Empty conformers of HLA-B preferentially bind CD8 and regulate CD8+ T cell function
Figures
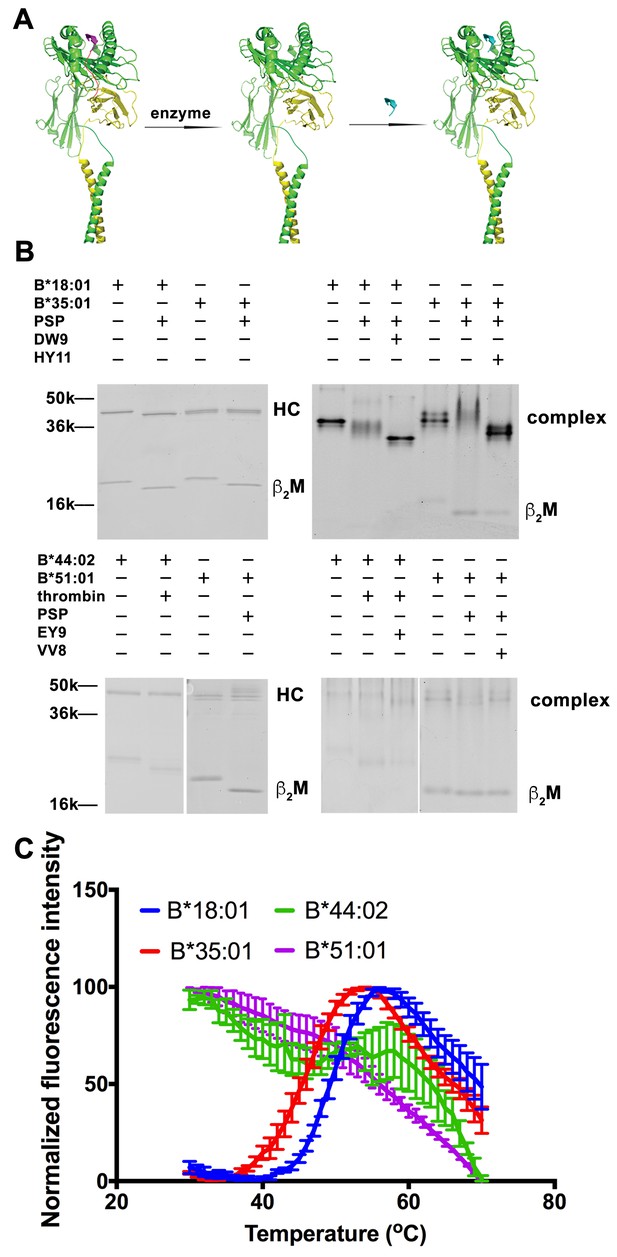
Peptide-deficient conformers of HLA-B molecules have different thermostabilities.
(A) Peptide-deficient HLA-B can be prepared by cleavage of engineered HLA-B (LZ-ELBM) with a specific enzyme and peptide-loaded versions by subsequent incubation with specific peptide as described in the scheme. (B) Representative SDS (left panels) and native (right panels)-PAGE gels showed cleavage and loading of B*18:01, B*35:01, B*44:02, and B*51:01 molecules with peptides DEVASTHDW (DW9), HPVGEADYFEY (HY11), EEIPDFAFY (EY9) and VPYEPPEV (VV8), respectively. (C) Averaged (n ≥ 3 replicates) normalized thermal shift assays were performed with peptide-deficient conformers of B*18:01, B*35:01, B*44:02 and B*51:01 molecules. B*18:01 and B*35:01 are more stable than B*44:02 and B*51:01.
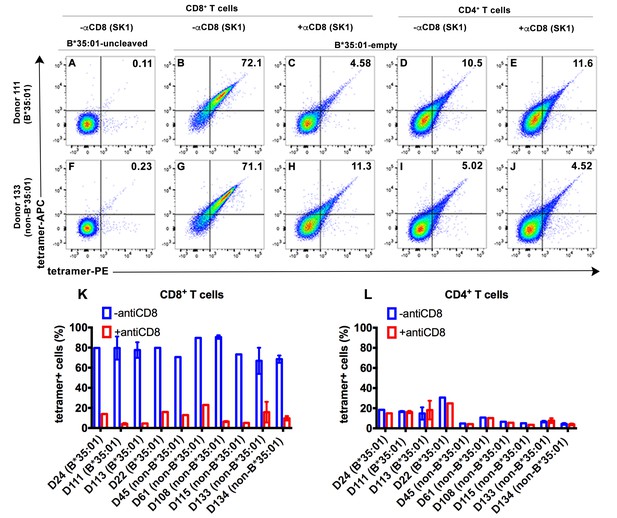
CD8-dependent binding of peptide-deficient HLA-B*35:01 tetramers to CD8+ T cells.
Uncleaved B*35:01 tetramer poorly stained CD8+ T cells (A and F), whereas peptide-deficient B*35:01 tetramers stained most (over 70%) CD8+ T cells from B*35:01-positive donors (such as Donor 111) (B) or B*35:01-negative donors (such as Donor 133) (G) in a manner sensitive to blockage by anti-CD8 (Clone SK1) (C and H). CD4+ T cells were, in comparison, poorly stained by peptide-deficient B*35:01 tetramers and the staining cannot be blocked by anti-CD8 (D, E, I and J). Peptide-deficient B*35:01 tetramer staining data from 10 tested donors (mean ± SEM of one to two assays) are shown in (K and L).
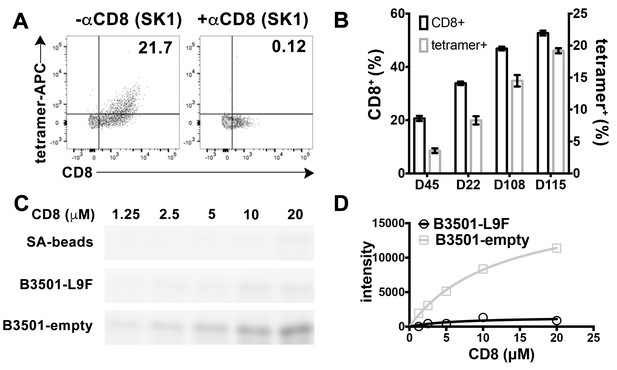
Preferential binding of peptide-deficient conformers of HLA-B*35:01 to CD8.
(A) Primary NK cells (CD3-CD56+) from Donor 115 were stained with peptide-deficient B*35:01 tetramers, demonstrating specific binding to the CD8+ NK cell fraction (left panel). NK cell staining by peptide-deficient B*35:01 tetramers was blocked by anti-CD8 (right panel). Representative data are shown based on two experiments each with four donors. (B) Primary NK cells from different donors have different CD8+ fractions and CD8-dependent binding of peptide-deficient B*35:01 tetramer to NK cells is proportional to the CD8+ fraction of NK cells among tested donors. The mean ± SEM of two experiments for each donor are shown. (C) Binding of SA-bead immobilized peptide-deficient or peptide-filled B*35:01 to the indicated concentrations of CD8-FITC. Proteins pulled-down were analyzed by SDS-PAGE gel and fluorimaging. (D) Quantified binding signals are plotted following background subtraction. Data are representative of four experiments.
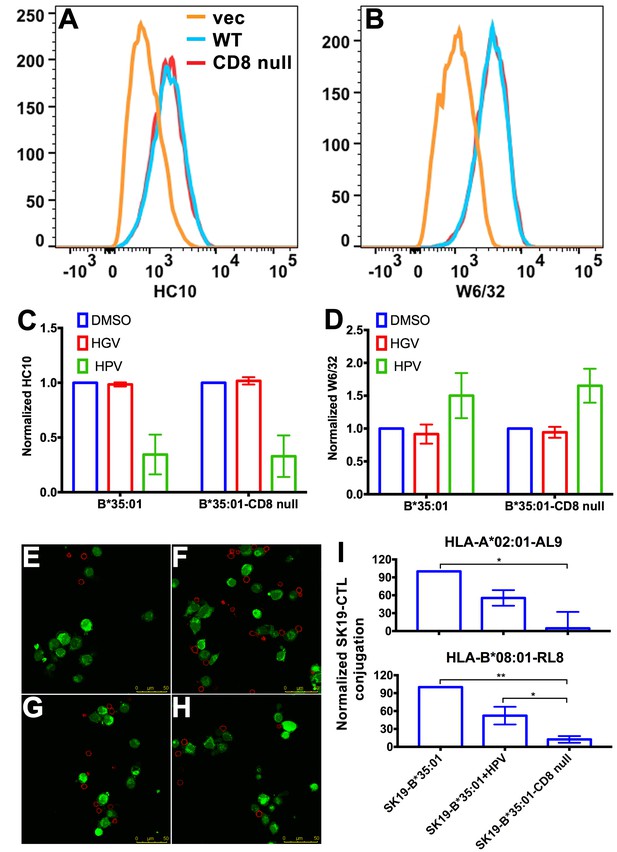
Binding of peptide-deficient conformers of HLA-B*35:01 to CD8 enhances cell adhesion.
HLA-B*35:01 and HLA-B*35:01-CD8 null were expressed by retroviral infection in the TAP1-deficient cell line, SK19. Similar levels of HLA-I in either peptide-deficient (A) or peptide-filled (B) versions were detected on the cell surface by flow cytometry. The peptide-deficient conformers can partly be blocked by the HLA-B*35:01-specific peptide (HPV) but not control peptide (HGV), which are indicated by reduced HC10 staining (C) and enhanced W6/32 staining (D). The mean ± SEM of two experiments are shown. Confocal microscopy (E–H) was used to test cell adhesion between SK19 cells expressing HLA-B*35:01 or HLA-B*35:01-CD8 null and a CTL line A2-AL9. A2-AL9 was incubated with preattached and CFSE-labeled SK19 cells (green) infected with retroviruses lacking HLA-B (E), or encoding HLA-B*35:01 (F and G) or HLA-B*35:01-CD8 null (H). For G, SK19-HLA-B*35:01 cells were preloaded with peptide HPV (100 μM). Cells were washed, fixed and stained with anti-CD8 (red) before analysis. Representative data are shown. Flow cytometry was used as a more quantitative assessment to test cell adhesion between SK19 cells expressing HLA-B*35:01 or HLA-B*35:01-CD8 null and CTL lines, A2-AL9 or B8-RL8 (I). CFSE and CD8 double positive cells were quantified as percentages of total SK19 cells. The condition with SK19 cells lacking HLA-B was subtracted as background. The mean ± SEM of three experiments are shown. Statistical analyses were undertaken using one-way ANOVA analysis with Fisher’s LSD test. *p<0.05, **p<0.01.
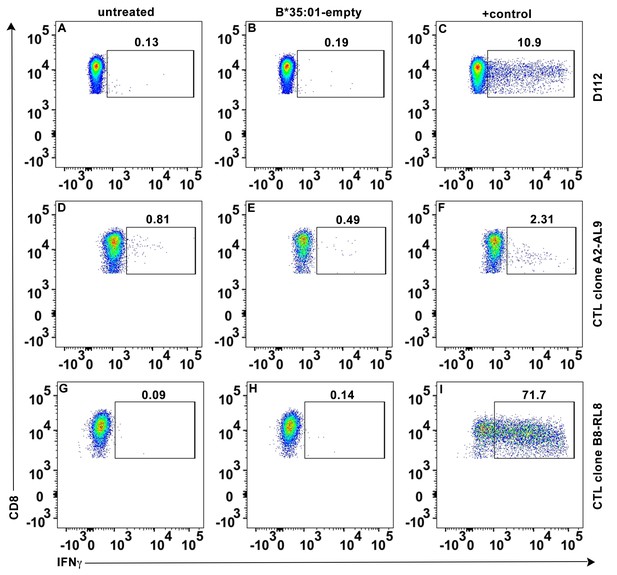
No direct activation of peptide-deficient HLA-B*35:01 tetramers on CTL activation.
Peptide-deficient HLA-B*35:01 tetramers (40 μg/ml) were incubated with primary CD8+ T cells (A–C), CTL line A2-AL9 (D–F) or CTL line B8-RL8 (G–I). Intracellular IFN-γ was tested with flow cytometry as CD8+ T cell activation marker. Untreated cells (A, D and G) or cells treated with PMA + ionomycin (C), cognate HLA-A*02:01-AL9 tetramer (F) or HLA-B*08:01-RL8 tetramer (I) were used as negative and positive controls, respectively. Representative data from n ≥ 3 independent experiments are shown.
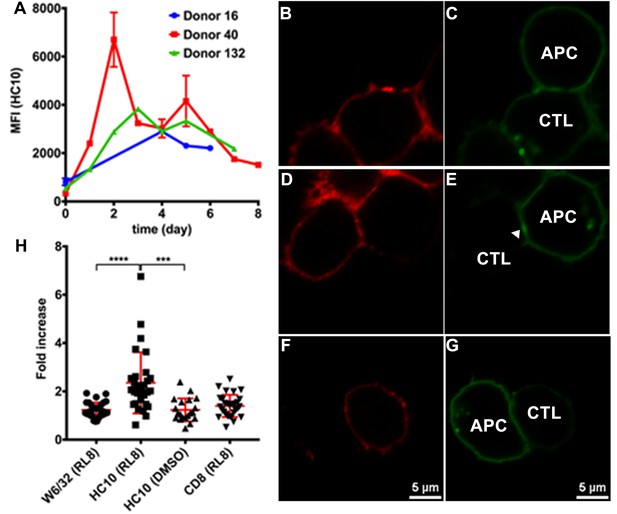
Clustering of peptide-deficient HLA-I in cognate peptide-induced immunological synapses.
Expression levels of HLA-I on the surface of activated CD4+ T cells isolated from PBMCs of three healthy donors were assessed by flow cytometry after staining with HC10 (A), confirming that activated lymphocytes express peptide-deficient conformers of HLA-I molecules. The mean ± SEM of two replicates for each donor are shown. CTL line B8-RL8 was incubated with activated CD4+ T cells from Donor 25 (carrying B*08:01 and B*35:01) loaded with peptide RL8 (B, C, D and E) or not (F and G). Cells were fixed and stained with anti-CD8 and W6/32 or HC10 before analysis by confocal microscopy. Peptides were used at a concentration of 100 μM. Anti-CD8 staining (B, D and F) were shown in red and W6/32 (C) or HC10 (E and G) in green. Arrowheads indicate peptide-deficient conformers of HLA-I clustering at the interface between CD4+ T cells and CTL line. The intensity of HLA-I staining of the CD4+ T cells at the interface was compared with the membrane at a noncontact area and plotted as the fold increase above background (H). The results with a total of 20–30 conjugates (mean ± SEM) per condition are shown. CD8 clustering was derived from 30 conjugates under conditions shown in D. Statistical analyses were undertaken using a one-way ANOVA analysis with Fisher’s LSD test. ***, p<0.001; ****, p<0.0001.
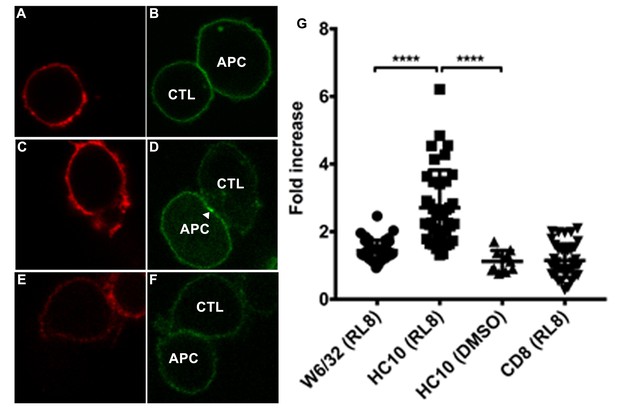
Clustering of peptide-deficient HLA-I in cognate peptide-induced immunological synapses.
CTL line B8-RL8 was incubated with activated PBMCs from Donor 25 (carrying B*08:01 and B*35:01) loaded with peptide RL8 (A–D) or not (E and F). Cells were fixed and stained with anti-CD8 and W6/32 or HC10 before analysis by confocal microscopy. Peptides were used at a concentration of 100 μM. Anti-CD8 staining (A, C and E) is shown in red and W6/32 (B) or HC10 (D and F) in green. Arrowheads indicate peptide-deficient conformers of HLA-I clustering at the interface between PBMCs and CTL line. The intensity of HLA-I staining of the PBMCs at the interface was compared with the membrane at a noncontact area and plotted as the fold increase above background (G). The results with a total of 39 conjugates (for B and D) or 10 conjugates (for F) per condition are shown. CD8 clustering was derived from 39 conjugates as condition shown in C. The mean ± SEM was shown. Statistical analyses were undertaken using one-way ANOVA analysis with Fisher’s LSD test. ****, p<0.0001.
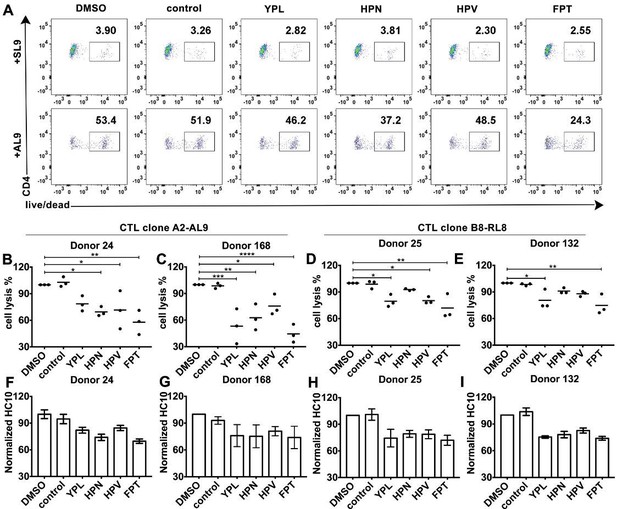
HLA-B*35:01 peptide-deficient conformers enhance cognate peptide-induced lysis of target cells by CD8+T cell activation.
CTL line A2-AL9 specifically killed activated primary CD4+ T cells (expressing HLA-A*02:01 and HLA-B*35:01 and used as antigen presenting cells) loaded with cognate peptide AL9 but not non-cognate peptide SL9 (A). Peptides that binds specifically to HLA-B*35:01 (YPL, HPN, HPV and FPT) could reduce AL9-induced cell lysis. Representative data with CD4+ T cells from Donor 24 are shown in Panel A and statistical analyses from Donors 24 and 168 in Panels B and C. Cell lysis assays were also performed with another CTL line B8-RL8 and activated primary CD4+ T cells (carrying HLA-B*08:01 and B*35:01) from Donors 25 and 132 loaded with cognate peptide RL8 were used as target cells (D and E). (B–E) The mean ± SEM of three independent assays are shown. Statistical analyses were undertaken using one-way ANOVA analysis with Dunnett test. *, p<0.05, **, p<0.01, ***, p<0.001, ****, p<0.0001. (F–I) Blocking of HLA-B*35:01 peptide-deficient conformers on the surface of CD4+ T cells by B*35:01-specific peptides (YPL, HPN, HPV and FPT), but not nonspecific control peptide (TW10 or HGV) was confirmed by flow cytometry after staining cells with HC10. The mean ± SEM of two to three independent experiments are shown.
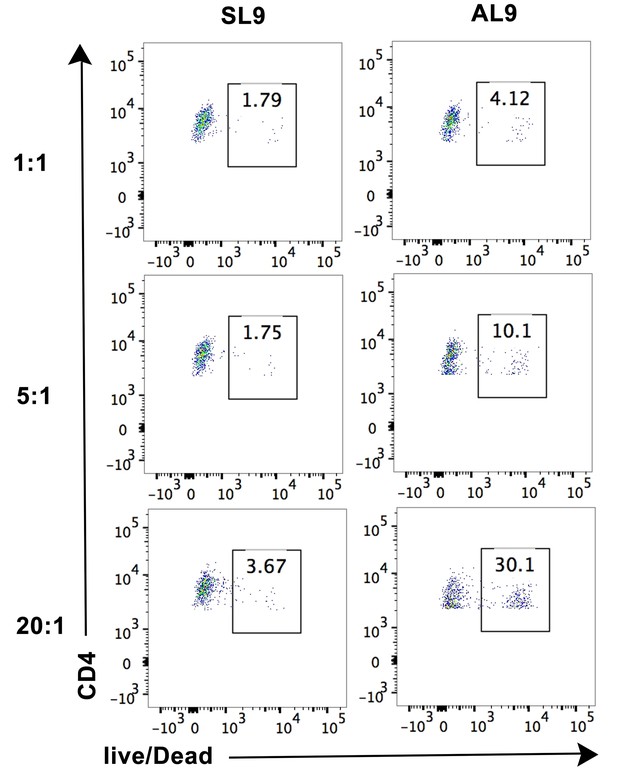
Cell lysis by CTL line A2-AL9 of activated primary CD4+ T cells expressing HLA-A*02:01.
Lysis was induced by pulsing cells with the cognate peptide AL9 but not the non-cognate peptide SL9. Cell lysis assay was performed at different effector: target cell ratios 1:1, 5:1 and 20:1, as described in the Materials and methods section. Representative data from n = 3 independent experiments are shown.
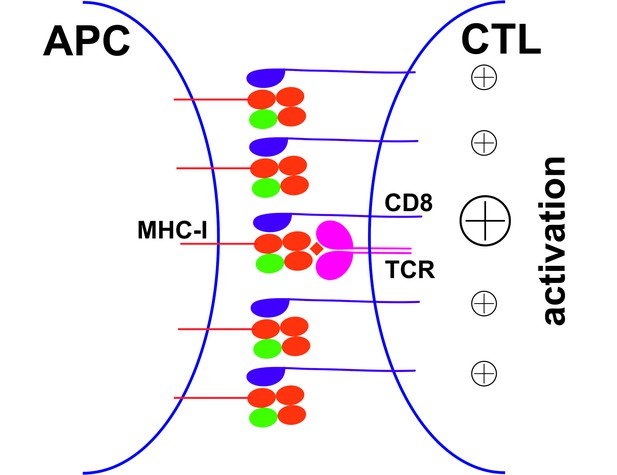
Peptide-deficient conformers of MHC-I molecules enhance CTL activation.
CTL activation is generally induced by the recognition of a specific peptide-MHC-I complex on the surface of antigen presenting cells (APC) by a T cell receptor (TCR) and co-receptor CD8. In this study, we show that peptide-deficient conformers of MHC-I molecules have increased binding to CD8 compared with peptide-filled conformers and therefore enhance the cell-cell contact between APC and CTL. Peptide-deficient conformers of MHC-I molecules are enriched in the immunological synapse and augment CTL activation.
Additional files
-
Transparent reporting form
- https://doi.org/10.7554/eLife.36341.013





