HCN2 channels in the ventral tegmental area regulate behavioral responses to chronic stress
Figures
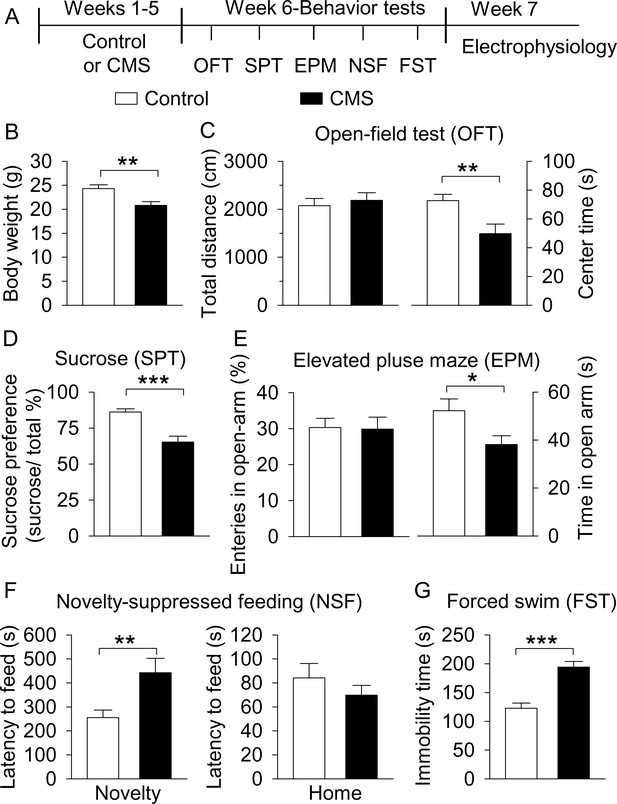
CMS produced depressive- and anxiety-like behaviors.
(A) The timeline of CMS, behavioral tests, and electrophysiology. (B) CMS significantly decreased body weight compared with non-stressed control mice (**p=0.004, control, n = 14 mice; CMS n = 12 mice from B to G). (C) CMS significantly decreased the center time (**p=0.007) without affecting total distance traveled in the OFT (p=0.615). (D) CMS significantly decreased sucrose preference compared to control (***p<0.001). (E) CMS did not affect entries into the open arms (p=0.919), but significantly decreased the time spent in the open arms (*p=0.036) in the EPM test. (F) CMS significantly increased the latency to feed in the novel environment (Novelty) in the NSF test (**p=0.008) but did not significantly affect the latency to feed in the home cage (Home) (p=0.345). (G) CMS significantly increased immobility time in the FST (***p<0.001).
-
Figure 1—source data 1
Body weight and behavior following CMS in Figure 1B–G.
- https://doi.org/10.7554/eLife.32420.004
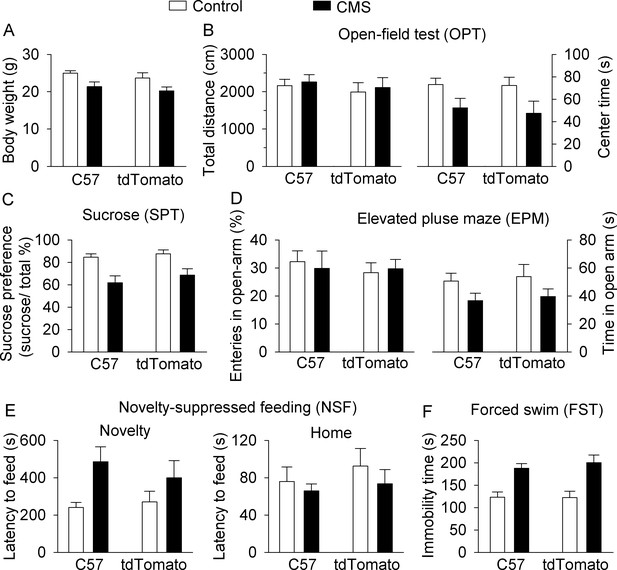
C57BL/6J and DAT-tdTomato mice do not significantly differ in body weight or behaviors at baseline or in response to CMS.
Figure 1 was reanalyzed to compare C57BL/6J (C57) and DAT-tdTomato (tdTomato) reporter mice for control and CMS-induced effects on body weight (A) and behavior in OPT (B) SPT (C) EPM (D) NSF (E) and FST (F). Detailed statistical analysis is presented in Supplementary file 1. Control-C57, n = 7 mice; control-tdTomato, n = 6 mice; CMS-C57, n = 7 mice; CMS-tdTomato, n = 6 mice.
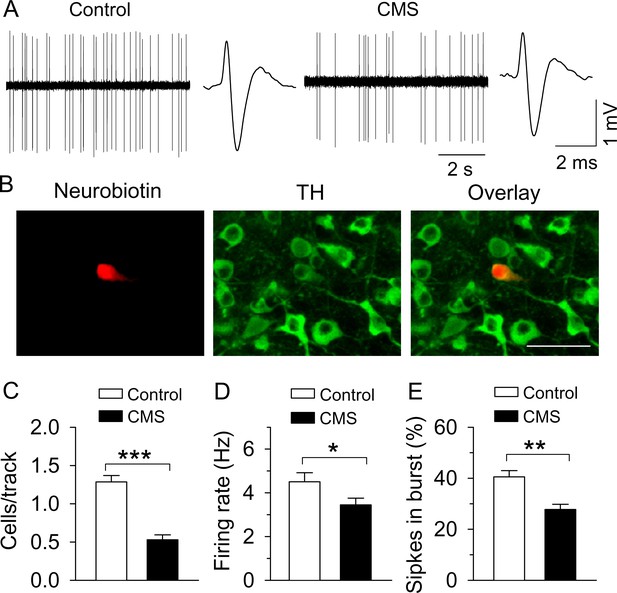
CMS decreased single-unit AP firing in VTA dopamine neurons in vivo.
(A) Sample traces of VTA dopamine neuron AP firing in control and CMS mice. Dopamine neurons were identified by a broad triphasic extracellular action potential of a width greater than 2 ms and a relatively slow firing rate (<10 Hz). (B) A recorded dopamine neuron was confirmed by neurobiotin (red) and TH (tyrosine hydroxylase, green) co-localization. (C–E) Population activity (C, ***p<0.001, control, n = 4 mice; CMS n = 5 mice), firing rate (D) *p=0.045, control, n = 15 cells from four mice; CMS, n = 17 cells from five mice), and the percent of spikes in burst (E, **p=0.006, control, n = 10 cells from four mice; CMS, n = 13 cells from five mice) were decreased in CMS mice.
-
Figure 2—source data 1
In vivo VTA dopamine neuron firing following CMS in Figure 2C–E.
- https://doi.org/10.7554/eLife.32420.006
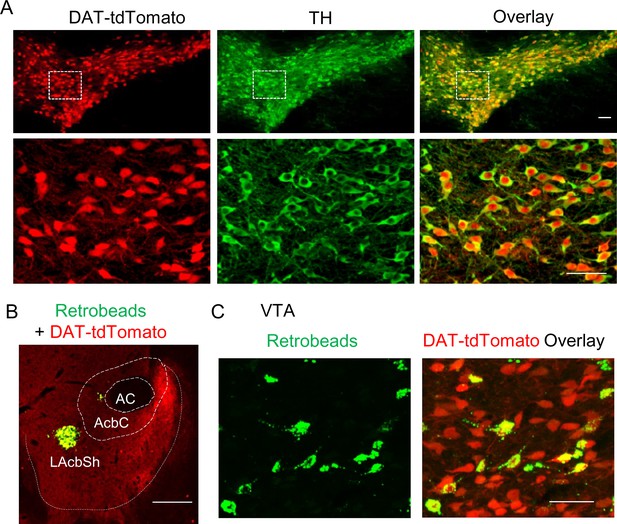
Retrobead labeling of VTA dopamine neurons that project to the lateral shell of the NAc.
(A) DAT-Cre mice were bred with Ai9 reporter mice, which express tdTomato in the presence of Cre, to produce DAT-tdTomato mice. TdTomato and TH (green) were completely co-localized, indicating that tdTomato expression provides faithful reporting of dopamine neurons for slice physiology. (B) Green Retrobeads were injected into the lateral shell of the NAc (LAcbSh) in DAT-tdTomato mice. (C) The Retrobeads were retrogradely transported to the VTA and were predominantly co-localized with tdTomato-positive VTA dopamine neurons.
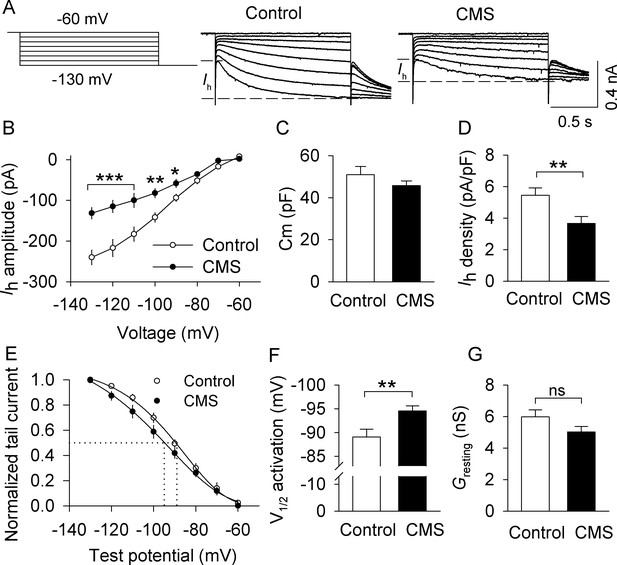
CMS decreased Ih currents in VTA dopamine neurons that project to the lateral shell of the NAc (LAcbSh).
(A) Left: Voltage protocol for recording Ih current. Right: Representative Ih current recorded from NAc-projecting VTA dopamine neurons in control and CMS mice. (B) Compared with the control group, Ih amplitude was significantly decreased in the CMS group at corresponding hyperpolarization potentials (*p<0.05, **p<0.01, ***p<0.001, control, n = 15 cells from five mice; CMS, 13 cells from three mice from B to G). Ih amplitude was calculated by subtracting the instantaneous current from the steady-state current achieved during the voltage step. (C) The membrane capacitance (Cm) was not significantly different between control and CMS mice (p=0.273). (D) Ih current density was significantly decreased in the CMS group compared with the control group (**p=0.009). (E) Ih activation curves in the control and CMS groups generated by the tail current protocol. Tail current amplitudes were fitted with a Boltzmann function. (F) CMS led to a significant hyperpolarizing shift of the half-activation potential (V1/2) compared with that of control (**p=0.007). (G) The resting membrane conductance (Gresting) was not significantly different between control and CMS mice (p=0.107).
-
Figure 4—source data 1
Ih amplitude and activation properties in VTA dopamine neurons following CMS in Figure 4B–G.
- https://doi.org/10.7554/eLife.32420.010
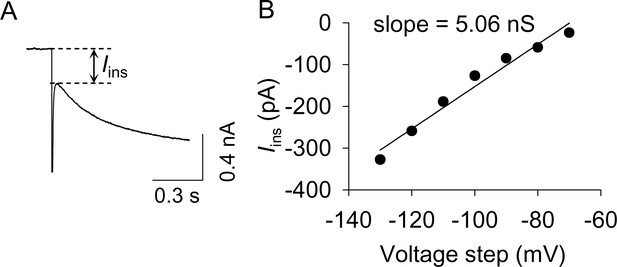
The measurement of resting membrane conductance.
(A) The instantaneous inward current (Iins) was measured as indicated by the dashed lines. (B) A representative example for calculating resting membrane conductance. Iins was plotted against the hyperpolarizing voltage steps. The slope of these I-V curves is indicative of the resting membrane conductance shown in Figure 4G.
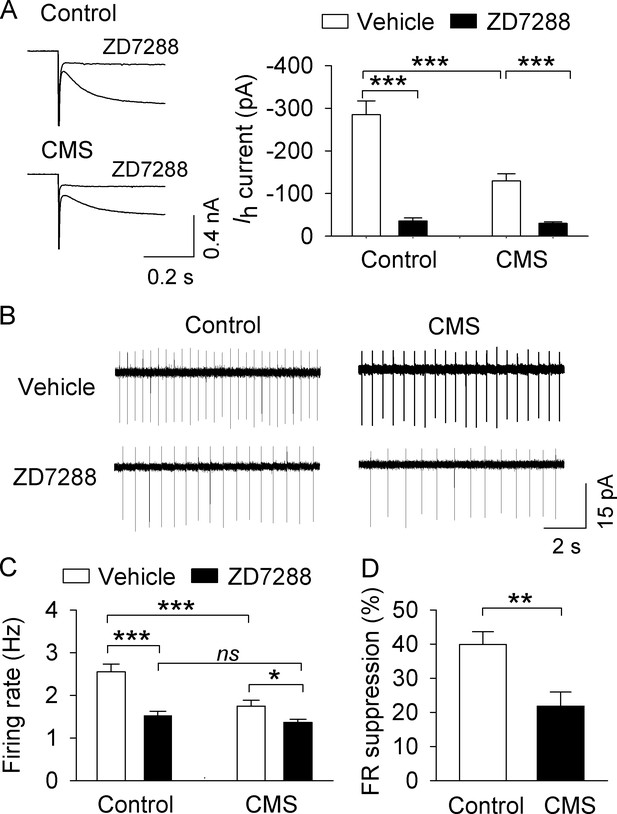
, CMS decreased AP firing in VTA dopamine neurons in midbrain slices.
(A) Ih currents recorded at −130 mV in NAc-projecting VTA dopamine neurons in both control (***p<0.001, n = 6 cells from three mice) and CMS groups (***p<0.001, n = 6 cells from three mice) were abolished by the Ih channel blocker ZD7288 (30 μM). (B) Representative AP firing in cell-attached recordings from NAc-projecting VTA dopamine neurons in control and CMS slices before and after ZD7288 (30 µM). (C) The AP firing rate was significantly decreased in the CMS group (n = 13 cells from three mice) compared with the control group (n = 12 cells from four mice; ***p<0.001). ZD7288 significantly decreased the firing rate in both control (n = 11 cells from three mice) and CMS (n = 12 cells from five mice) groups (*p<0.5, ***p<0.001). The mean firing rate was not significantly different between control (n = 11 cells from three mice) and CMS (n = 12 cells from four mice) groups following ZD7288 (p=0.382). (D) The firing rate (FR) suppression (%) by ZD7288 in the control group (n = 11 cells from three mice) was significantly higher than that of the CMS group (n = 12 cells from five mice; **p=0.004).
-
Figure 5—source data 1
Effects of ZD7288 on Ih current and ex vivo AP firing in VTA dopamine neurons following CMS in Figure 5A,C,D.
- https://doi.org/10.7554/eLife.32420.012
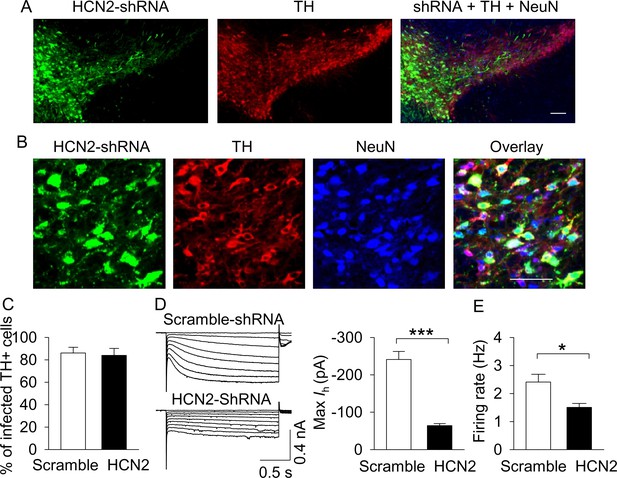
AAV-mediated shRNA knockdown of HCN2 in the VTA.
(A,B), Immunofluorescence labeling showing the expression of AAV2-HCN2-shRNA-eGFP (green), TH (dopamine neuron marker, red) and NeuN (neuronal marker, blue) in the midbrain under low magnification (A) and high magnification (B). Scale bars: 50 µm. (C), The percentage of TH+ VTA dopamine neurons that were infected with AAV2-HCN2-shRNA-eGFP or scramble-shRNA (n = 3 mice/group). (D), Maximal Ih current amplitude was significantly decreased in AAV2-HCN2-shRNA-eGFP-infected dopamine neurons (n = 14 from three mice) compared with AAV2-scramble-shRNA-eGFP-infected dopamine neurons (n = 15 from four mice; ***p<0.001). (E), AP firing frequency was decreased in AAV2-HCN2-shRNA-GeFP-infected neurons (n = 10 from three mice) compared with AAV2-scramble-shRNA-eGFP-infected neurons (n = 9 from four mice; *p=0.036).
-
Figure 6—source data 1
Ih current and AP firing following shRNA-mediated HCN2 knockdown in Figure 6C–E.
- https://doi.org/10.7554/eLife.32420.015
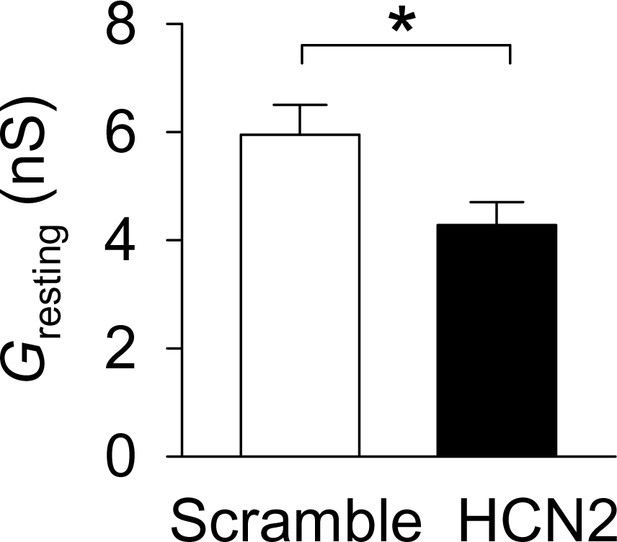
The resting conductance (Gresting) was significantly decreased in the HCN-shRNA group (n = 14) compared with the scrambled group (n = 15, *p=0.022).
https://doi.org/10.7554/eLife.32420.014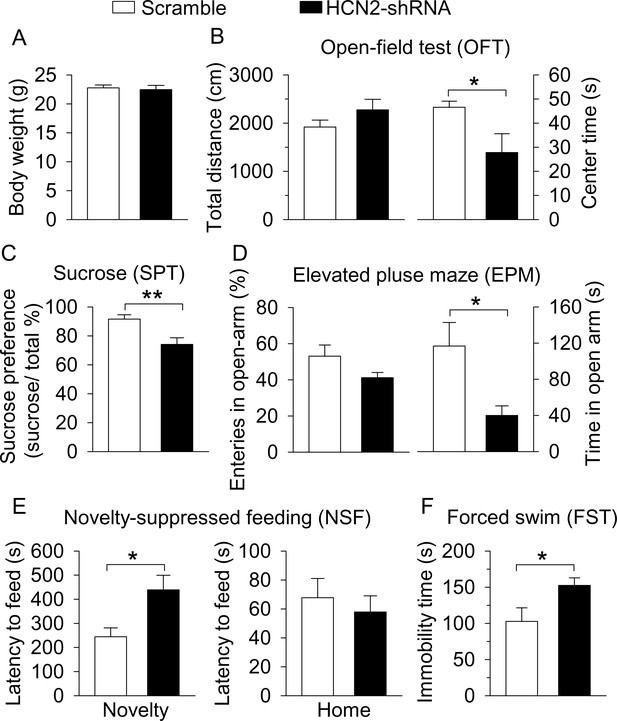
ShRNA knockdown of HCN2 in the VTA produced anxiety- and depressive-like behaviors.
(A) VTA-specific HCN2 knockdown did not significantly affect the body weight of mice (control, n = 7 mice; CMS, n = 7 mice; p=0.727). (B) HCN2 knockdown significantly decreased the center time (*p=0.042) without affecting the total distance traveled (p=0.197) in the OFT. (C) HCN2 knockdown significantly decreased sucrose preference (**p=0.003). (D) HCN2 knockdown did not affect entries into the open arms (p=0.104) but significantly decreased time spent in the open arms (*p=0.018) in the EPM test. (E) HCN2 knockdown increased the latency to feed in the novel environment (Novelty) in the NSF test (*p=0.017) but did not significantly affect the latency to feed in the home cage (Home; p=0.583). (F) HCN2 knockdown increased immobility time in the FST (*p=0.039).
-
Figure 7—source data 1
Body weight and behavior following VTA HCN2 knockdown in Figure 7A–F.
- https://doi.org/10.7554/eLife.32420.017
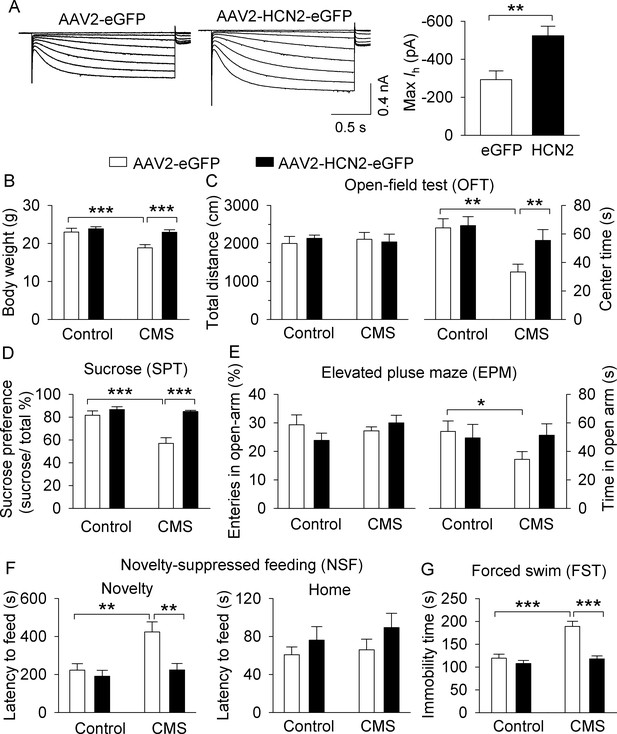
Overexpression of HCN2 in the VTA prevented the development of CMS-induced depressive-like behavior.
(A) The maximal amplitude of Ih current in AAV2-HCN2-eGFP-infected VTA dopamine neurons (n = 15 from four mice) was significantly increased compared with that in AAV2-eGFP-infected VTA dopamine neurons (n = 14 cells from five mice; **p=0.002). (B) CMS significantly decreased the body weight of mice in the AAV2-eGFP injection group (***p<0.001, control, n = 9 mice; CMS, n = 10 mice from B to G), whereas CMS did not decrease the body weight of mice in the HCN2 overexpression group (p=0.419, control, n = 10 mice; CMS, n = 8 mice from B to G). (C) Neither CMS nor HCN2 overexpression affected the total distance traveled in the OFT test (p>0.05). Compared with non-stressed control mice, CMS significantly decreased the time spent in the center square of the open field in the AAV2-eGFP group (**p=0.002), and this decrease was prevented by HCN2 overexpression (**p=0.004). (D) CMS significantly decreased sucrose preference in the AAV2-eGFP injection group (***p<0.001), whereas HCN2 overexpression prevented this decrease (***p<0.001). (E) In the AAV2-GFP group, CMS significantly decreased time spent in the open arms (*p=0.023) but did not affect open arm entries (p>0.05). HCN2 overexpression did not significantly affect open arm time nor entries (p=0.057) compared to the AAV2-GFP group. (F) CMS induced a significant increase in the latency to feed in the novel environment in the NSF test (**p=0.002), which was prevented by HCN2 overexpression (**p=0.001). Neither CMS nor HCN2 overexpression affected the latency to feed in the home cage (p>0.05). (G) CMS produced a significant increase in immobility in the FST (***p<0.001), and this increase was prevented by HCN2 overexpression (***p<0.001).
-
Figure 8—source data 1
VTA HCN2 overexpression effects on Ih current and CMS-induced changes in body weight and behavior in Figure 8A–G.
- https://doi.org/10.7554/eLife.32420.019
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Strain, strain background (Mus musculus) | male C57BL/6J mice | The Jackson Laboratory | Stock#: 000664 RRID:IMSR_JAX:000664 | |
| Genetic reagent (Mus musculus) | male heterozygous Slc6a3Cre+/− (DAT-Cre) mice | The Jackson Laboratory | Stock#: 006660 RRID:IMSR_JAX:006660 | maintained on the C57BL/6J background |
| Genetic reagent (Mus musculus) | male Ai9 reporter mice | The Jackson Laboratory | Stock#: 007909 RRID:IMSR_JAX:007909 | maintained on the C57BL/6J background |
| Strain, strain background (Adeno-associated virus) | AAV2.shRNA.U6.ShRLuc. CMV.eGFP.SV40 | shRNA provided by Dr. Han-gang Yu (PMID: 19236845), packed into AAV2 at Penn Vector Core | ||
| Strain, strain background (Adeno-associated virus) | AAV2.scramble.U6.ShRLuc. CMV.eGFP.SV40 | Penn Vector Core | ||
| Strain, strain background (Adeno-associated virus) | AAV2.CMV.PI.HCN2. WPRE.eGFP.SV40 | HCN2 plasmid packed into AAV2 at Penn Vector Core | HCN2 plasmid provided by Dr. Dane Chetkovich at Northwestern University | |
| Strain, strain background (Adeno-associated virus) | AAV2.CMV.PI.eGFP.WPRE.bGH | Penn Vector Core | ||
| Antibody | Mouse anti-TH | Santa Cruz Biotechnology | SC-136100, Lot: G1309, RRID:AB_2287193 | monoclonal, 1:300 |
| Antibody | Rabbit anti-TH | Santa Cruz Biotechnology | SC-14007, Lot: C2707, RRID:AB_671397 | polyclonal, 1:300 |
| Antibody | Rabbit anti-NeuN | Millipore | ABN78, Lot: 2702139, RRID:AB_10807945 | polyclonal, 1:400 |
| Antibody | anti-mouse IgG Alexa Fluor 555 | Cell Signaling | Stock #4409, RRID:AB_1904022 | Goat anti-mouse, 1:300 |
| Antibody | anti-rabbit IgG Alexa Fluor 647 | Invitrogen | A21245, Lot: 1445259, RRID:AB_141775 | Goat anti-rabbit, 1:100 |
| Antibody | anti-mouse IgG Alexa Fluor 488 | Cell Signaling | Stock #4408, RRID:AB_10694704 | Goat anti-mouse, 1:300 |
| Antibody | anti-rabbit IgG Alexa Fluor 488 | Cell Signaling | Stock #4412, RRID:AB_1904025 | Goat anti-rabbit, 1:500 |
| Other | neurobiotin tracer | Vector Laboratories | SP-1120, RRID:AB_2336606 | 1.5% |
| Other | Texas Red avidin D | Vector Laboratories | A-2006, RRID:AB_2336751 | 1:100 |
| Software (Sigmaplot 11.2) | RRID:SCR_003210 |
Experimental schedule for the chronic mild stress (CMS) procedure in mice
https://doi.org/10.7554/eLife.32420.020| Week | Monday | Tuesday | Wednesday | Thursday | Friday | Saturday | Sunday |
|---|---|---|---|---|---|---|---|
| 1 | Cold Wet bedding | Restraint No bedding | Light inversion Cage tilt | Cage rotation Strobe | Cold Food and water deprivation | Restraint Overcrowding | Light inversion Wet bedding |
| 2 | Cold Cage tilt | Cage rotation Food and water deprivation | Restraint Wet bedding | Rat bedding Strobe | Light inversion No bedding | Cage rotation Food and water deprivation | Cold Wet bedding |
| 3 | Rat bedding Strobe | Restraint Light inversion | Cage rotation No bedding | Light inversion Food and water deprivation | Cold Wet bedding | Cage tilt Strobe | Light inversion Overcrowding |
| 4 | Cold No bedding | Restraint Food and water deprivation | Cage rotation Strobe | Rat bedding Light inversion | Cold Cage tilt | Restraint Wet bedding | Cage rotation No bedding |
| 5 | Cold Food and water deprivation | Cage rotation Strobe | Light inversion Wet bedding | Cold Cage tilt | Cage rotation No bedding | Light inversion Overcrowding | Restraint Cage tilt |
Additional files
-
Supplementary file 1
Statistical results for Figure 1—figure supplement 1.
Abbreviations: CMS, chronic mild unpredictable stress; EPM, elevated plus maze; FST, forced swimming test; NSF, novelty-suppressed feeding; OFT, open field test; SPT, sucrose preference test
- https://doi.org/10.7554/eLife.32420.021
-
Transparent reporting form
- https://doi.org/10.7554/eLife.32420.022





