Radical and lunatic fringes modulate notch ligands to support mammalian intestinal homeostasis
Figures
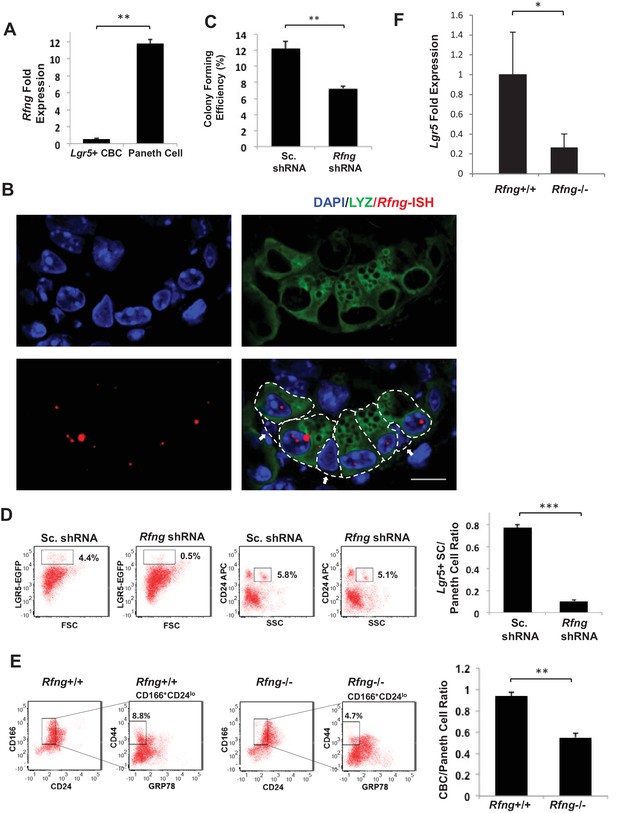
Rfng supports Lgr5+ stem cell self-renewal.
(A) RT-qPCR quantification of Rfng in Lgr5+ CBC and Paneth cells isolated from Lgr5-GFP mouse intestines. The experiment was performed in triplicate and presented as mean ± s.d. (standard deviation) (B) Representative image showing Rfng transcripts (red) and Lysozyme protein (green) expression at the bottom of the crypt of Lgr5-GFP mouse intestine. DAPI (Blue) labels the nuclei and scale bar represents 10 μm. Arrows point to CBCs. (C–D) Single Lgr5-GFP CBCs were transduced with either Sc. shRNA or Rfng shRNA. The experiment was performed in triplicate. (C) Colony forming efficiency measured after 7 days. Quantitative analysis calculated from 1000 cells/replicate presented as mean ± s.d. (D) Left: Representative flow cytometry plots indicating gated percentage of Lgr5+ (GFPhigh) and Paneth cells (CD24high/SSChigh). Right: Ratio of Lgr5-GFP+ CBCs/Paneth cells as determined by flow cytometry and presented as mean ± s.d. (E) Left: Representative plots indicating gated population of CBCs (CD166+CD24loCD44+GRP78-) from the intestine of Rfng+/+ and Rfng-/- mice. Percentage reflects fraction of total population. Right: Ratio of number of CBCs to Paneth cells of n = 3 mice and presented as mean ± s.d. (F) RT-qPCR quantification of Lgr5 in crypts extracted from Rfng+/+ and Rfng-/- mice. n = 3 mice. Data is presented as mean ± s.d. (*p<0.05; **p<0.01; ***p<0.001).
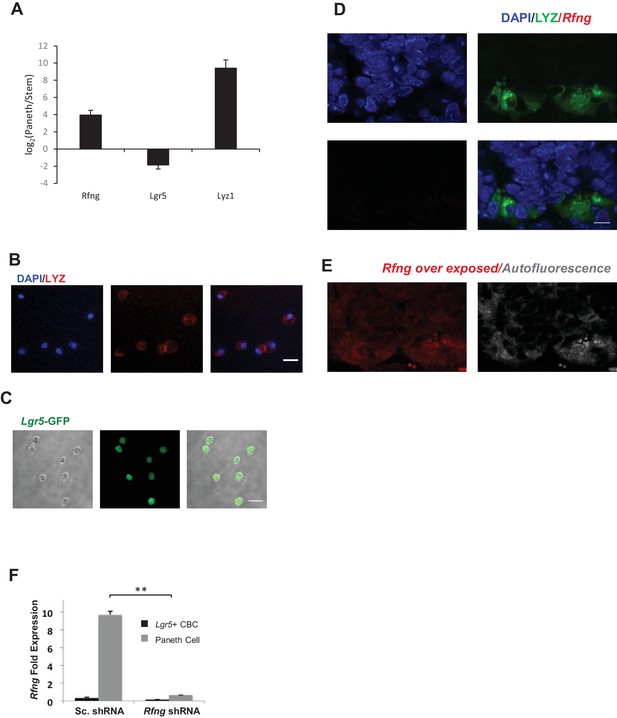
Paneth Cells express Rfng.
(A) Ratio of gene expression in Lgr5+ CBC and Paneth cells from two independent microarrays published previously (Sato et al., 2011). Data presented as mean ± s.d. (B) FACS Sorted Paneth cells were plated in Matrigel and stained for Lysozyme (red). DAPI (Blue) shows the nuclei and scale bar represents 10 μm. (C) Sorted Lgr5-GFP cells were imaged. Endogenous GFP expression is shown in green. Scale bar represents 20 μm. (D–E) Representative image from RNAscope assay for Rfng on Rfng−/− small intestinal section. DAPI (blue) shows nuclei and Lysozyme protein (green) marks the Paneth cells. Scale bar represents 10 μm. (D) No signal is seen in the red channel (Rfng channel) confirming the specificity of Rfng probes. (E) The region was then significantly overexposed to show the background signal. An additional image was taken by over exposing the region in the far-red channel (shown in grey; no probe/antibody present in this channel) to show tissue auto-fluorescence. (F) Lgr5 + CBC and Paneth cell populations were isolated from organoids that were infected with scrambled or Rfng shRNA. The experiment was performed in triplicate. RT-qPCR quantification of Rfng presented as mean ± s.d. in CBC and Paneth cells. (**p<0.01).
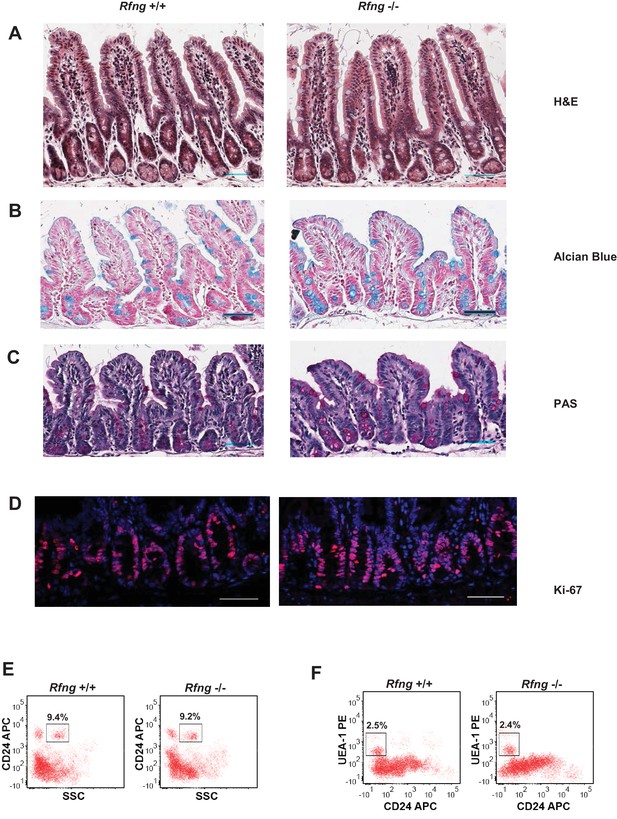
Histological and flow cytometric analysis of Rfng null intestines.
(A–D) Representative images from the small intestine of Rfng+/+ and Rfng-/- mouse strains stained using (A) Haematoxylin and Eosin (H and E) (B) Alcian Blue and Nuclear Fast Red (C) Periodic Acid-Schiff (PAS) and Haematoxylin and (D) Ki-67 (red) and DAPI (blue). Scale bar represents 50 μm. (E–F) Representative flow cytometry plots indicating gated percentage of Paneth cells (E) or goblet cells (F) from the small intestine of Rfng+/+ and Rfng−/− mice.
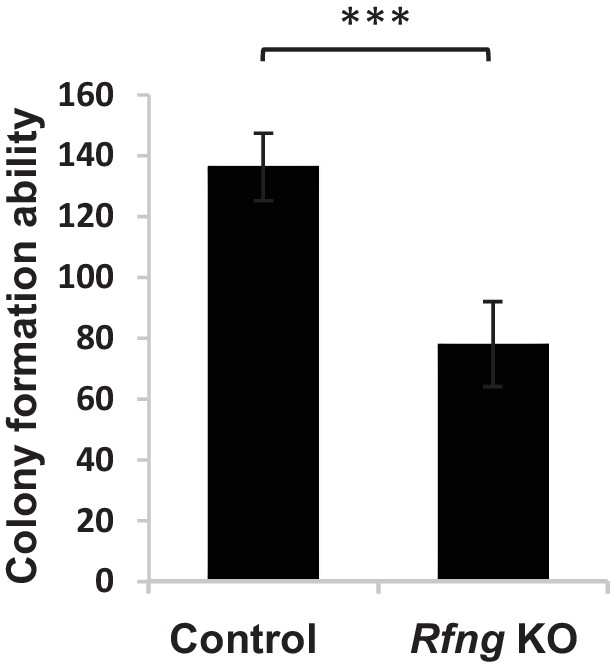
Colony formation ability of Lgr5+ CBCs when mixed with Paneth cells from control or Rfng null mice.
n = 4 replicates with 8000 CBCs per replicate. Data is presented as mean ± s.d. (***p<0.001).
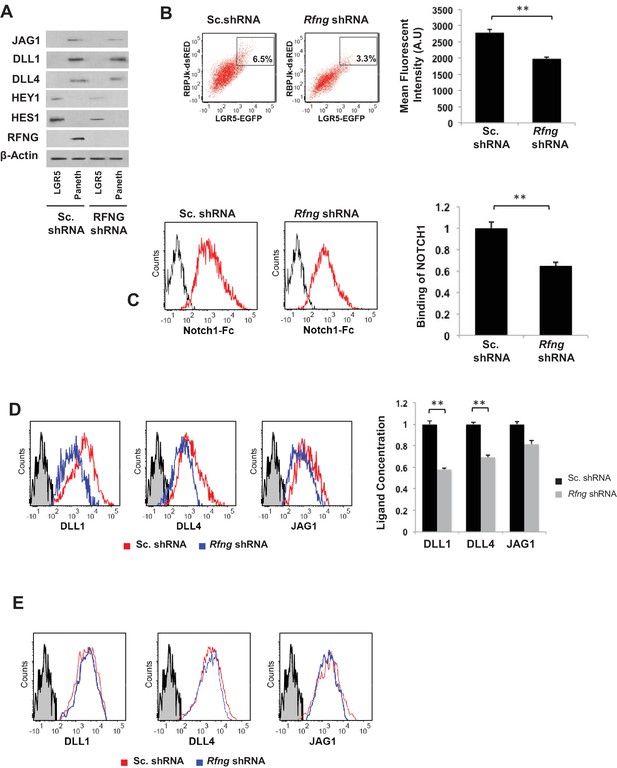
Rfng promotes Notch signaling in Lgr5+ CBC.
(A) Western blot analysis of Notch signaling components in CBCs and Paneth cells FACS sorted from Rfng KD and control organoids. (B) Left: Representative plots for RBPJκ-dsRed and Lgr5-GFP expression indicating a gated double positive fraction for Rfng KD and control CBCs transfected with RBPJκ-dsRed reporter. Right: Mean fluorescence intensity (MFI) of RBPJκ-dsRed expression. The experiment was performed in triplicate and presented as mean ± s.d. (C) Ligand availability on Rfng KD and control Paneth cells. Representative traces (left) and MFI (right) showing ligand binding to NOTCH1 measured by flow cytometry. Unstained Paneth cells were used as a negative control. The experiment was performed in triplicate and presented as mean ± s.d. (D) Cell surface DLL1, DLL4, and JAG1 concentration on Rfng KD and control unpermeabilised Paneth cells. Left: Representative traces measured by flow cytometry. Right: MFI measurements. The experiment was performed in triplicate and presented as mean ± s.d. (E) Cell surface DLL1, DLL4, and JAG1 concentration on Rfng KD and control permeabilised Paneth cells. (**p<0.01).
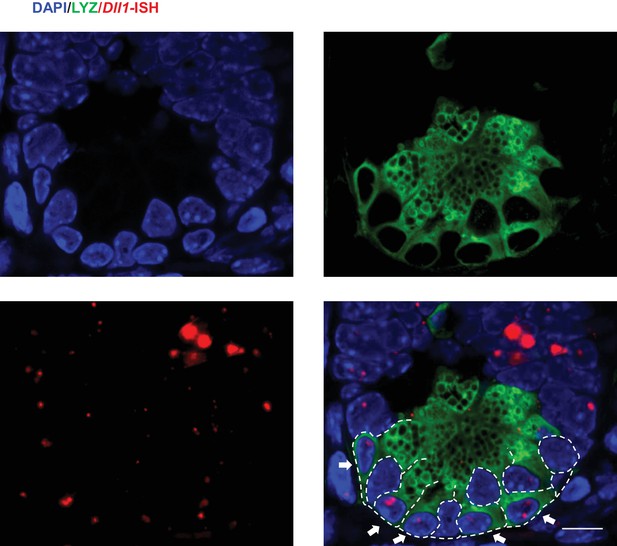
Dll1 expression in the crypts.
Representative image showing Dll1 transcripts (red) and Lysozyme protein (green) expression at the bottom of the crypt of Lgr5-GFP mouse intestine.DAPI (Blue) labels the nuclei and scale bar represents 10 μm. Arrows point to Paneth cells.
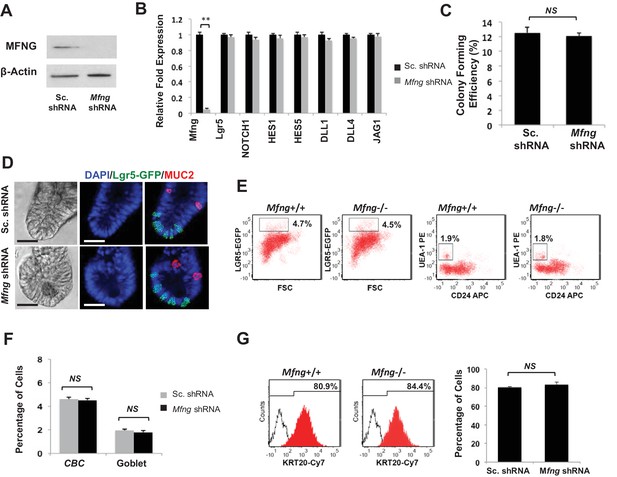
Mfng plays an insignificant role.
Single Lgr5-GFP CBCs were transduced with either Sc. shRNA or Mfng shRNA. The experiment was performed in triplicate. (A) Western blot for Mfng expression. (B) RT-qPCR quantification of Mfng and Notch components in organoids. (C) Colony forming efficiency measured after 7 days. Quantitative analysis from 1000 cells/replicate. (D) Representative bright field and co-IF images indicating Lgr5-GFP (green) expression. MUC2 (red) marks Goblet cells. DAPI (blue) labels nuclei and scale bar represents 25 μm. (E) Representative flow cytometry plots indicating gated percentage of Lgr5+ CBCs (GFPhigh) and goblet cells (UEA-1+/CD24-). (F) Percentage of Lgr5+ CBCs and goblet cells as determined by flow cytometry and presented as mean ± s.d. (G) Left: Representative flow cytometry histograms indicating KRT20+ (CK20+) cells. Right: Percentage of KRT20+ cells and presented as mean ± s.d.
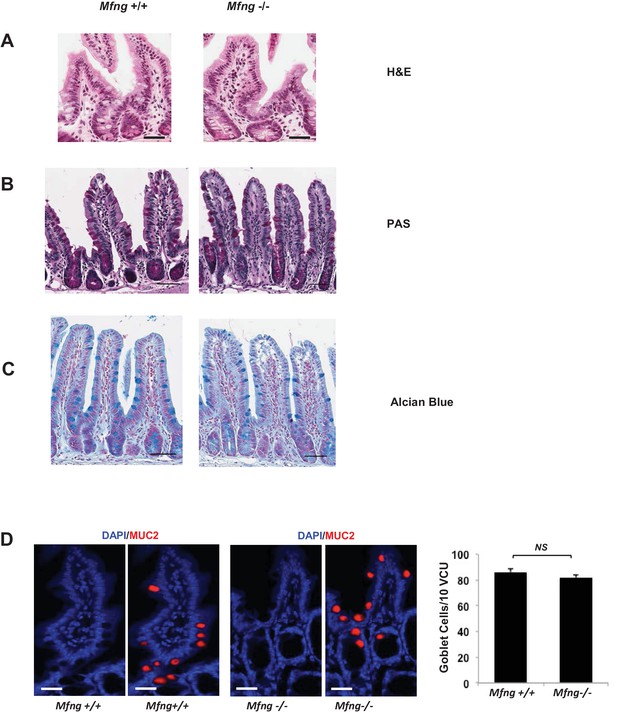
Histological analysis of Mfng null intestines.
(A–C) Representative images from the small intestine of Mfng+/+ and Mfng-/- mouse strains stained using (A) Haematoxylin and Eosin (H and E) (B) Periodic Acid Schiff (PAS) and Haematoxylin (C) Alcian Blue and Nuclear Fast Red. Scale bar represents 50 μm. (D) Left: Representative IF images of villus-crypt units (VCU) from the small intestine of Mfng+/+ and Mfng-/- mouse strains. MUC2 (red) marks goblet cells. DAPI (blue) labels nuclei. Right: Quantification of the number of goblet cells of n = 4 mice/condition. Data represents mean ± s.d of n = 500 VCU per mouse. (NS (not significant)).
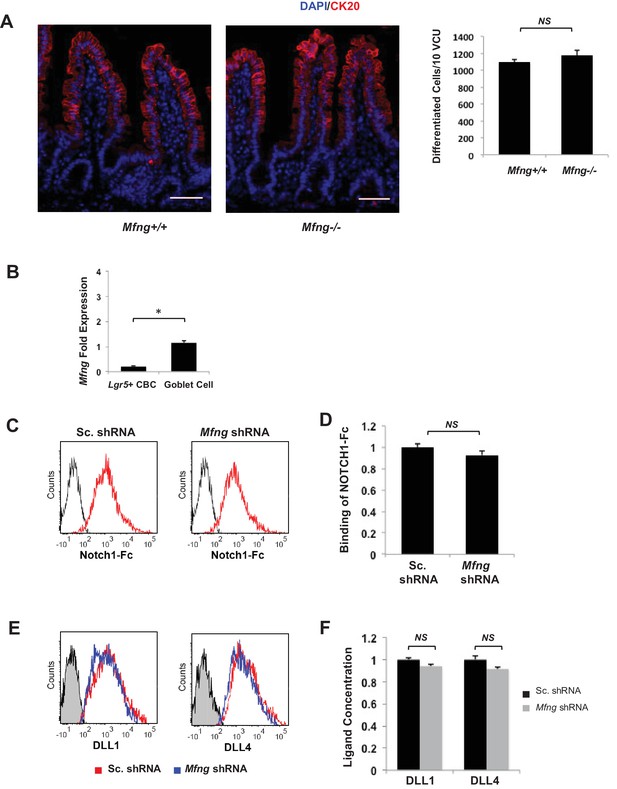
No significant phenotype detected upon loss of Mfng.
(A) Left: Representative IF images of villus-crypt units (VCU) from the small intestine of Mfng+/+ and Mfng-/- mouse strains. CK20 (red); DAPI (blue) labels nuclei; scale bar represents 50 μm. Right: Quantification of CK20+ differentiated cells in VCU of Mfng+/+ and Mfng-/- mice based on IF. The data represents 500 VCU/mouse of n = 4 mice/condition and is presented as mean ± s.d. (NS) (not significant); Student t-test). (B) RT-qPCR quantification of Mfng in Lgr5+ CBC and goblet cell populations from small intestinal tissue of Lgr5-GFP mice. The experiment was performed in triplicate and presented mean ± s.d. (C) Single Lgr5-GFP CBCs were transduced with either Sc. shRNA or Mfng shRNA and propagated as organoids for seven days. Isolated goblet cells were incubated with 0.5 μg/ml NOTCH1-Fc. Shown are representative traces indicating ligand binding to Notch1 measured by flow cytometry in goblet cells. Unstained goblet cells were used as a negative control. (D) Ligand binding data to NOTCH1-Fc in Mfng shRNA-expressing goblet cells normalized to Sc. shRNA-transfected goblet cells using flow cytometry mean fluorescent intensity (MFI) measurements. The experiment was performed in triplicate and presented mean ± s.d. (NS). (E) Single Lgr5-GFP CBCs were transduced with either Sc. shRNA or Mfng shRNA and propagated as organoids for 7 days. Isolated goblet cells were incubated with antibodies directed against DLL1 and DLL4 ligands. Shown are representative traces showing surface ligand concentration measured by flow cytometry on goblet cells. Unstained goblet cells were used as a negative control. (F) Surface DLL1 and DLL4 ligand concentration on Mfng shRNA-expressing goblet cells normalized to Sc. shRNA-transfected goblet cells using flow cytometry mean fluorescent intensity (MFI) measurements. The experiment was performed in triplicate and presented mean ± s.d. (NS). (*p<0.05; **p<0.01).
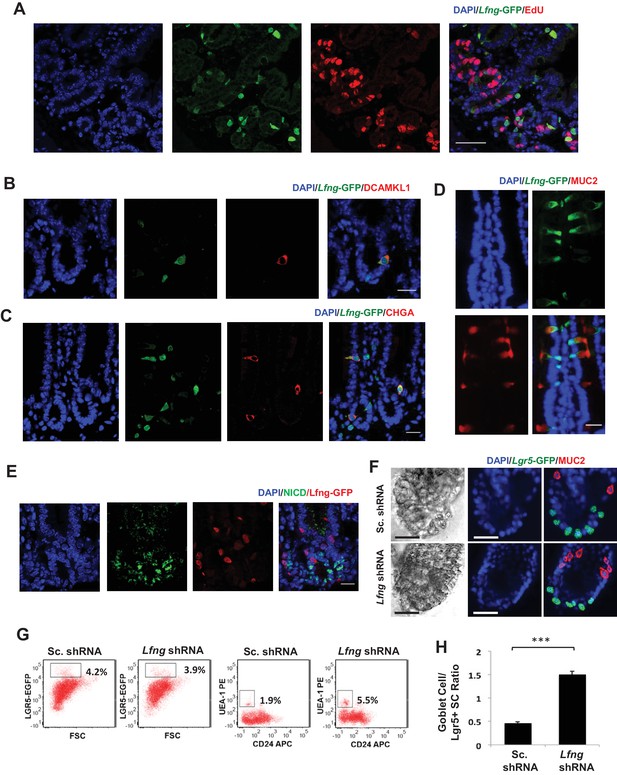
Lfng loss results in increased goblet cell differentiation in vitro.
(A–E) Representative IF images of the small intestine of Lfng-GFP reporter mice. (A) GFP (green) shows the Lfng expression and EdU (red) marks the proliferating cells. DAPI (blue) labels nuclei. Scale bar represents 50 μm. (B) GFP (green) shows the Lfng expression and DCAMKL1 (red) marks the Tuft cells. Scale bar represents 20 μm. (C) GFP (green) shows the Lfng expression and CHGA (red) marks the enteroendocrine cells. Scale bar represents 20 μm. (D) GFP (green) shows the Lfng expression and MUC2 (red) marks the goblet cells. Scale bar represents 20 μm. (E) GFP (red) shows the Lfng expression and NICD (green) identifies the cells with NOTCH1 activity. Scale bar represents 20 μm. (F) Representative bright field and co-IF images of Lfng KD and control organoids indicating Lgr5-GFP (green) expression. MUC2 (red) marks goblet cells. DAPI (blue) labels nuclei and scale bar represents 25 μm. (G) Representative plots indicating gated percentage of Lgr5+ (GFPhigh) and goblet cells (UEA-1+/CD24-) of Lfng KD and control organoids. (H) Ratio of goblet cells to Lgr5-GFP + CBCs as determined by flow cytometry. The experiment was performed in triplicate and presented mean ± s.d. (***p<0.001).
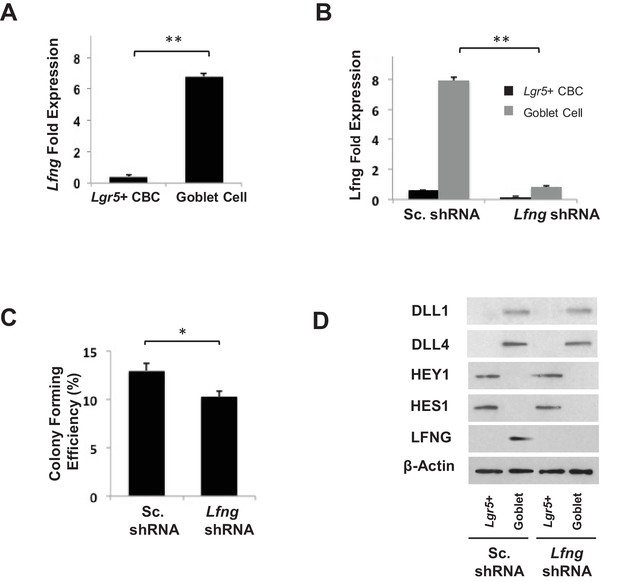
Characterisation of Lfng KD organoids.
(A) RT-qPCR quantification of Rfng in Lgr5+ CBC and goblet cell populations from Lgr5-GFP murine intestinal tissue. The experiment was performed in triplicate and presented mean ± s.d. (B) RT-qPCR quantification of Lfng in Lgr5+ CBC and goblet cell populations after organoids were infected with Scrambled shRNA (Sc. shRNA) or Lfng shRNA. The experiment was performed in triplicate and presented mean ± s.d. (C) Single Lgr5-GFP CBCs were transduced with either Sc. shRNA or Lfng shRNA. Shown is colony forming efficiency measured after seven days. Quantitative analysis calculated from 1000 cells/replicate. The experiment was performed in triplicate and presented mean ± s.d. (D) Single Lgr5-GFP CBCs were transduced with either Sc. shRNA or Lfng shRNA and propagated as organoids for seven days. Shown is Western blot analysis for LFNG and Notch signaling components. Actin was used as a loading control. (*p<0.01; **p<0.01).
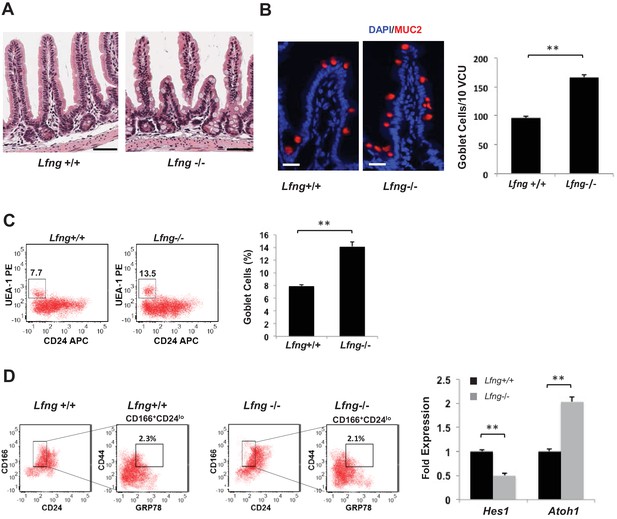
Lfng loss results in increased goblet cell differentiation in vivo.
(A) Representative H and E sections from the small intestine of Lfng+/+ and Lfng−/− mice. Scale bar represents 50 μm. (B) Left: Representative IF images of intestine of Lfng+/+ and Lfng−/− mice. MUC2 (red) marks goblet cells. DAPI (blue) labels nuclei. Right: Quantification of the number of goblet cells of n = 4 mice/condition and n = 500 VCU per mouse presented as mean ± s.d. (C) Left: Representative plots indicating gated percentage of goblet cells (UEA-1+/CD24-) from intestinal tissue derived from Lfng+/+ or Lfng−/− mice. Right: Percentage of goblet cells presented as mean ± s.d. The data represent n = 3 mice/condition. (D) Left: Representative plots indicating gated population of intestinal progenitors from the intestine of Lfng+/+ and Lfng−/− mice. Percentage reflects fraction of total population. Right: RT-qPCR measurements in progenitor cells from Lfng+/+ and Lfng−/− mice. The experiment was performed in triplicate presented as mean ± s.d. (**p<0.01).
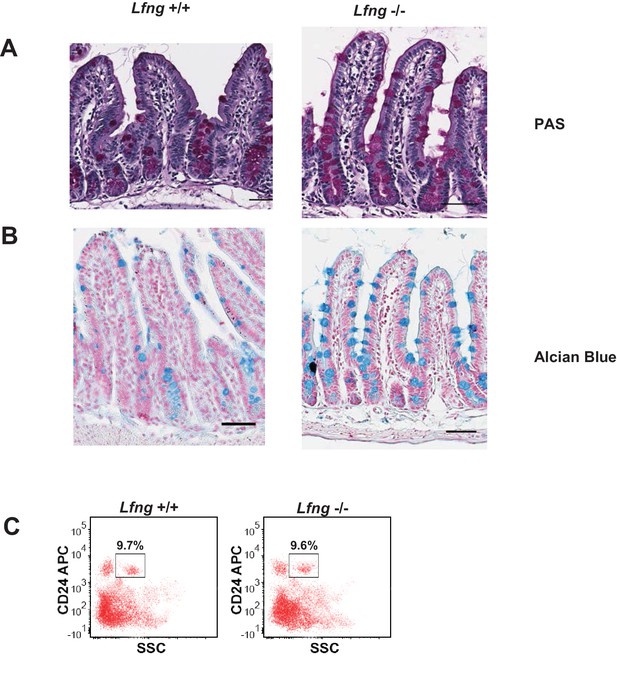
Histological and flow cytometric analysis of Lfng null intestines.
(A-B)Representative images from the small intestine of Lfng+/+ and Lfng-/- mouse strains stained using (A) PAS and Haematoxylin (B) Alcian Blue and Nuclear Fast Red.
Scale bar represents 50 μm. (C) Representative flow cytometry plots indicating gated percentage of Paneth cells from the small intestine of Lfng+/+ and Lfng-/- mice.
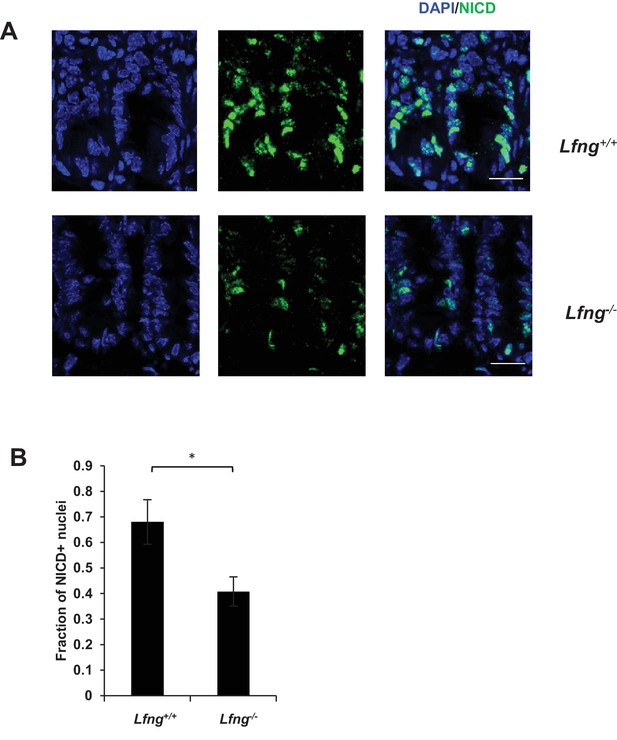
Lfng loss results in reduced Notch activity.
(A) Representative images from the small intestine of Lfng+/+ and Lfng-/- mouse strains stained for NICD. Scale bar represents 20 μm. (B) Fraction of NICD + nuclei per crypt quantified from small intestines of Lfng+/+ and Lfng-/- mice presented as mean ± s.d. Data represents n = 3 mice/condition. (*, p<0.05).
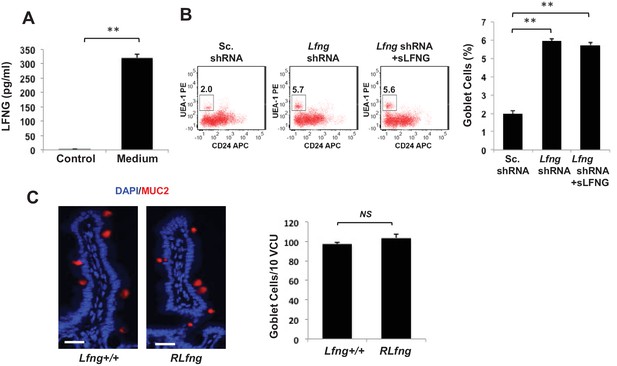
Secreted LFNG plays no apparent function.
(A) ELISA of the secretion of LFNG in culture medium from Lgr5-GFP organoids. Culture medium (T = 0 days) was used as a control. The experiment was performed in triplicate and presented as mean ± s.d. (B) Left: Representative plots indicating gated percentage of goblet cells (UEA-1+/CD24-) for organoids under Sc. shRNA control, Lfng KD and Lfng KD treated with sLFNG conditions. Right: Percentage of goblet cells in each condition. The experiment was performed in triplicate and presented as mean ± s.d. (C) Left: Representative IF images of intestine of Lfng+/+ and LfngRLfng/+ mice. MUC2 (red) marks goblet cells. DAPI (blue) labels nuclei. Right: Quantification of the number of goblet cells of n = 4 mice/condition and n = 500 VCU/mouse. Data presented as mean ± s.d. (**p<0.01).
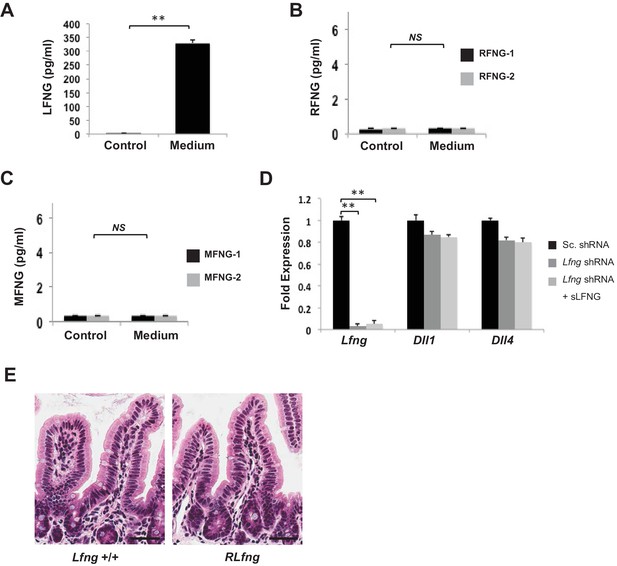
Secretion of Fringe proteins.
(A) ELISA of the secretion of LFNG in culture medium from murine Lgr5-GFP+ CBCs propagated as organoids for 7 days using an additional primary LFNG antibody (LFNG-2). Culture medium (T = 0 days) was used as a control. The experiment was performed in triplicate and presented mean ± s.d. (B) ELISA of the secretion of RFNG in culture medium from murine Lgr5-GFP+ CBCs propagated as organoids for 7 days using two separate primary RFNG antibodies (RFNG-1, RFNG-2). Culture medium (T = 0 days) was used as a control. The experiment was performed in triplicate and presented mean ± s.d. (C) ELISA of the secretion of MFNG in culture medium from murine Lgr5-GFP+ CBCs propagated as organoids for 7 days using two separate primary MFNG antibodies (MFNG-1, MFNG-2). Culture medium (T = 0 days) was used as a control. The experiment was performed in triplicate and presented mean ± s.d. (NS). (D) RT-PCR quantification of Lfng and Notch ligands (DLL1 and DLL4) in Lgr5-GFP+ organoids that were infected with Scrambled shRNA (Sc. shRNA) or Lfng shRNA. Lfng shRNA-expressing CBC organoids were subsequently incubated in conditioned medium containing soluble LFNG (sLFNG) for 24 hr. The experiment was performed in triplicate and presented mean ± s.d. (E) Representative H and E sections from the small intestine of Lfng+/+and RLfng mice. Scale bar represents 50 μm. (**p<0.01).
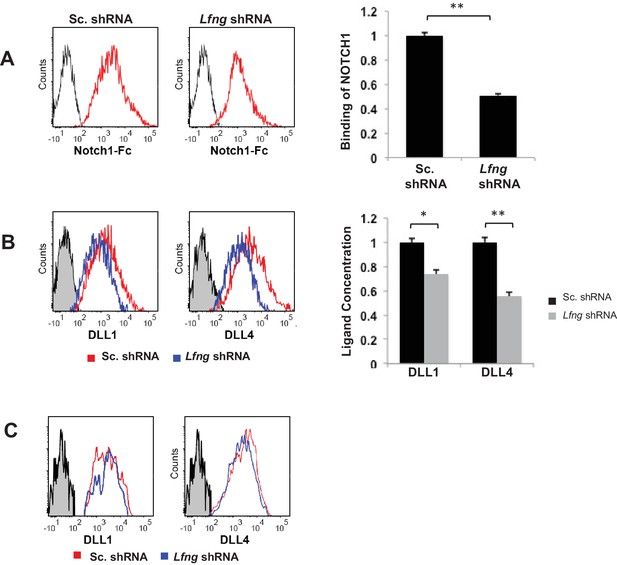
LFNG promotes cell surface expression of DLL.
(A) Ligand availability on Lfng KD and Sc. Control goblet cells. Representative traces (left) and MFI (right) showing ligand binding to NOTCH1 measured by flow cytometry. Unstained goblet cells were used as a negative control. The experiment was performed in triplicate and presented as mean ± s.d. (D) Cell surface DLL1 and DLL4 concentration on Lfng KD and Sc. Control unpermeabilised goblet cells. Left: Representative traces measured by flow cytometry. Right: MFI measurements. The experiment was performed in triplicate and presented as mean ± s.d. (E) Cell surface DLL1 and DLL4 concentration on Lfng KD and Sc. Control permeabilised goblet cells. (**p<0.01).

Summary.
Rfng is enriched in the Paneth cells and promotes cell surface expression of DLL1 and DLL4. This promotes Notch activity in the neighbouring Lgr5+ CBCs assisting their self-renewal. Mfng does not appear to contribute significantly in maintaining the epithelium. Lfng on the other hand is expressed by enteroendocrine, Tuft, and goblet cells and suppresses the secretory lineage.
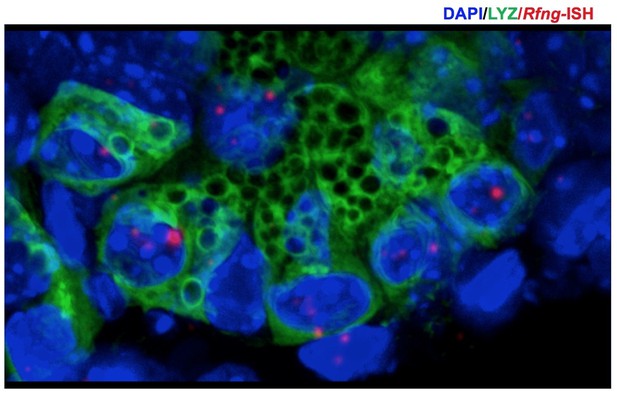
3D projection (from z-stack spanning 9μm) was used to correctly identify the Paneth cells in Figure 1B.
Rfng transcripts (red) and Lysozyme protein (green) expression can be seen at the bottom of the crypts. Dapi (Blue) labels the nuclei.
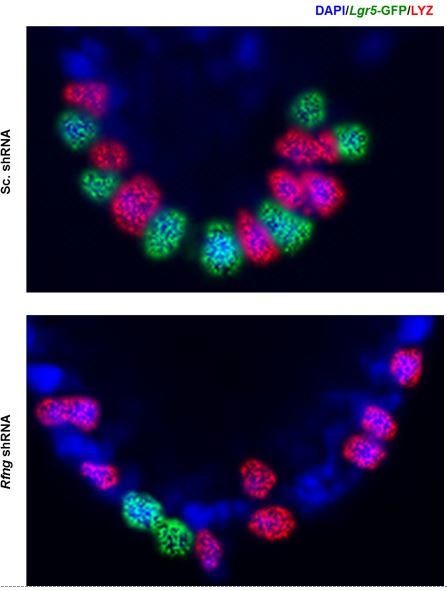
Lysozyme (red) and GFP (green) staining in Paneth cells and CBCs respectively are cytoplasmic as expected.
Figure 1D (previously Figure 1C) has been enlarged and brightness and contrast have been increased.
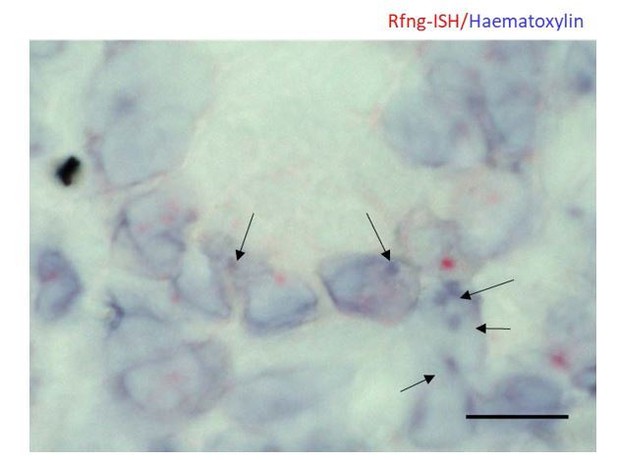
Representative image of RNAscope ISH for Rfng counterstained using haematoxylin on Lgr5-GFP mouse intestine.
Arrows indicate regions with strong haematoxylin staining. Note that they do not overlap with the ISH signal. Scale bar represents 10μm.

Representative image of in situ HCR for Rfng on Lgr5-GFP mouse intestines shows signal primarily in the nuclei.
Arrows point to nuclei with HCR signal. Dotted line separates the epithelium from the mesenchyme. Scale bar represents 10μm. Assay was performed as per manufacturer’s recommendation (Choi, Beck et al., 2014, Shah, Lubeck et al., 2016). V3.0 Probes for Rfng were purchased from Molecular Instruments. Tissues were pretreated using RNAscope reagents. Probes were hybridised to the section overnight at 400C, washed using 5x SSCT buffer (5x SSC with 0.1% Tween-20) and signal was amplified overnight at 400C. Slides were washed in SSCT, mounted and imaged using a confocal microscope. Spectral imaging and linear unmixing using Zen (Zeiss) software was performed to detect Rfng signal.
Additional files
-
Supplementary file 1
Supplementary Methods.
List of antibodies used for Immunofluorescence and Western blotting.
- https://doi.org/10.7554/eLife.35710.020
-
Transparent reporting form
- https://doi.org/10.7554/eLife.35710.021





