Functional limb muscle innervation prior to cholinergic transmitter specification during early metamorphosis in Xenopus
Figures
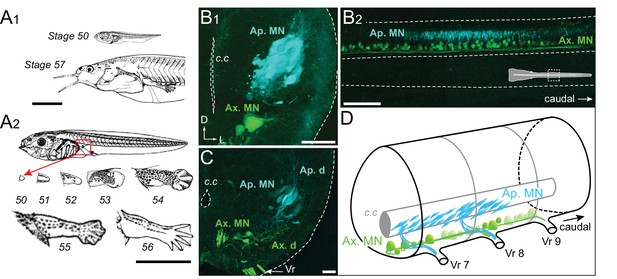
Developmental stages of X. laevis and anatomical organization of the spinal motor columns.
(A) Relative size differences between stage 50 and 57 larvae (A1) and associated morphological changes of the hindlimb bud during metamorphosis onset (A2), from Nieuwkoop and Faber (1956). (B, C) Segmental (B1, C) and rostrocaudal (B2) organization of the spinal motor column region containing limb MNs at stages 50–52 (B1, B2) and 55 (C). Inset in B2 shows the location of labeled MNs in the mid-region of the spinal cord. (D) Schematic representation of the segmental organization of the appendicular and axial motor columns in the larval spinal cord. Ap./Ax. MNs, appendicular/axial motoneurons; Ap./Ax. d., Ap./Ax. dendrites; Vr, ventral root; c.c, central canal; D, dorsal; L, lateral. Scale bars: A1 and A2 = 5 mm; B1 and C = 20 µm; B2 = 100 µm.
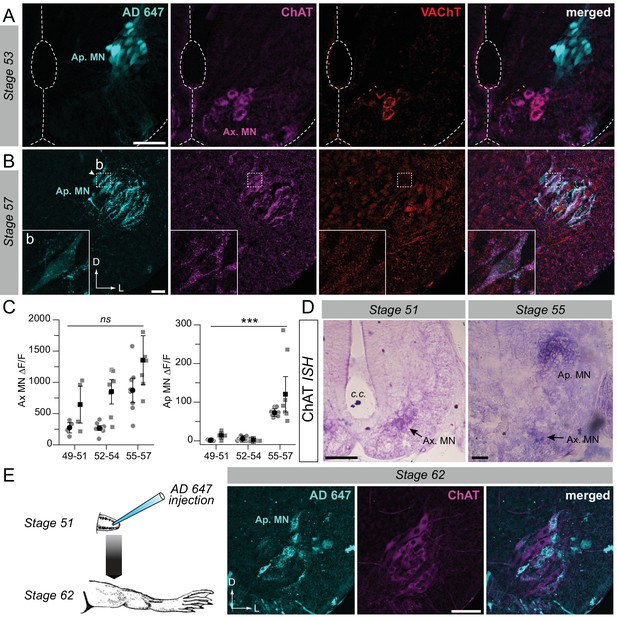
Developmental emergence of the molecular ACh phenotype in appendicular MNs.
(A, B) Examples of fluorescence immunolabeling against ChAT (magenta) and VAChT (red) in appendicular MNs (Ap. MN) labeled with retrograde Alexa Fluor dextran 647 (AD 647, cyan) in stages 53 (A) and 57 (B) tadpoles. Insets (b) in B show x60 magnification of stage 57 appendicular MNs. (C) Variation of fluorescence (∆F/F) of axial (left plot) and appendicular (right plot) MNs for ChAT and VAChT immuno-signals at stages 49–51 (n = 4), stages 52–54 (n = 7) and stage 55–57 (n = 8). Grey dots represent the averaged ∆F/F values for ChAT (squares) and VAChT (circles) in each preparation, and the black dots are ∆F/F grand means ± SEM for all preparations in a given developmental group. ns non significant, ***p<0.001, Kruskall-Wallis test. (D) Examples of in situ hybridization (ISH) labeling for ChAT mRNA in the appendicular spinal column at stages 51 and 55. (E) Example of fluorescence immunolabeling against ChAT in stage 62 appendicular MNs that were previously labeled with AD 647 injected into the hindlimb at stage 51 (see schematic at left). All scale bars = 50 µm; D, dorsal; L, lateral.
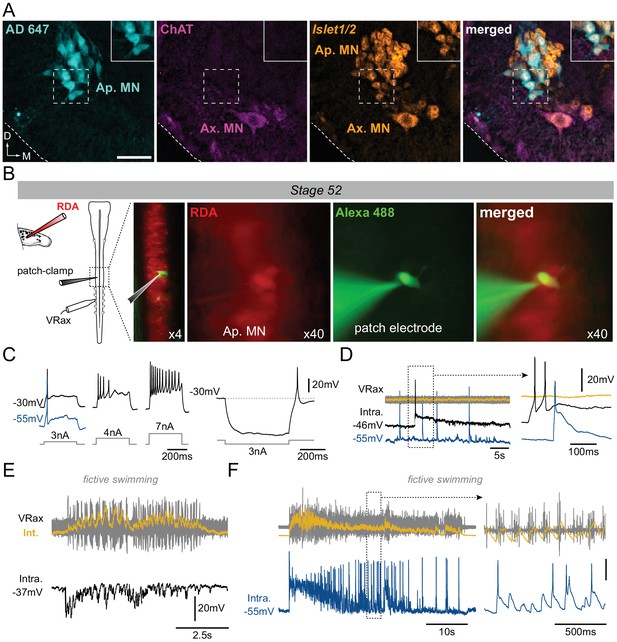
Motoneuronal identification of non-cholinergic limb projecting neurons.
(A) Example of fluorescence immunolabeling against ChAT (magenta) and Islet1/2 (orange) in appendicular and axial MNs (Ap. MN, Ax. MN) in a stage 52 tadpole. Appendicular MNs were previously labeled with retrograde Alexa Fluor dextran 647 (AD 647, cyan). Scale bar = 50 µm; D, dorsal; M, medial. (B) Protocol for intracellular recordings from appendicular MNs. Stage 52 MNs were identified by prior rhodamine dextran amine (RDA) retrograde labeling from the hindlimb bud and recorded with cell-attached patch-clamp in whole CNS preparations in order to preserve spinal locomotor circuitry. Alexa Fluor 488 added into the recording pipette allowed verifying the motoneuronal identity of the recorded cell. Recordings were made with either a low (black traces in C-F) or high [Cl−] (blue traces in C-F) intra-pipette solution. (C) Increasing steps of injected depolarizing current elicited increasing spike discharge whereas release from hyperpolarizing current injection evoked rebound spiking. (D) Spontaneous MN (Intra) firing in the absence of axial ventral root (VRax) activity. Extended time scale is shown at right. (E-F) MNs received strong rhythmic synaptic input during spontaneous episodes of fictive axial locomotion, which triggered locomotor-related spiking in high [Cl−]-recorded MNs (F). Extended time scale is shown at right. VRax traces (in yellow) are integrated transforms (Int.) of raw extracellular VR recordings.
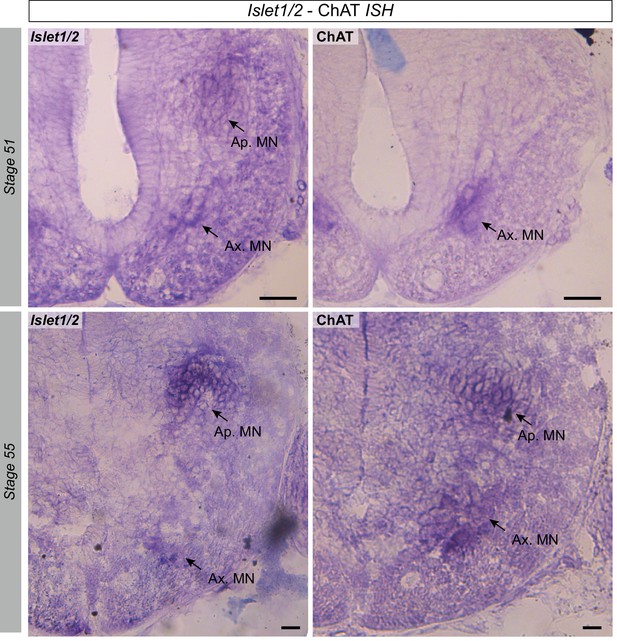
Presence of spinal Islet1 and ChAT mRNAs at different developmental stages.
In situ hybridization of the two mRNAs was performed with the same protocol (see Materials and methods). The Islet1 probe was synthesized according to Shi et al (2009) and involved amplifying a PCR fragment of 798 pb by the primers 5’-AAGTGCAACATCGGCTTCAG-3’ and 5’-GCTGTTTGGGGTATCTGGGA-3’. Despite the occasional appearance of a widespread background signal (due to differences in colorimetric reaction times), Islet1 mRNAs in darkened clusters were clearly evident in both the axial and appendicular motor columns at all developmental stages examined (stage 51 (upper left panel) and 55 (lower left panel) are illustrated), whereas ChAT mRNAs were found in limb MNs only from stage 55 onwards (c.f., upper and lower right panels).
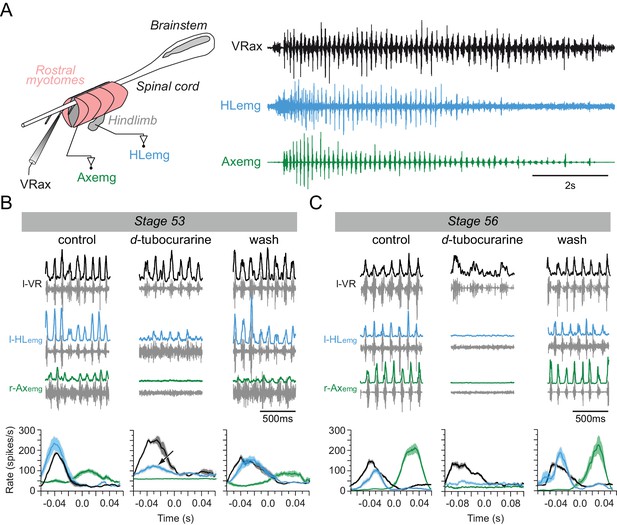
Switch from non-cholinergic to cholinergic limb muscle activation during axial swimming at different developmental stages.
(A) Rhythmic burst discharge (right panel) recorded from an axial ventral root (VRax) together with a hindlimb bud muscle (HLemg) and axial myotome (Axemg) during a spontaneous fictive swim episode in a semi-isolated stage 53 preparation (left panel). (B-C) Examples of recordings before (control), during and after (wash) bath application of d-tubocurarine to a stage 53 (B) and a stage 56 (C) preparation. Lower plots in B and C show instantaneous l-HLemg (blue) and r-Axemg (green) discharge rates (spikes/s) averaged (±SEM) over 10–20 l-VRax locomotor cycles (black). Black arrow in the middle graph of B indicates persistent limb bud EMG activity occurring in phase with the ipsilateral ventral root during d-tubocurarine application, and which was no longer present at the later developmental stage (c.f., middle plot in C).
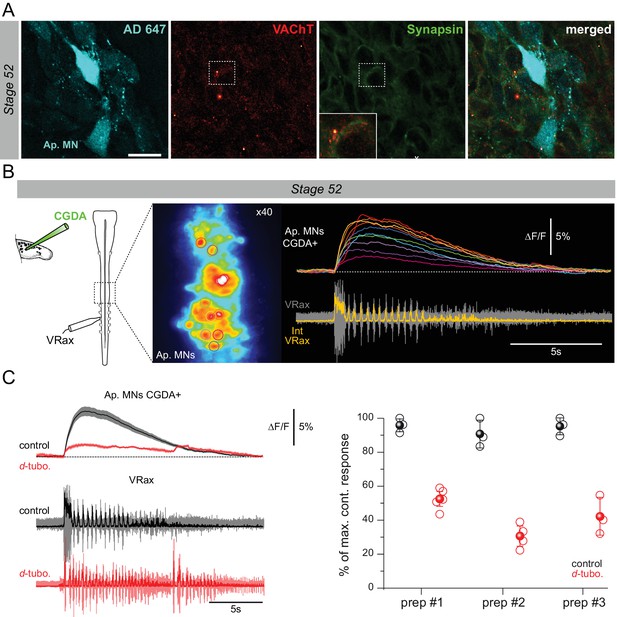
Appendicular MNs are activated centrally by cholinergic inputs.
(A) Immuno-labeling of putative cholinergic synapses on limb MNs retrogradely labeled with Alexa Fluor dextran 647 (AD 647), using antibodies against VAChT and the synaptic marker synapsin [mouse anti-synapsin (1:200; Synaptic Systems, Germany) revealed by secondary donkey anti-mouse antibodies coupled to Alexa Fluor 488 (1:500; Thermo Fischer)]. Both VAChT and synapsin fluorescent signals were found in close apposition surrounding MN cell bodies, indicating the presence of cholinergic input synapses. Inset: enlargement of synapsin and VAChT signals in the vicinity of an appendicular MN cell body. Scale bar = 20 µm (B) Calcium imaging optical recordings (20 fps) from stage 52 limb MNs retrogradely filled with Calcium Green Dextran Amine 3kD (CGDA, Invitrogen; see schematic at left) during fictive swimming (n = 3). Right panel: individual calcium signals (∆F/F) recorded simultaneously from 10 MN somata (encircled in x40 image at left) during a swim episode recorded from an axial VR (lower trace). (C) Left panel: average fluorescence variation (black trace; ± SEM, shaded area; top) for the 10 MNs illustrated in B during fictive swimming (bottom) in control (black) and in the presence of 50 µM d-tubocurarine (d-tubo.; red). Right panel: mean calcium transient amplitudes in control (black) and under d-tubocurarine (red) expressed as a percentage of the maximal control amplitude (i.e., ∆F/F peak soon after swim episode onset) for three stage 52 preparations. Unfilled circles represent single acquisitions; filled circles denote grand means ± SEM. Long term calcium recordings were performed in control experiments without drug application to avoid any fluorescence bleaching effect.
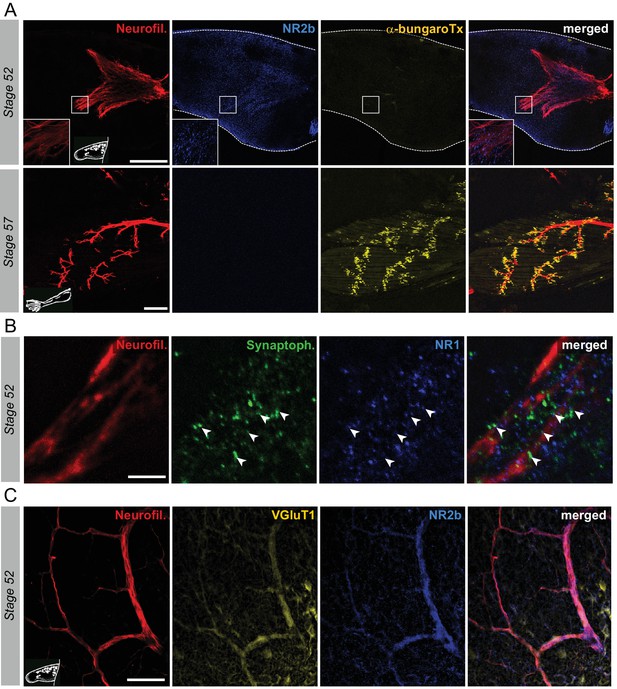
Switch from glutamate to acetylcholine receptors in hindlimb muscles.
(A) Examples of hindlimb innervation patterns and distribution of ACh nicotinic receptors in a whole-mount hindlimb bud, revealed by fluorescence immunolabeling of neurofilament associated protein (Neurofil., red), glutamate receptor (NR2b, blue) and α-bungarotoxin labeling (α-bungaroTx, yellow) respectively, at stages 52 (upper panels) and 57 (lower panels). Inset drawings at bottom left of each panel show bud morphology at the two representative larval stages. Scale bars = 200 µm for stage 52, 100 µm for stage 57. (B) Examples of fluorescence immunolabeling against neurofilament associated protein (red), synaptophysin (Synaptoph., green) and NMDA glutamate receptor subunit 1 (NR1, blue) in whole-mount limb bud at stage 52. White arrowheads indicate sites of apposition of all three markers. Scale bar = 20 µm. (C) Examples of fluorescence immunolabeling against neurofilament associated protein (red), the glutamate vesicular transporter 1 (VGluT1, yellow) and the glutamate receptor subunit NR2b (blue) in whole-mount limb at stage 52. Scale bar = 50 µm.
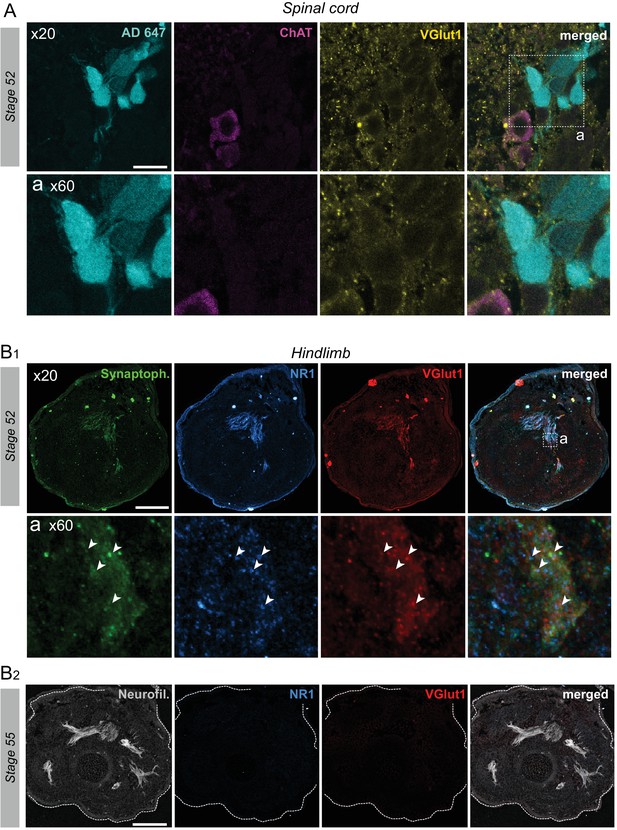
VGluT1 expression appendicular MNs.
(A) Fluorescence immunolabeling against ChAT (red) and VGluT1 (yellow) in limb MNs retrogradely labeled with Alexa Fluor dextran 647 (AD 647, cyan) at stage 52. Whereas ChAT is absent from retrogradely labeled MNs, VGluT1 is expressed throughout almost the entire ventral horn, including in limb MN somata. Bottom images are magnifications of the area (a) delineated by the dotted line square in the merged image at top right. Punctiform VGluT1 expression occurred around MN somata (indicating labeling of glutamatergic presynaptic terminals) and a more diffuse VGluT1 signal also appeared in MN cytoplasm surrounding cell nuclei. Scale bar = 20 µm. (B) Fluorescence immunolabeling against synaptophysin (Synaptoph., green), NMDA glutamate receptor subunit 1 (NR1, blue), VGluT1 (red) and neurofilament (Neurofil., grey) in hindlimb muscle tissue at stage 52 (B1) and stage 55 (B2). VGluT1 expression colocalized with synaptophysin and postsynaptic glutamate receptors were detected at stage 52, but not at stage 54. The bottom images in B1 are magnifications of the area (a) indicated in the merged image at top right. Scale bars = 200 µm for stage 52 (B1) and 400 µm for stage 55 (B2).
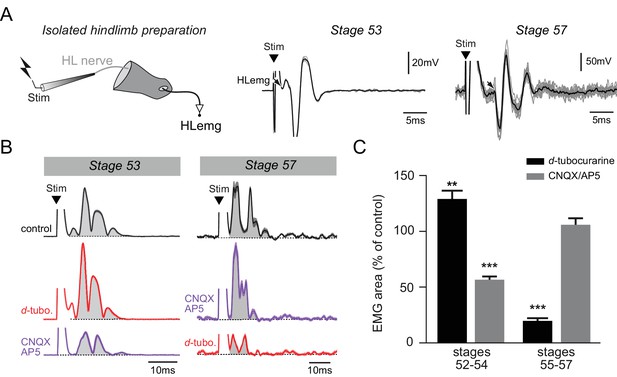
Functional switch in hindlimb neuromuscular transmission.
(A) EMG recordings (HLemg) from an isolated hindlimb preparation (schematic at left) in response to single pulse (10 µs) electrical stimulation (Stim) of a limb motor nerve (HL nerve) in stages 53 and 57 larvae. In each case, the black arrow indicates the beginning of the EMG response and the black trace represents the mean profile of 6 superimposed responses. Note that the trace illustration for stage 53 was taken from a 1 mM Mg2+ saline experiment. (B) Integrated motor nerve-evoked HLemg responses in control (black), and under d-tubocurarine (d-tubo.; red) or CNQX + AP5 (purple) bath application to stage 53 and 57 preparations. Thick lines represent the mean response profile (± SEM); the area under each curve (grey) was used to measure the response size. (C) Mean (± SEM) EMG response area as percentage of control response during d-tubocurarine (black) or CNQX/AP5 (grey) bath application at stages 52–54 and 55–57, respectively. **p<0.01 and ***p<0.001, Mann–Whitney U-test.
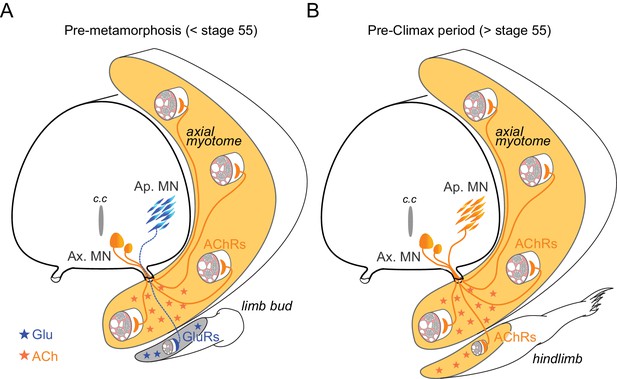
Schematic representation of neurotransmitter phenotype switching associated with the establishment of functional limb muscle innervation at different stages of Xenopus metamorphic development.
See text for further explanation. AChRs: nicotinic ACh receptors; GluRs: glutamate receptors; Ap. MN: appendicular MNs; Ax. MN: axial MNs; c.c: central canal.
Additional files
-
Transparent reporting form
- https://doi.org/10.7554/eLife.30693.012





