Cyclin-dependent kinase control of motile ciliogenesis
Figures
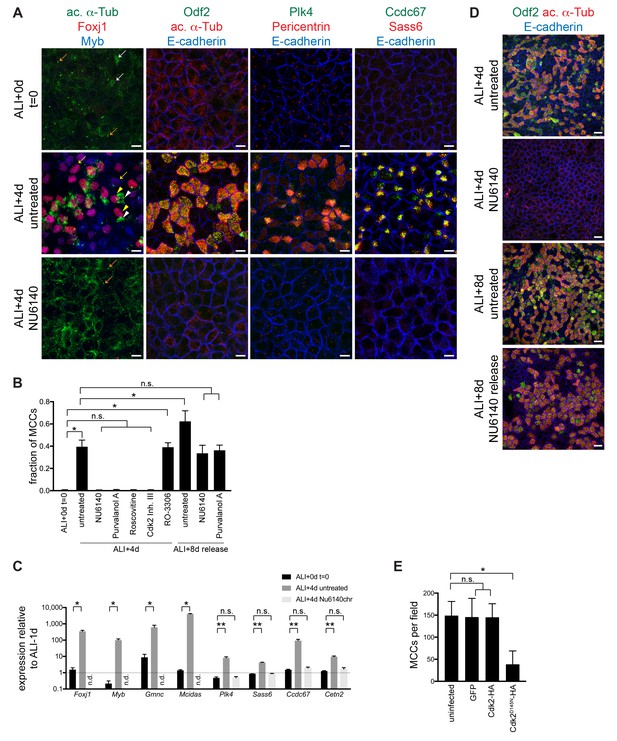
Chronic Cdk inhibitor treatment blocks motile ciliogenesis.
(A) MTECs were treated with NU6140 from ALI + 0 to 4d. They were fixed at ALI + 0 and +4 d and labeled with antibodies to monitor ciliogenesis: left, ac. α-Tub (green), Foxj1 (red) and Myb (blue); center left, Odf2 (green), ac. α-Tub (red) and E-cadherin (blue); center right, Plk4 (green), Pericentrin (red) and E-cadherin (blue); and right, Ccdc67 (green), Sass6 (red) and E-cadherin (blue). MTECs are confluent without any sign of motile ciliogenesis at ALI + 0 d. Untreated ALI + 4 d cells are robustly ciliating, but NU6140 treatment blocks all signs of motile ciliogenesis. Ac. α-Tub marks cytoplasmic microtubules in non-MCCs (white arrow) and motile ciliary axonemal tufts in MCCs (white arrowhead); when MCCs are present, the much weaker cytoplasmic signal is not discernible. Foxj1 and Myb mark both ciliating (yellow arrow) MCCs without ac. α-Tub + axonemes and Foxj1 marks mature MCCs (yellow arrowheads) with axonemes. NU6140 treatment has no effect on primary cilium formation (orange arrow) or on apical cell junctions. Scale bar, 10 μm. (B) Quantitation of the Cdki block and the release for Cdki treatment. MCCs were identified by ac. α-Tub labeling. n.s., not significant; *p<0.0001 (C) Realtime PCR results show that the expression of MCC TFs (Foxj1, Myb, Gmnc and Mcidas) is suppressed and the expression of ciliary components (Plk4, Sass6, Ccdc67 and Cetn2) is not upregulated in cells treated with NU6140 from ALI + 0 to 4d. Levels were normalized to Gapdh expression and compared to values obtained for MTECs at ALI-1d (n.d. = none detected). n.s., not significant, *p<0.05, **p<0.0001 (D) MTECs were treated with NU6140 from ALI + 0 to 4d, then cultured without Nu6140 until ALI + 8 d. Cells were fixed at ALI + 4 and+8 d and labeled with Odf2 (green), ac. α-Tub (red) and E-cadherin (blue) antibodies to show that MTECs ciliate robustly after release from Cdki treatment. Scale bar, 20 μm. (E) MCCs were quantitated based on ac. α-Tub labeling in MTECs infected with GFP, Cdk2-HA or Cdk2D145N-HA lentivirus. Cdk2D145N, but not wildtype Cdk2 expression blocks ciliogenesis. Ectopic wildtype Cdk2 expression in MTECs is not sufficient to drive motile ciliogenesis. n.s., not significant; *p<0.000.
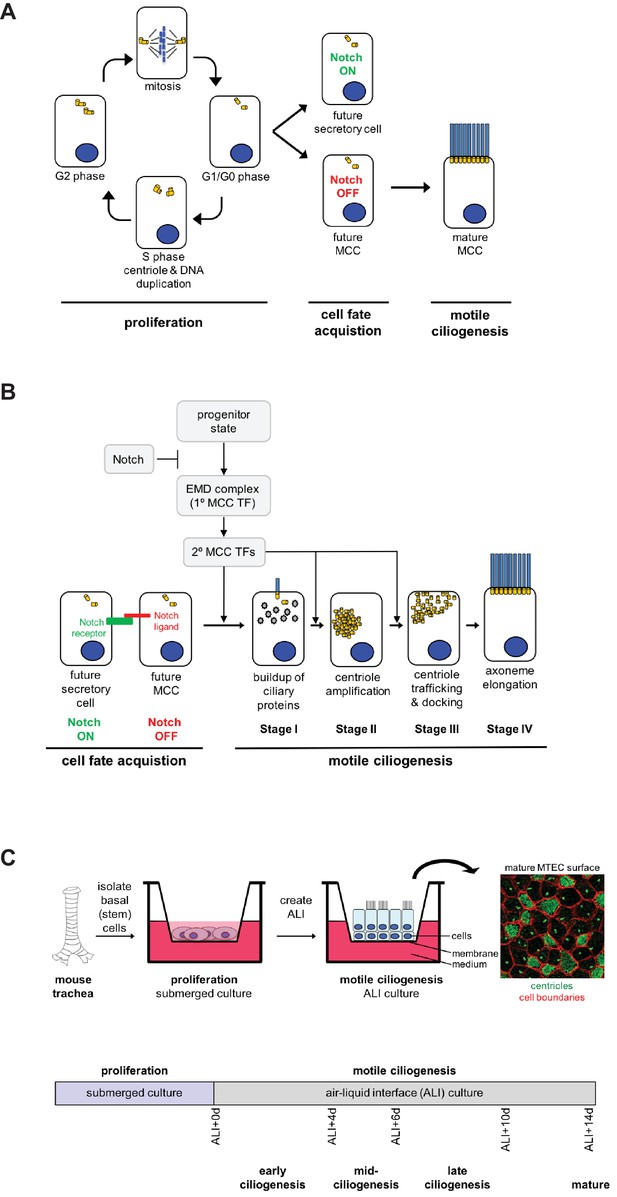
The motile ciliogenesis pathway and the MTEC culture system.
(A) Progenitor basal cells proliferate during development or regeneration to establish or repair the airway epithelial layer, then exit the cell cycle and experience Notch signaling to distinguish MCC vs. secretory cell fates. Future MCCs then undergo motile ciliogenesis by amplifying centrioles to build motile cilia for airway clearance. (B) Future MCCs and secretory cells are selected out in a Notch-dependent manner such that the future secretory cell expresses the Notch receptor and activates the Notch pathway, whereas future MCCs do not experience Notch activation, but express ligand. Downstream of the Notch signaling event, nascent MCCs undergo the motile ciliogenesis pathway. During Stage I, MCCs launch the MCC gene expression program to express regulatory and structural ciliary genes, which build up in the cytoplasm (grey shapes). The MCC transcriptional program is controlled by the primary EMD complex, which turns on multiple secondary transcription factors. At Stage I, MCCs also briefly possess a primary cilium. During Stage II, cells generate hundreds of centrioles in the cytoplasm, which then traffic to and dock with the apical plasma membrane during Stage III. Stage IV represents a mature MCC in which centrioles act as basal bodies and elongate the motile ciliary axoneme. Centrioles, yellow cylinders; axonemes, blue rods. (C) The MTEC system faithfully models the establishment of the multiciliated airway epithelium. Progenitor basal cells are isolated by protease digestion from adult mouse tracheas and seeded onto porous Transwell membranes. Basal cells proliferate under submerged conditions. After they have formed a confluent, postmitotic columnar epithelium, the air-liquid interface (ALI) is created by supplying medium only from the basal compartment of the culture vessel. Culture days 1–5 comprise the submerged, proliferative phase, and the differentiation of the MCCs and other cell types commences upon ALI culture. MCC fate determination and motile ciliogenesis occur asynchronously, but early ALI culture days are strongly enriched for young MCCs at the early stages of the pathway and by ALI + 14 days the filter contains only mature MCCs. The immunofluorescence image shows centrioles in both MCCs and nonMCCs in green and cell boundaries in red.
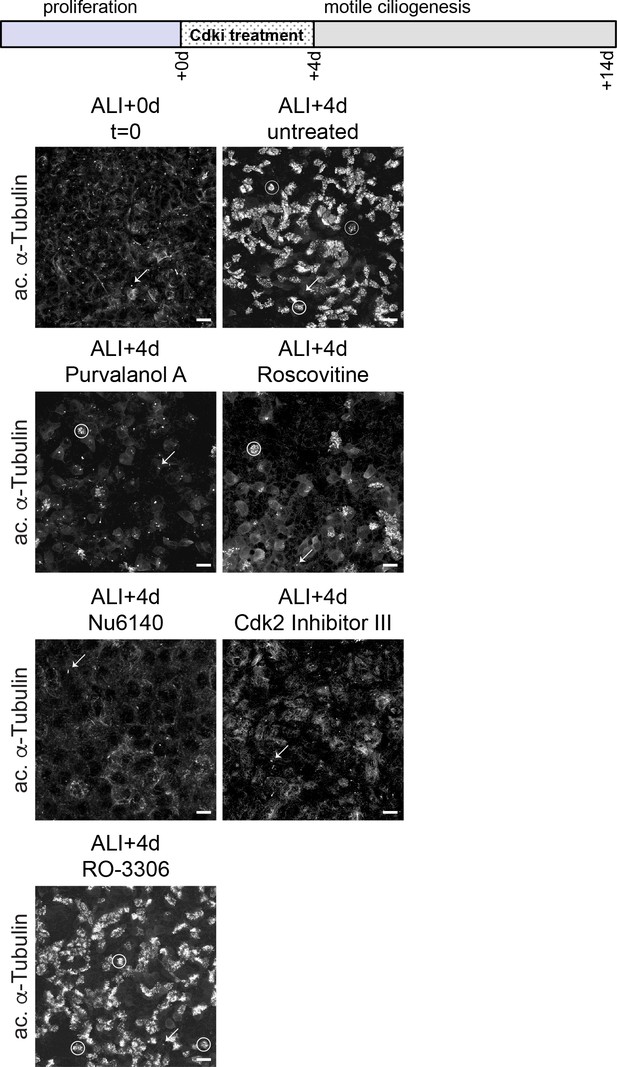
Cdk inhibitor treatment blocks MCC differentiation in MTECs.
MTECs were treated with Cdkis from ALI + 0 to 4d. They were fixed at ALI + 0 and ALI + 4 d and labeled with anti-ac. α-Tub antibody to mark MCCs (some individual MCCs circled). MTECs treated with Cdki, with the exception of RO3306, show a block to MCC formation. Primary cilia (arrows) are visible on some cells in all panels. Scale bar, 20 μm.
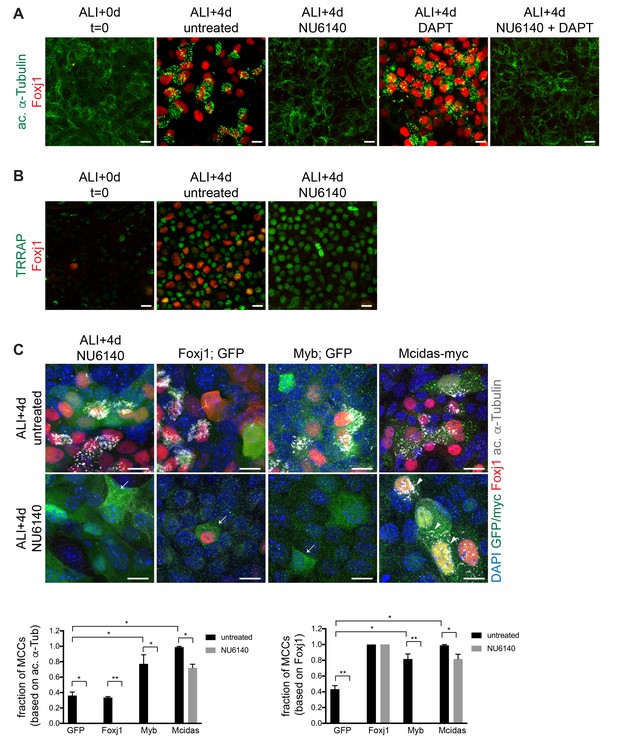
Cdk2 acts downstream of Notch signaling and upstream of the MCC gene expression program.
(A) MTECs were treated with NU6140 and/or DAPT from ALI + 0 to 4d. They were fixed at ALI + 0 and +4 d and labeled with ac. α-Tub (green) and Foxj1 (red) antibodies. DAPT induces MCC formation but not in the presence of NU6140. Scale bar, 10 μm. (B) MTECs were treated with NU6140 from ALI + 0 to 4d. They were fixed at ALI + 0 and +4 d and labeled with TRRAP (green) and Foxj1 (red) antibodies. Scale bar, 20 μm. (C) MTECs were infected with lentivirus encoding GFP, or Foxj1 or Myb and GFP from separate promoters, or myc-tagged Mcidas at ALI-2d, then treated with NU6140 from ALI + 0 to 4d. They were fixed at ALI + 4 d and labeled with GFP or myc (green), Foxj1 (red) and ac. α-Tub (white) antibodies and stained with DAPI (blue) to mark nuclei. Only Mcidas, but not GFP, Foxj1 or Myb expression can drive the complete motile ciliogenesis pathway (arrows indicate GFP+ cells without ac. α-Tub+ cilia, arrowheads point to mature myc+ MCCs) in NU6140 treated MTECs. Foxj1 expression leads to nuclear Foxj1 accumulation, but not ac. α-Tub+ cilia. MCCs were quantitated based on Foxj1 and ac. α-Tub expression. Scale bar, 10 μm. *p<0.001, **p<0.0001.
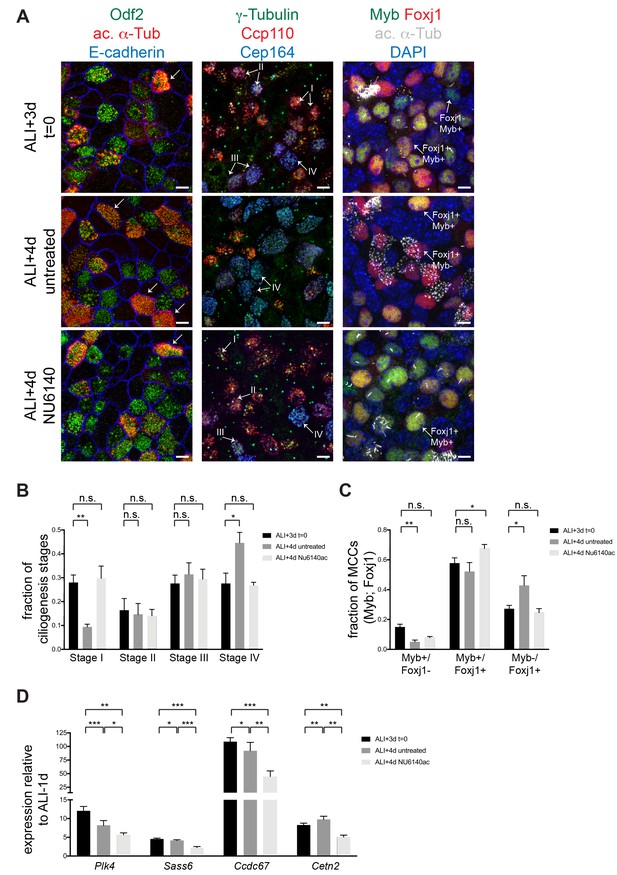
Cdk2 activity is required to sustain motile ciliogenesis.
(A) MTECs were treated with NU6140 from ALI + 3 to 4d. They were fixed at ALI + 3 and+4 d and labeled with antibodies to monitor ciliogenesis: left, Odf2 (green), ac. α-Tub (red) and E-cadherin (blue); center, γ-Tubulin (green) Ccp110 (red) and Cep164 (blue); and right, DAPI (blue), Myb (green), Foxj1 (red) and ac. α-Tub (white). MTECs at ALI + 3 d contain predominantly ciliating cells. Many mature MCCs emerge in untreated ALI + 4 d MTECs, but not in the presence of NU6140. Left panels: Stage IV MCCs are marked by ac. α-Tub + cilia (arrow). Center panels: Stage I MCCs are Ccp110+, Cep164-; Stage II MCCs are Ccp110+, Cep164+; Stage III MCCs are Ccp110 low, Cep164 high and Stage IV MCCs are Ccp110-, Cep164+. Right panels: Stage I MCCs are Foxj1-, Myb+; Stage II MCCs are Foxj1+, Myb+; and Stage III-IV MCCs are Foxj1+, Myb-. Scale bar, 10 μm. n.s., not significant; *p<0.01, **p<0.001 (B) Quantitation of MCCs at Stages I-IV (see Figure 1—figure supplement 1B) with and without acute NU6140 treatment. n.s., not significant; *p<0.05 (C) Quantitation of Foxj1 and Myb positive cells with and without acute NU6140 treatment. n.s., not significant; *p<0.05, **p<0.01 (D) Realtime PCR showing that the expression of ciliary components Plk4, Sass6, Ccdc67 and Cetn2 decreases in cells treated with NU6140 from ALI + 3 to 4d. Note that as ciliogenesis peaks expression levels for some components also decrease in untreated MTECs from ALI + 3 to 4d. Levels were normalized to Gapdh expression and compared to values obtained for MTECs at ALI-1d. *p<0.05; **p<0.01; ***p<0.001.
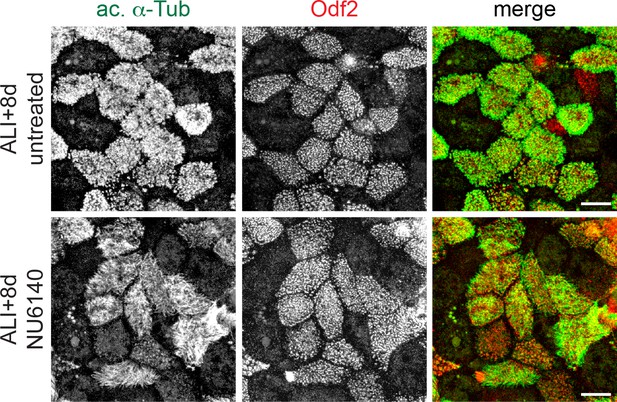
NU6140 treatment does not disrupt cilia on mature MCCs.
MTECs were treated with NU6140 from ALI + 4 to 8d. They were fixed at ALI + 8 d and labeled with antibodies against ac. α-Tub (green) and Odf2 (red) to mark MCCs. Intact MCCs are present in NU6140 treated MTECs. Scale bar, 10 μm.
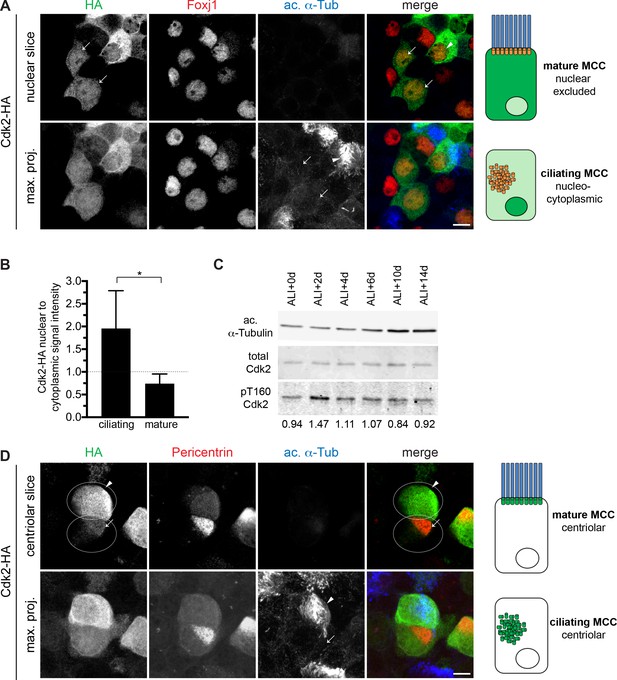
Cdk2 is nuclear and active during motile ciliogenesis.
(A) MTECs were infected with lentivirus encoding HA-tagged Cdk2 at ALI-2d, then labeled at ALI + 4 d with HA (green), Foxj1 (red) and ac. α-Tub (blue) antibodies. A single image slice through the nuclear region (top panel) shows that Cdk2-HA is present in the nucleus in ciliating MCCs (Foxj1+, ac. α-Tub-, arrow) and nuclear excluded in mature MCCs (Foxj1+, ac. α-Tub+, arrowhead). Ac. α-Tub signal visible on the maximum projection (bottom panel) shows cilia on the mature (arrowhead) or lack thereof on the ciliating cells (arrow). Scale bar, 10 μm. (B) Quantitation of nuclear to cytoplasmic ratio of Cdk2-HA signal intensity in MCCs indicating nuclear enrichment in ciliating cells. Ciliating vs. mature MCCs were identified based on ac. α-Tub and Foxj1 signal. *p<0.0005 C. Western blot of MTEC timecourse lysates shows that total Cdk2 levels are equally abundant at all times, but phospho-T160 Cdk2 is enriched during early ciliogenesis; ratio of phospho/total Cdk2 is indicated under each lane. α-Tubulin signal reflects increasing ciliogenesis and is not to be interpreted as loading control; see Figure 4—figure supplement 1 for Ponceau S stain for blot which serves loading control. (C) MTECs were infected with lentivirus encoding HA-tagged Cdk2 at ALI-2d, then labeled at ALI + 4 d with HA (green), Pericentrin (red) and ac. α-Tub (blue) antibodies. Image slices through the basal body/centriolar region (top panel) shows that Cdk2-HA is centriolar in both ciliating (arrow) and mature (arrowhead) MCCs. Pericentrin signal is strong on ciliating centrioles and weak on mature basal bodies. Ac. α-Tub signal indicating the mature MCC (arrowhead) is visible on the maximum projection (bottom panel). Scale bar, 5 μm.
-
Figure 4—source data 1
Quantitation of Cdk2-HA nucleo-cytoplasmic localization in ciliating and mature MCCs.
- https://doi.org/10.7554/eLife.36375.010
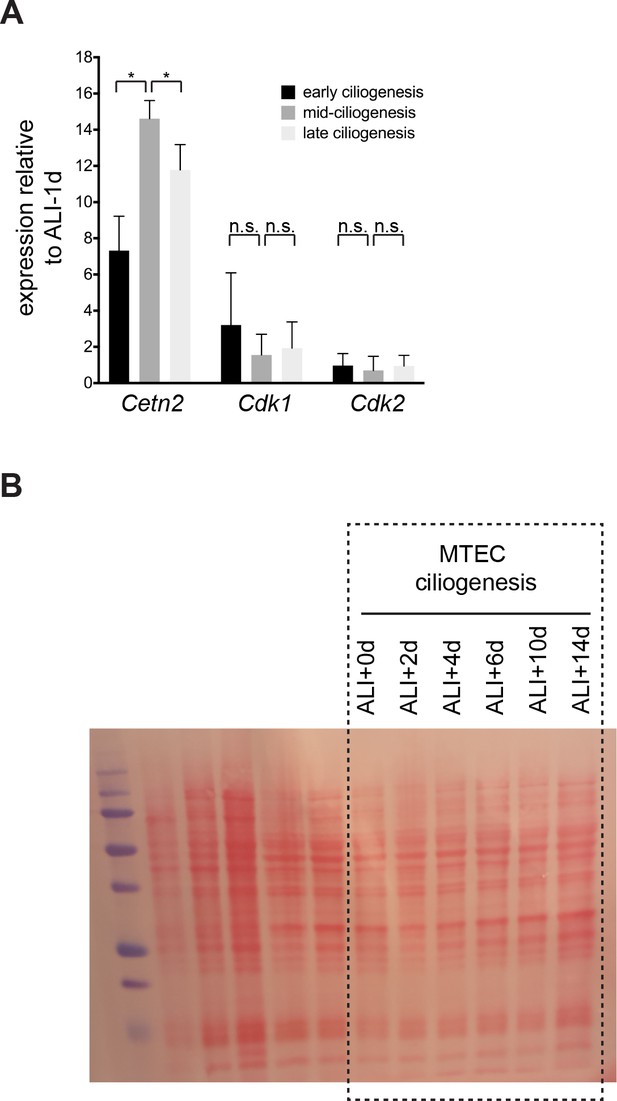
Cdk1 and Cdk2 gene expression levels do not change during motile ciliogenesis.
(A) Quantitative realtime PCR was used to assess Cdk gene expression during motile ciliogenesis. Unlike the ciliary component Cetn2, Cdk1 and Cdk2 do not show the typical profile of induction followed by a decline in gene expression during motile ciliogenesis. Levels were normalized to Gapdh expression and compared to values obtained from confluent pre-ALI samples. n.s., not significant; *p<0.05 (B) Ponceau-S stained membrane for Western blot in Figure 4C serving as loading control. The boxed area indicates the portion of the blot presented in the main figure. The first five lanes contain unrelated samples.
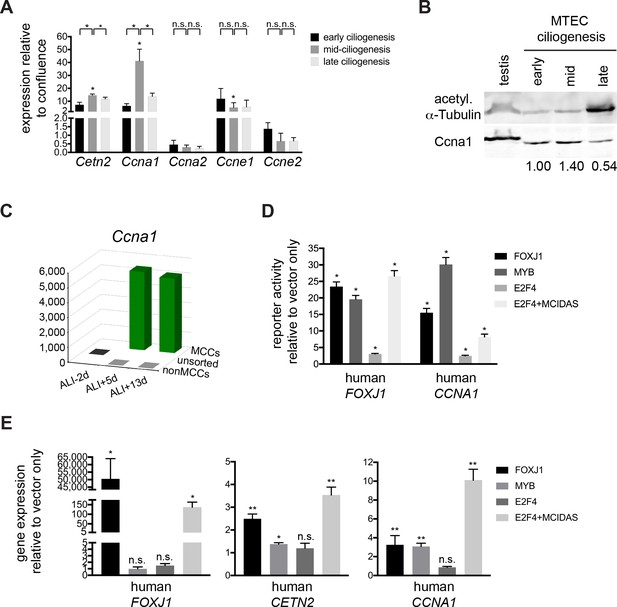
Ccna1 is enriched in MCCs and is a target of the MCC gene expression program during ciliogenesis.
(A) Quantitative realtime PCR was used to assess A and E-type cyclin gene expression during motile ciliogenesis. Similar to the ciliary component Cetn2, Ccna1 and Ccne1 are enriched during ciliogenesis. Ccna2 and Ccne2 are not enriched. Levels were normalized to Gapdh expression and compared to values obtained from confluent, ALI-1d samples. Brackets indicate comparison between ciliogenesis timepoints; asterisk above bar indicates significant increase in expression during mid-ciliogenesis compared to confluent, ALI-1d sample. n.s., not significant; *p<0.05 (B) Western blot with MTEC lysates using Ccna1 antibody indicates that Ccna1 is enriched in ciliating MTECs. Testis lysate serves as control of Ccna1 expression. Values normalized to early ciliogenesis indicated under each lane. Acetylated α-Tubulin signal reflects increasing ciliogenesis and should not be interpreted as loading control; see Figure 5—figure supplement 1 for Ponceau S stain for blot, which serves loading control. (C) Realtime PCR for Ccna1 expression in MCCs vs. non-MCCs (sorted from Foxj1-EGFP MTECs) shows that it is restricted to MCCs and Ccna1 expression is higher mid-ciliogenesis (ALI +5 d). (D) Luciferase reporter assay using the human FOXJ1 (left) or CCNA1 promoter (right) in 293T/17 cells shows that they are responsive to MCC transcriptional regulators compared to vector only control. Promoters are only responsive to E2F4 in the presence of the Mcidas transcriptional activator. *p<0.01 (E) 293T/17 cells were infected with lentivirus expressing MCC TFs. Realtime PCR indicates that at least one or more MCC TFs can activate endogenous FOXJ1 (left), CETN2 (center) and CCNA1 (right) gene expression. E2F4 can only activate MCC-specific gene expression in the presence of the Mcidas transcriptional activator. n.s., not significant; *p<0.05, **p<0.01. .
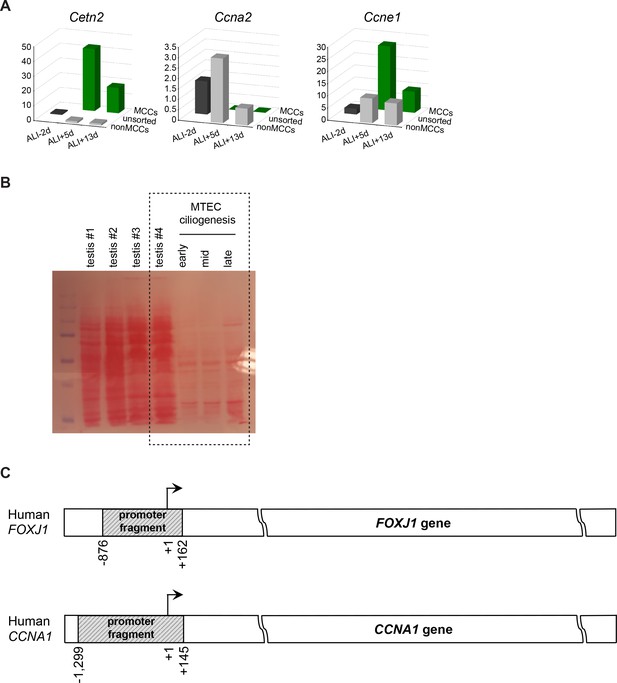
Ccna1 expression is restricted to MCCs and is a target of the MCC gene expression program.
(A) Realtime PCR values for Cetn2, Ccna2, Ccne1 expression in MCCs vs. non-MCCs (sorted from Foxj1-EGFP MTECs) shows MCC enrichment for Cetn2, non-MCC enrichment for Ccna2 and MCC and non-MCC expression for Ccne1. Levels were normalized to Gapdh expression and compared to values obtained from ALI + 0 d samples. (B) Ponceau-S stained membrane for Western blot in Figure 5B serving as loading control. The first four lanes (testis #1–4) are four identical testis lysate preps from four different mice. The boxed area indicates the portion of the blot presented in the main figure. (C) Schematics showing the human FOXJ1 and CCNA1 promoter genomic fragments tested in the Luciferase assay.
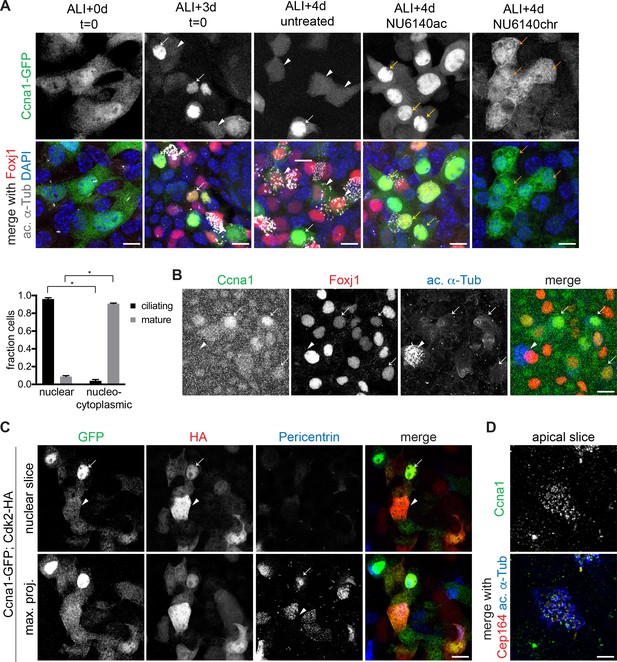
Ccna1 is nuclear and localization depends on Cdk activity during ciliogenesis.
(A) MTECs were infected with lentivirus encoding GFP-tagged Ccna1 at ALI-2d, treated with NU6140 from ALI + 3 to+4 d (NU6140ac, acute treatment) or from ALI + 0 to+4 d (NU6140chr, chronic treatment) then labeled at ALI + 4 d with DAPI (blue), GFP (green), Foxj1 (red) and ac. α-Tub (white) antibodies. The ALI + 3 and 4d untreated panels shows that Ccna1-GFP is nuclear in ciliating MCCs (white arrow) and present in low amounts everywhere in mature MCCs (white arrowhead). Ccna1-GFP is retained in the nucleus in cells arrested during ciliogenesis due to acute NU6140 treatment (yellow arrow). Ccna1-GFP remains nucleo-cytoplasmic in cells blocked from ciliogenesis due to chronic NU6140 treatment (orange arrow). MCC fraction was quantitated based on ac. α-Tub antibody labeling. Scale bar, 10 μm. *p<0.001 (B) ALI + 4 d MTECs labeled with Ccna1 (green), Foxj1 (red) and ac. α-Tub (blue) antibodies shows nuclear Ccna1 (arrow) in ciliating MCCs and no discernible specific signal in mature MCCs (arrowhead). Scale bar, 10 μm. (C) MTECs were infected with equal amounts of lentivirus encoding Ccna1-GFP and Cdk2-HA at ALI-2d, then labeled at ALI + 4 d with GFP (green), HA (red) and Pericentrin (blue) antibodies. A single image slice through the nuclear region (top panel) shows that Ccna1-GFP and Cdk2-HA are nuclear in ciliating MCCs (arrow) and nucleo-cytoplasmic in mature MCCs (arrowhead). Centriolar Pericentrin signal shows a tight cluster of centrioles (arrow) in a ciliating MCC and centrioles distributed at the apical surface in a mature MCC. Scale bar, 10 μm. (D) ALI + 4 d MTECs labeled at ALI + 4 d with Ccna1 (green), Cep164 (red) and ac. α-Tub (blue) antibodies shows a mature MCC with centriolar Ccna1 signal. Scale bar, 5 μm. .
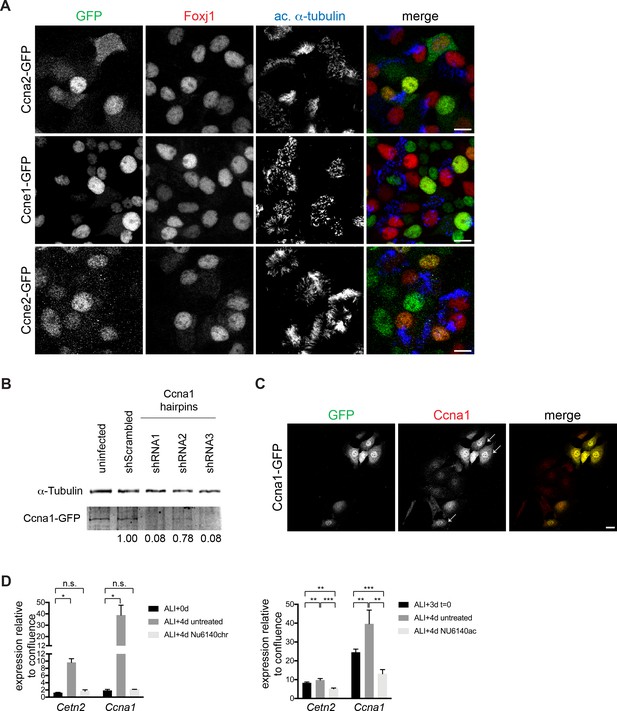
Cyclin localization and expression during motile ciliogenesis and Ccna1 antibody validation.
(A) MTECs were infected with GFP-tagged Ccna2, Ccne1 and Ccne2 lentiviruses at ALI-2d and labeled at ALI + 4 d with GFP (green), Foxj1 (red) and ac. α-Tub (blue) antibodies. Ccna2-GFP showed nuclear and cytoplasmic localization regardless of ciliogenesis state. Ccne1 and Ccne2-GFP constructs were nuclear in all cells. Scale bar, 10 μm. (B) mIMCD3 cells stably expressing Ccna1-GFP were infected with three different lentiviral Ccna1 RNAi constructs. Cell lysates were harvested 96 hr after infection, and Ccna1 levels were assessed by Western blotting with the Ccna1 and α-Tub antibodies. Protein levels normalized to the α-Tub loading control show that two hairpins (shRNAs 1 and 3) have nearly complete and one hairpin (shRNA2) has a modest level of Ccna1 depletion and support the specificity of the Ccna1 antibody for western blotting. (C) mIMCD3 cells transiently transfected with Ccna1-GFP were labeled with GFP and Ccna1 antibodies. Ccna1 antibody shows increased signal in GFP+ cells (arrow) to support the specificity of the Ccna1 antibody for immunolabeling. Scale bar, 20 μm. (D) Realtime PCR shows that Ccna1, like Cetn2, fails to increase under chronic (ALI + 0 to 4d, top) and decreases under acute (ALI + 3 to 4d, bottom) NU6140 treatment. Levels were normalized to Gapdh expression and compared to values obtained for MTECs at ALI-1d. n.s., not significant; *p<0.05; **p<0.01; ***p<0.001.
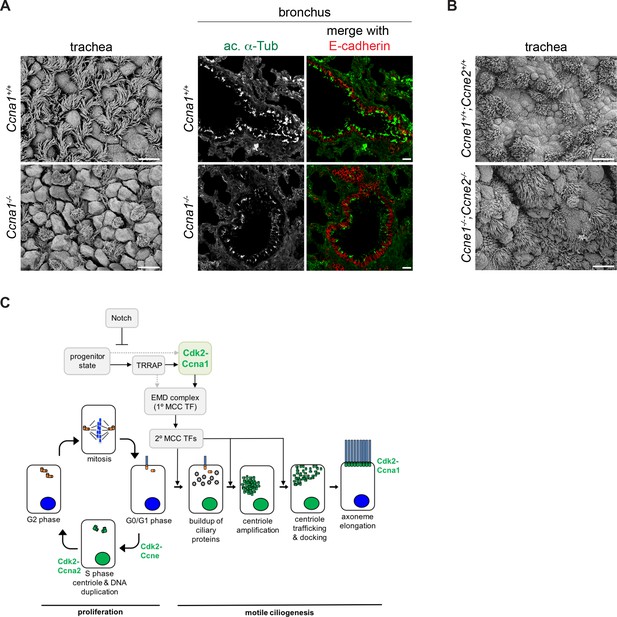
Ccna1, but not Ccne1;Ccne2 mutant mice have fewer MCCs.
(A) SEM of adult Ccna1-/- trachea (left panels) and ac. α-Tub (green) antibody labeling of bronchi (epithelium marked with E-cadherin antibody labeling, red) on cryosectioned lung tissue (right panels) show fewer MCCs with shorter, sparser cilia (bottom) compared to wildtype littermates (top). See Figure 7—figure supplement 1A for quantitation of MCC fraction for SEM. Scale bar, 10 μm for trachea, 20 μm for lung. (B) SEM of Ccne1-/-;Ccne2-/- trachea shows no difference in the number and distribution of MCCs (bottom) compared to wildtype littermates (top). Scale bar, 10 μm. (C) Schematic of Cdk2 function during cell cycle progression and proposed Cdk2-Ccna1 activity in the motile ciliogenesis pathway. Solid and dotted arrows linking the progenitor state to EMD activation represent alternative potential pathways that have yet to be distinguished. .
-
Figure 7—source data 1
Quantitation of MCC fraction in Ccna1 mutant and control mice.
- https://doi.org/10.7554/eLife.36375.017
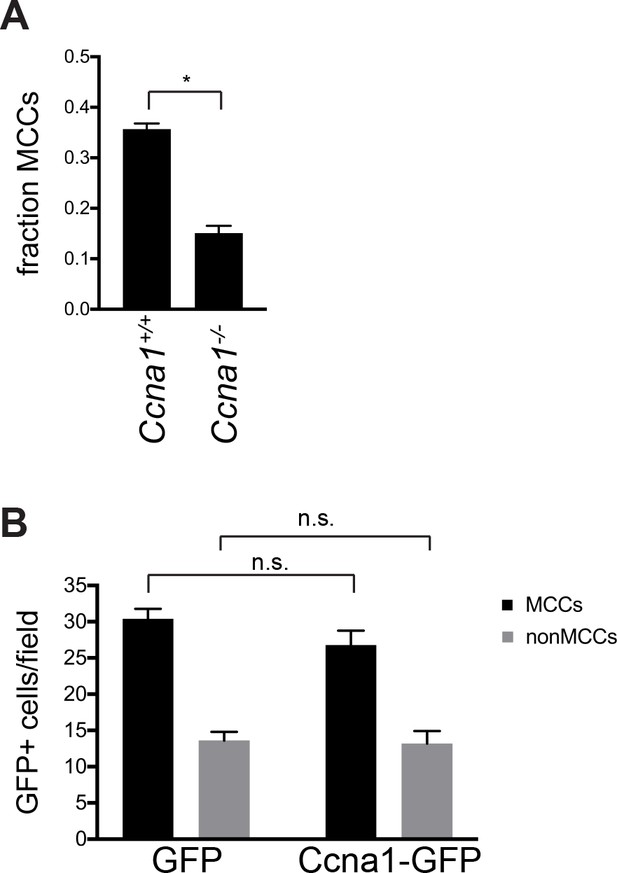
Ectopic Ccna1 expression is not sufficient to drive motile ciliogenesis.
(A) Quantitation of MCC fraction from n > 400 cells from SEM images of the middle of the ventral surface of the trachea from n = 3 mice. *p<0.0001 (B) MTECs were infected with GFP or GFP-Ccna1 lentivirus at ALI-2d and labeled at ALI + 4 d with GFP and ac. α-Tub antibodies. Quantitation of GFP+ MCCs and non-MCCs shows that Ccna1 expression is not sufficient to induce motile ciliogenesis. n.s., not significant.
Additional files
-
Supplementary file 1
Supplementary Table 1 Antibodies used.
Supplementary Table 2 Quantitative realtime PCR primers used. All primers amplify the mouse target unless otherwise specified. Supplementary Table 3 Statistical test and replicate information for data.
- https://doi.org/10.7554/eLife.36375.018
-
Transparent reporting form
- https://doi.org/10.7554/eLife.36375.019





