A resource for the Drosophila antennal lobe provided by the connectome of glomerulus VA1v
Figures
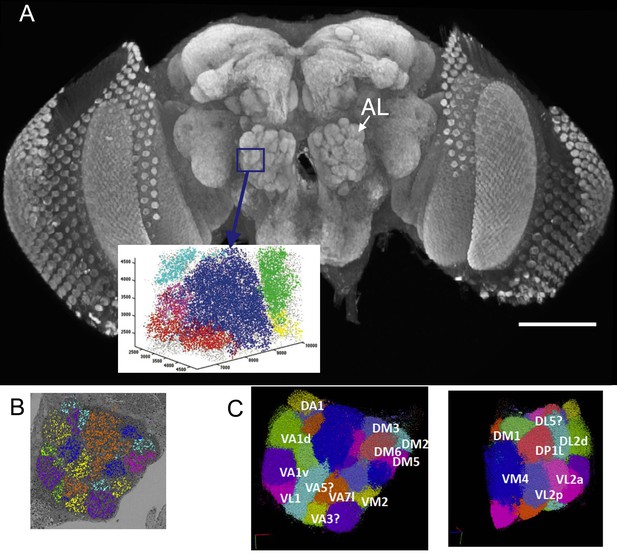
(A) The right antennal lobe of a female Drosophila melanogater wholemounted and immunolabelled with nc82 to detect Bruchpilot at synaptic sites, revealing the neuropiles of the brain.
These included the glomeruli of the antennal lobe (AL). Va1v of the right antennal lobe (blue) is enclosed by a box in A, identified in synapse cloud images. Scale bar 100 µm. (B) Image of a single plane with synapses identified as clouds by synapse detection on a corresponding FIB-SEM image stack, enclosing most of the right antennal lobe within which different glomeruli are identified and colour coded. Glomerular borders are visible in this single image from local rarefactions in the density of recognized synapse puncta. (C) A cloud of synaptic puncta in two image planes parallel and corresponding to the one in (B), with different glomeruli – including VA1v (left panel) – identified.

Single grey-scale image of the densely reconstructed glomerulus VA1v with colour-coded profiles of different cells (key).
Confirming the denseness of reconstruction, note that essentially all profiles are labelled, the few remaining dark grey belonging to orphan elements. Most profiles fall in the range of 0-5-1.5µm in diameter, with larger diameter axons giving rise to tiny dendritic neurites. Scale bar: 5 µm.
-
Figure 2—source data 1
List of quantitative features for all cells of the dataset.
Name - putative name based on Tanaka et al., 2012; Cell_ID - unique id given by NeuTu program; soma - location of soma, or soma tract (t); vol – volume of neurite in glomerulus; T-bars - number of presynaptic ribbons; Pre - targets of pre-synapse; Post – postsynaptic site; PSDs per T-bar – Average number of targets of pre-synapse; pre/post – ratio of targets of pre-synapses to postsynaptic site; T-bar/vol – number of T-bars per neurite vol (µm−3). Figure illustrates designation of pre- and postsynaptic sites.
- https://doi.org/10.7554/eLife.37550.005
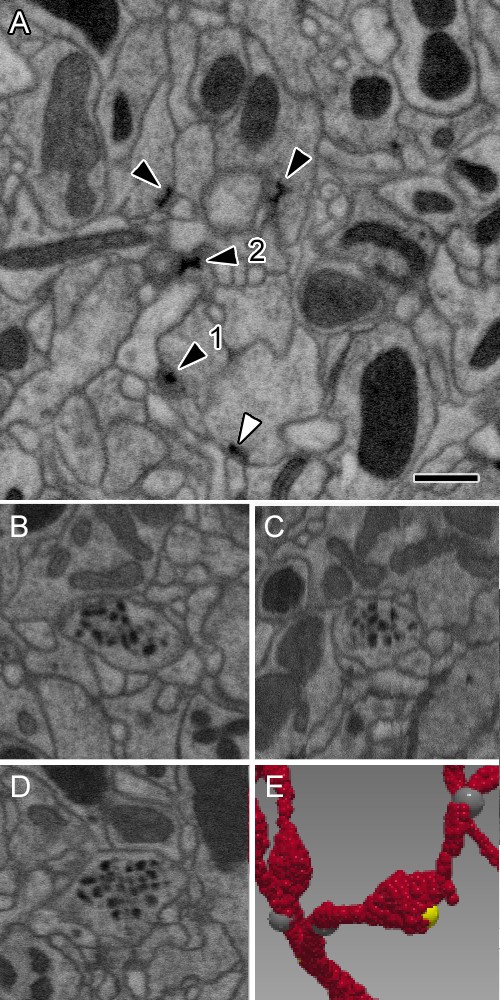
Representative synaptic profiles seen in FIB-SEM at 8 nm resolution.
(A) Five electron-dense profiles of presynaptic sites, four in ORNs and one (open arrowhead) in an LN, reveal a range of shapes, from a clear T-shape, in canonical cross section (1), to cruciform (2), in an en face view of the pedestal, with a range of other profiles that cut the organelle in different planes. Unifying their common identity, all have the same electron density that is clearly visible after FIB-SEM imaging. Postsynaptic densities are not well resolved. The profiles exhibit a wide range of shapes because the neurites contributing them are not aligned, as are the columns of the medulla (Takemura et al., 2015) and mushroom body output lobes (Takemura et al., 2017), and other neuropiles analysed with this imaging method. (B–D) Single neurite profile with ~84 nm diameter dense-core vesicles (dcv) viewed in three orthogonal planes, revealing each dcv as approximately circular in all three planes, and thus a sphere. (E) Same neurite reconstructed to show a varicosity with a single presynaptic site (yellow). Scale bars: 500 nm.
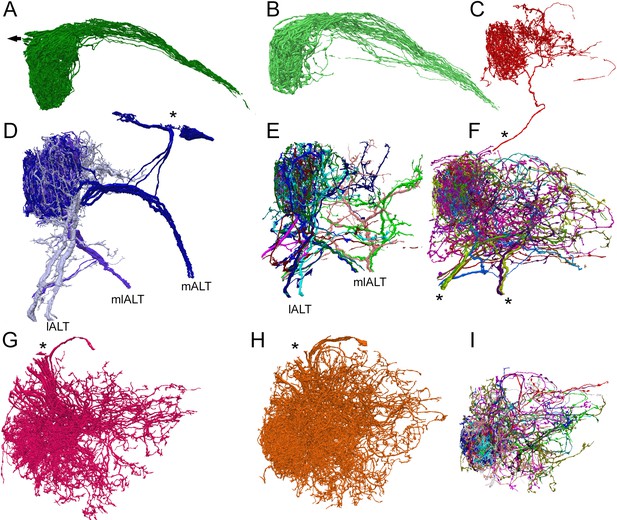
Reconstructions of the three main types of antennal lobe neurons in glomerulus VA1v, for comparison with published single-cell reporter expression lines (Tanaka et al., 2012).
Dorsal (A–F) and frontal (G–I) views. (A, B) Composite of 51 ipsilateral (A) and 56 contralateral (B) ORNs. Individual cells shown in Figure 4—source data 4 (see also library of cell types in Tobin et al., 2017, their Figure 1—figure supplement 1). Arrow in (A) represents neuron path to antennal nerve. (C) A single anomalous LN (possibly LN2V in Figure 2B in Tanaka et al., 2012). Asterisk shows path to cell body beyond region of segmentation. (D) The three mPN1 (dark purple) are monoglomerular in Va1v with axons that exit via the medial tract mALT (cf Figure 3A in Tanaka et al., 2012). Soma locations marked with an asterisk. Two monoglomerular mlPN1 cells (intermediate purple) with axons that exit via the mediolateral tract mlALT (cf Figure 4A in Tanaka et al., 2012). Three lPN4 (light purple) that arborize in Va1v and its sister glomerulus Va1d, have axons that exit via the lateral tract lALT (cf Figure 6D2 in Tanaka et al., 2012). (E) Additional multiglomerular PN types. For all PNs see Figure 4—source data 2. (G) 17 LN1 cells with somata in the dorsolateral cortex (cf Figure 2A in Tanaka et al., 2012). (H) 24 LN2L cells (cf Figure 2B in Tanaka et al., 2012). (F, I) Composite images of other cells identified as LNs of types LN3-6 and six multiglomerular local interneurons (LNs) differ in the position of their somata and the incoming tract of their axon; these are not illustrated by Tanaka et al., 2012. For a library of densely reconstructed cell types that arborize in VA1v, see , Figure 4—source data 1, Figure 4—source data 2, Figure 4—source data 3, Figure 4—source data 4 ); most are incompletely traced to other glomeruli.
-
Figure 4—source data 1
Library of reconstructed ORNs.
- https://doi.org/10.7554/eLife.37550.008
-
Figure 4—source data 2
Library of reconstructed PNs, some partially so.
- https://doi.org/10.7554/eLife.37550.009
-
Figure 4—source data 3
Library of partially reconstructed LNs.
- https://doi.org/10.7554/eLife.37550.010
-
Figure 4—source data 4
Library of other reconstructed cells.
- https://doi.org/10.7554/eLife.37550.011
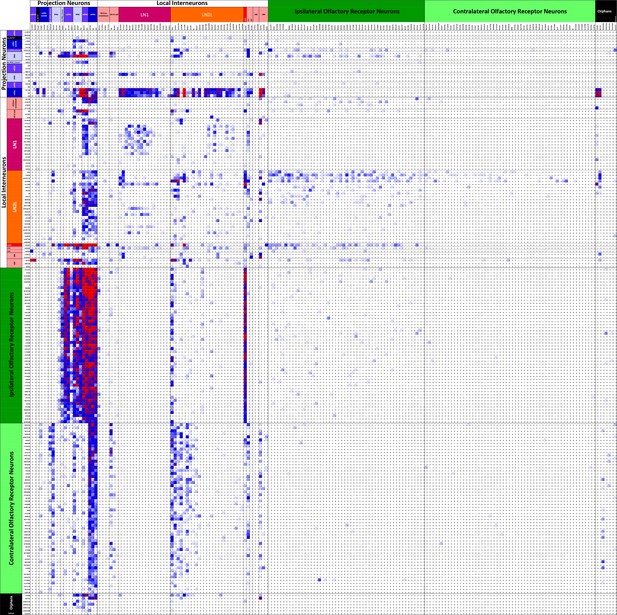
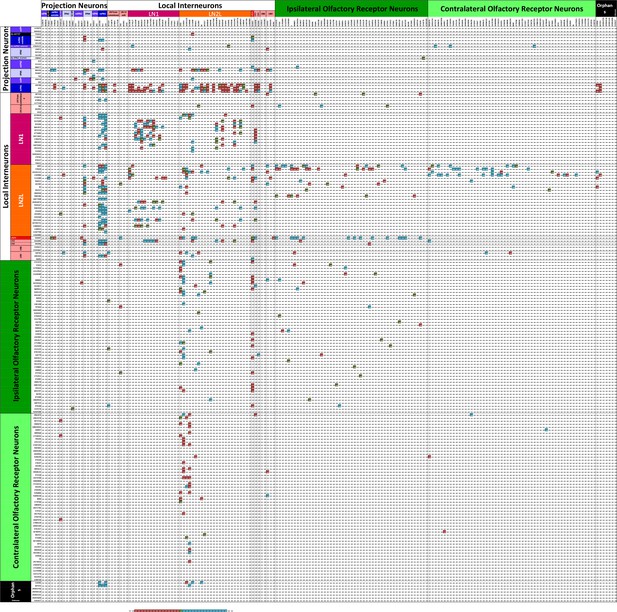
Connectivity matrix of VA1v cell types.
Register of cells with presynaptic sites (x axis, ordinate) plotted against the same cells having postsynaptic sites and colour-coded intercepts denoting the number of synaptic contacts between each pair of cell types (key), and thus the anatomical strength of their connection. Cells are arranged from the top left origin as, first, outputs (PNs), then interneurons (LNs), and finally inputs (ORNs) and further organized within those groups by the particular cell. Among the total of 192 cells, dense pathways occupy few intercepts, mostly concentrated in ORN to PN, and PN to LN intercepts. Only cells with more than 50 pre- or postsynaptic contacts are included. For the complete matrix, as a spreadsheet, see Figure 5—source data 1.
-
Figure 5—source data 1
Connectivity matrix as an Excel spreadsheet file for all 192 antennal lobe glomerulus VA1v cells having >50 contacts.
Data are the same as contribute to the matrix in Figure 5, but presented cell by cell. Register of cells with presynaptic sites (x axis, ordinate) plotted against the same cells having postsynaptic sites and colour-coded intercepts denoting the number of synaptic contacts between each pair (key), and thus the anatomical strength of their connection. Cells are arranged from the top left origin as, first, outputs (PNs), then interneurons (INs), and finally inputs (ORNs), and further organized within those groups by the particular cell. Among the total of 192 cells, dense pathways occupy few intercepts, mostly concentrated in ORN to PN, and PN to LN intercepts. Only cells with more than 50 pre- or postsynaptic contacts are included.
- https://doi.org/10.7554/eLife.37550.014
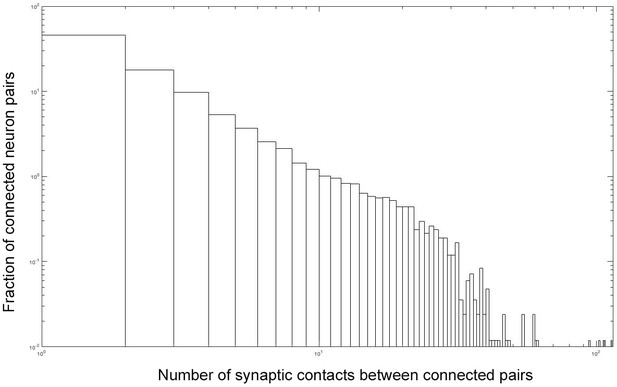
Fraction (ordinate, log) of pathway strengths between connected neuron pairs (abscissa, log) from data in the connectivity matrix (Figure 5—source data 1).
More than 32,700 synaptic partnerships exist between 8221 pairs of which 45% constitute one, 18% two, and 9.9% three, synaptic contacts.
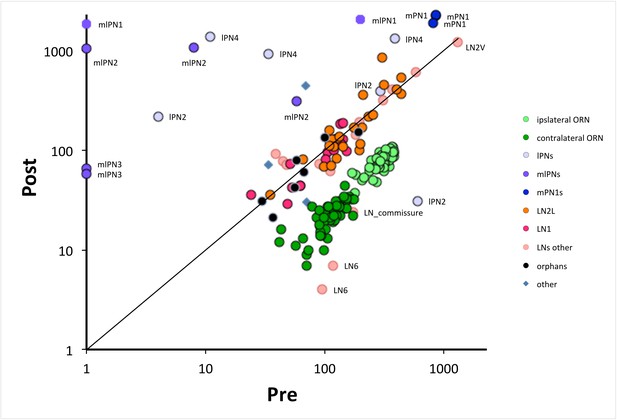
The numbers of pre- and postsynaptic sites for each cell, log-log plot.
Only cells with >50 contacts are shown. Cells are colour-coded for their type (ORN, LN, PN: key); ORNs are either ipsi or contralateral, PNs are labeled as well as anomalous LNs, which do not cluster with other LNs. Cells can be identified from their pre:post synapse ratio. Almost all LNs have equal numbers of pre and postsynaptic contacts; ORNs have more presynaptic than postsynaptic contacts, with a ratio of 3.9 ± 0.6 for ipsilateral and 5.3 ± 1.5 for contralateral (Figure 2—source data 1). PNs are predominately postsynaptic but have more variable pre/post ratios as well as number of contacts than the other two cell types; mPN1s have most presynaptic sites with an average ratio of 0.4 ± 0.03 and mlPN2s are almost completely postsynaptic with a ratio of 0.06 ± 0.11; some PNs have >1000 contacts. Unknown cells, some orphans (not traced to identified neurons), cluster with local neurons, their suspected source. A few cells are anomalous, some (especially LNs) falling outside their cluster are possibly mis-assigned.
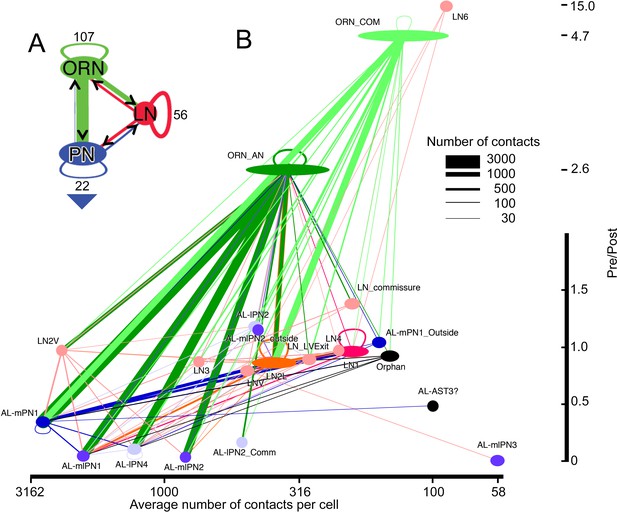
Network diagram for all cells and cell types within glomerulus VA1v having >50 pre/post contacts and reported in the overall matrix (Figure 5).
Edges show connections by classes of cells, not those of their individual constituent neurons. The width of each pathway arrow shows the number of anatomical synaptic contacts (key). (A) Aggregate network for three major classes of VA1v cells. The direction of pre- to postsynaptic contact is shown by the arrow and colour of the line connection between synaptic partner cells. ORN inputs to PNs are numerically predominant. Most LNs are predicted to be inhibitory, and provide pathways that integrate both ORN and PN activity; LNs also form their own network. Numbers indicate cells per class. Recurrent loops are feedback between different neurons of the same class. Large arrowhead indicates PN output to higher olfactory centres in the mushroom body and lateral horn. (B) Network diagram, as in A, plotted for subgroups of their type (see Figure 4 and Figure 4—Source data 4). Node width represents cell number per group, colour-coded as in Figure 6. Vertical: pre-/post- axis, note that this is interrupted at 2.0; horizontal: number of connections per cell plotted on a log scale.
Matrix of the 192 cells in Figure 5 highlighting those pairs that are strongly reciprocal.
Pairs have connections of at least three synapses in the pre:post and post:pre directions so having at least six pre- and postsynaptic contacts in total. Each intercept shows the ratio for the pair of partner neurons of its pre- to post- contacts. Ratios of between 0.25 and 4 are highlighted in a graded fashion (see scale). These represent those partner neurons at which the numerical strength of pre and postsynaptic pathways is at least 4:1, where four represents a balance in favour of the presynaptic pathway, 0.25 represents a balance in favour of the postsynaptic pathway, and a value of 1.0 represents equal numerical balance in both directions. Numerical values are reflected about the diagonal of the matrix, changed inversely between the two sectors, 0.25 in one direction corresponding to four in the other.
-
Figure 8—source data 1
Reciprocity matrix for all 192antennal lobeglomerulus VA1v cells having >50 contacts.
- https://doi.org/10.7554/eLife.37550.018
Tables
Nomenclatures for antennal lobe neurons
https://doi.org/10.7554/eLife.37550.002| *Tanaka et al. (2012) | Gal four enhancer -trap strain | Cell count | Va1v cells, this study | |
|---|---|---|---|---|
| LN1 | NP1227 | 18 | 17 | |
| LN2L | NP2426 | 40 | 24 | |
| LN2V‡ | NP2427 | 3 | 1 | |
| LN3 | NP1326 | 4.5 | 1 | |
| LN4 | NP842 | 3 | 1 | |
| LN6 | NP1587 | 1 | 2 | |
| mPN1† | 59 | 3 | ||
| mlPN1 | NP5288 | 9.5 | 2 | |
| mlPN2 | 23 | 3 | ||
| mlPN3 | 3 | 2 | ||
| t3PN1 | 2 | |||
| t4PN1 | 5.5 | |||
| lPN2 | 9.3 | 3 | ||
| lPN4 | 1 | 3 | ||
| AST1 | 1 | |||
| AST2 | 1 | |||
| AST3 | 1 | 1 | ||
| MBDL1 | 1 | |||
| VUMa5 | 1 | |||
| 5HT | 1 | 1§ | ||
| Cells not reconciled with those reported by Tanaka et al. (2012) | ||||
| LNV | 3 | |||
| LN_LVExit | 3 | |||
| LN_commissure | 4 | |||
| Total | 10 | |||
| Ipsilateral ORNs | 51 | |||
| contralateral ORNs | 56 | |||
-
*Column lists all those cells reported by Tanaka et al. (2012) to enter Va1v
**†Tanaka labelled only non-specifically for several cell classes including those in Va1v
-
***‡Tanaka did not find in Va1v at all
****§Not included in matrix
Additional files
-
Transparent reporting form
- https://doi.org/10.7554/eLife.37550.019





