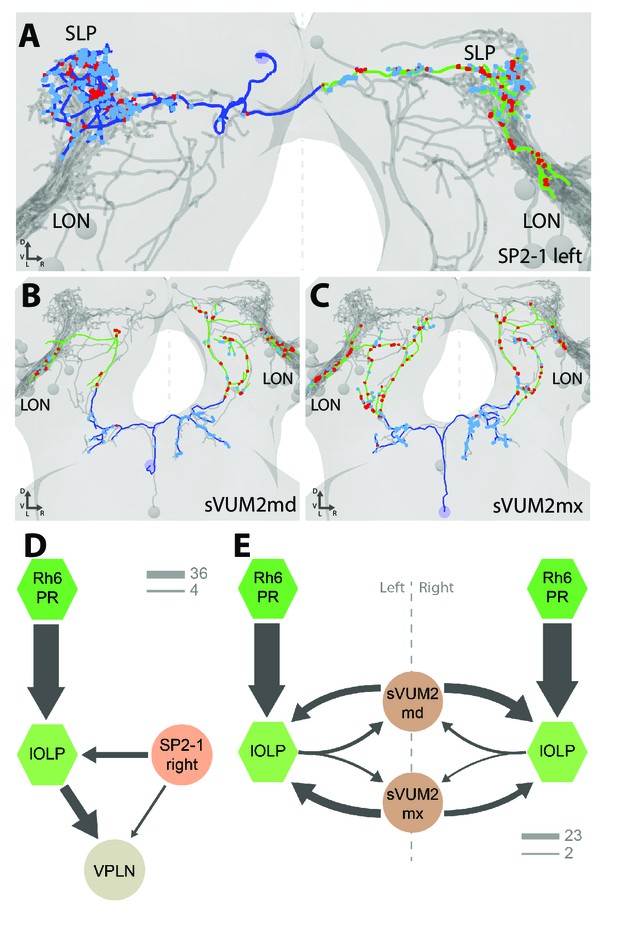
Aminergic modulatory inputs of the larval visual system.
(A-C) 3D reconstructions from ssTEM dataset, posterior view, dendrites in blue, axons in green, presynaptic sites in red, postsynaptic sites in cyan, other LON neurons in gray, dashed line represent brain midline. (A) The SP2-1 neuron from the left hemisphere innervates the ipsilateral SLP and the contralateral LON. sVUM2md (B) and sVUM2mx (C) neurons are located along the midline in the SEZ with their neurit splitting and innervating both hemispheres in a symmetric fashion. Their bilaterally symmetrical branches receive synaptic input in the SEZ and extend their axon towards the protocerebrum prior to turning laterally and entering the LON. Branches within the protocerebrum and LON contain presynaptic and postsynaptic sites. (D) Connectivity graph showing the SP2-1 neuron (orange) of the right hemisphere connecting with the lOLPs (light green) and the VPLN (light brown) of the left hemisphere. Connections between the lOLPs and the VPLN are also displayed, as well as lOLPs inputs from Rh6-PRs (dark green). (E) Connectivity graph of sVUM2mx and sVUM2md (brown) showing that their only partners are the lOLPs (light green) but in both hemispheres. (D–E) Hexagons represent group of cells, circles represent single cell, arrow thickness weighted by the square root of the number of synapses, arrow thickness scales shows minimum and median.
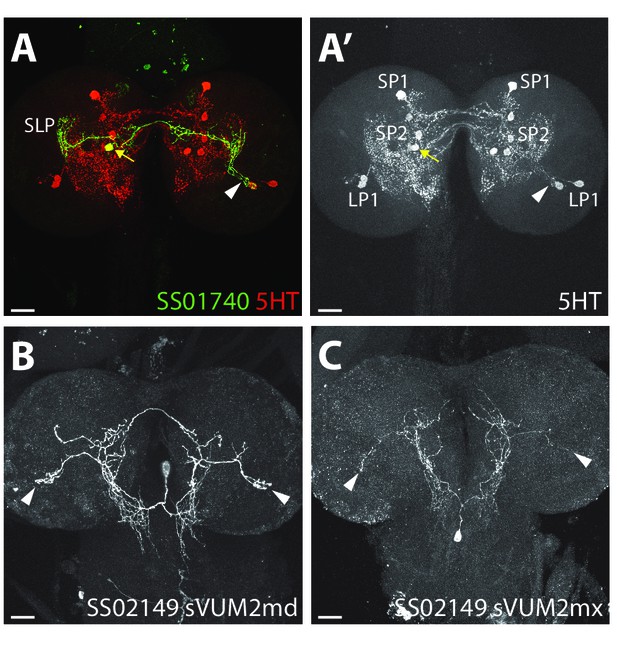
Confocal microscopy of the three aminergic neurons of the LON.
(A) Confocal z-projection of SS01740 > UAS-myr::GFP (green) showing a stochastic single clone of SP2-1 neuron (yellow arrow) innervating the contralateral LON (arrowhead, serotonin (5-HT) in red). (A’) anti-5-HT channel shows the three 5-HT clusters from the lower and superior protocerebrum (LP and SP) and innervation in the LON (arrowhead). (B–C) Confocal z-projection of SS02149 > UAS-myr::GFP (white) showing stochastic single clone expression of sVUM2md (B) and sVUM2mx (C) innervating both LON (arrowheads). Scale bars: 20 μm.
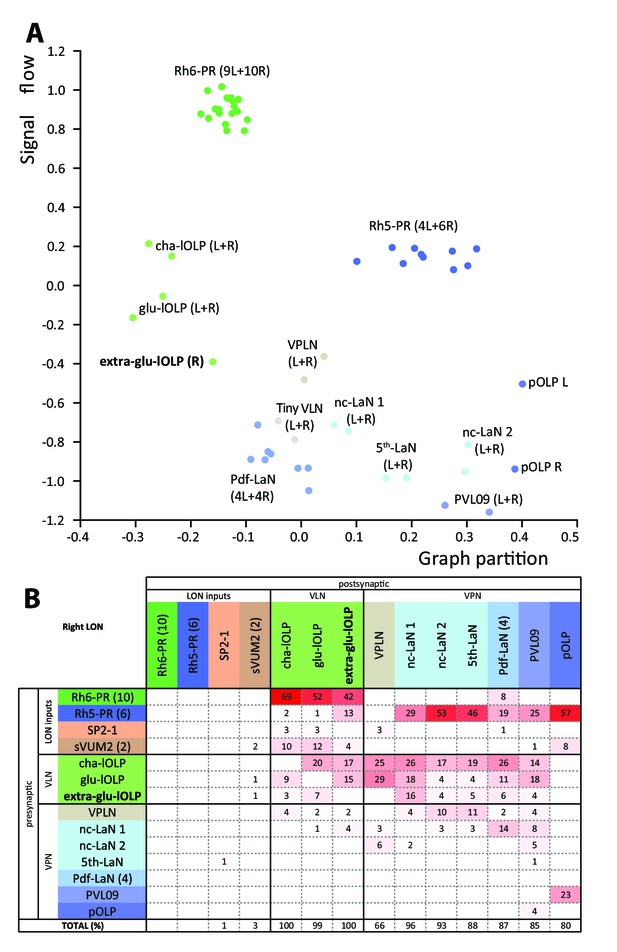
Larval optic neuropil architecture is maintained despite a variable number of neurons.
Color code as in Figure 1B. (A) We compared the structure of the left (L) and right (R) LON circuits with spectral graph analysis. For the left and right connectivity matrices, we plotted the components of the first nonzero eigenvector of the graph Laplacian associated with each neuron (x axis) as a function of the signal flow metric (y axis), measures that reflect the topological role of neurons in the circuit (Varshney et al., 2011). We excluded the neuromodulatory neurons SP2-1, which are weakly connected, and the sVUM2md and sVUM2mx, which project bilaterally. Visual information flows from PRs at the top towards VPNs at the bottom. In this representation, bilaterally homologous neurons share a similar position revealing that both sides have similar network topology (Schneider-Mizell et al., 2016). Note how the extra-glu-lOLP of the right hemisphere (bold) positioned closely with the other lOLPs. (B) Connectivity table of the right LON with the percentage of postsynaptic sites of a neuron in a column from a neuron in a row. Only connections with at least two synapses found in both hemispheres were used (except for the extra-glu-lOLP).
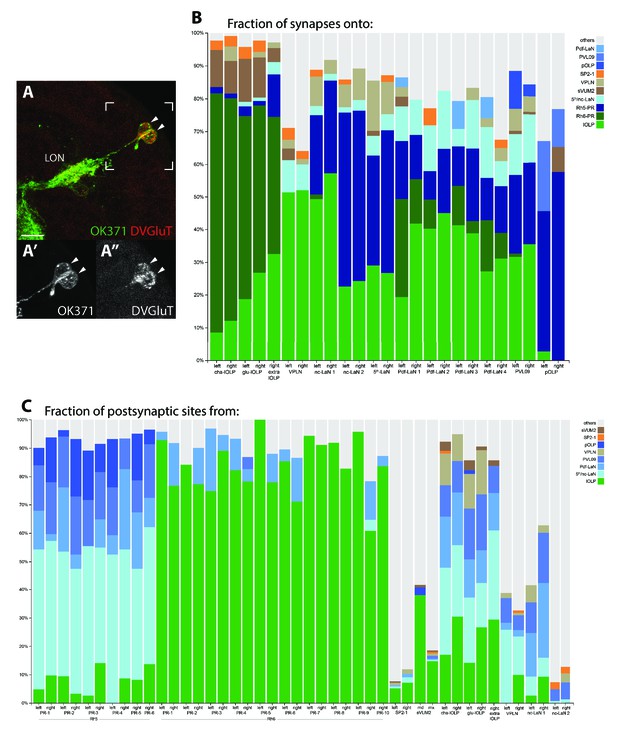
Stability of the connections of LON neurons despite the variability in neuron number.
(A) Confocal z-projection of OK371-Gal4 > UAS-myr::GFP (green) stained with anti-DVGluT (red) with insets onto two cells bodies in the OLP region and projecting into the LON and which are both DVGluT positive (arrow heads, close up (A’), (A’’)). Dorsal view, scale bar: 10 μm. (B) Percentage of synapses onto all visual interneurons of both hemispheres (two bars per neuron except for the extra-lOLP of the right hemisphere). lOLPs main inputs are from the Rh6-PRs. The VPLN does not receives from either PRs but its main inputs come from the lOLPs. All other VPNs receive from Rh5-PRs and only the Pdf-LaNs receives from both PRs-subtypes. Except for pOLP, the lOLPs connections represent an important fraction of each neuron inputs. The VPLN inputs are most significant on the two nc-LaNs and the 5th-LaN. (C) Percentage of synapses from all PRs, aminergic modulatory neurons and visual interneurons with significant roles in local processing (lOLPs, VPLN and nc-LaNs). Additional PRs from the right hemisphere have clear connectivity profiles of either Rh5 or Rh6 subtypes. Strong involvement of the aminergic modulatory neurons in other neural circuits is visible.
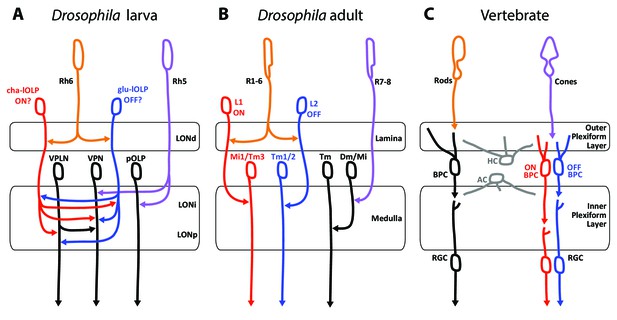
Comparison of the structural organization and putative functions of the Drosophila larval visual circuit with the Drosophila adult compound eye and the vertebrate visual circuit.
(A) Larval visual circuit as described in this paper. Two main groups of VPNs receive input from Rh5-PRs (purple): one corresponds to the pOLP that only receives inputs from Rh5-PRs, whereas the second group (VPN) also receives inputs from cha and glu -lOLPs (red and blue) that are targets of Rh6-PRs (orange). The third-order neuron VPLN does not receive from either larval PRs but is downstream of the two lOLPs and connect onto the VPN group in top of its projection beyond the LON. We propose that cha and glu -lOLPs control light intensity increment and decrement (ON/OFF) detection respectively and transfer these information to the VPLN and the VPN group. Moreover, the VPLN may potentially keep the ON/OFF responses brief. (B) Model of a single unit of the fly compound eye where R1-6 PRs (orange) are well known to be involved in contrast and motion detection, whereas R7-8 PRs (purple) are involved in color sensing (Sanes and Zipursky, 2010; Clark and Demb, 2016, for reviews). In the lamina, R1-6 PRs make connections to the glutamatergic L1 neuron controlling the ON pathway (red) and to the cholinergic L2 neuron controlling the OFF pathway (blue). In the deeper medulla, L1 and L2 reach their targets (Mi1, Tm1/2), whereas R7-8 PRs connect to medullar neurons (Dm/Mi). (C) Model of the vertebrate visual circuit (Sanes and Zipursky, 2010; Clark and Demb, 2016), for reviews). Cones (purple), which are also the color sensors of the retina, connect to bipolar retinal cells (BPC) which constitute the ON or OFF pathways depending on the glutamate receptor they express (ON BPC and pathway in red, OFF BPC and pathway in blue). Rods (orange) also connect to BPC and control vision in dim light conditions. LONd, LONi and LONp: distal, intermediate and proximal larval optic neuropil. Mi: medulla intrinsic neurons; Tm: transmedulla neurons; Dm: dorsal medulla neurons. RGC: retina ganglion cells; HC: horizontal cells; AC: amacrine cells.
Additional files
-
Transparent reporting form
- https://doi.org/10.7554/eLife.28387.019





