Sex-dependent effects of in utero cannabinoid exposure on cortical function
Figures
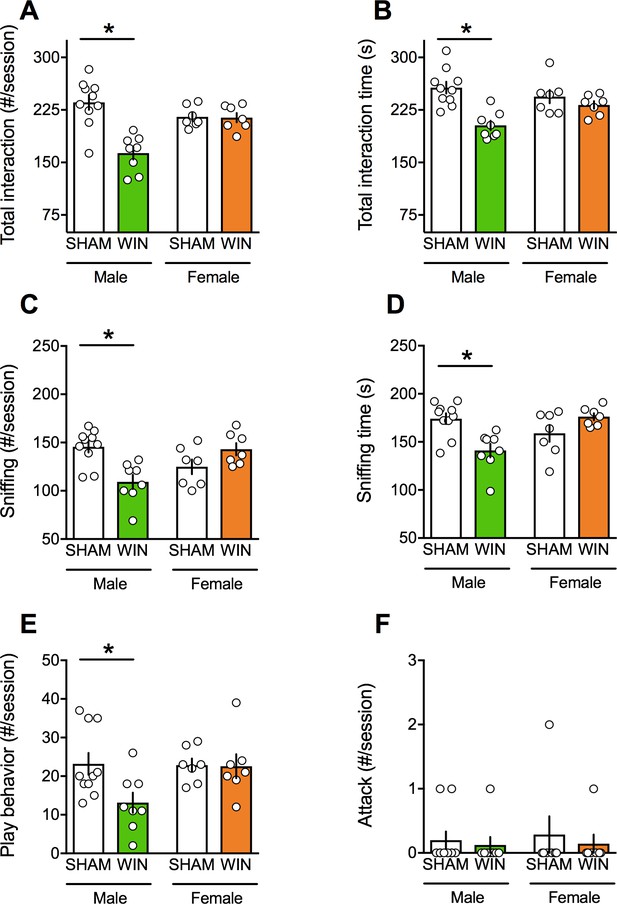
Prenatal cannabinoid exposure induces sex-specific social deficits at adulthood.
(A–B) Adult male progeny from dams exposed during gestation to WIN had less contact (A): F(sex x treat)1,28=15.54, p<0.05, two-way ANOVA) and spent less time interacting with their congeners than SHAM-animals (B): F(sex x treat)1,28=7.09, p<0.05, two-way ANOVA). In contrast, the social behavior of female littermates was normal. During the social session, the number (C): F(sex x treat)1,28=16.30, p<0.05, two-way ANOVA) and the time (D): F(sex x treat)1,28=14.87, p<0.05, two-way ANOVA) of sniffing events and the frequency of play behavior (E): F(sex x treat)1,28=3.217, p>0.05, two-way ANOVA) were exclusively reduced in male rats exposed to WIN during gestation. Prenatal cannabinoid exposure did not induce aggressive behavior in adult rats (F): F(sex x treat)1,28=0.037, p>0.05, two-way ANOVA). Scatter dot plot represents a pair of animals. Error bars indicate SEM. *p<0.05. Student–Newman–Keuls test.
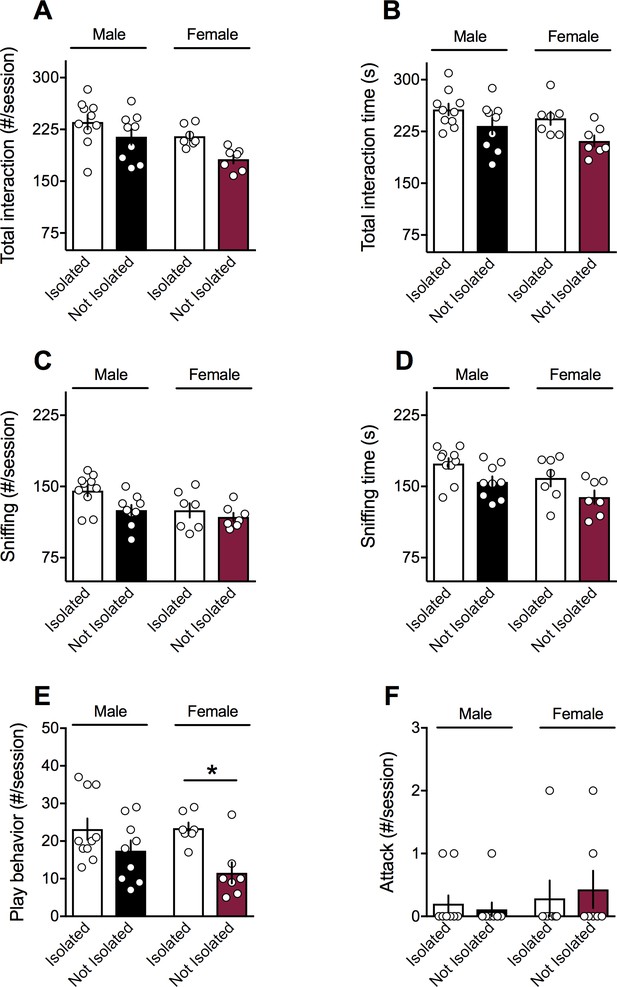
Social isolation before testing did not induce sex-specific social changes in adult animals.
(A-B) Adult male and female naïve animals not isolated 24 hr before testing showed no difference in contact (A): F(sex x isolation)1,29=0.203, p>0.05, two-way ANOVA) and time to interact with their congeners than male and female SHAM-animals isolated before testing (B): F(sex x isolation)1,29=0.372, p>0.05, two-way ANOVA). During the social session, the number (C): F(sex x isolation)1,29=0.003, p>0.05, two-way ANOVA) and the time (D): F(sex x isolation)1,28=1.253, p>0.05, two-way ANOVA) of sniffing events were unchanged; the frequency of play behavior (E): F(sex x isolation)1,29=1.289, p>0.05, two-way ANOVA) were exclusively reduced in female naïve rats not isolated before testing. Social isolation did not change aggressive behavior in adult naïve rats (F): F(sex x isolation)1,29=0.325, p>0.05, two-way ANOVA). Scatter dot plot represents a pair of animals. Error bars indicate SEM. *p<0.05. Student–Newman–Keuls test.
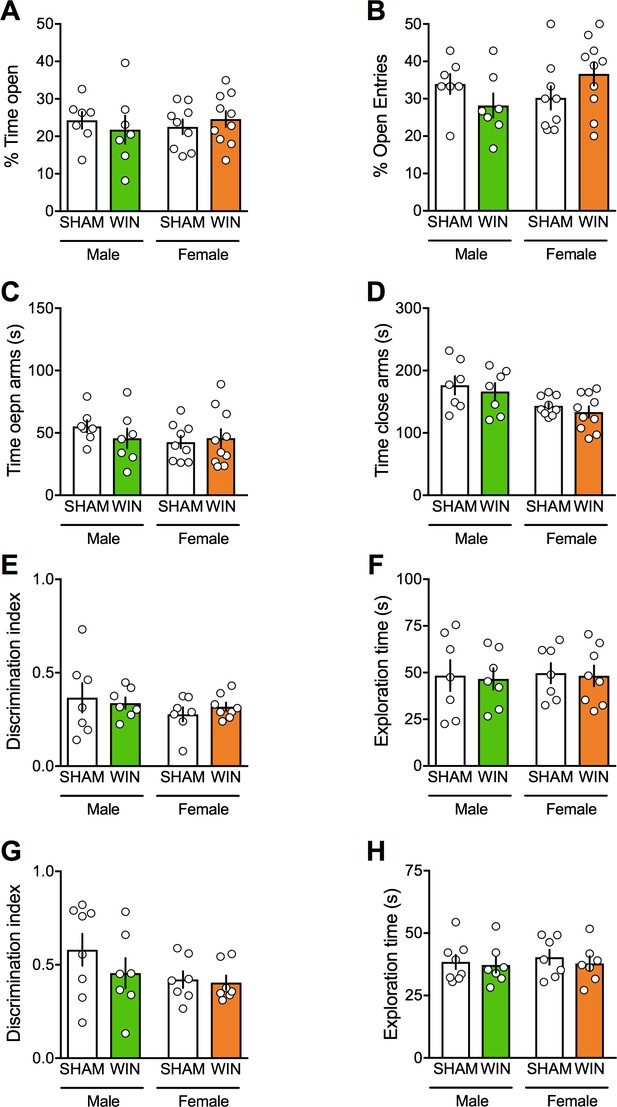
Prenatal cannabinoid exposure does not modify adult cognitive and anxious behaviors in both sexes.
(A–B) Prenatal cannabinoid exposure did not alter anxiety behavior in adult rats of either sex. No differences between WIN-exposed and SHAM animals were found in the elevated plus maze test as expressed in the percentage of time spent in the open arms (A): F(treat x sex)1,29=0.818, p>0.05, two-way ANOVA), the percentage of open arm entries (B): F(treat x sex)1,29=3.762, p>0.05, two-way ANOVA), the time spent exploring the open (C): F(treat x sex)1,29=0.04, p>0.05, two-way ANOVA) and the closed arm (D): F(treat x sex)1,29=0.944, p>0.05, two-way ANOVA). (E–F), Prenatal cannabinoid exposure did not impair cognitive performance. (E–F), In the temporal order memory task, no differences were found in the discrimination index (E): F(treat x sex)1,25=0.549, p>0.05, two-way ANOVA) and in the total time exploring objects during the test trial (F): F(treat x sex)1,25=0.001, p>0.05, two-way ANOVA). (G–H) In the object recognition test, male and female rats exposed to the cannabinoid WIN in-utero showed no differences in their discrimination index (G): F(treat x sex)1,25=0.639, p>0.05, two-way ANOVA) or the time exploring objects (H): F(treat x sex)1,25=0.04, p>0.05, two-way ANOVA) from the SHAM group, suggesting no deficits in the recognition memory for objects. Scatter dot plot represents each animal. Error bars indicate SEM. *p<0.05. Student–Newman–Keuls test.
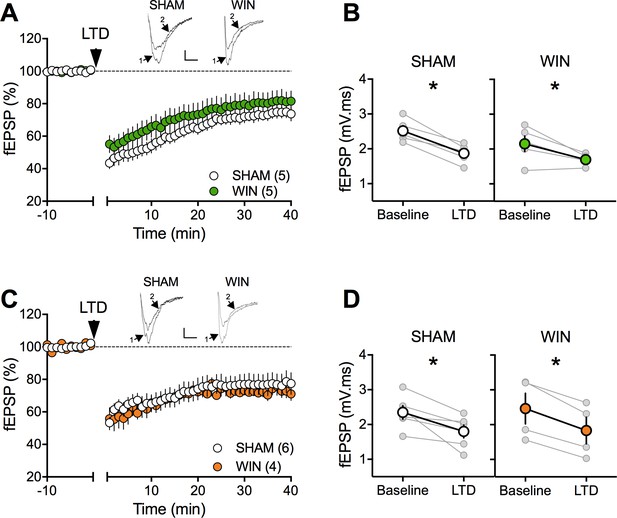
Prenatal cannabinoid exposure does not modify LTD in NAc of adult rats.
(A) Average time-courses of mean fEPSPs showing that low-frequency stimulation (indicated by arrow) induces LTD at accumbens synapses in both SHAM- (white circles, n = 5) and WIN- (green circles, n = 5) exposed male rats. Above: example traces, baseline (1) and 40 min poststimulation (2). (B) Individual experiments (grey) and group average (tan), before (baseline) and after (40 min) LTD induction showing that PCE does not affect eCB-LTD in male accumbens. In SHAM male rats: 2.518 ± 0.145 mV.ms before LTD versus 1.872 ± 0.124 mV.ms after LTD induction (n = 5, p<0.05, paired t-test). In WIN male: 2.152 ± 0.224 mV.ms before LTD versus 1.696 ± 0.073 mV.ms after LTD induction (n = 5, p<0.05, paired t-test). (C) Average time-courses of mean EPSPs showing similar low-frequency LTD in SHAM (n = 6; white circles) and WIN (n = 4; orange circles) in utero-exposed female rats. Above: example traces, baseline (1) and 40 min poststimulation (2). (D) Individual experiments (grey) and group average (white represents SHAM; orange represents WIN), before (baseline) and after (40 min) LTD induction showing that prenatal WIN exposure does not alter LTD in the female exposed group. In SHAM female: 2.350 ± 0.191 mV.ms before LTD versus 1.800 ± 0.182 mV.ms after LTD induction (n = 6, p<0.05, paired t-test). In WIN female: 2.461 ± 0.439 mV.ms before LTD versus 1.833 ± 0.381 mV.ms after LTD induction (n = 4, p<0.05, paired t-test). n = individual rats. Error bars indicate SEM. Scale bar: 10 ms, 0.1 mV.
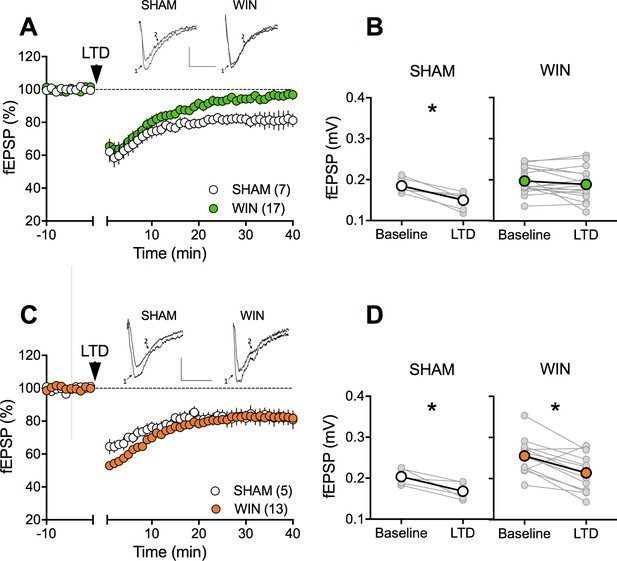
Sexual divergent ablation of LTD in the PFC of adult rats prenatally exposed to WIN.
(A) Average time-courses of mea field EPSPs showing that low-frequency stimulation (10 min at 10 Hz, arrow) induced LTD of evoked fEPSPs recorded in the mPFC in SHAM (n = 7; white circles), but not in WIN (n = 17; green circles) prenatally-exposed male rats. Above: example traces, baseline (1) and 40 min post-stimulation (2). (B) Individual experiments (grey) and group average (white represents SHAM; green represents WIN), before (baseline) and after (40 min) LTD induction showing the lack of LTD in the WIN exposed group. In SHAM rats: 0.185 ± 0.006 mV before LTD versus 0.150 ± 0.007 mV after LTD induction (n = 7, p<0.05, paired t-test). In WIN animals: 0.197 ± 0.008 mV before LTD versus 0.189 ± 0.009 mV after LTD induction (n = 17, p>0.05, paired t-test). (C) Average time-courses of mean EPSPs showing similar low-frequency LTD in SHAM (n = 5; white circles) and WIN (n = 13; orange circles) in utero-exposed female rats. Above: example traces, baseline (1) and 40 min post-stimulation (2). (D) Individual experiments (grey) and group average (white represents SHAM; orange represents WIN), before (baseline) and after (40 min) LTD induction showing that prenatal WIN exposure does not alter LTD in the female exposed group. In SHAM female rats: 0.204 ± 0.009 mV before LTD versus 0.168 ± 0.010 mV after LTD induction (n = 5, p<0.05, paired t-test). In WIN female rats: 0.255 ± 0.012 mV before LTD versus 0.214 ± 0.013 mV after LTD induction (n = 13, p<0.05, paired t-test). *p<0.05. n = individual rats. Error bars indicate SEM. Scale bar: 10 ms, 0.1 mV.
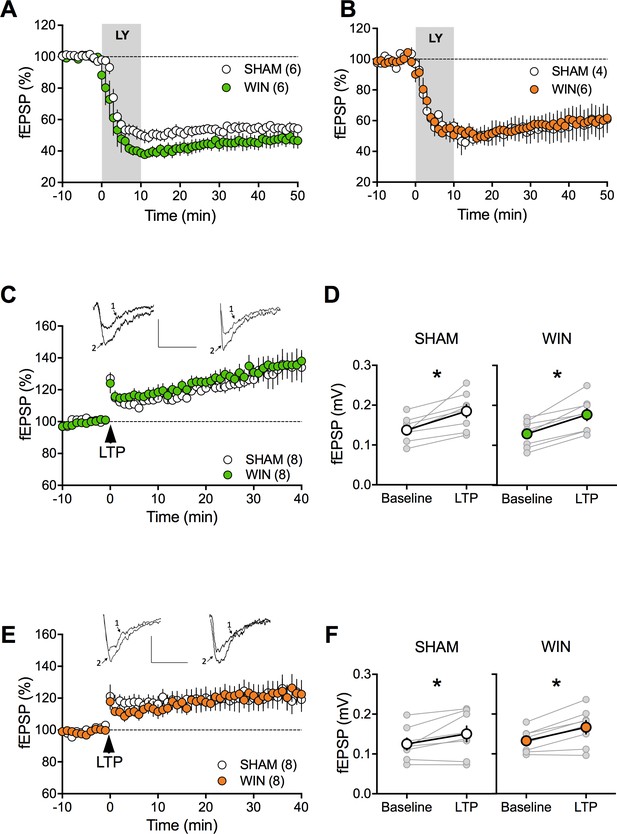
mGlu2/3-LTD and long-term potentiation are spared by prenatal WIN exposure.
(A, B) Prenatal exposure to cannabinoids does not alter mGlu2/3-LTD. Average time of mean fEPSPs showing that mGlu2/3-agonist induced LTD (bath application of LY379268, 30 nM, 10 min) is identical in SHAM and WIN exposed male (A): SHAM, n = 6, white circles; WIN, n = 6, green circles) and female (B): SHAM, n = 4, white circles; WIN, n = 6, orange circles). (C) Average time-courses of mean fEPSPs showing that theta burst stimulation (indicated by arrow) induces LTP at layer V/VI mPFC synapses in both SHAM- (white circles, n = 8) and WIN- (green circles, n = 8) exposed male rats. (D) Individual averaged fEPSP amplitude measurements before (baseline) and 40 min after LTP induction. Grey, individual experiments; tan, group averages. In SHAM rats: 0.138 ± 0.011 mV before LTP versus 0.185 ± 0.016 mV after LTP induction (n = 8, p<0.05, paired t-test). In WIN male rats: 0.129 ± 0.012 mV before LTP versus 0.177 ± 0.015 mV after LTP induction (n = 13, p<0.05, paired t-test). (E) Identical TBS-LTP induction protocol but recording from female prenatally-exposed animals (SHAM, white circles, n = 8; WIN, orange circles, n = 8). (F) Peak amplitude measurements before (baseline) and after (LTP) stimulation protocol from individual experiments (grey) and group averages in SHAM (white; 0.125 ± 0.014 mV before LTP versus 0.150 ± 0.020 mV after LTP; p<0.05, paired t-test) and WIN (orange; 0.133 ± 0.010 mV before LTP versus 0.167 ± 0.016 mV after LTP; p<0.05, paired t-test) animals. *p<0.05. n = individual rats. Error bars represent SEM.
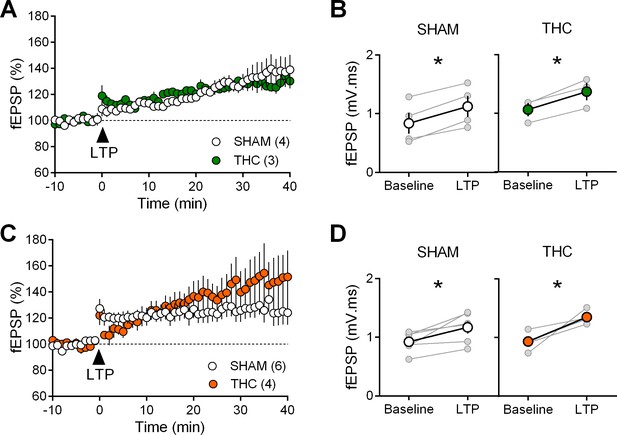
Prenatal THC does not alter long-term potentiation.
(A) Average time-courses of mean fEPSPs showing that theta burst stimulation (indicated by arrow) induces LTP at layer V/VI mPFC synapses in both SHAM- (white circles, n = 8) and THC- (green circles, n = 8) exposed male rats. (B) Individual averaged fEPSP amplitude measurements before (baseline) and 40 min after LTP induction. Grey, individual experiments; tan, group averages. In SHAM rats: 0.138 ± 0.011 mV before LTP versus 0.185 ± 0.016 mV after LTP induction (n = 8, p<0.05, paired t-test). In THC male rats: 0.129 ± 0.012 mV before LTP versus 0.177 ± 0.015 mV after LTP induction (n = 13, p<0.05, paired t-test). (C) Identical TBS-LTP induction protocol but recording from female prenatally-exposed animals (SHAM, white circles, n = 8; THC, orange circles, n = 8). (D) Peak amplitude measurements before (baseline) and after (LTP) stimulation protocol from individual experiments (grey) and group averages in SHAM (white, 0.125 ± 0.014 mV before LTP versus 0.150 ± 0.020 mV after LTP; p<0.05, paired t-test) and THC (orange; 0.133 ± 0.010 mV before LTP versus 0.167 ± 0.016 mV after. LTP; p<0.05, paired t-test) animals. *p<0.05. n = individual rats. Error bars represent SEM.
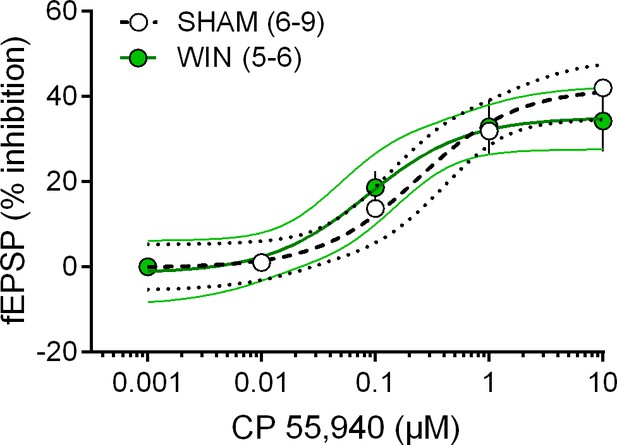
Prenatal cannabinoid exposure does not modify synaptic CB1R potency or efficiency.
Dose-response curve for the cannabimimetic CP55,940 in SHAM (white symbols, n = 6–9 animals, EC50 = 0.240 μM, top value 42.01%, 95% CI for EC50 = 0.088–0.658) and WIN-exposed (green symbols, n = 5–6, EC50 = 0.085 μM, top value 35.09%, 95% CI for EC50 = 0.025–0.292) male rats. fEPSP amplitudes were measured 30 min after application of CP55,940. Each point is expressed as the percentage of inhibition of its basal value. Error bars indicate SEM.
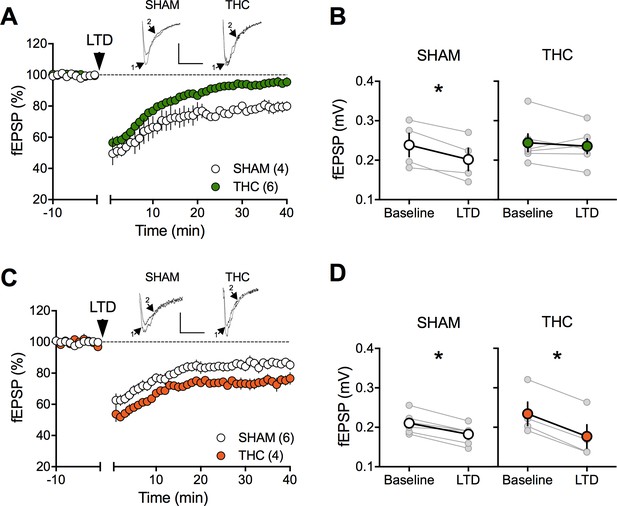
Sexual divergent ablation of LTD in the PFC of adult rats prenatally exposed to THC.
(A) Average time-courses of men field EPSPs showing that low-frequency stimulation (10 min at 10 Hz, arrow) induced LTD of evoked fEPSPs recorded in the mPFC in SHAM (n = 4; white circles), but not in THC (n = 6; green circles) prenatally-exposed male rats. Above: example traces, baseline (1) and 40-min poststimulation (2). (B) Individual experiments (grey) and group average (white represents SHAM; green represents THC), before (baseline) and after (40 min) LTD induction showing the lack of LTD in the THC exposed group. In SHAM rats: 0.239 ± 0.030 mV before LTD versus 0.202 ± 0.028 mV after LTD induction (n = 4, p<0.05, paired t-test). In THC animals: 0.244 ± 0.023 mV before LTD versus 0.236 ± 0.019 mV after LTD induction (n = 6, p>0.05, paired t-test). (C) Average time-courses of mean EPSPs showing similar low-frequency LTD in SHAM (n = 6; white circles) and THC (n = 4; orange circles) in utero-exposed female rats. Above: example traces, baseline (1) and 40 min poststimulation (2). (D) Individual experiments (grey) and group average (white represents SHAM; orange represents THC), before (baseline) and after (40 min) LTD induction showing that prenatal THC exposure does not alter LTD in the female exposed group. In SHAM female rats: 0.211 ± 0.011 mV before LTD versus 0.183 ± 0.010 mV after LTD induction (n = 6, p<0.05, paired t-test). In THC female rats: 0.234 ± 0.030 mV before LTD versus 0.177 ± 0.030 mV after LTD induction (n = 4, p<0.05, paired t-test). *p<0.05. n = individual rats. Error bars indicate SEM. Scale bar: 10 ms, 0.1 mV.
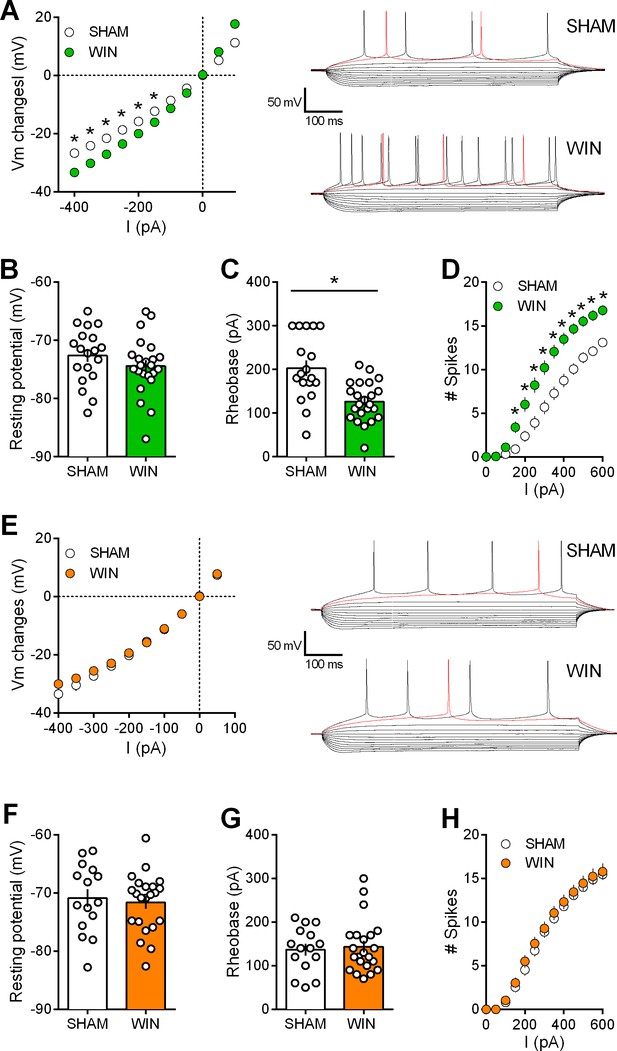
Prenatal WIN exposure induces sex-specific alteration of intrinsic properties of layer five prefrontal pyramidal neurons in adult rats.
(A) Left, Current-voltage (I–V) curves recorded in SHAM (n = 19 cells/10 rats, white circles) and WIN-exposed rats (n = 23 cells/12 rats, green circles) showing altered membrane potentials in male from the in utero cannabinoid group (F(10, 400)=10.07, p<0.05, two-way ANOVA). Right, typical membrane response to somatic current steps from SHAM (up) and WIN- (down) exposed male. (B) The resting membrane potential was similar in both SHAM and WIN male groups (p>0.05, Mann-Whitney U test). (C) The rheobase was reduced in WIN-exposed male rats compared to the SHAM male (p<0.05, Mann-Whitney U test). (D) The number of evoked action potentials in response to increasing depolarizing current steps was higher in WIN in-utero treated male compared to SHAM animals (F(18, 720)=6.652, p<0.05, two-way ANOVA). (E) Left, In contrast, I-V curves were similar in SHAM (n = 15 cells/9 rats, white circles) and WIN-exposed female (n = 22 cells/12 rats, orange circles; F(9, 315)=1.091, p>0.05, two-way ANOVA). Right, typical membrane response to somatic current steps from SHAM (up) and WIN- (down) exposed females. (F,G) The resting membrane potential and the rheobase were not impacted in females (F,G) p>0.05, Mann-Whitney U test). (H) The number of action potentials was not altered in females after an in utero treatment (F(12, 455)=0.079, p>0.05, two-way ANOVA). *p<0.05. Error bars represent SEM.
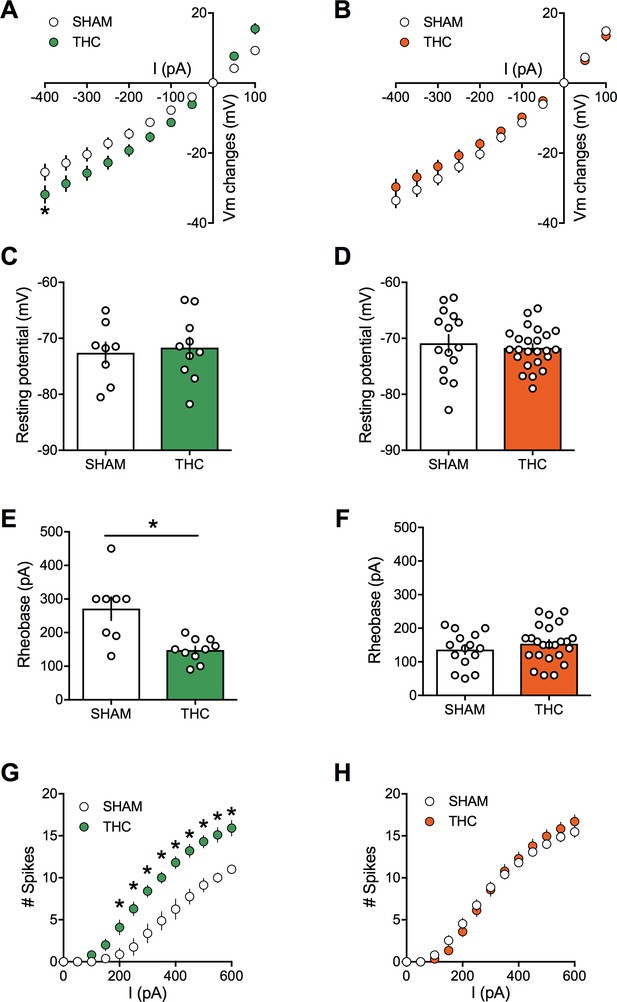
Prenatal THC exposure induces sex-specific alteration of intrinsic properties of layer five prefrontal pyramidal neurons in adult rats.
(A) Current-voltage (I–V) curves recorded in SHAM (n = 8 cells/5 rats, white circles) and THC-exposed rats (n = 10 cells/5 rats, green circles) showing altered membrane potentials in male after an in utero cannabinoid exposure (F(26, 416)=1.987, p<0.05, two-way ANOVA). (B): In contrast, I-V curves were similar in SHAM group (n = 15 cells/7 rats, white circles) and THC-exposed group (n = 24 cells/7 rats, orange circles; F(26, 962)=0.534, p>0.05, two-way ANOVA). (C,D): The resting membrane potential was similar in both male (C) and female (D) rats after an in utero exposure to THC (p>0.05, Mann-Whitney U test). (E,F): The rheobase was reduced in THC-exposed male rats compared to the SHAM male (E); p<0.05, Mann-Whitney U test) whereas it was not altered in female (F); p>0.05, Mann-Whitney U test). (G): The number of evoked action potentials in response to several depolarizing current steps was higher in THC in utero treated male compared to SHAM animals (F(18, 288)=3.764, p<0.05, two-way ANOVA). (H): The number of action potential was also altered in female after an in utero treatment with THC (F(18, 666)=1.320, p<0.05, two-way ANOVA). Error bars represent SEM.
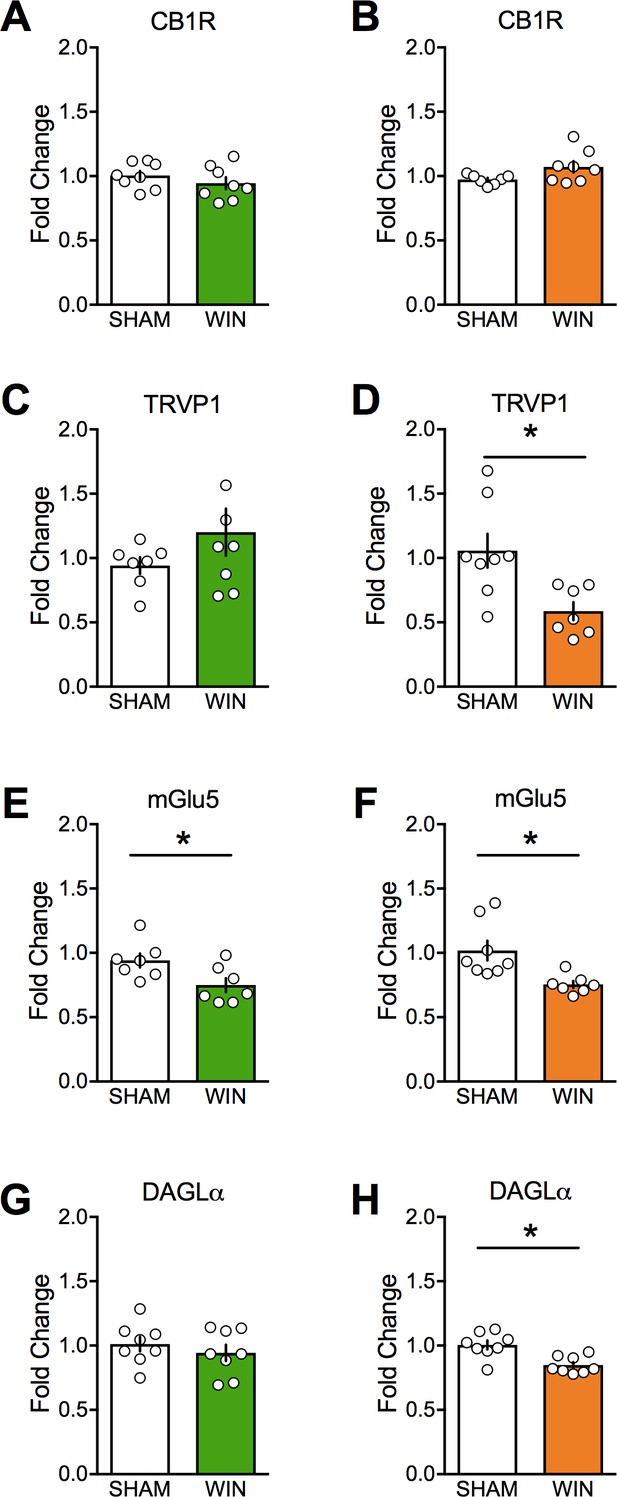
Prenatal cannabinoid treatment affects expression of endocannabinoid system components in a sex-dependent fashion.
CB1, TRPV1, mGlu5, and DAGLα mRNA levels in medial prefrontal cortex at PND90 were determined by real-time PCR in male (A, C, E, and G) and female (B, D, F, and H) rats using Taqman probes and a QuantStudio7 thermocycler. Treatment of dams with WIN from GD5 to GD20 decreased TRPV1, mGlu5, and DAGLα mRNA in female offspring. However, the same treatment decreased only mGlu5 in male offspring. *p<0.05. Error bars represent SEM.
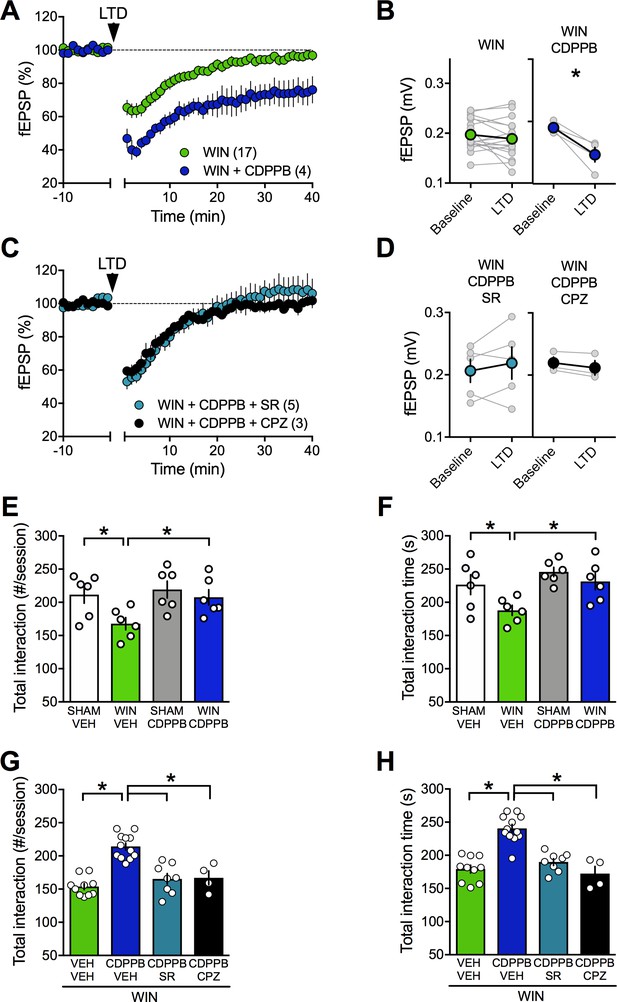
Positive allosteric modulation of mGlu5 restores LTD and normalizes social interaction in male rats prenatally exposed to WIN.
(A) Average time-courses of mean field EPSPs showing that, in WIN-exposed males (WIN, n = 17, green circle), LTD can be restored with the mGlu5positive allosteric modulator CDPPB (10 μM, n = 4, blue navy circle). (B) Peak amplitude measurements before (baseline) and after (LTD) stimulation protocol from individual experiments in absence or presence of CDPPB. After 45 min CDPPB preincubation: 0.190 ± 0.004 mV before and 0.147 ± 0.012 after LTD induction (n = 4, p<0.05, paired t-test). (C) Time course of fEPSP during 10 Hz stimulus evoked LTD (arrow, 10 min) in presence of CB1R antagonist SR141716 (SR, 4 μM, n = 5, blue circles) or TRPV1 antagonist Capsazepine (CPZ, 10 μM, n = 3, black circles) in WIN-exposed males. (D) Individual averaged fEPSP amplitude experiments before (baseline) and 40 min after 10 Hz LTD stimulation (LTD). CB1R were necessary for the CDPPB effect on LTD: SR141716A preincubation blocked LTD induction in the presence of CDPPB: 0.206 ± 0.019 mV before and 0.217 ± 0.026 (blue circle, n = 5, p>0.05, paired t-test). Similarly, the TRPV1 antagonist inhibits the restorative effects of CDPPB: 0.220 ± 0.009 mV before LTD and 0.212 ± 0.012 mV after LTD induction (black circle, n = 3, p<0.05, paired t-test). (E–F) Systemic administration of CDPPB (0.75 mg/Kg, i.p.) normalized the altered social behavior in male rats prenatally exposed to WIN (number of contacts: E: F(WIN in utero x CDPPB)1,20=1.867, p=0.187, two-way ANOVA; time interaction: F: F(WIN in utero x CDPPB)1,20=1.607, p=0.219, two-way ANOVA). (G-H) Pre-treatment with SR141716A (1 mg/kg, i.p.) prevents the ameliorative actions of CDPPB (0.75 mg/Kg, i.p.) on social interaction. Similarly, the TRPV1 antagonist blocks the restorative effects of CDPPB on social interaction in adult rats prenatally exposed to WIN (number of contacts: G: F3,30=22.36, p<0.05, one-way ANOVA; time interaction: H: F3,30=25.95, p<0.05, one-way ANOVA). Scatter dot plot represents a pair of animals. Error bars indicate SEM. *p<0.05. Student–Newman–Keuls test.
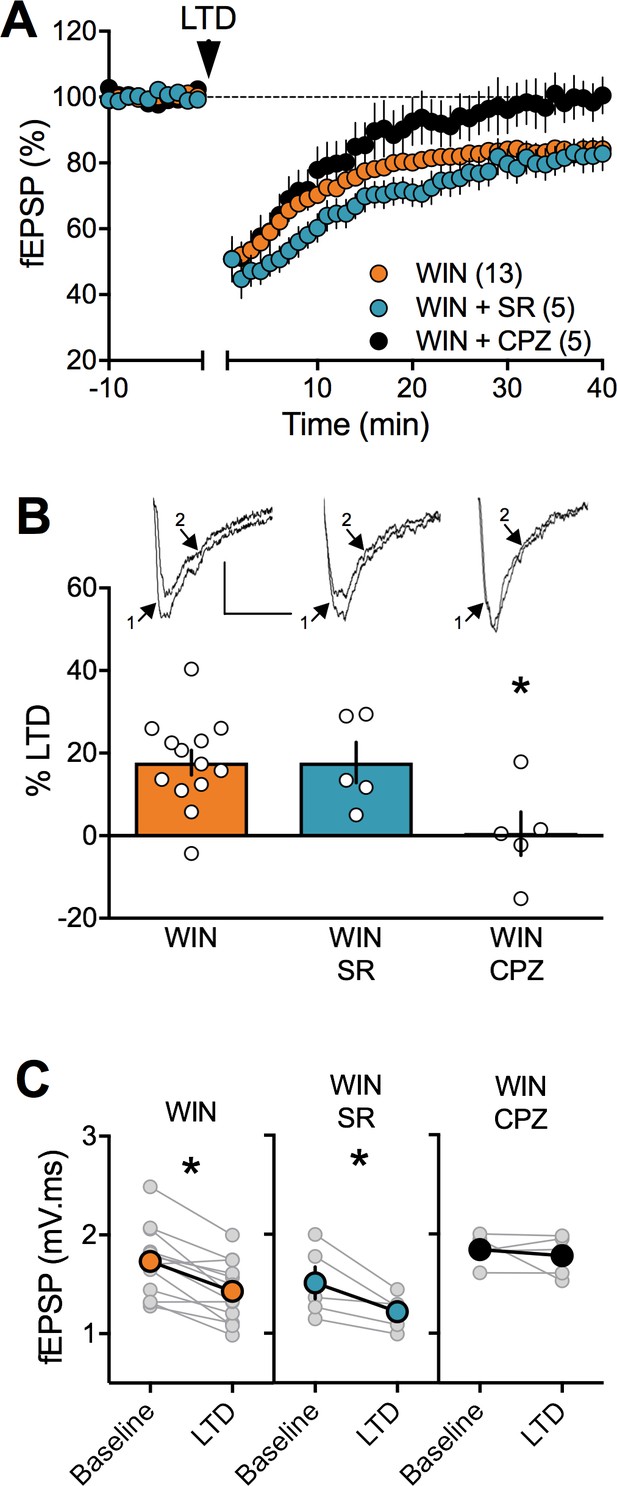
In prenatally exposed female, TRPV1 receptors mediate LTD, not CB1R.
(A) Average time course of mean fEPSPs showing that LTD (arrow) is inhibited by the selective TRPV1 antagonist capsazepine (CPZ, black circles, n = 5) but not the CB1R antagonist SR141617A (SR, blue circles, n = 5) in WIN in utero exposed female rats. (B) Summary bar chart of percent LTD calculated from normalized fEPSPs measured 40 min after 10 Hz protocol showing that LTD is blocked by TRPV1 antagonist (black bar) but not by CB1R antagonist (blue bar; p>0.05, one-way ANOVA). Above: example traces, baseline (1) and 40 min poststimulation (2). (C) Plot of individual experiments (grey) showing fEPSP areas before (baseline) and 40 min after stimulation protocol (LTD) in absence (orange, n = 13; p<0.05, paired t-test) and presence of SR141716A (SR, blue, n = 5; p<0.05, paired t-test) or Capsazepine (CPZ, black, n = 5; p>0.05, paired t-test) from WIN-exposed female rats. *p<0.05. n = individual rats. Error bars represent SEM. Scale bar: 10 ms, 0.1 mV.
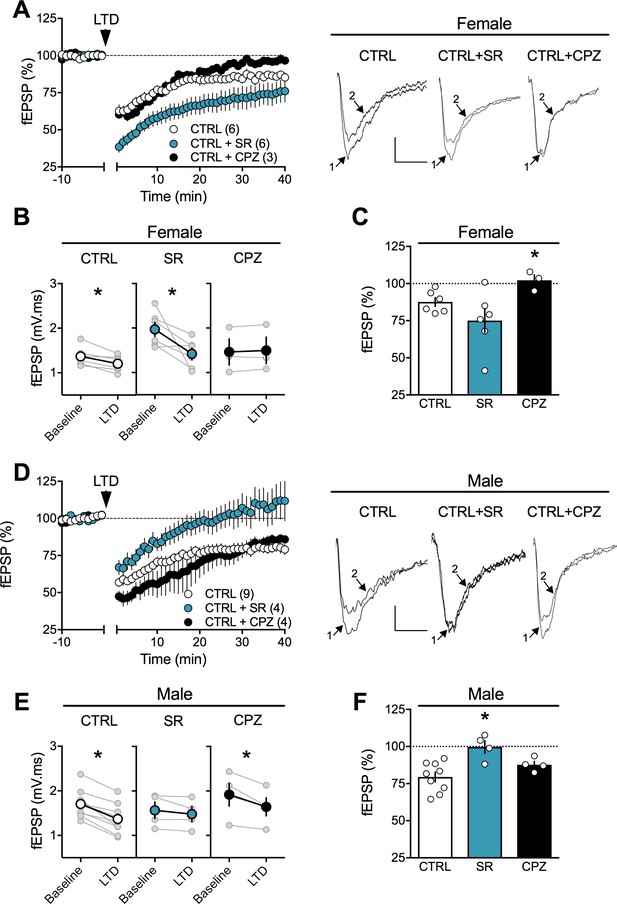
Sex-specific substrates of LTD in adult mPFC.
(A) Time course of normalized field EPSPs recordings from layer 5 mPFC after 10 Hz stimulation protocol (10 min, arrow) in presence of CB1R antagonist SR141716 (SR, n = 6, blue circles) or TRPV1 antagonist Capsazepine (CPZ, n = 3, black circles) in naïve female rats. Right: example traces, baseline (1) and 40 min poststimulation (2)(B) Individual experiments (grey) and group average (tan circles), before (baseline) and after (40 min) LTD induction (arrow) showing that, in control female rat (CTRL, left), LTD is blocked by the TRPV1 antagonist (CPZ, right) but not by the CB1R antagonist (SR, middle). In control rats: 1.368 ± 0.089 mV.ms before LTD versus 1.205 ± 0.074 after LTD induction (n = 6; p<0.05, paired t-test). After SR141716A preincubation: 1.970 ± 0.160 mV.ms before LTD versus 1.420 ± 0.133 after LTD induction (n = 6; p<0.05, paired t-test). In control female, LTD induction requires activation of TRPV1 receptors: 1.463 ± 0.295 mV.ms before LTD versus 1.497 ± 0.300 after LTD induction (n = 3; p>0.05, paired t-test). (C) Bar chart summary of fEPSP percentage change from baseline 38–40 min after 10 Hz stimulation in control female (white bar) after treatment with CB1R antagonist (SR, blue bar) or TRPV1 antagonist (CPZ, black bar) (F(2, 12)=3.928; p<0.05, one-way ANOVA). (D) Time course of normalized field EPSPS recordings from layer 5 mPFC after 10 Hz stimulation protocol (10 min, arrow) in presence of CB1R antagonist SR141716 (SR, n = 4, blue circles) or TRPV1 antagonist Capsazepine (CPZ, n = 4, black circles) in naive male rats. Right: example traces, baseline (1) and 40 min poststimulation (2). (E) Individual experiments (grey) and group average (colors), before (bsl) and after (40 min) LTD induction showing that LTD in control male rats is CB1R-mediated (middle) rather than TRPV1-mediated (right). Low-frequency stimulation of mPFC slices triggers LTD in male (left, white): 1.704 ± 0.107 mV.ms before 10 Hz stimulation versus 1.367 ± 0.112 after LTD induction (n = 9; p<0.05, paired t-test). This LTD is blocked by CB1R antagonist SR141716A: 1.565 ± 0.185 mV.ms before and 1.480 ± 0.167 after LTD induction (n = 4; p>0.05, paired t-test). In contrast, in male rats, LTD is not blocked by TRPV1 antagonist: 1.917 ± 0.256 mV.ms before LTD versus 1.644 ± 0.204 after LTD induction (n = 4; p<0.05, paired t-test). (F) Bar chart summary of fEPSP percentage change from baseline 38–40 min after LTD induction in control male (white bar) after treatment with CB1R antagonist (SR, blue bar) or TRPV1 antagonist (CPZ, black bar) (F(2, 14)=7.623; p<0.05, one-way ANOVA). mPFC slices were preincubated for 45 min in 5 μM SR141716A and 10 μM Capsazepine. *p<0.05. n = individual rats. Error bars indicate SEM. Scale bar: 10 ms, 0.1 mV.
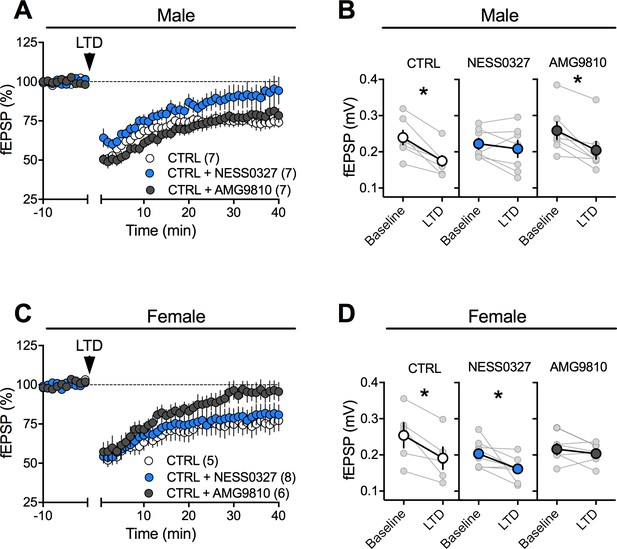
Sex-specific substrates of LTD in naïve adult mPFC.
(A) Time course of normalized field EPSPs recordings from layer 5 mPFC after 10 Hz stimulation protocol (10 min, arrow) in presence of the selective CB1R antagonist NESS0327 (n = 7, blue circles) or TRPV1 antagonist AMG9810 (n = 7, grey circles) in naïve male rats. (B) Individual experiments (grey) and group average (tan circles), before (baseline) and after (40 min) LTD induction (arrow) showing that, in control male rats (CTRL, left), LTD is blocked by the CB1R antagonist (NESS0327, middle) but not by the TRPV1 antagonist (AMG9810, right). In control rats: 0.239 ± 0.019 mV before LTD versus 0.175 ± 0.015 after LTD induction (n = 7; p<0.05, paired t-test). After NESS0327 preincubation: 0.223 ± 0.015 mV before LTD versus 0.208 ± 0.024 after LTD induction (n = 7; p>0.05, paired t-test). In naive males, LTD induction did not require activation of TRPV1 receptors: 0.258 ± 0.024 mV before LTD versus 0.204 ± 0.024 after LTD induction (n = 7; p<0.05, paired t-test). (C) Time course of normalized field EPSPs recordings from layer 5 mPFC after 10 Hz stimulation protocol (10 min, arrow) in presence of CB1R antagonist NESS0327 (n = 8, blue circles) or TRPV1 antagonist AMG9810 (n = 6, grey circles) in naive female rats. (D) Individual experiments (grey) and group average (colors), before (baseline) and after (40 min) LTD induction showing that LTD in control female rats is TRPV1R-mediated (right) rather than CB1R-mediated (middle). Low-frequency stimulation of mPFC slices triggers LTD in female (left, white): 0.254 ± 0.034 mV before 10 Hz stimulation versus 0.191 ± 0.031 after LTD induction (n = 5; p<0.05, paired t-test). This LTD is blocked by TRPV1R antagonist AMG9810: 0.216 ± 0.016 mV before and 0.204 ± 0.012 after LTD induction (n = 6; p>0.05, paired t-test). In contrast, in female rats, LTD is not blocked by CB1R antagonist: 0.204 ± 0.013 mV before LTD versus 0.162 ± 0.011 after LTD induction (n = 8; p<0.05, paired t-test). mPFC slices were preincubated for 45 min in 1 μM NESS0327 and 3 μM AMG9810. *p<0.05. n = individual rats. Error bars indicate SEM. Scale bar: 10 ms, 0.1 mV.
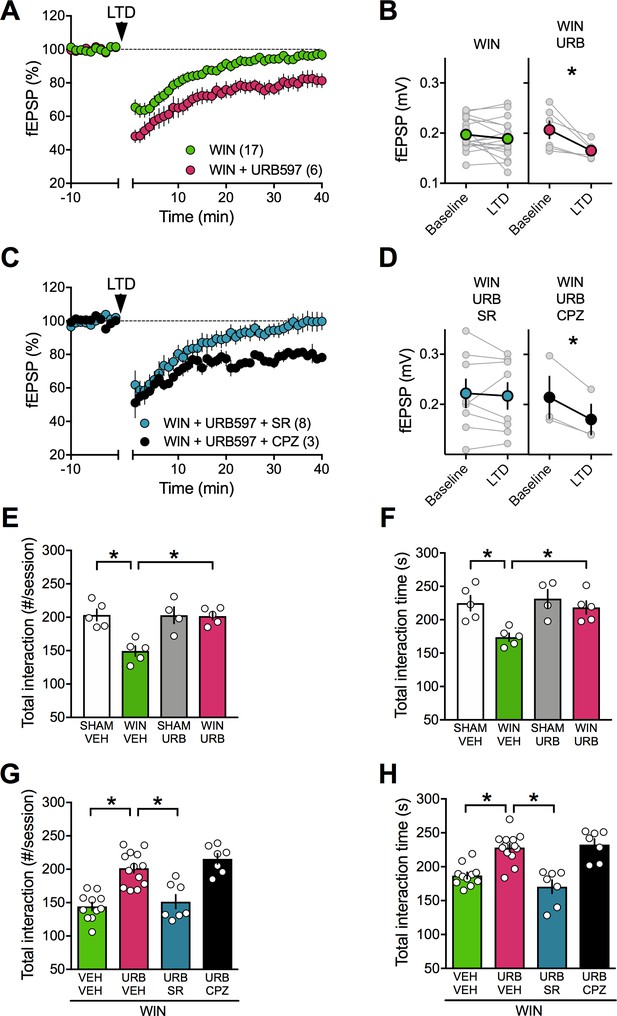
FAAH inhibition restores LTD and social interaction in male rats prenatally exposed to cannabinoids.
(A) Average tie-courses of mean fEPSPs after 10 Hz LTD protocol (1à min, arrow) in WIN-exposed mPFC slices preincubated for 45 min in 2 μM URB597, a FAAH inhibitor. (B) Individual experiments (grey) and group averages (tan circles), before (baseline) and after (40 min) LTD induction showing that, in WIN-exposed male (WIN, n = 17, green circle), LTD is restored following bath-application of the FAAH inhibitor URB597 (2 μM, 45 min preincubation, URB, n = 6; pink circle): 0.207 ± 0.017 mV before and 0.165 ± 0.007 after LTD induction (n = 6; p<0.05, paired t-test). (C) The URB597 LTD rescue requires CB1R. Time course of fEPSP during 10 Hz stimulus evoked LTD (arrow, 10 min) in presence of CB1R antagonist SR141716 (SR, 4 μM, n = 8, blue circles) or TRPV1 antagonist Capsazepine (CPZ, 10 μM, n = 3, black circles) in WIN-prenatally exposed males. (D) Individual experiments: SR141716A preincubation prevented from the URB597 restoration of LTD: 0.222 ± 0.028 mV before and 0.217 ± 0.026 after LTD induction (blue circle, n = 8; p>0.05, paired t-test). In contrast, the TRPV1 antagonist did not prevent from URB597 rescue: 0.214 ± 0.042 mV before LTD and 0.170 ± 0.030 mV after LTD induction (black circle, n = 3; p<0.05, paired t-test). (E–F) Systemic administration of URB597 (0.1 mg/kg, i.p.) normalized the altered social behavior in male rats prenatally exposed to WIN (numbers of contacts: E: F(WIN in utero x URB)1,15=9.341. p<0.05, two-way ANOVA; time interaction: F: F(WIN in utero x URB)1,15=3.421, p=0.08, two-way ANOVA). (G-H) Pre-treatment with the CB1R antagonist SR141716A (1 mg/kg, i.p.) prevented the curating actions of URB597 (0.1 mg/kg, i.p.) on social interaction. In contrast, the TRPV1 antagonist capsazepine did not block the curating effects of URB597 on social interaction in adult rats prenatally exposed to WIN55,212–2 (number of contacts: G: F3,34=21.02, p<0.05, one-way ANOVA; time interaction: H: F3,34=18.34, p<0.05, one-way ANOVA). Scatter dot plot represents each animal. Error bars indicate SEM. *p<0.05 Student–Newman–Keuls test.
Tables
Statistical significance of different behavioral tasks in SHAM and Naive animals.
Prenatal treatment from GD 5 to GD 20 with vehicle solution in control dams (SHAM group) did not alter sociability (social interaction), anxiety (elevated plus maze), and cognition (temporal order and object recognition memory tasks) in adult rats of either sex. Statistical significance was determined using the unpaired Student’s t-test after outliers were detected and removed from the dataset using Grubbs’ test.
| Behavior | Parameter | Treatment | Sex | n | Mean | SEM | P value (Unpaired t test) |
|---|---|---|---|---|---|---|---|
| Social Interaction | # of contacts | NAIVE | Male | 5 | 216.200 | 28.420 | 0.439 |
| SHAM | 10 | 235.800 | 10.550 | ||||
| NAIVE | Female | 4 | 211.800 | 21.600 | 0.85 | ||
| SHAM | 7 | 215.100 | 5.713 | ||||
| Elevated Plus Maze | % Time Open | NAIVE | Male | 6 | 25.900 | 2.988 | 0.665 |
| SHAM | 7 | 24.270 | 2.245 | ||||
| NAIVE | Female | 9 | 25.530 | 2.464 | 0.359 | ||
| SHAM | 9 | 22.540 | 1.978 | ||||
| % Open Entries | NAIVE | Male | 6 | 27.740 | 3.248 | 0.164 | |
| SHAM | 7 | 33.950 | 2.678 | ||||
| NAIVE | Female | 9 | 33.160 | 2.520 | 0.475 | ||
| SHAM | 9 | 30.230 | 3.111 | ||||
| Temporal order | Discrimination index | NAIVE | Male | 8 | 0.320 | 0.085 | 0.702 |
| SHAM | 7 | 0.366 | 0.078 | ||||
| NAIVE | Female | 8 | 0.258 | 0.132 | 0.896 | ||
| SHAM | 7 | 0.277 | 0.038 | ||||
| Object Recognition | Discrimination Index | NAIVE | Male | 10 | 0.547 | 0.087 | 0.792 |
| SHAM | 8 | 0.580 | 0.086 | ||||
| NAIVE | Female | 7 | 0.323 | 0.126 | 0.481 | ||
| SHAM | 7 | 0.421 | 0.045 |
Statistical significance of gene expression changes in adult rat medial prefrontal cortex after in utero WIN treatment.
The levels of mRNA’s of interest were determined from medical prefrontal cortex punches using qPCR (See Materials and methods for details). Statistical significance was determined using the unpaired Student’s t-test after outliers were detected and removed from the dataset using Grubbs’ test. p-Values of less than 0.05 are identified by bold text in the table. Relative expression levels for CB1, TRPV1, mGluR5 and DAGL-alpha are shown in Figure 10.
| MALE | Female | |||||
|---|---|---|---|---|---|---|
| Targets | Treatment | N | p-Value (Unpaired t-test) | Treatment | N | p-Value (Unpaired t-test) |
| CB1 | SHAM | 8 | 0.3279 | SHAM | 7 | 0.0643 |
| WIN | 8 | WIN | 8 | |||
| CB2 | SHAM | 7 | 0.9379 | SHAM | 6 | 0.1427 |
| WIN | 7 | WIN | 7 | |||
| TRPV1 | SHAM | 7 | 0.2281 | SHAM | 8 | 0.0098 |
| WIN | 8 | WIN | 7 | |||
| mGlu5 | SHAM | 7 | 0.0273 | SHAM | 8 | 0.0092 |
| WIN | 7 | WIN | 7 | |||
| mGlu1 | SHAM | 8 | 0.0400 | SHAM | 7 | 0.3910 |
| WIN | 8 | WIN | 8 | |||
| DAGL alpha | SHAM | 8 | 0.4370 | SHAM | 8 | 0.0024 |
| WIN | 8 | WIN | 8 | |||
| DAGL beta | SHAM | 7 | 0.4218 | SHAM | 6 | 0.3783 |
| WIN | 7 | WIN | 7 | |||
| NAPE-PLD | SHAM | 7 | 0.4258 | SHAM | 7 | 0.7518 |
| WIN | 7 | WIN | 7 | |||
| MAGL | SHAM | 8 | 0.5097 | SHAM | 8 | 0.2147 |
| WIN | 8 | WIN | 8 | |||
| FAAH | SHAM | 8 | 0.5505 | SHAM | 8 | 0.4096 |
| WIN | 8 | WIN | 7 | |||
| ABHD6 | SHAM | 7 | 0.2437 | SHAM | 6 | 0.6014 |
| WIN | 7 | WIN | 7 | |||
| CRIP1a | SHAM | 7 | 0.6414 | SHAM | 7 | 0.1887 |
| WIN | 7 | WIN | 7 | |||
Reproduction data and pup weight after in utero WIN exposure.
Dam weight gain was calculated from GD 1 to GD 21 for n = 10 dams per treatment group. Pup weight at different postnatal days (PND) was calculated for n = 9–10 male and female pups from different litters. Data represent mean values ± SEM. Statistical significance was determined using the one-way ANOVA test.
| p-Value (ANOVA) | |||||
|---|---|---|---|---|---|
| Naive | SHAM | WIN | |||
| mean ± SEM | mean ± SEM | mean ± SEM | |||
| Dam weight gain (%) | |||||
| 33.8 ± 0.93 | 34.3 ± 1.91 | 33.1 ± 1.42 | 0.745 | ||
| Pregnancy length (days) | |||||
| Reproduction data | 22.4 ± 0.14 | 22.6 ± 0.29 | 22.6 ± 0.17 | 0.923 | |
| Litter size | 13 ± 0.82 | 12.9 ± 0.78 | 12.7 ± 0.67 | 0.854 | |
| PND 1 | 6.8 ± 0.2 | 6.7 ± 0.1 | 6.8 ± 0.2 | 0.965 | |
| PND 10 | 23.5 ± 0.89 | 24.6 ± 0.65 | 22.6 ± 0.72 | 0.207 | |
| Pup weight (grams) | PND 13 | 31.8 ± 0.62 | 30.3 ± 0.82 | 30.5 ± 0.75 | 0.339 |
| PND 25 | 64.5 ± 2.24 | 65.6 ± 0.77 | 66.7 ± 1.55 | 0.647 | |
| PND 90 | 452.7 ± 12.28 | 484.2 ± 12.52 | 481.7 ± 17.33 | 0.261 |
Additional files
-
Transparent reporting form
- https://doi.org/10.7554/eLife.36234.022





