Activation of Discs large by aPKC aligns the mitotic spindle to the polarity axis during asymmetric cell division
Figures
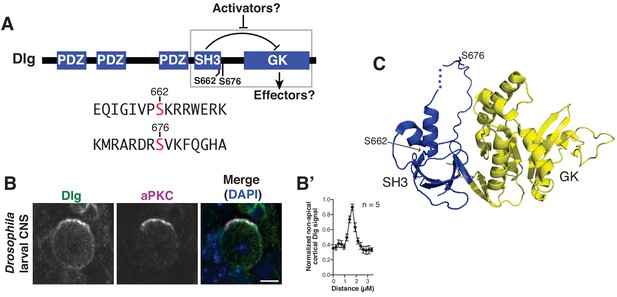
Discs large localizes uniformly to the neuroblast cortex and is phosphorylated by atypical Protein Kinase C.
(A) Dlg domain structure. An intramolecular interaction between the SH3 and GK domains inhibits GK binding to certain effectors. Dlg activators may function by disrupting the intramolecular interaction, allowing a necessary spindle orientation factor to bind. S662 and S676 denote the two sites of modification by aPKC identified by mass spectrometry. The sequences surrounding each phosphorylated serine are shown. (B) Localization of Dlg and aPKC in a metaphase Drosophila larval brain neuroblast showing that Dlg, while enriched at the apical cortex with aPKC, is also found in significant amounts at non-apical regions of the cortex. (B') Quantification of non-apical Dlg signal. (C) Location of aPKC phosphorylation sites mapped on to the structure of the SH3GK module from PSD-95 (PDB ID: 1KJW).
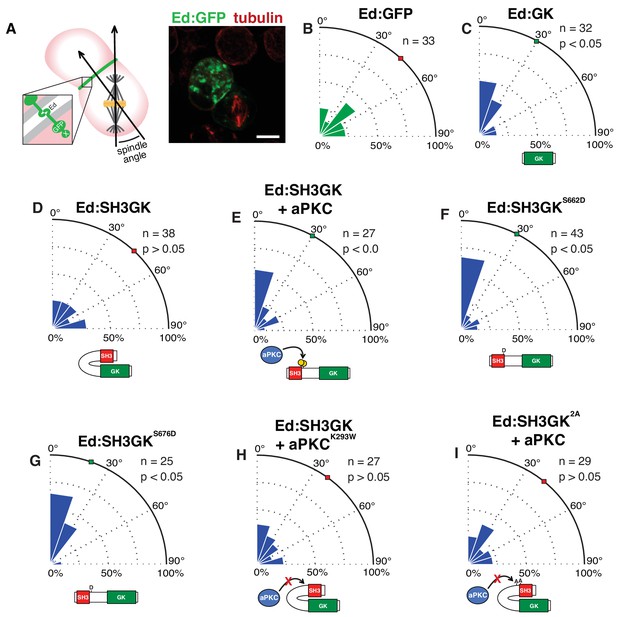
The atypical protein kinase C induces spindle orientation by Dlg SH3GK.
(A) Induced polarity spindle orientation assay. S2 cells clustered by the Echinoid adhesion protein (green) polarize proteins attached to the Ed COOH-terminus (‘X’). The spindle angle is measured between axes defined along the spindle and the middle of the polarized crescent and cell. A representative image (anti-tubulin, red; GFP, green) of a typical analyzed cell cluster is shown. Spindle angle measurements (polar histogram; green represents control and blue represents experimental) for cells expressing (B) Ed fused to GFP (Ed:GFP) (C) Ed fused to the Discs large GK domain (Ed:GK) (D) Ed:SH3GK (E) Ed:SH3GK co-expressed with aPKC (F) Ed:SH3GK with a phosphomimetic aspartic acid at position 662 (G) Ed:SH3GK with a phosphomimetic aspartic acid at position 676 (H) Ed:SH3GK co-expressed with aPKC containing the K293W mutation in its ATP-binding pocket that prevents kinase activity (I) Ed:SH3GK with both aPKC phosphorylation sites mutated to alanine co-expressed with aPKC. For each condition, the number of measurements is shown along with a p-value calculated using a K-S test. Individual measurements are included in comma separated value format in Figure 2—source data 1.
-
Figure 2—source data 1
Individual spindle angle measurements in comma separated value format (corresponding figure panel is included in the column header).
- https://doi.org/10.7554/eLife.32137.004
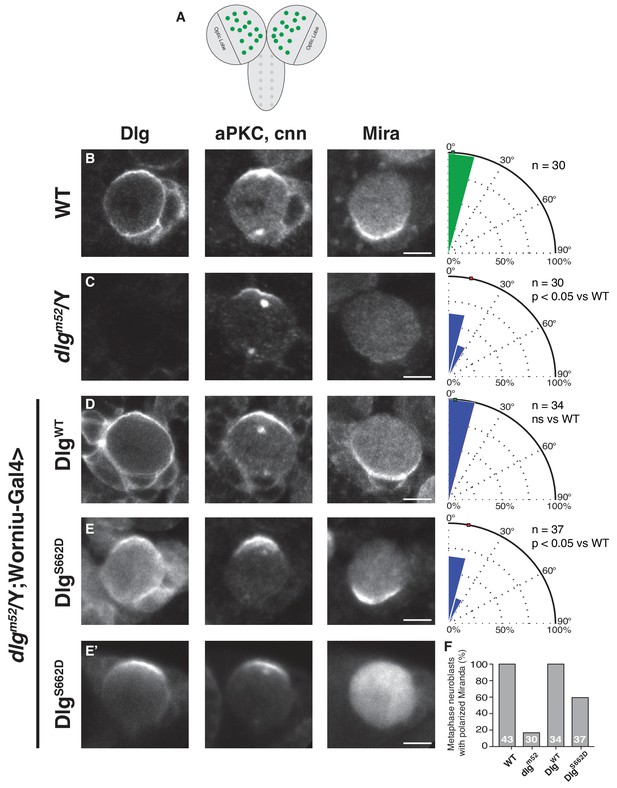
Constitutive Dlg activity disrupts spindle orientation in Drosophila larval brain neuroblasts.
(A) Schematic of Drosophila larval central nervous system showing brain lobes and ventral nerve cord. In this study we analyzed neuroblasts (green circles) from the brain lobes outside of the optic lobes. Neuroblasts were stained for Dlg, aPKC, the centrosome marker centrosomin (cnn) to measure spindle angle, and the polarized neural fate determinant Miranda (Mira), after dissection from animals with the following genotypes (B) Wild-type (C) dlgm52 null mutant (D) dlgm52 Worniu-Gal4 >UAS WT Dlg (E) dlgm52 Worniu-Gal4 >UAS Dlg with a phosphomimetic mutation at site S662 (E') the same genotype as in panel D but showing the variability in Mira polarization we observed. For each condition, a polar histogram with the measured spindle angles is shown along with a p-value calculated by comparison with the wild-type condition using the K-S test. (F) Quantification of neuroblasts exhibiting polarized Mira for the conditions shown in panels B-E'. Individual measurements are included in comma separated value format in Figure 3—source data 1.
-
Figure 3—source data 1
Individual spindle angle measurements in comma separated value format (corresponding figure panel is included in the column header).
- https://doi.org/10.7554/eLife.32137.006
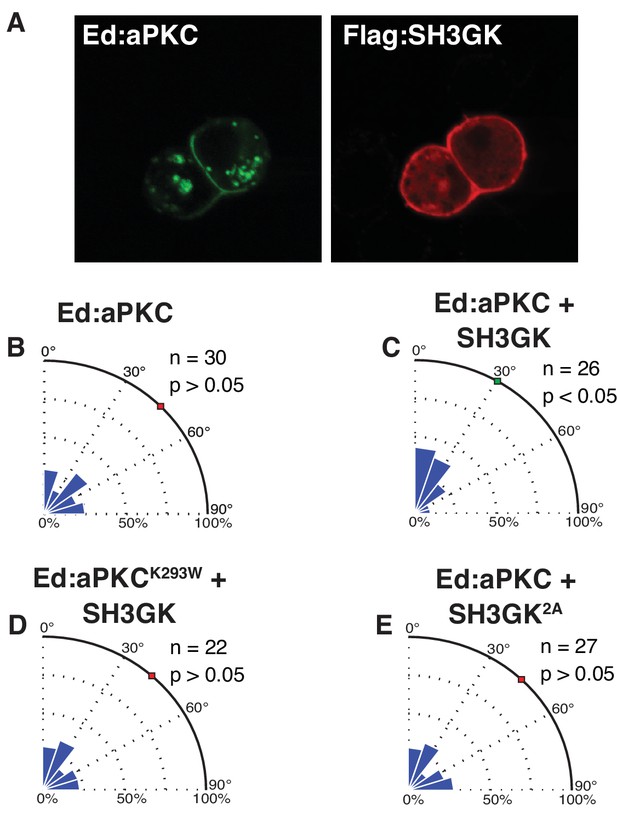
Dlg mediates spindle alignment by aPKC.
Ed:aPKC was polarized in S2 cells and in some cases Dlg was co-expressed. (A) Localization of Ed:aPKC and SH3GK in S2 cells. Spindle measurements were made for the following conditions (B) Ed:aPKC (C) Ed:aPKC co-expressed with Dlg SH3GK (D) Ed:aPKC containing the ‘kinase-dead’ K293W mutation co-expressed with Dlg SH3GK. (E) Ed:aPKC co-expressed with Dlg SH3GK containing alanine substitutions at the two aPKC phosphorylation sites. For each condition, a polar histogram of measured angles is shown along with a p-value calculated by comparison against Ed:GFP (Ed:aPKC) or Ed:aPKC (all others) using the K-S test. Individual measurements are included in comma separated value format in Figure 4—source data 1.
-
Figure 4—source data 1
Individual spindle angle measurements in comma separated value format (corresponding figure panel is included in the column header).
- https://doi.org/10.7554/eLife.32137.008
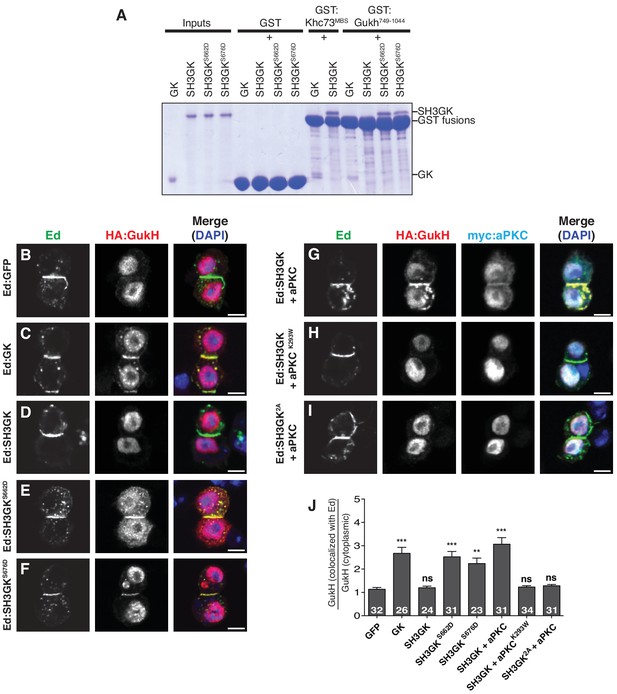
aPKC phosphorylation activates GukHolder binding and recruitment by Dlg SH3GK.
(A) GK versus SH3GK binding of Khc73 and GukHolder. ‘Inputs’ are approximately 2 µg of purified Dlg GK and SH3GK proteins. ‘GST’ lanes are the results of affinity chromatography with GST alone to measure the amount of background binding. ‘GST-Khc73MBS’ lanes are the Kinesin Khc73's ‘MAGUK Binding Stalk’ with the Dlg and GK SH3GK, showing no qualitative effect on binding by the autoinhibitory SH3GK interaction. ‘GST-GukH749-1044’ lanes are the GukH COOH-terminus with Dlg GK and SH3GK. Binding to the GK but not the SH3GK indicates that GukH is inhibited by the SH3GK intramolecular interaction. Binding to SH3GKs containing phosphomimetic mutations suggests that aPKC phosphorylation overcomes autoinhibition. (B–J) Cultured S2 cells expressing full-length GukH and the following Ed fusions with Dlg stained for Ed, GukH and DAPI (B) Ed fused to GFP (C) Ed fused to Dlg GK (D) Ed fused to Dlg SH3GK (E) Ed:SH3GK with phosphomimetic mutation at S662 (F) Ed:SH3GK with phosphomimetic mutation at S676 (G) Ed:SH3GK co-expressed with aPKC (H) Ed:SH3GK co-expressed with ‘kinase-dead’ aPKC (I) Ed:SH3GK containing alanine mutations at the aPKC phosphorylation sites co-expressed with aPKC (J) Quantification of GukH recruitment to Ed-fusion proteins. The number of measurements is shown in each bar and significance (**p<0.01, ***p<0.001) calculated by comparison with the GFP condition calculated using ANOVA with Dunnett's post-test.
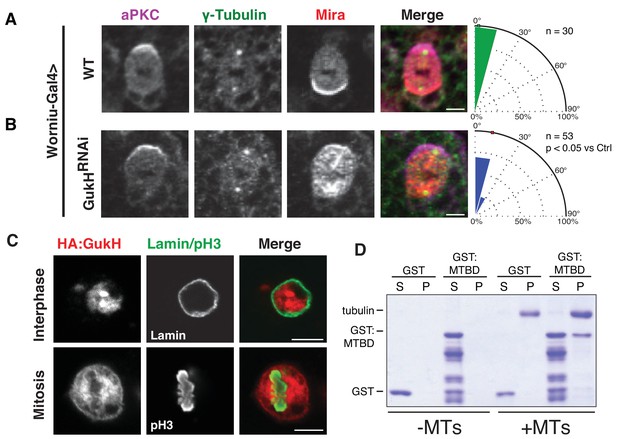
Gukholder is a microtubule-binding protein that mediates neuroblast spindle orientation.
Spindle angle measurements from larval brain neuroblasts stained for aPKC, γ-tubulin (used to measure spindle angles), and Mira from animals with the following genotypes (A) Worniu-Gal4 >WT (no RNA expressed) (B) Worniu-Gal4 >GukH RNAi. Polar histograms of angle measurements are shown along with the p-value calculated by comparison with the control RNAi condition using the K-S test. (C) Localization of GukH in interphase and mitotic S2 cells. Cells were stained for HA to detect tagged full-length GukH, Lamin for the nuclear membrane, and pH3 for mitotic chromatin. (D) GST and a GST fusion of the putative GukH microtuble-binding domain (MTBD; residues 404-534) were subjected to ultracentrifugation both in the absence (–MTs) and presence (+MTs) of microtubules. The supernatant and pellet from these experiments are shown. Individual measurements are included in comma separated value format in Figure 6—source data 1.
-
Figure 6—source data 1
Individual spindle angle measurements in comma separated value format (corresponding figure panel is included in the column header).
- https://doi.org/10.7554/eLife.32137.012
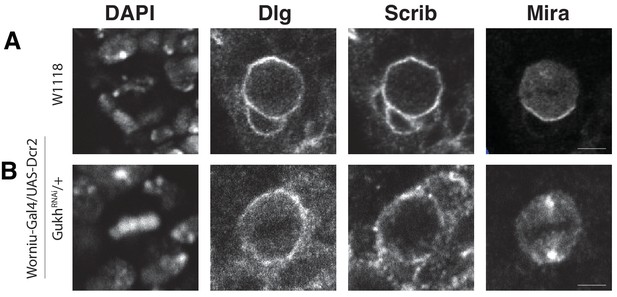
GukHolder is not required for localization of its binding partner, the polarity protein and tumor suppressor Scribble.
Neuroblasts were stained with DAPI (DNA), anti-Dlg, anti-Scrib, and anti-Mira antibodies (A) Worniu-Gal4 > WT (no RNAi) (B) Worniu-Gal4 > GukH RNAi.
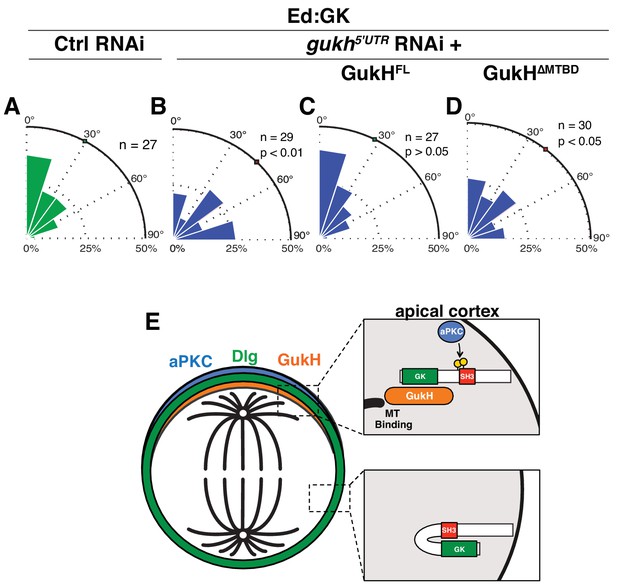
The GukHolder microtubule binding domain mediates spindle orientation.
Spindle angles measured in S2 cells expressing Ed fused to Dlg GK exposed to the following RNAi conditions (A) Control RNAi (B) RNAi directed against the gukh 5' UTR (C) gukh5'UTR RNAi with expression of full-length GukH (D) gukh5'UTR RNAi with expression of full-length GukH lacking its microtuble-binding domain (MTBD). For each condition, a polar histogram of the measured angles is shown and a p-value calculated by comparison to the control RNAi condition using the K-S test. (E) Model for coupling of polarity and spindle orientation during asymmetric cell division. When Dlg is not co-localized with aPKC, it is autoinhibited by the SH3GK intramolecular interaction and cannot recruit GukH. Phosphorylation of Dlg by aPKC relieves autoinhibition allowing it to interact with GukH and recruit it to sites containing aPKC. The GukH interaction with microtubules mediates spindle orientation. Individual measurements are included in comma separated value format in Figure 7—source data 1.
-
Figure 7—source data 1
Individual spindle angle measurements in comma separated value format (corresponding figure panel is included in the column header).
- https://doi.org/10.7554/eLife.32137.014
Additional files
-
Transparent reporting form
- https://doi.org/10.7554/eLife.32137.015





