RETRACTED: Targeting posttranslational modifications of RIOK1 inhibits the progression of colorectal and gastric cancers
Figures
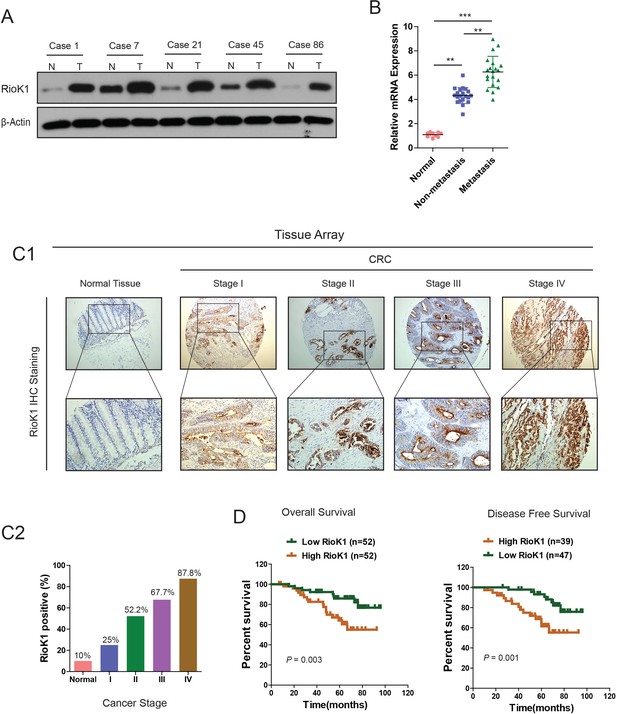
RIOK1 is significantly upregulated in CRC and associated with an aggressive and poor survival.
(A) RIOK1 expression in five paired human CRC biopsies and matched normal mucosa analyzed by Western-blot. (B) Comparison of RIOK1 mRNA expression level in human CRC tissues (with and without metastasis) and matched normal mucosa. RIOK1 mRNA expression was quantified by qPCR and normalized to the matched adjacent normal tissues. (C1) IHC analysis of RIOK1 on a tissue micro array of CRC patients (n = 110) and healthy adjacent tissue (n = 10) using the Allred score. (C2) The IHC signals were scored as 0, 1, 2, and 3; a score ≧1 + indicated positive detection. (D) Kaplan-Meier curves for overall survival and disease free survival of 104 and 86 CRC patients stratified by RIOK1 expression respectively.
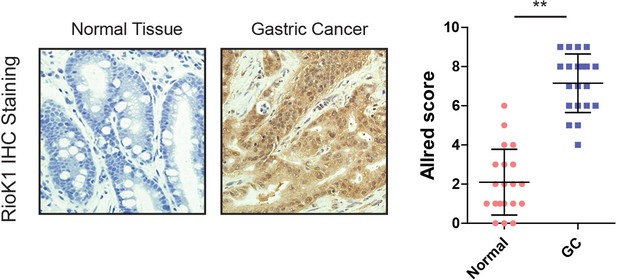
RIOK1 expression in GC patients.
Immunohistochemical analysis and statistic calculation of RIOK1 on a group of patients with GC (n = 20) and healthy adjacent tissue (n = 20).
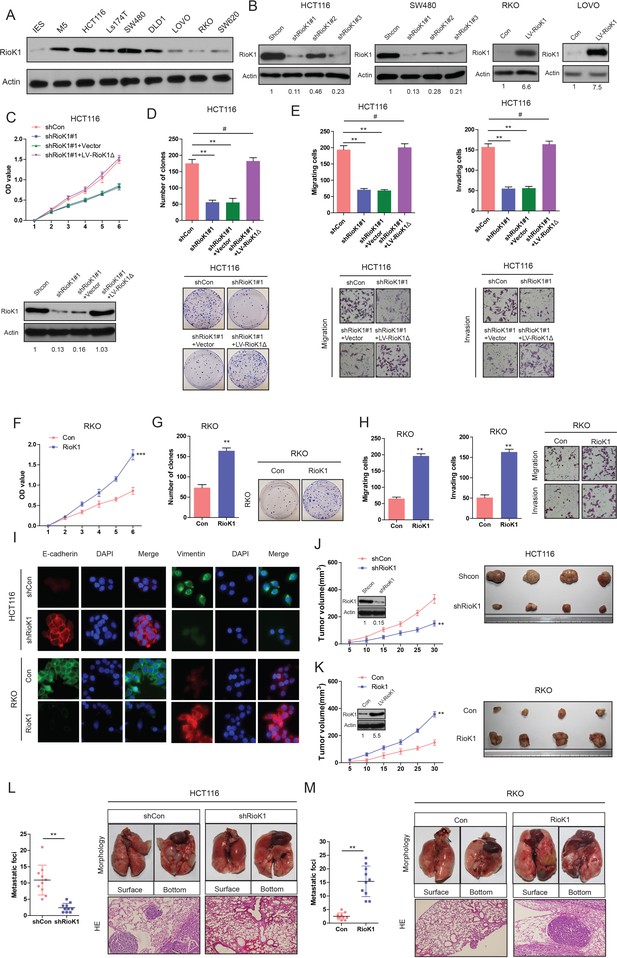
RIOK1 promotes growth and metastasis of CRC cells in vitro and in vivo.
(A) The expression levels of RIOK1 in 7 CRC cell lines and normal intestinal epithelial cells (IECs) were analyzed by Western blot. (B) Western blot for RIOK1 in HCT116 and SW480 cells infected with shRIOK1, or control shRNA lentiviral vector, as well as in RKO and LOVO infected with the RIOK1-expressing or empty control vector (n = 4).And the intensity of the western blot bands was quantified using NIH ImageJ software. (C) Western blot for RIOK1 in the indicated HCT116 cells transfected with shRIOK1#1 or the shRNA-resistant expression construct, RIOK1Δ. RIOK1 expression was recovered in the HCT116-shRIOK1 cells transfected with RIOK1Δ (lower-panel).And the intensity of the western blot bands was quantified using NIH ImageJ software. RIOK1Δ almost restored the proliferative ability of the cells (upper-panel). (D) RIOK1 knockdown inhibited proliferation of HCT116 cells determined by CCK-8 and colony formation assays. (E) RIOK1 knockdown inhibited migration and invasion of HCT116 cells determined by transwell assays. (F) and (G) Ectopic expression of RIOK1 promoted proliferation of RKO cells determined by CCK-8 and colony formation assays. (H) Ectopic expression of RIOK1 promoted migration and invasion of RKO cells determined by transwell assays. (I) Representative immunofluorescence images demonstrated that RIOK1 level has an effect on the expression of EMT proteins in CRC cells. (J) Growth curve of subcutaneous injection of HCT116/Scramble and HCT116/shRIOK1 in NOD/SCID mice (n = 6 per group, left panel). NOD/SCID mice were injected subcutaneously into opposite flanks with 1.5 × 106 control cells and RIOK1-knocked down HCT116 cells. The mice were sacrificed and the tumors were then removed, weighed and compared (right panel). And knockdown of RIOK1 in a xenograft tumor model was analyzed with western blotting, and the intensity of the western blot bands was quantified using NIH ImageJ software. (K) Growth curve of subcutaneous injection of RKO/Vector and RKO/RIOK1 in NOD/SCID mice (n = 6 per group, left panel). The tumors were then removed, weighed and compared (right panel). Over-expression of RIOK1 in a xenograft tumor model was analyzed with western blotting, and the intensity of the western blot bands was quantified using NIH ImageJ software. Effects of RIOK1 Knockdown (L) or overexpression (M) on lung metastasis of indicated cells in NOD/SCID mice (n = 10 per group): the number of metastatic nodules in the lung (left-panel); representative morphological observation of lung metastases (right-upper panel); and histopathological observation of lung sections (right-lower panel). The results are presented as are means ±SD (n = 3 for each panel). Statistical significance was concluded at *p<0.05, **p<0.01, ***p<0.001; # represents no statistical significance.
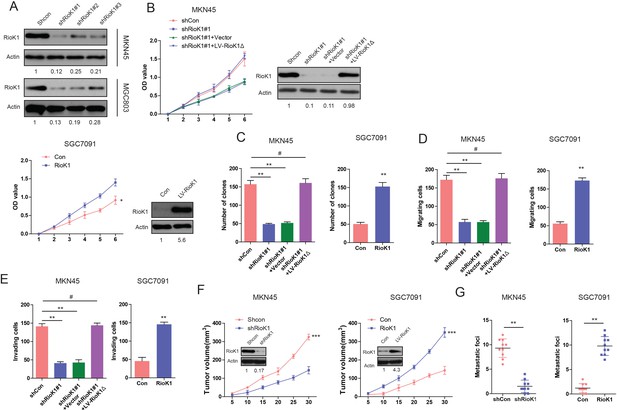
RIOK1 promotes the proliferation, invasion and metastasis of GC in vitro and in vivo.
(A) Western blot for RIOK1 in MKN45 cells infected with shRIOK1, or control shRNA lentiviral vector, as well as in SGC7091 infected with the RIOK1-expressing or empty control vector (n = 4).And the intensity of the western blot bands was quantified using NIH ImageJ software. (B) Western blot for RIOK1 in the indicated MKN45 cells transfected with shRIOK1#1 or the shRNA-resistant expression construct, RIOK1Δ. RIOK1 expression was recovered in the MKN45-shRIOK1 cells transfected with RIOK1Δ.And the intensity of the western blot bands was quantified using NIH ImageJ software. RIOK1Δ almost restored the proliferative ability of the cells. The effect of RIOK1 on proliferation of MKN45 and SGC7091 cells was determined by CCK-8 and (C) colony formation assays. (D and E) The effect of RIOK1 on migration and invasion of MKN45 cells was determined by transwell assays. (F) Growth curve of subcutaneous injection of indicated plasmids in NOD/SCID mice (n = 6 per group). The mice were sacrificed and the tumors were then removed, weighed and compared. And knockdown or overexpression of RIOK1 in a xenograft tumor model was analyzed with western blotting, and the intensity of the western blot bands was quantified using NIH ImageJ software. (G) Effects of RIOK1 knockdown or over-expression on tumor metastasis of indicated cells in NOD/SCID mice (n = 10 per group): the number of metastatic nodules in the lung. The results are presented as are means ±SD (n = 3 for each panel). Statistical significance was concluded at *p<0.05, **p<0.01, ***p<0.001; # represents no statistical significance.
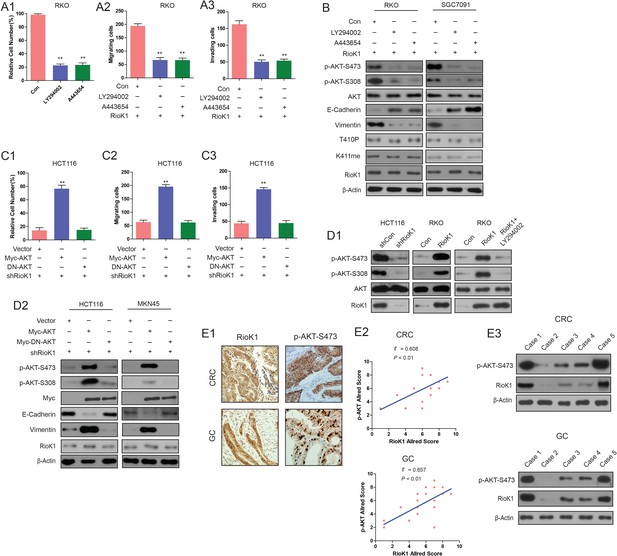
PI3K/AKT pathway is required for RIOK1-mediated CRC and GC cell proliferation and migration.
(A) CRC and GC cells were transfected with RIOK1 plasmid, and treated with LY29004 (30 μM) or A443654 (1 μM). Then cell proliferation (A1), migration(A2) and invasion (A3) were determined. (B) The whole-cell lysates in (A) were analyzed by Western blot with indicated antibodies. (C) The RIOK1-knockdown-HCT116 and MKN45 cells were transfected with indicated plasmids. The cell proliferation (C1), migration(C2) and invasiveness (C3) was tested. (D1 and D2) The cells in (C) were harvested at 48 hr for western blot analysis with indicated antibodies. The data represent the mean ±SEM from three independent experiments. (E1) Immunohistochemistry staining, (E2) correlation analysis and (E3) WB analysis for RIOK1 and phospho-AKT-Ser473 in human CRC and GC tissues(n = 20).
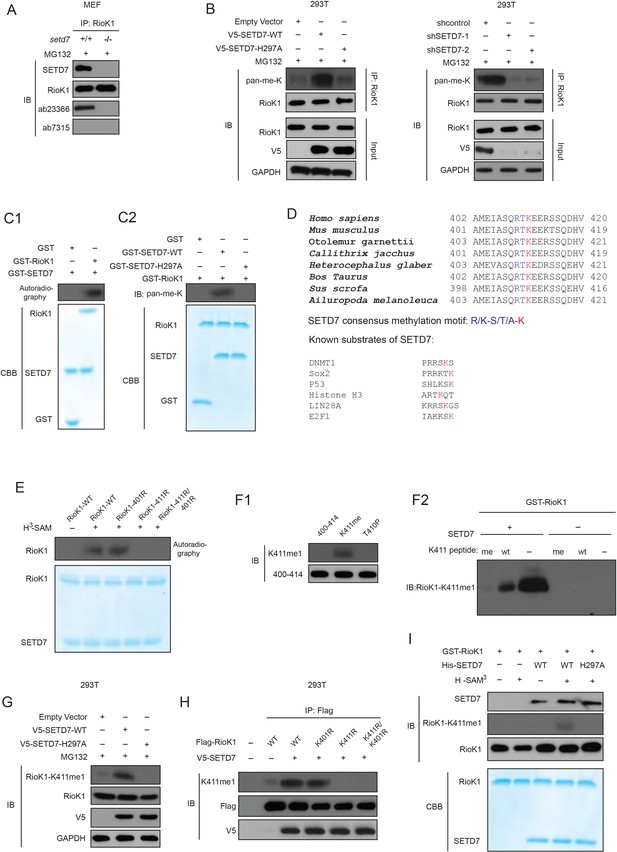
SETD7 monomethylates RIOK1 on lysine 411 in vitro and in vivo.
(A) Anti-RIOK1 immunoprecipitates from high-density Setd7+/+ and Setd7−/− MEFs were immunoblotted for RIOK1, SETD7, methyl-lysine (mono-, dimethylated) (ab23366), and dimethyl-lysine (ab7315) antibodies, respectively. (B) SETD7 methylates RIOK1 in vivo. Stably expressed control, SETD7-WT, or SETD7-H297A HEK293T cells were generated. The cell lysates were then precipitated with an anti–RIOK1 antibody and probed with an anti–pan-me-K antibody to detect methylation of RIOK1 (left panel). Stably expressed shcontrol or sh-SETD7 HEK293T cells were generated. The cell lysates were then precipitated with an anti–RIOK1 antibody and probed with an anti–pan-me-K antibody to detect methylation of RIOK1 (right panel). (C1) An in vitro methylation assay was performed with 3H-SAM, recombinant SETD7, and RIOK1. Autoradiography and Coomassie brilliant blue R-250 staining (CBB) were used to show methylation and protein levels, respectively. (C2) In vitro methylation analysis using indicated proteins. An anti–pan-methyl-lysine antibody (pan-me-K) was used to detect the methylation of RIOK1. CBB was used to show the protein levels. (D) Alignment of the consensus amino acid residues adjacent to lysine targeted by SETD7, and identification of a putative SETD7 methylation site in RIOK1 (upper panel). The consensus SETD7 recognition sequence (middle panel). The lysines targeted for methylation by SET7 in known substrates are shown (lower panel). In each case, the methylated lysine is shown in red. (E) In vitro methylation of various purified RIOK1 mutants by SETD7. (F1) An anti-RIOK1-K411me1-specific antibody is characterized. RIOK1 peptide substrates: 400–414 SKAMEIASQRTKEER(Lane 1), K411me SKAMEIASQRTKmeEER(Lane 2), T410p SKAMEIASQRTpKEER(Lane 3), p: phosphorylation; me: methylation. The above peptides were immunoblotted with indicated antibodies. (F2) GST-RIOK1 (1 μg) was methylated in vitro in the presence or absence of recombinant SETD7 (0.5 μg). The reaction was probed with the anti-RIOK1-K411me1 antibody, in the presence of 10 μg/ml competing wild-type (wt) or mono-methylated K411 peptide (me). (G) The effect of ectopically expressed SETD7 and SETD7-H297A on K411me1 of endogenous RIOK1 in HEK293T cells. (H) Methylation analysis of Flag-RIOK1 and mutants by SETD7 in transfected HEK293T cells. (I) In vitro methylation was performed with GST-RIOK1, S-adenosylmethionine (SAM), recombinant His-SETD7, and His-SET7-H297A. Immunoblotting and CBB were used to show methylation and protein levels, respectively.
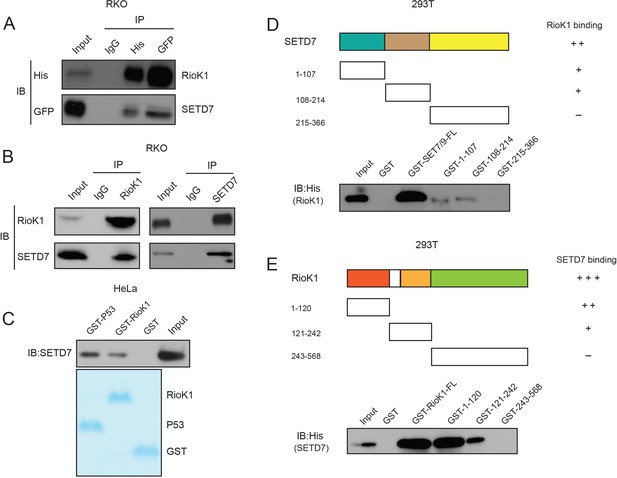
SET7/9 directly interacts with RIOK1 in vitro and in vivo.
(A) Whole-cell lysates of RKO cells transfected with GFP-SET7/9 and His-RIOK1 were precipitated with an anti-GFP or anti-His antibody, and the interactive components were analyzed by Western blot. (B) RKO cells were extracted and immunoprecipitated with an anti–RIOK1 (left) or anti-SET7/9 (right) antibody. IP with rabbit IgG was used as the negative control. Western blot analysis was performed with the antibodies indicated. (C) GST (negative control), GST-RIOK1, or GST-p53 (positive control) was incubated with whole-cell lysates of HeLa cells, and Western blot analysis was performed to detect the interaction between SET7/9 and RIOK1. (D) GST-SET7/9 FL or fragments were incubated with His-RIOK1, and Western blotting was performed to detect the interaction with an anti-His antibody. (E) The reciprocal pull-down assay between GST-RIOK1 FL or fragments and His-SET7/9.
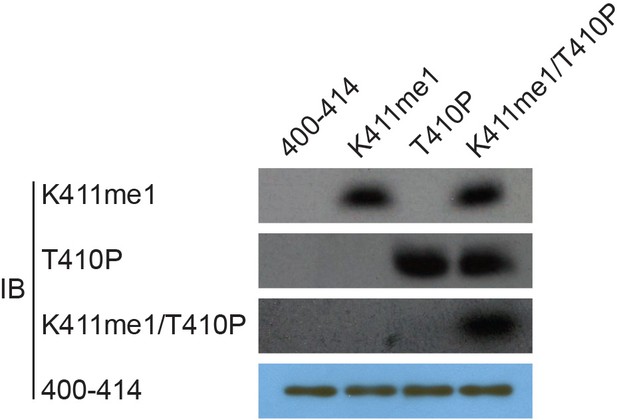
Validation of the antibodies’ specificity.
To easily find whether methylation of RIOK1 occurred in cells, we prepared a modification-specific antibody, anti-RIOK1-K411me1, which specifically recognized monomethylated RIOK1-K411 but not the unmethylated peptide. This antibody also recognizes a pT410.K411me1 doubly modified peptide as efficiently as the K411me1 peptide. Conversely, the anti-pT410 antibody recognizes a pT410.K411me1 peptide as efficiently as the pT410 peptide. These specific antibodies are characterized. RIOK1 peptide substrates: 400–414 SKAMEIASQRTKEER(Lane 1), K411me SKAMEIASQRTKmeEER(Lane 2), T410p SKAMEIASQRTpKEER(Lane 3), K411me/T410p SKAMEIASQRTpKmeEER(Lane 4), p: phosphorylation; me: methylation.
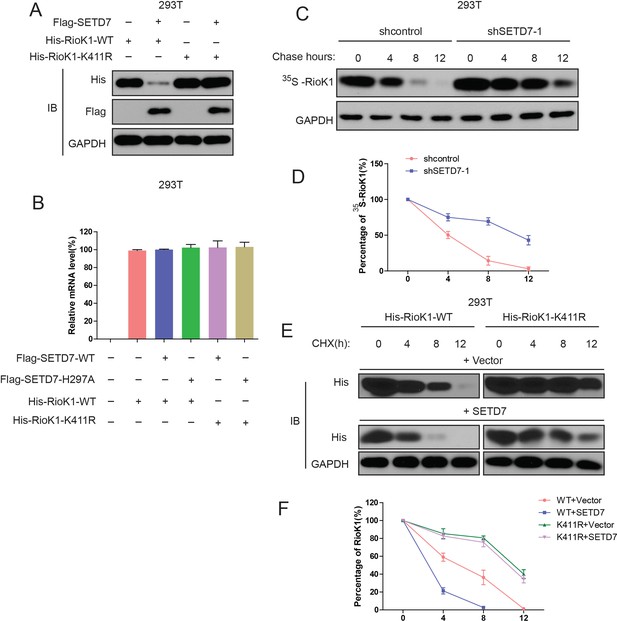
SET7/9 significantly reduces the stability of RIOK1.
(A) His-RIOK1-WT or His-RIOK1-K411R was cotransfected into HEK293T cells for 48 hr with or without Flag-SET7/9. Cell lysates were examined by western blot with indicated antibodies. (B) His-RIOK1-WT was cotransfected with or without Flag-SET7/9 or mutant Flag-SET7/9-H297A into HEK293T cells for 48 hr. Real-time PCR was used to detect the mRNA levels of RIOK1. (C) HEK293T -shcontrol and HEK293T -sh-SET7/9-1 cells were metabolically labeled with [35S]-methionine, as indicated, and followed by chasing with standard medium. Total lysates were examined with anti-RIOK1 antibody. (D) Quantification analysis of endogenous RIOK1 levels in (C). (E) HEK293T cells were transfected with indicated plasmids, and 24 hr later, treated with cycloheximide (CHX), and probed with anti-His antibody. (F) Quantification analysis of the results in (E).
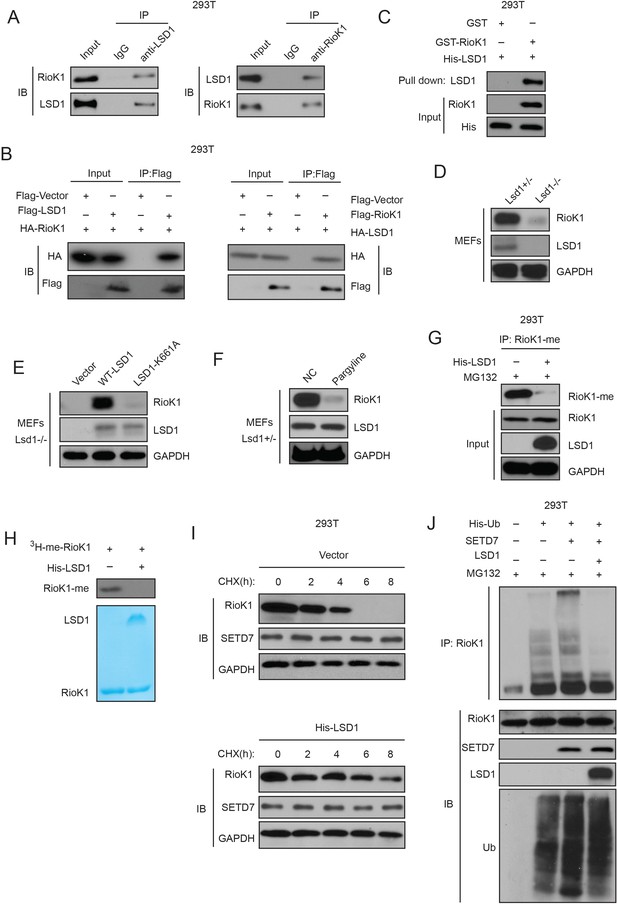
SET7/9-mediated methylation of RIOK1 is reversed by LSD1, which increases RIOK1 stability.
(A) Endogenous RIOK1 co-immunoprecipitated reciprocally with endogenous LSD1 from HEK293T cells and subjected to immunoprecipitation followed by immunoblotting with indicated antibodies. (B) HA-RIOK1 co-immunoprecipitated reciprocally with Flag-LSD1. Total cell lysates were extracted from HEK 293T cells transiently cotransfected with expression constructs as indicated and probed with antibodies as indicated. (C) RIOK1 directly interacted with LSD1. Bacterially produced GST-RIOK1 was used to pull down bacterially produced His-LSD1 in vitro. (D) RIOK1 protein levels in Kdm1a+/− and Kdm1a−/− MEFs were compared. (E) Kdm1a−/− MEFs were reconstituted with either WT or a catalytically inactive Mutant (K661A) of LSD1. RIOK1 protein levels were monitored. (F) Kdm1a+/−MEFs were pretreated with pargyline for 12 hr, RIOK1 protein levels were evaluated. (G) Overexpressing LSD1 with MG132 treatment significantly decreases RIOK1 methylation in HEK293T cells. (H) LSD1 reverses SET7/9-dependent RIOK1 methylation. In vitro demethylation assays using purified His-LSD1 were performed and RIOK1 methylation was detected by autoradiography. (I) HEK293T-LV-SETD7 cells transfected with indicated plasmids were treated with CHX (20 μg /ml), collected at the indicated times, and analyzed by western blot. (J) Overexpressing LSD1 with MG132 treatment from HEK293T cells co-transfected with the indicated plasmids. And cell lysates were subjected to pull-down with Ni2+-NTA beads. RIOK1 ubiquitination was assessed by anti-RIOK1 antibody in the presence of MG132.
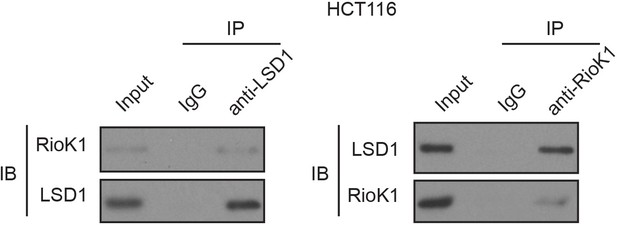
LSD1 interacts with RIOK1 endogenously.
HCT116 cells were extracted and immunoprecipitated with an anti–LSD1 (left) or anti-RIOK1 (right) antibody. IP with rabbit IgG was used as the negative control. Western blot analysis was performed with the antibodies indicated.
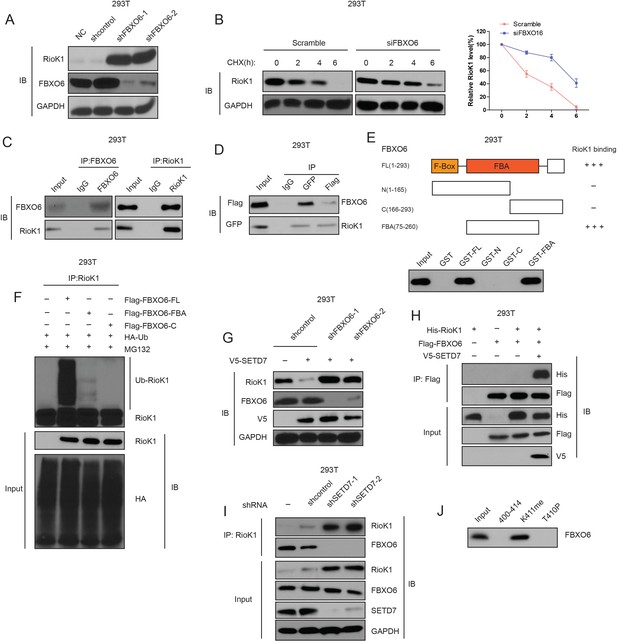
K411 methylation of RIOK1 by SETD7 promotes the interaction between FBXO6 and RIOK1.
(A) The effect of FBXO6 knockdown on the levels of RIOK1 proteins in HEK293T cells. (B) HEK293T cells were transfected with the indicated siRNAs, and after 48 hr, treated with 160 μM CHX, and representative RIOK1 expression result is shown in the left panel. Right panel indicates quantitation of the RIOK1 blots. (C) HEK293T cells were extracted and immunoprecipitated with an anti–FBXO6 (left) or anti-RIOK1 (right) antibody. Western blot analysis was performed with indicated antibodies. (D) Whole-cell lysates of HEK293T cells transfected with GFP-FBXO6 and Flag-RIOK1 were precipitated with an anti-GFP or anti-Flag antibody, and the interactive components were analyzed by Western blot. (E) Binding of several different domains of human FBXO6 to RIOK1. Numbers represent the amino acid residues in human FBXO6. FL, N, C, and FBA represent the full-length, amino terminus, carboxyl terminus, and the FBA domain only of FBXO6, respectively. The extent of the interaction between RIOK1 and FBXO6 domains is indicated by the number of plus signs (+). (F) HEK293T cells were transfected with HA-RIOK1, Myc-tagged FBXO6 FL or mutants with His-ubiquitin for 48 hr. Cells were lysed and blotted with indicated antibodies. (G) Knockdown of FBXO6 blocked V5-SETD7-induced RIOK1 degradation. (H) The interaction between Flag-FBXO6 and HA-RIOK1 was dependent on SETD7. (I) SETD7 knockdown disrupted the interaction between endogenous FBXO6 and RIOK1 in HEK293T cells. (J) The binding of FBXO6 to unmodified, T410p, or K411me RIOK1 aa 400–414 peptides was analyzed using in vitro peptide pull-down assay.
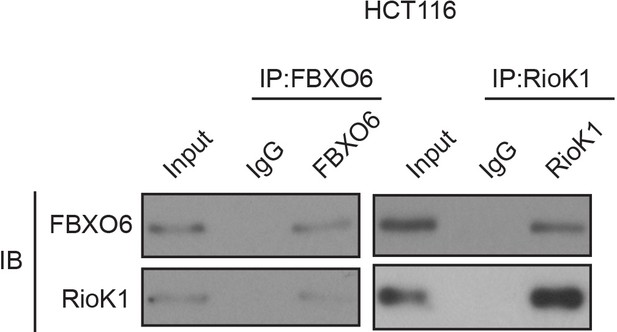
FBXO6 interacts with RIOK1 endogenously.
HCT116 cells were extracted and immunoprecipitated with an anti–FBXO6 (left) or anti-RIOK1 (right) antibody. IP with rabbit IgG was used as the negative control. Western blot analysis was performed with the antibodies indicated.
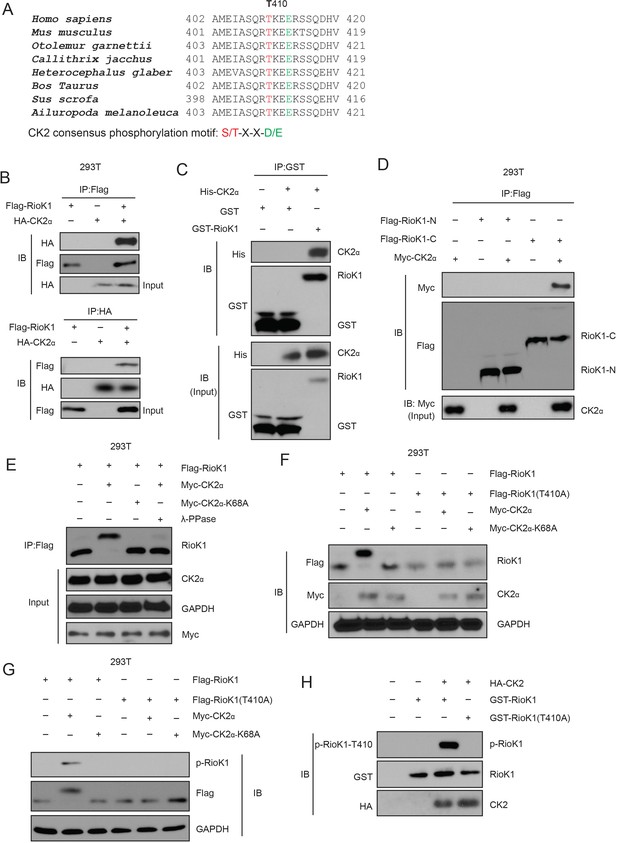
CK2 phosphorylates RIOK1 at T410 both in vitro and in vivo.
(A) RIOK1 consensus phosphorylation sites (shown in red) corresponding to the CK2 consensus motifs S/TXXE/D are presented. These consensus phosphorylation sites are exactly located within the consensus sequences of SETD7. (B) FLAG-RIOK1 was present in the HA-CK2 immunocomplex. Total cell lysates were extracted from 293T cells transiently co-transfected with FLAG-RIOK1 and HA-Vec and HA-CK2α subjected to immunoprecipitation with an anti-HA antibody followed by immunoblotting with indicated antibodies. (C) In vitro glutathione S-transferase (GST)-precipitation assay of no CK2α, or purified His-tagged CK2α combined with GST alone or GST-RIOK1. (D) Immunoprecipitation and immunoblot analysis of lysates of HEK293T cells expressing no plasmid (−) or plasmid encoding the Flag-tagged amino terminus (amino acids 1–242) of RIOK1 (Flag–RIOK1-N) or carboxyl terminus (amino acids 243–568) of RIOK1 (Flag–RIOK1-C), plus Myc-tagged CK2α, probed with anti-Myc and/or anti-Flag. (E) Lysates of HEK293T cells expressing Flag-tagged wild-type RIOK1 and Myc-tagged wild-type CK2α or CK2α-K68A; far right (λ-PPase), was analyzed using Western blot. GAPDH serves as a loading control throughout. (F) Immunoblot analysis of RIOK1 and CK2α in lysates of HEK293T cells expressing Flag-tagged wild-type RIOK1 or RIOK1 (T410A) and Myc-tagged wild-type CK2α or CK2α-K68A. (G) Immunoblot analysis of phosphorylated p-RIOK1-T410 and total RIOK1 and CK2 in lysates of HEK293T cells. (H) In vitro kinase assay of purified recombinant GST-tagged RIOK1 or RIOK1-T410A with HA-tagged CK2.
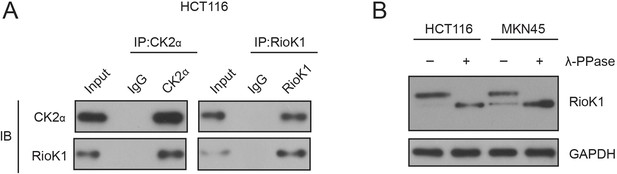
CK2 interacts with RIOK1 endogenously.
(A) HCT116 cells were extracted and immunoprecipitated with an anti–CK2 (left) or anti-RIOK1 (right) antibody. IP with rabbit IgG was used as the negative control. Western blot analysis was performed with the antibodies indicated. (B) Prior to development with anti-RIOK1 antibodies, HCT116 and MKN45 cell lysates were incubated with or without λ-PPase and subsequently probed with RIOK1.
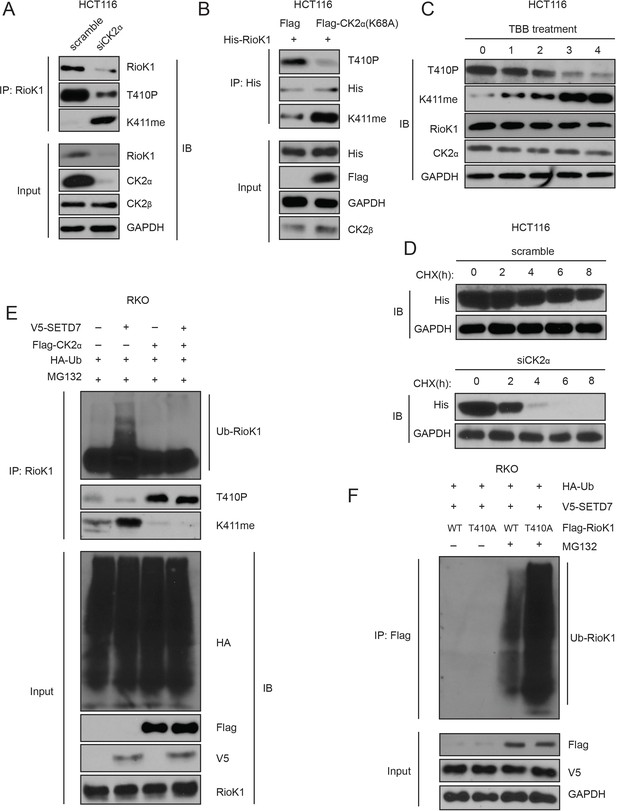
T410 phosphorylation of RIOK1 antagonists SETD7-mediated K411 methylation, which stabilizes RIOK1 in CRC and GC cells.
(A) The effect of knockdown of CK2α in HCT116 cells on the levels of RIOK1 proteins, T410p, and K411me. (B) The effect of the CK2α-K68A mutant on the levels of RIOK1 proteins, T410p, and K411me. (C) The effect of the CK2 kinase inhibitor TBB on the levels of RIOK1 proteins, T410p, and K411me. (D) The effect of CK2 knockdown in HCT116 cells on the RIOK1 protein stability. (E) CK2 inhibits K411me by SETD7 and its consequent RIOK1 ubiquitination in RKO cells. Indicated cells were cotransfected with HA-Ub and treated with MG132 for 12 hr. (F) Wild-type RIOK1 or the RIOK1-K411me1 mutant was overexpressed in cells, along with HA-ubiquitin and V5-SET7, in the presence or absence of the proteasome inhibitor MG132. Cell lysates were analyzed with indicated antibodies.
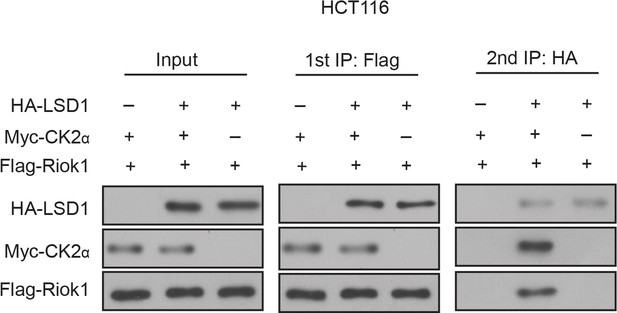
LSD1, CK2 and RIOK1 were in the same complex.
Two-step co-IP assay. The HCT116 cells were transfected with the HA-LSD1, Flag-RIOK1 and Myc-CK2 plasmids as indicated. Cells were lysed. The first immunoprecipitation was performed with an anti-Flag antibody. The complex was eluted with the Flag peptide, followed by the second step of immunoprecipitation with an anti-HA antibody. Protein samples from each step were subjected to western blot analysis.
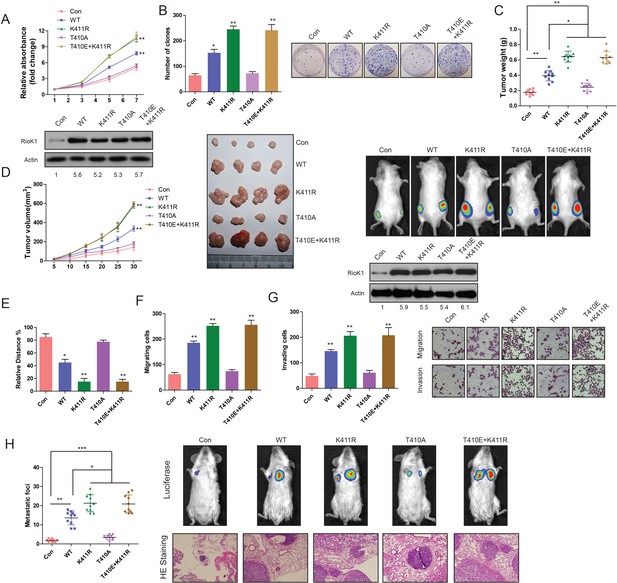
Methylation of RIOK1 at K411 and phosphorylation of RIOK1 at T410 oppositely regulated the CRC growth and metastasis.
(A) MTT assays in RKO cells expressing indicated proteins (upper panel). Western blotting was used to detect expression of RIOK1 in RKO cells using anti-RIOK1 antibody (lower panel), and the intensity of the western blot bands was quantified using NIH ImageJ software. (B) Photography and statistic results of colony formation assays in RKO cells expressing indicated proteins. (C and D) Statistic, tumor growth curves, photography results and representative bioluminescence images of tumor size in RKO cells expressing indicated proteins. Over-expression of RIOK1 in a xenograft tumor model was analyzed with western blotting, and the intensity of the western blot bands was quantified using NIH ImageJ software. (E-G) Migration ability evaluated by wound healing assay (E) and transwell migration assay (F). Invasiveness evaluated by matrigel invasion assay (G) in RKO cells expressing indicated proteins. (H) Statistic results, HE staining and representative bioluminescence images of lung metastasis in RKO cells expressing indicated proteins.
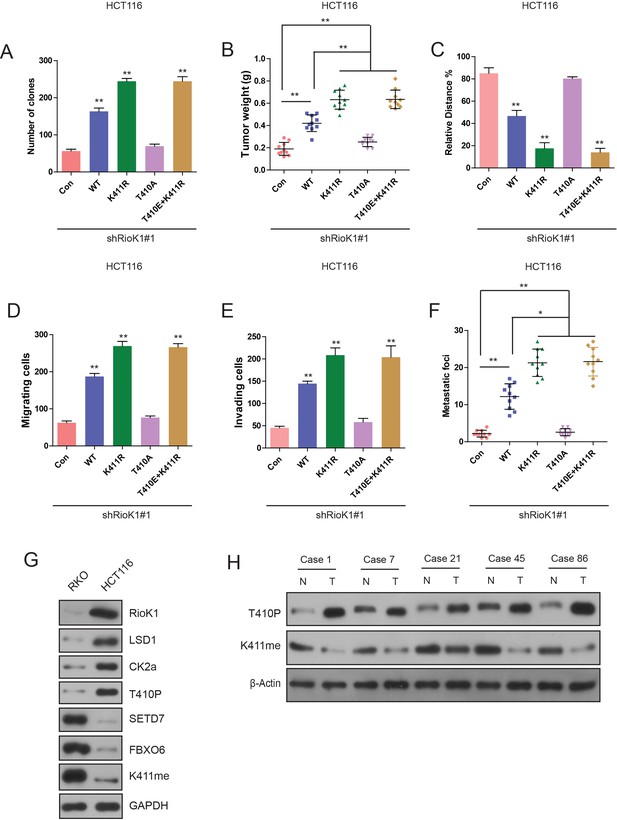
Methylation of RIOK1 at K411 and phosphorylation of RIOK1 at T410 oppositely regulated the CRC growth and metastasis.
(A) Photography and statistic results of colony formation assays in HCT116-shRIOK1#1 cells expressing indicated proteins. (B) Photography and statistic results of tumor weight in HCT116-shRIOK1#1 cells expressing indicated proteins. (C–E) Migration ability evaluated by wound healing assay (C) and transwell migration assay (D). Invasiveness evaluated by matrigel invasion assay (E) in HCT116-shRIOK1#1 cells expressing indicated proteins. (F) Photography and statistic results of lung metastasis in HCT116-shRIOK1#1 cells expressing indicated proteins. (G) WB analysis of indicated proteins in RKO and HCT116 cells. (H) WB analysis of T410P and K411me in CRC samples.
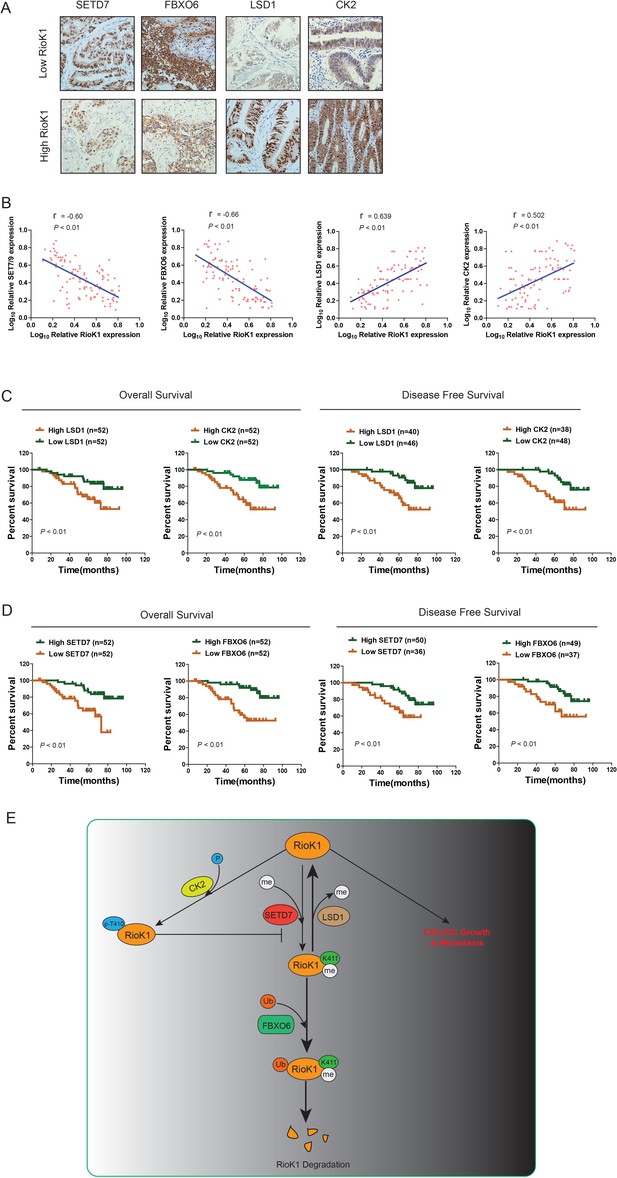
Clinical relevance of RIOK1, SET7/9, LSD1, FBXO6 and CK2 Expression in patients with CRC.
(A) Immunohistochemical staining of 104 human CRC for LSD1, CK2, SET7/9, and FBXO6 was performed. Representative photos of stains are shown in the groups with high (staining score, 5–8.0) and low (staining score, 0–4.0) expression of RIOK1. (B) Correlation between RIOK1 expression and LSD1, CK2, SET7/9, and FBXO6 expression in 104 clinical samples, respectively. (C and D) Kaplan–Meier plots of the overall survival and disease free survival in the patients (n = 104) with CRC in the groups with high (staining score, 5–8.0) and low (staining score, 0–4.0) expression of LSD1, and CK2 and SET7/9 and FBXO6. (E) A proposed model illustrating that the RIOK1 methyl-phospho switch by SET7/9- CK2-LSD1 axis dictates the stability of RIOK1 and its role in CRC and GC growth and metastasis.
Additional files
-
Supplementary file 1
Univariate analysis and multivariate analysis between RioK1 expresssion and Clinicopathologic Features of CRC Patients (n = 104)
- https://doi.org/10.7554/eLife.29511.022
-
Supplementary file 2
Identification of RioK1-interacting proteins via MS
- https://doi.org/10.7554/eLife.29511.023
-
Transparent reporting form
- https://doi.org/10.7554/eLife.29511.024





