Patronin governs minus-end-out orientation of dendritic microtubules to promote dendrite pruning in Drosophila
Figures
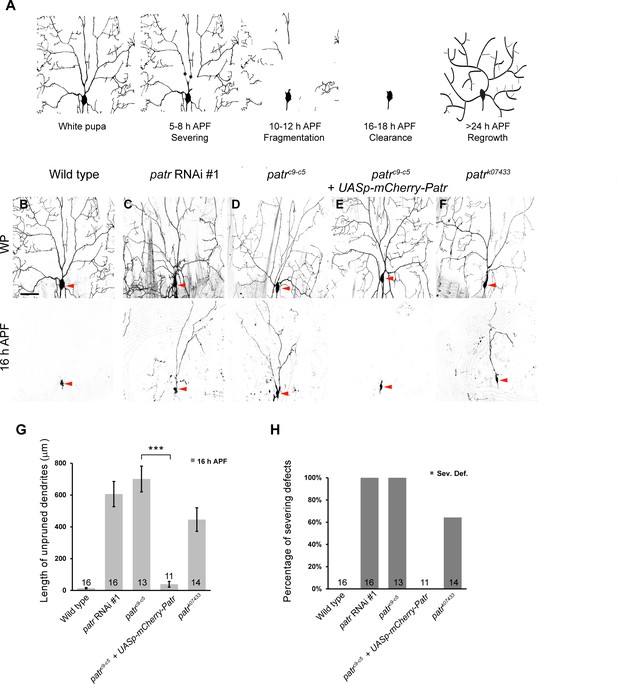
Patronin is required for dendrite pruning in ddaC neurons.
(A) A schematic representation of dendrite pruning in ddaC neurons. (B–F) Live confocal images of ddaC neurons expressing mCD8-GFP driven by ppk-Gal4 at WP and 16 hr APF. While the wild-type neurons pruned all the dendrites (B), ddaC neurons overexpressing patronin RNAi #1 (C), patroninc9-c5 (D) or patronink07433 (F) MARCM ddaC clones exhibited simple arbors at WP stage and dendrite pruning defects at 16 hr APF. Low-level expression of mCherry-Patronin under the control of the UASp promoter fully restored the elaborate arbors at WP and rescued the pruning defects at 16 hr APF in patroninc9-c5 MARCM ddaC clones (E). Red arrowheads point to the ddaC somas. (G) Quantification of total length of unpruned ddaC dendrites at 16 hr APF. (H) Quantification of severing defects at 16 hr APF. Scale bar in (B) represents 50 μm. Error bars represent SEM. The number of samples (n) in each group is shown on the bars. ***p<0.001 as assessed by two-tailed Student’s T test.
-
Figure 1—source data 1
Extended statistical data as Microsoft Excel spreadsheet.
- https://doi.org/10.7554/eLife.39964.005
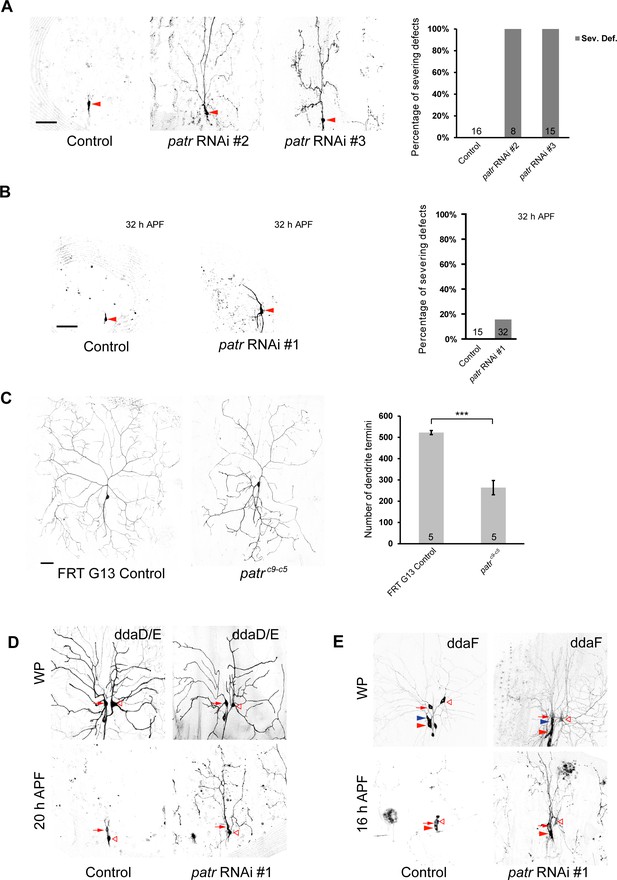
Patronin is required for dendrite pruning and arborization in sensory neurons.
(A–E) Live confocal images of da neurons expressing mCD8-GFP at w3L, WP, 16 hr and 20 hr, 32 hr APF. (A) While the control neurons pruned all the dendrites at 16 hr APF, ddaC neurons overexpressing patronin RNAi #2 or patronin RNAi #3 by ppk-Gal4 exhibited dendrite pruning defects at 16 hr APF. Red arrowheads point to the ddaC somas. Quantification of the severing defects in control and mutant ddaC neurons at 16 hr APF. (B) ddaC neurons overexpressing patronin RNAi #1 by ppk-Gal4 pruned away most of their dendrites at 32 hr APF. Red arrowheads point to the ddaC somas. Quantification of the severing defects in control and mutant ddaC neurons at 32 hr APF. (C) Compared to FRT G13 control, patroninc9-c5 ddaC MARCM clones exhibited simple dendrite arbors at wL3 stage. Quantification of number of dendrite termini in control and patroninc9-c5 ddaC MARCM clones. (D) While the control class I ddaD/ddaE neurons labelled by Gal42-21-driven mCD8-GFP pruned their larval dendrites at 20 hr APF, patronin RNAi #1 ddaD/ddaE neurons failed to do so. Red arrows point to the ddaD somas and open arrowheads to the ddaE somas. (E) Like control ddaF neurons, patronin RNAi #1 ddaF neurons labelled by Gal4109(2)80-driven mCD8-GFP were also eliminated by 16 hr APF. Red arrowheads point to the ddaC somas, and blue arrowheads point to the ddaF somas. Error bars represent S.E.M. The number of samples (n) in each group is shown on the bars. Scale bars represent 50 µm. ***p<0.001 as assessed by two-tailed Student’s t-test.
-
Figure 1—figure supplement 1—source data 1
Extended statistical data as Microsoft Excel spreadsheet.
- https://doi.org/10.7554/eLife.39964.004
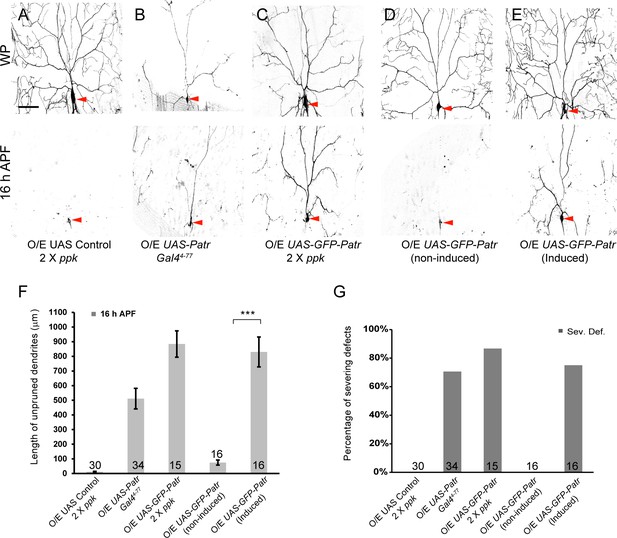
Overexpression of Patronin causes dendrite pruning defects in ddaC neurons.
(A–E) Live confocal images of ddaC neurons expressing mCD8-GFP driven by ppk-Gal4, Gal44-77 or ppk-CD4-tdGFP at WP and 16 hr APF. While ddaC neurons overexpressing the UAS control construct pruned all the dendrites (A), ddaC neurons overexpressing Patronin (B) driven by Gal44-77 or GFP-Patronin (C) driven by two copies of ppk-Gal4 at a higher level exhibited simple arbors at WP stage and consistent dendrite pruning defects at 16 hr APF. ddaC neurons, in which GFP-Patronin was expressed by GeneSwitch-Gal4-2295 in RU486-induced conditions (E), exhibited normal arbors at WP stage but severe dendrite pruning defects at 16 hr APF, compared to those in non-induced conditions (D). Red arrowheads point to the ddaC somas. (F) Quantification of total length of unpruned ddaC dendrites at 16 hr APF. (G) Quantification of severing defects at 16 hr APF. Scale bar in (A) represents 50 μm. Error bars represent SEM. The number of samples (n) in each group is shown on the bars. ***p<0.001 as assessed by two-tailed Student’s T test.
-
Figure 2—source data 1
Extended statistical data as Microsoft Excel spreadsheet.
- https://doi.org/10.7554/eLife.39964.009
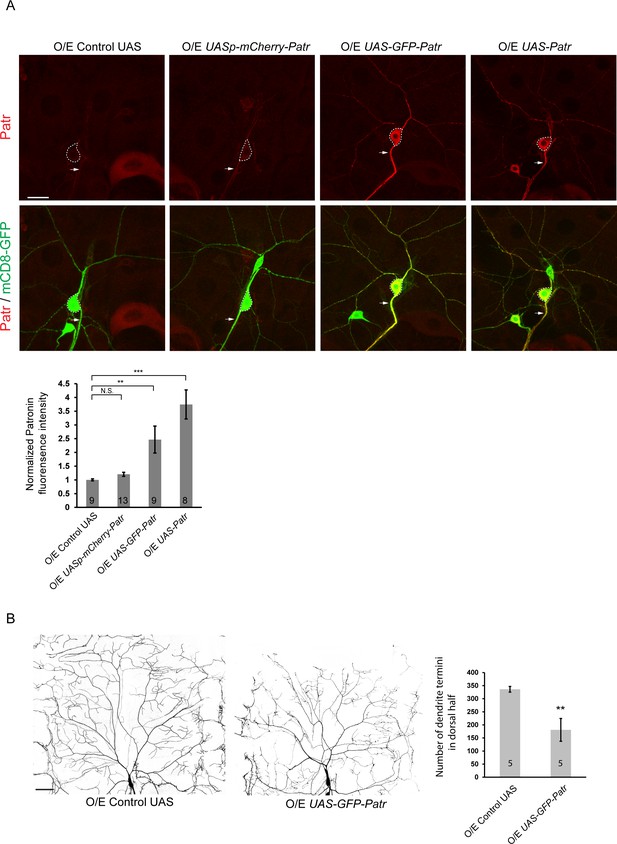
Overexpression of Patronin causes dendrite arborization defects in ddaC neurons.
(A) Confocal images of ddaC neurons expressing mCD8-GFP and immunostained for anti-Patronin at w3L stage. ddaC somas are marked by dashed lines, and axons by arrows. Endogenous Patronin was detected in the ddaC neurons expressing an unrelated UAS control. Exogenous Patronin was expressed at a low level in ddaC neurons using a UASp-mCherry-Patronin transgene, but at much higher levels using UAS-GFP-Patronin or UAS-Patronin transgenes. Quantification of normalized Patronin fluorescence intensity in the somas of ddaC neurons. (B) Live confocal images of ddaC neurons expressing mCD8-GFP driven by two copies of ppk-Gal4 at w3L stage. Compared to the control neurons, GFP-Patronin-overexpressing ddaC neurons showed simple dendrite arbors at wL3 stage. Quantification of number of dendrite termini in the dorsal half of ddaC neurons. Error bars represent SEM. The number of neurons (n) in each group is shown on the bars. Scale bars in (A) and (B) represent 20 μm and 50 μm, respectively. N.S., not significant; **p<0.01; ***p<0.001 as assessed by one-way ANOVA test or two-tailed Student’s T test.
-
Figure 2—figure supplement 1—source data 1
Extended statistical data as Microsoft Excel spreadsheet.
- https://doi.org/10.7554/eLife.39964.008
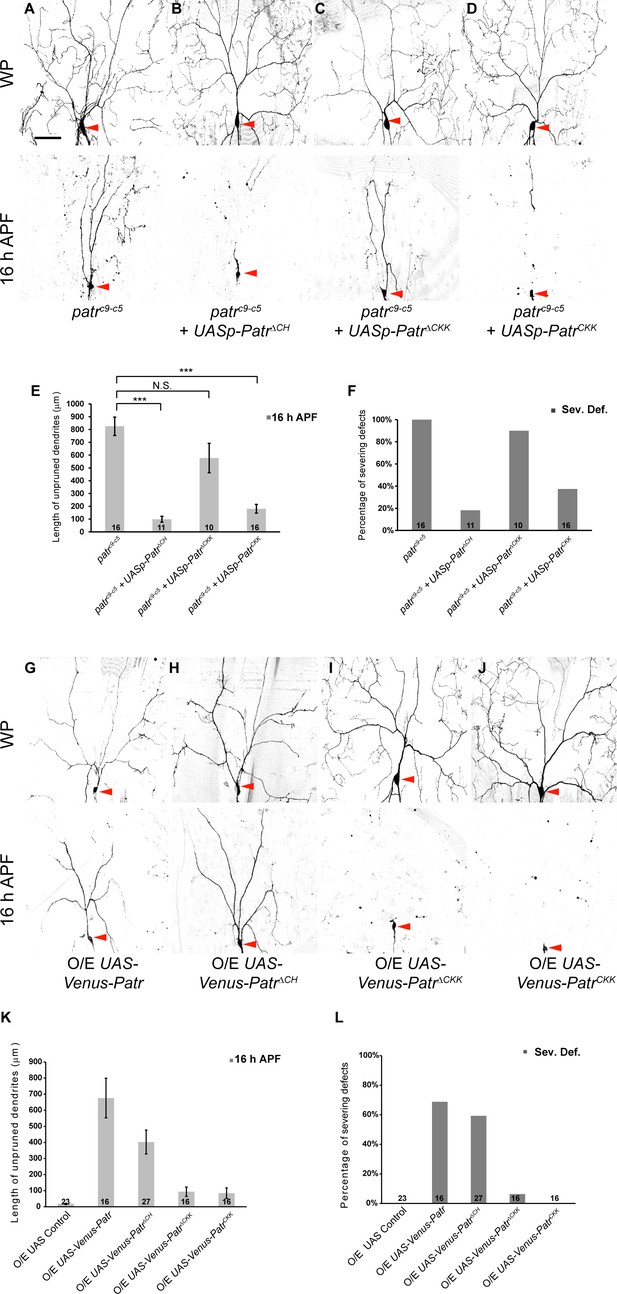
The CKK domain is important for Patronin to govern dendrite pruning.
(A–D, G–J) Live confocal images of ddaC neurons expressing mCD8-GFP driven by ppk-Gal4 or Gal44-77 at WP and 16 hr APF. Low-level expressions of PatroninΔCH (B) and PatroninCKK (D), but not PatroninΔCKK (C), strongly rescued the dendrite arborization defects at WP stage and the pruning defects at 16 hr APF in patroninc9-c5 (A) MARCM ddaC clones. (G–J) ddaC neurons overexpressing Venus-Patronin (G) or Venus-PatroninΔCH (H) by Gal44-77 exhibited simple arbors at WP stage and the dendrite pruning defects at 16 hr APF. However, overexpression of Venus-PatroninΔCKK (I) or Venus-PatroninCKK (J) had negligible effect on dendrite arborization and pruning. (E, K) Quantification of total length of unpruned ddaC dendrites at 16 hr APF. (F, L) Quantification of severing defects at 16 hr APF. Scale bar in (A) represents 50 μm. Error bars represent SEM. The number of samples (n) in each group is shown on the bars. N.S., not significant; ***p<0.001 as assessed by one-way ANOVA test.
-
Figure 3—source data 1
Extended statistical data as Microsoft Excel spreadsheet.
- https://doi.org/10.7554/eLife.39964.013
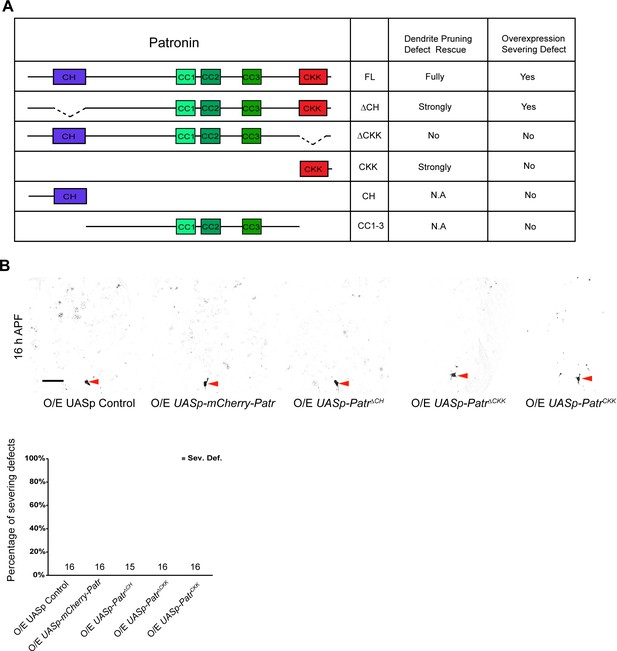
Structure-function analysis of the Patronin protein.
(A) A schematic representation of full-length and truncated Patronin proteins and a summary of structure-function analysis of Patronin. Patronin contains multiple domains: a calponin homology (CH) domain at the N-terminus, three coiled-coil (CC) repeats at the central region, and a signature CKK domain at the C-terminus. N.A., not available. (B) Confocal images of ddaC neurons expressing mCD8-GFP driven by two copies of ppk-Gal4 at 16 hr APF. Similar to the control neurons, UASp-mCherry-Patr, UASp-PatroninΔCH, UASp-PatroninΔCKK or UASp-PatroninCKK-expressing ddaC neurons pruned away their larval dendrites by 16 hr APF. Red arrowheads point to the ddaC somas. Quantification of the severing defects in the control and mutant ddaC neurons at 16 hr APF. The number of samples (n) in each group is shown on the bars. Scale bar in (B) represents 50 µm.
-
Figure 3—figure supplement 1—source data 1
Extended statistical data as Microsoft Excel spreadsheet.
- https://doi.org/10.7554/eLife.39964.012
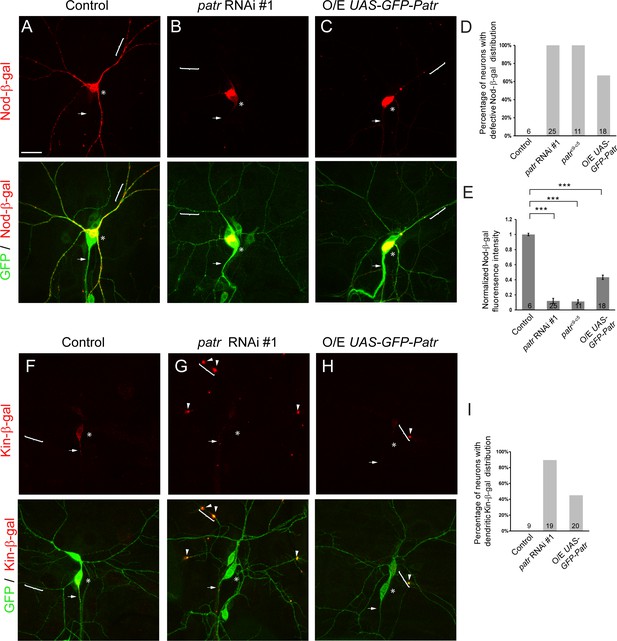
Patronin is required for proper distribution of dendritic and axonal MT markers.
(A–C, F–H) Confocal images of ddaC neurons expressing mCD8-GFP, Nod-β-gal or Kin-β-gal and immunostained for β-galactosidase at wL3 stage. ddaC somas are marked by asterisks, axons by arrows and dendrites by curly brackets. Nod-β-gal levels were reduced in the dendrites but enriched in the somas in patronin RNAi #1 (B) or GFP-Patronin-overexpressing (C) ddaC neurons, compared to the control neurons (A). (D) Quantification of the percentage of neurons with defective Nod-β-gal distribution. (E) Quantification of normalized Nod-β-gal intensity in the dendrites. Moreover, Kin-β-gal mis-localized to the dendrites in both patronin RNAi #1 (G) or GFP-Patronin-overexpressing (H) ddaC neurons, compared to the control neurons (F). Arrowheads point to ectopic Kin-β-gal aggregates in the dendrites. (I) Quantification of the percentage of neurons with dendritic Kin-β-gal distribution. Scale bar in (A) represents 20 μm. Error bars represent SEM. The number of samples (n) in each group is shown on the bars. ***p<0.001 as assessed by one-way ANOVA test.
-
Figure 4—source data 1
Extended statistical data as Microsoft Excel spreadsheet.
- https://doi.org/10.7554/eLife.39964.016
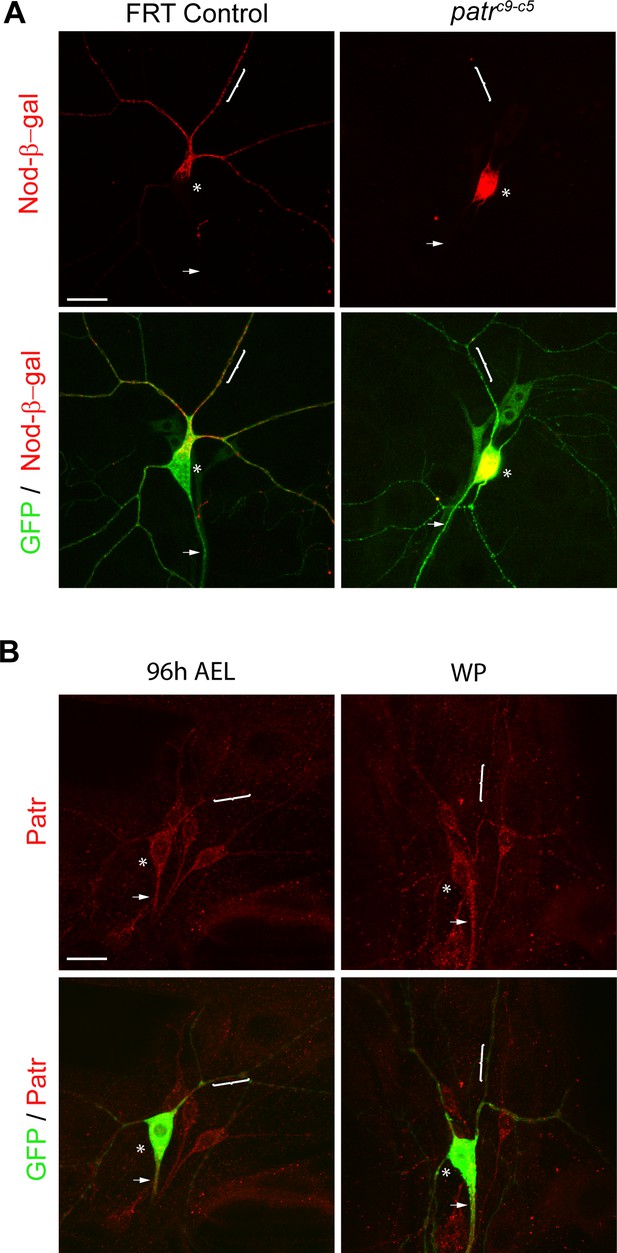
Patronin is required for proper distribution of the MT marker Nod-β-gal in ddaC dendrites.
(A) Confocal images of ddaC neurons expressing mCD8-GFP, Nod-β-gal and immunostained for β-galactosidase at wL3 stage. Nod-β-gal levels were reduced in the dendrites but enriched in the somas of patroninc9-c5 MARCM clones, compared to those in the control clones. (B) Confocal images of ddaC neurons expressing mCD8-GFP and immunostained for anti-Patronin at 96 hr AEL and WP stages. Patronin localized to the dendrites, axons and somas at both stages. ddaC somas are marked by asterisks, axons by arrows and proximal dendrites by curly brackets. Scale bars represent 20 μm.
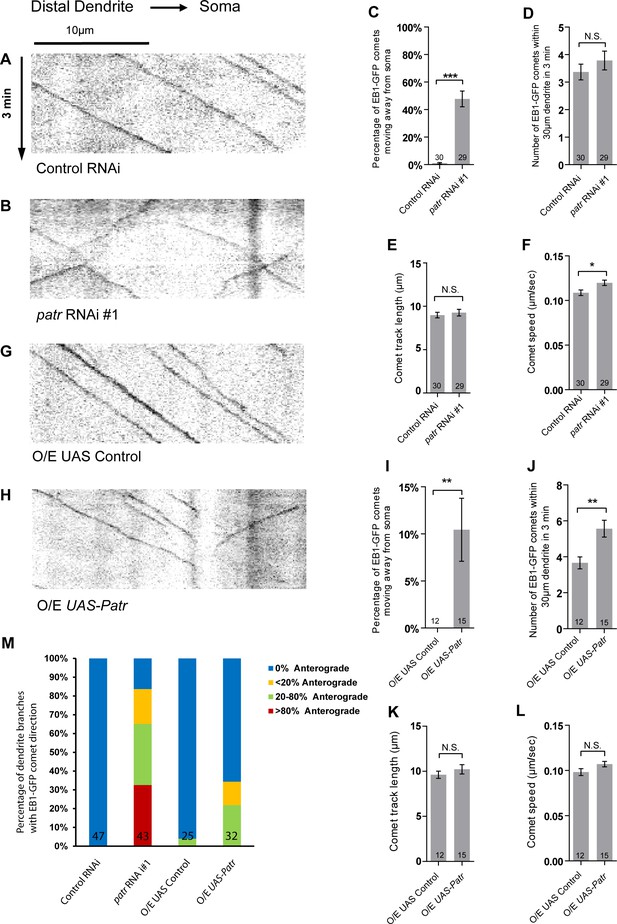
Patronin is required for the minus-end-out orientation of dendritic MTs in ddaC neurons.
(A–B, G–H) Representative kymographs of EB1-GFP comets in control or mutant ddaC dendrites. In control RNAi (A) or UAS control (G) ddaC neurons, dendritic EB1-GFP comets predominantly moved retrogradely towards the somas. However, in patronin RNAi #1 (B) or Patronin-overexpressing (H) ddaC neurons, dendritic EB1-GFP comets moved bidirectionally (anterogradely and retrogradely). Horizontal arrow indicates the direction towards the soma. Scale bar in (A) represents 10 μm, and each movie was taken for 3 min. EB1-GFP was expressed by Gal44-77. (C, I) Quantification of percentage of dendritic EB1-GFP comets moving away from soma (anterogradely). (D, J) Quantification of average numbers of EB1-GFP comets along 30 µm of dendrites within 3 min. (E, K) Quantification of average track length of EB1-GFP comets along 30 µm of dendrites within 3 min. (F, L) Quantification of average speed of EB1-GFP comets along 30 µm of dendrites within 3 min. (M) Quantification of the percentage of dendrite branches showing different levels of anterograde EB1-GFP comets. The number of dendrite branches (n) examined in each group is shown on the bars. Error bars represent SEM. N.S., not significant; *p<0.05; **p<0.01; ***p<0.001 as assessed by two-tailed Student’s T test.
-
Figure 5—source data 1
Extended statistical data as Microsoft Excel spreadsheet.
- https://doi.org/10.7554/eLife.39964.020
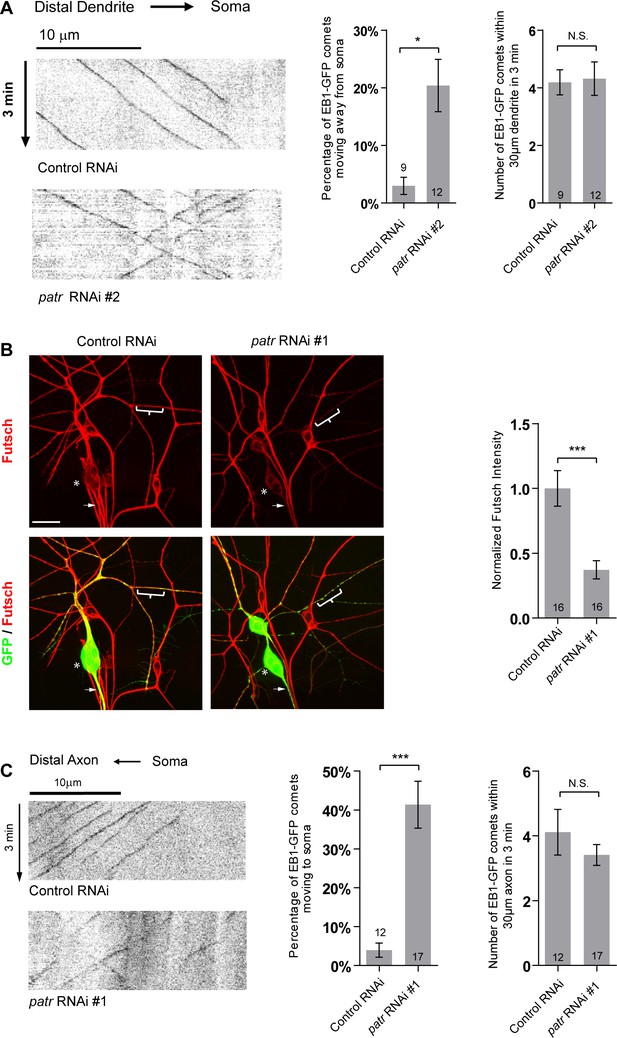
Patronin is required for dendritic and axonal MT orientations in ddaC neurons.
(A) Representative kymographs of EB1-GFP comets in control or mutant ddaC dendrites. In control RNAi ddaC neurons, dendritic EB1-GFP comets predominantly moved retrogradely towards the somas. However, in patronin RNAi #2 ddaC neurons, dendritic EB1-GFP comets moved bidirectionally (anterogradely and retrogradely). Horizontal arrow indicates the direction towards the soma. Scale bar represents 10 μm, and each movie was taken for 3 min. Quantification of percentage of dendritic EB1-GFP comets moving away from soma (anterogradely). Quantification of average numbers of EB1-GFP comets along 30 µm of dendrites within 3 min. (B) Confocal images of ddaC neurons expressing mCD8-GFP and immunostained for anti-Futsch antibody (22C10) at wL3 stage. ddaC somas are marked by asterisks, axons by arrows and dendrites by curly brackets. Curly brackets mark the dendritic regions where fluorescence intensity of Futsch were measured. Bar chart on the right depicts normalized Futsch intensity for 20 μm of major dendrite located 30 μm away from the soma. Scale bar represents 20 μm. (C) Representative kymographs of EB1-GFP comets in control or mutant ddaC axons. In the control neurons, axonal EB1-GFP comets predominantly moved away from the somas (anterogradely). In patronin RNAi #1 ddaC neurons, axonal EB1-GFP comets moved bidirectionally. Horizontal arrows indicate the direction towards the axons. Scale bar represents 10 μm, and each movie was taken for 3 min. Quantification of percentage of axonal EB1-GFP comets moving towards the somas (retrogradely). Quantification of average numbers of EB1-GFP comets along 30 µm of axons within 3 min. The number of neurons (n) in each group is shown on the bars. Error bars represent SEM. N.S., not significant; *p<0.05; ***p<0.001 as assessed by two-tailed Student’s T test.
-
Figure 5—figure supplement 1—source data 1
Extended statistical data as Microsoft Excel spreadsheet.
- https://doi.org/10.7554/eLife.39964.019
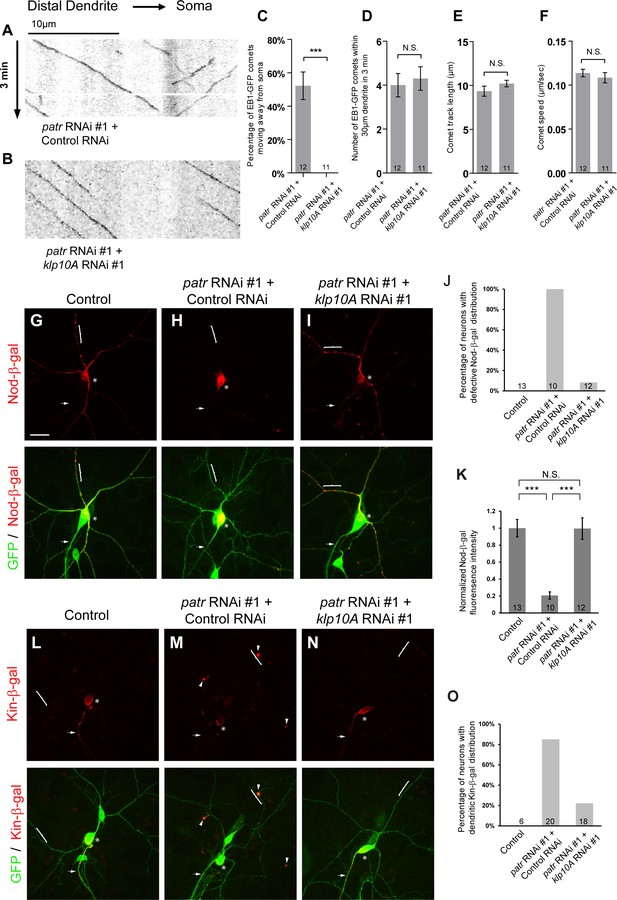
Attenuation of Klp10A, a kinesin-13 MT depolymerase, suppresses the patronin phenotypes in MT orientation in ddaC dendrites.
(A, B) Representative kymographs of EB1-GFP comets in control or mutant ddaC dendrites. In patronin RNAi #1 and control RNAi co-expressing ddaC neurons (A), dendritic EB1-GFP comets moved bidirectionally (anterogradely and retrogradely). However, knockdown of klp10A via RNAi #1 completely restored the retrograde movement of EB1-GFP comets in the patronin RNAi #1 ddaC dendrites (B). Horizontal arrow indicates the direction towards the somas. Scale bar in (A) represents 10 μm, and each movie was taken for 3 min. EB1-GFP was expressed by Gal44-77. (C) Quantification of percentage of dendritic EB1-GFP comets moving away from soma (anterogradely). (D) Quantification of average numbers of EB1-GFP comets along 30 µm of dendrites within 3 min. (E) Quantification of average track length of EB1-GFP comets along 30 µm of dendrites within 3 min. (F) Quantification of average speed of EB1-GFP comets. (G–I, L–N) Confocal images of ddaC neurons expressing mCD8-GFP, Nod-β-gal or Kin-β-gal and immunostained for β-galactosidase at wL3 stage. ddaC somas are marked by asterisks, axons by arrows and dendrites by curly brackets. Nod-β-gal levels were reduced in the dendrites but enriched in the somas of ddaC neurons co-overexpressing patronin RNAi #1 and control RNAi (H), compared to the control ddaC neurons (G). However, knockdown of klp10A (RNAi #1) almost completely restored dendritic distribution of Nod-β-gal in patronin RNAi #1 ddaC neurons (I). (J) Quantification of the percentage of neurons with defective Nod-β-gal distribution. (K) Quantification of normalized Nod-β-gal intensity in the dendrites. Moreover, Kin-β-gal mis-localization defects in patronin RNAi #1 ddaC neurons were drastically rescued by knockdown of klp10A (RNAi #1), compared to the patronin, control RNAi neurons (M). (O) Quantification of the percentage of neurons with dendritic Kin-β-gal distribution. Scale bar in (G) represents 20 μm. Error bars represent SEM. The number of samples (n) in each group is shown on the bars. N.S., not significant; ***p<0.001 as assessed by one-way ANOVA test or two-tailed Student’s T test.
-
Figure 6—source data 1
Extended statistical data as Microsoft Excel spreadsheet.
- https://doi.org/10.7554/eLife.39964.024
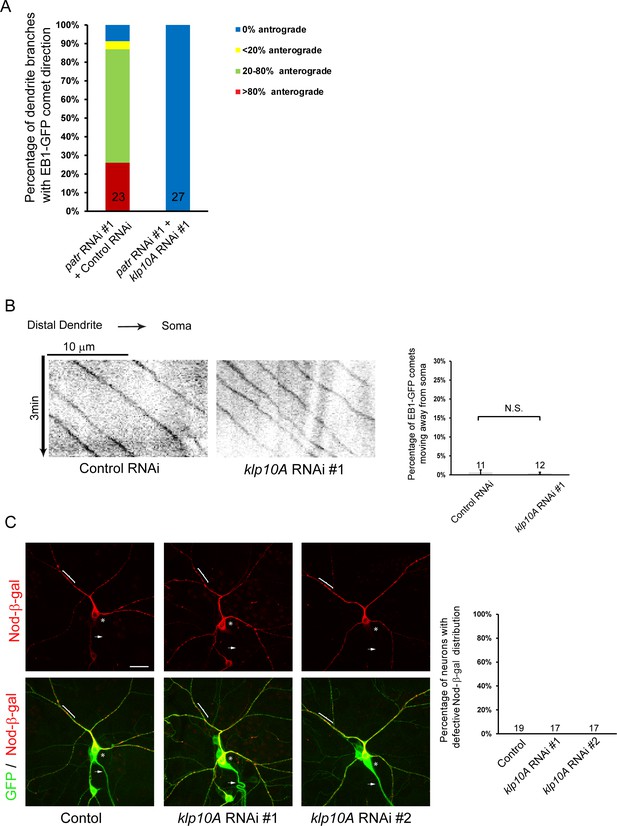
Attenuation of Klp10A, a kinesin-13 MT depolymerase, suppresses the patronin phenotype in MT orientation in ddaC dendrites.
(A) Quantification of the percentage of dendrite branches showing different levels of anterograde EB1-GFP comets in the control and mutant ddaC neurons. (B) Representative kymographs of EB1-GFP comets in control or mutant ddaC dendrites. In control RNAi and klp10A RNAi #1 ddaC neurons, dendritic EB1-GFP comets predominantly moved retrogradely towards the somas. Horizontal arrow indicates the direction towards the soma. Scale bar represents 10 μm, and each movie was taken for 3 min. EB1-GFP was expressed by Gal44-77. Quantification of percentage of dendritic EB1-GFP comets moving away from somas (anterogradely). (C) Confocal images of ddaC neurons expressing mCD8-GFP, Nod-β-gal and immunostained for β-galactosidase at wL3 stage. ddaC somas are marked by asterisks, axons by arrows and dendrites by curly brackets. Nod-β-gal was normally distributed in the control, klp10A RNAi #1 or klp10A RNAi #2 ddaC neurons. Quantification of the percentage of neurons with defective Nod-β-gal distribution. Scale bar in (C) represents 20 μm. The number of samples (n) in each group is shown on the bars. N.S., not significant, as assessed by two-tailed Student’s T test.
-
Figure 6—figure supplement 1—source data 1
Extended statistical data as Microsoft Excel spreadsheet.
- https://doi.org/10.7554/eLife.39964.023
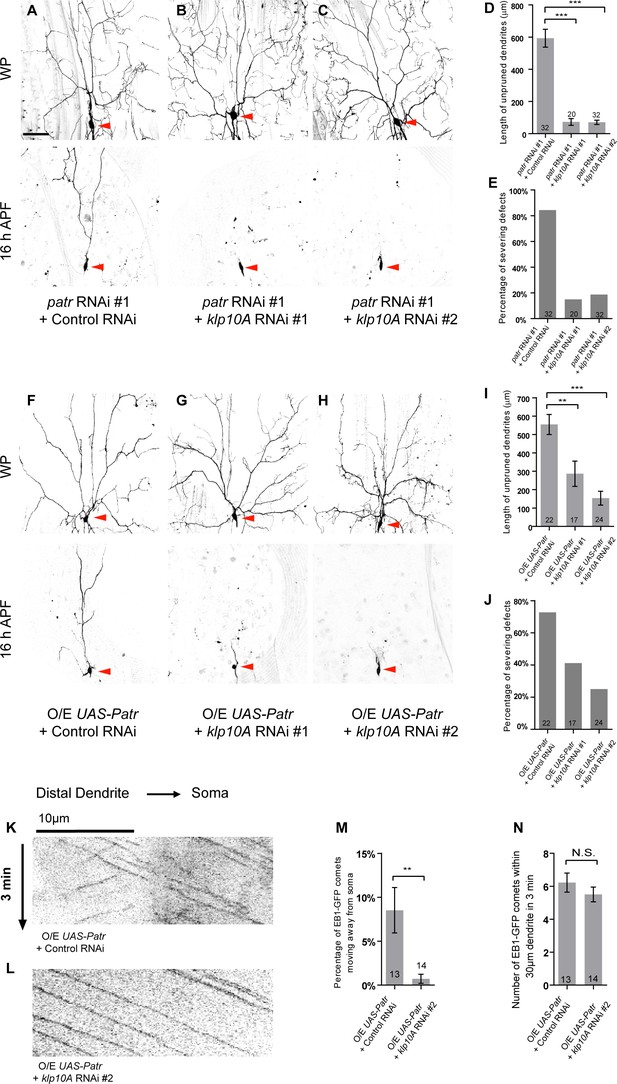
Patronin promotes dendrite pruning by orienting uniform minus-end-out MT arrays in dendrites.
(A–C, F–H) Live confocal images of ddaC neurons expressing mCD8-GFP driven by ppk-Gal4 at WP and 16 hr APF. Red arrowheads point to the ddaC somas. (A–C) ddaC neurons co-expressing patronin RNAi #1 and control RNAi exhibited strong pruning defects at 16 hr APF (A). The pruning defects in patronin RNAi #1 ddaC neurons were significantly suppressed by knockdown of klp10A via either RNAi #1 (B) or RNAi #2 (C). (F–H) ddaC neurons co-expressing Patronin and the control RNAi construct (F) exhibited strong pruning defects at 16 hr APF. These pruning defects were significantly suppressed by knockdown of klp10A via either RNAi #1 (G) or RNAi #2 (H). (D, I) Quantification of total length of unpruned ddaC dendrites at 16 hr APF. (E, J) Quantification of the severing defects at 16 hr APF. Scale bar in (A) represents 50 μm. (K–L) Representative kymographs of EB1-GFP comets in control or mutant ddaC dendrites. In ddaC neurons co-overexpressing Patronin and the control RNAi construct, EB1-GFP comets moved bidirectionally in the dendrites (K). However, klp10A knockdown via RNAi #2 (L) significantly restored the retrograde movement of EB1-GFP comets in Patronin-overexpressing ddaC dendrites. Horizontal arrow indicates the direction towards the soma. Scale bar in (K) represents 10 μm, and each movie was taken for 3 min. (M) Quantification of percentage of dendritic EB1-GFP comets moving away from soma (anterogradely). (N) Quantification of average numbers of EB1-GFP comets along 30 µm of dendrites within 3 min. Error bars represent SEM. The number of samples (n) in each group is shown on the bars. N.S., not significant; **p<0.01; ***p<0.001 as assessed by one-way ANOVA test or two-tailed Student’s T test.
-
Figure 7—source data 1
Extended statistical data as Microsoft Excel spreadsheet.
- https://doi.org/10.7554/eLife.39964.028
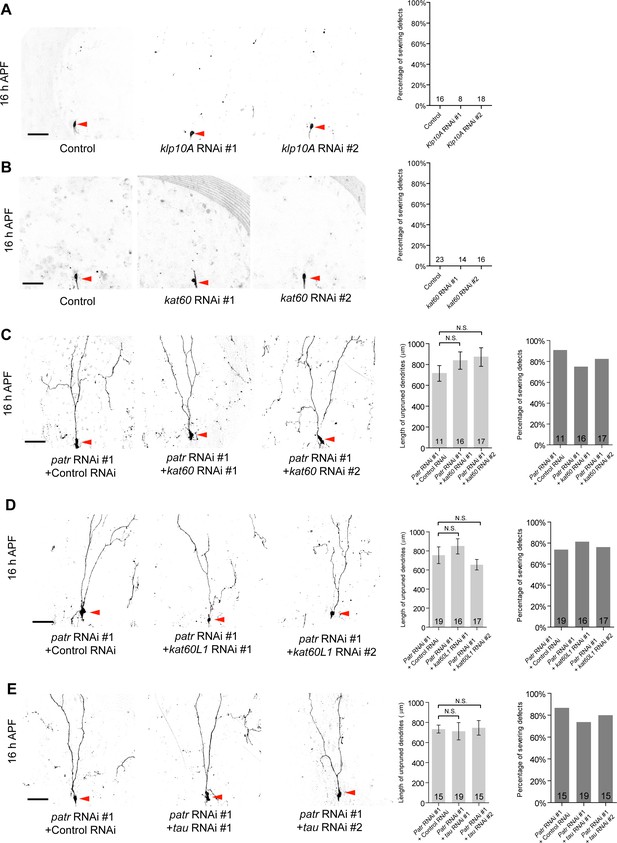
patronin appears not to genetically interact with kat-60, kat-60L1 or tau in dendrite pruning.
(A–E) Live confocal images of ddaC neurons expressing mCD8-GFP driven by ppk-Gal4 at 16 hr APF. Similar to the control neurons, klp10A RNAi (#1 or #2) or kat-60 RNAi (#1 or #2) ddaC neurons pruned away their larval dendrites by 16 hr APF (A–B). The pruning defects in patronin RNAi #1 ddaC neurons were not suppressed or enhanced by knockdown of kat-60 (RNAi #1 or #2) (C), kat-60L1 (RNAi #1 or #2) (D) or tau (RNAi #1 or #2) (E). Red arrowheads point to the ddaC somas. Bar charts on the right depict quantification of total length of unpruned ddaC dendrites and/or the severing defects at 16 hr APF. Scale bars represent 50 μm. Error bars represent SEM. The number of samples (n) in each group is shown on the bars. N.S., not significant, as assessed by one-way ANOVA test.
-
Figure 7—figure supplement 1—source data 1
Extended statistical data as Microsoft Excel spreadsheet.
- https://doi.org/10.7554/eLife.39964.027
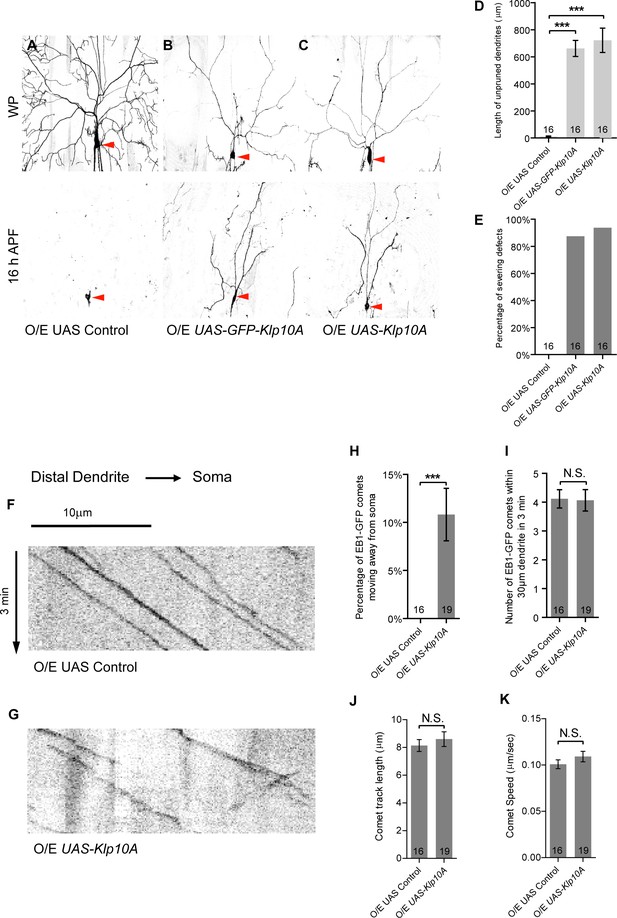
Klp10A overexpression phenocopies patronin knockdown in dendrite pruning and dendritic MT orientation.
(A–C) Live confocal images of ddaC neurons expressing mCD8-GFP driven by ppk-Gal4 at WP and 16 hr APF. Red arrowheads point to the ddaC somas. ddaC neurons overexpressing the UAS control construct pruned all the dendrites (A), whereas ddaC neurons overexpressing GFP-Klp10A (B) or Klp10A (C) via two copies of ppk-Gal4 exhibited simple arbors at WP stage and dendrite pruning defects at 16 hr APF. (D) Quantification of total length of unpruned ddaC dendrites at 16 hr APF. (E) Quantification of the severing defects at 16 hr APF. Scale bar in (A) represents 50 μm. (F–G) Representative kymographs of EB1-GFP comets in UAS control or Klp10A-overexpressing ddaC dendrites. In the dendrites of the control ddaC neurons (F), EB1-GFP comets moved towards the somas. However, in ddaC neurons overexpressing Klp10A (G), EB1-GFP comets moved bidirectionally in the dendrites. Horizontal arrow indicates the direction towards the soma. Scale bar in (F) represents 10 μm, and each movie was taken for 3 min. (H) Quantification of the percentage of EB1-GFP comets moving away from soma (anterogradely) in ddaC dendrites. (I) Quantification of average numbers of EB1-GFP comets along 30 µm of dendrites within 3 min. (J) Quantification of average track length of EB1-GFP comets along 30 µm of dendrites within 3 min. (K) Quantification of average speed of EB1-GFP comets. Error bars represent SEM. The number of samples (n) in each group is shown on the bars. N.S., not significant; ***p<0.001 as assessed by one-way ANOVA test or two-tailed Student’s T test.
-
Figure 8—source data 1
Extended statistical data as Microsoft Excel spreadsheet.
- https://doi.org/10.7554/eLife.39964.036
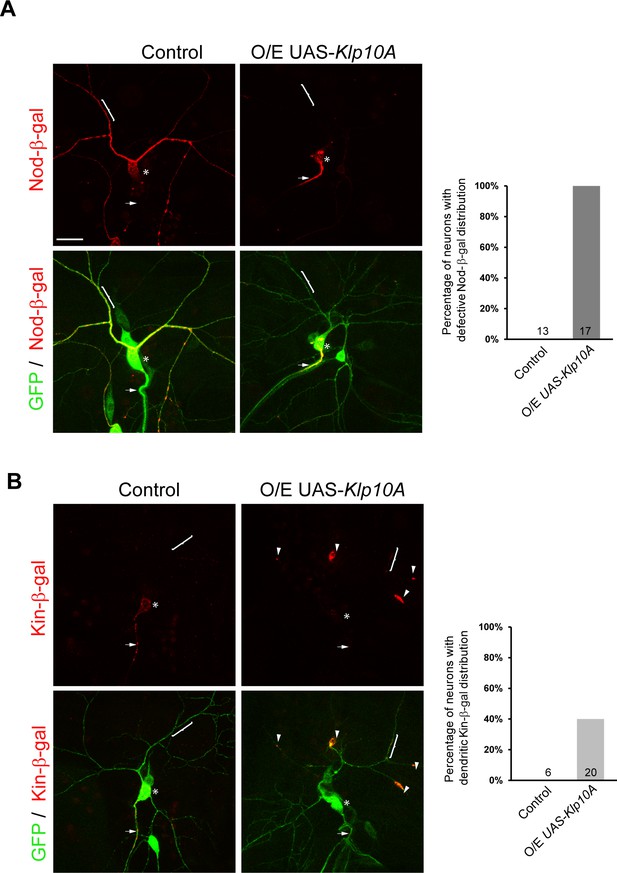
Overexpression of Klp10A causes dendritic MT orientation defects in ddaC neurons.
(A–B) Confocal images of ddaC neurons expressing mCD8-GFP, Nod-lacZ or Kin-lacZ and immunostained for β-galactosidase at wL3 stage. ddaC somas are marked by asterisks, axons by arrows and dendrites by curly brackets. (A) Nod-β-gal levels were reduced in the dendrites but enriched in the somas and proximal axons in all Klp10A-overexpressing ddaC neurons. Quantification of the percentage of neurons with defective Nod-β-gal distribution. (B) Kin-β-gal mis-localized to the dendrites in Klp10A-overexpressing ddaC neurons. Quantification of the percentage of neurons with dendritic Kin-β-gal distribution. Scale bar in (A) represents 20 μm. The number of samples (n) in each group is shown on the bars.
-
Figure 8—figure supplement 1—source data 1
Extended statistical data as Microsoft Excel spreadsheet.
- https://doi.org/10.7554/eLife.39964.031
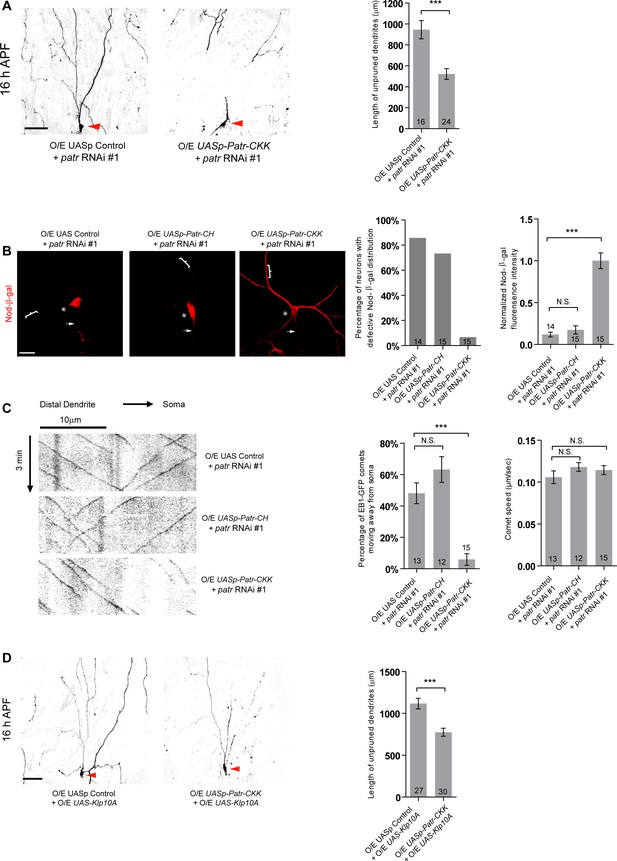
The CKK domain of Patronin is important for dendrite pruning and dendritic MT orientation.
(A, D) Live confocal images of ddaC neurons expressing mCD8-GFP driven by ppk-Gal4 at 16 hr APF. Low-level expression of PatroninCKK under the control of UASp promoter significantly rescued the pruning defects at 16 hr APF in patronin RNAi #1 (A) or Klp10A-overexpressing (D) ddaC neurons. Bar charts on the right depict quantification of total length of unpruned ddaC dendrites in ddaC neurons at 16 hr APF. Scale bars in (A) and (D) represent 50 μm. (B) Confocal images of ddaC neurons expressing Nod-β-gal and immunostained for β-galactosidase at wL3 stage. ddaC somas are marked by asterisks, axons by arrows and dendrites by curly brackets. Nod-β-gal levels were reduced in the dendrites but enriched in the somas of ddaC neurons co-overexpressing patronin RNAi #1 and UAS control. Low-level expression of PatroninCKK but not PatroninCH restored dendritic distribution of Nod-β-gal in patronin RNAi #1 ddaC neurons. Quantification of the percentage of neurons with defective Nod-β-gal distribution and normalized Nod-β-gal intensity in the dendrites. Scale bar in (B) represents 20 μm. (C) Representative kymographs of EB1-GFP comets in control or mutant ddaC dendrites. In patronin RNAi #1 and UAS control co-expressing ddaC neurons, dendritic EB1-GFP comets moved bidirectionally (anterogradely and retrogradely). Low-level expression of PatroninCKK but not PatroninCH significantly restored the retrograde movement of EB1-GFP comets in patronin RNAi #1 ddaC dendrites. Horizontal arrow indicates the direction towards the somas. Scale bar represents 10 μm. EB1-GFP was expressed by Gal44-77. Bar charts on the right depict quantification of the percentage of dendritic EB1-GFP comets moving away from somas (anterogradely) and average speed of EB1-GFP comets along 30 µm of dendrites within 3 min. The number of neurons (n) in each group is shown on the bars. Error bars represent SEM. N.S., not significant; ***p<0.001 as assessed by one-way ANOVA test or two-tailed Student’s T test.
-
Figure 8—figure supplement 2—source data 1
Extended statistical data as Microsoft Excel spreadsheet.
- https://doi.org/10.7554/eLife.39964.033
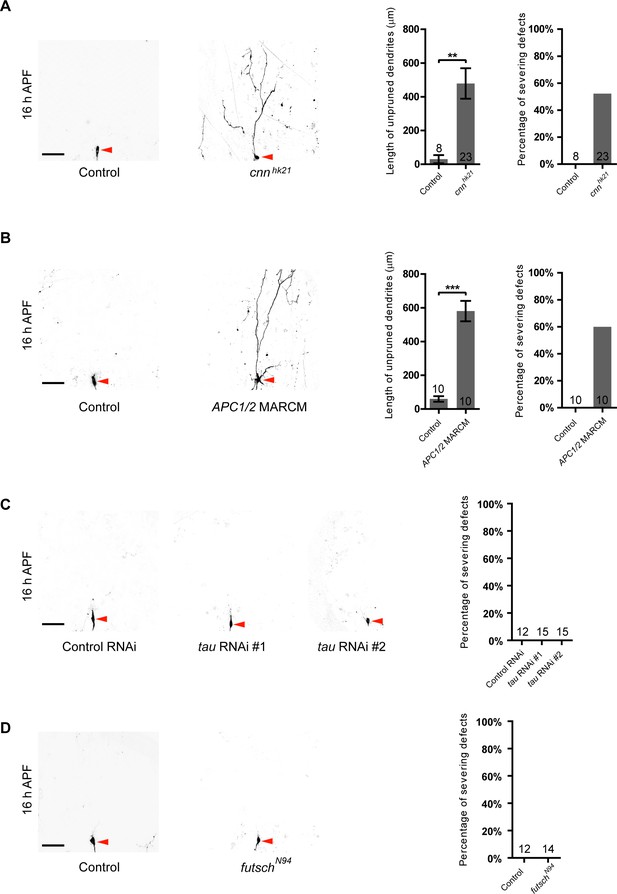
CNN and APC1/2 appear to be required for dendrite pruning of ddaC neurons.
(A–D) Live confocal images of ddaC neurons expressing mCD8-GFP driven by ppk-Gal4 at 16 hr APF. (A–B) While the control neurons pruned all the dendrites, cnnhk21 neurons (A), or APC2N175K, APC1Q8 double MARCM (B) ddaC clones exhibited dendrite pruning defects at 16 hr APF. (C–D) ddaC neurons overexpressing tau RNAi #1, #2 (C), or futschN94 (D) ddaC neurons exhibited normal dendrite pruning at 16 hr APF. Red arrowheads point to the ddaC somas. Bar charts on the right depict quantification of total length of unpruned ddaC dendrites and/or the severing defects at 16 hr APF. Scale bars represent 50 μm. Error bars represent SEM. The number of samples (n) in each group is shown on the bars. **p<0.01; ***p<0.001 as assessed by two-tailed Student’s T test.
-
Figure 8—figure supplement 3—source data 1
Extended statistical data as Microsoft Excel spreadsheet.
- https://doi.org/10.7554/eLife.39964.035
Tables
| Reagent type | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Genetic reagent (D. melangoaster) | UAS-MicalN-ter | other | (Terman et al., 2002) | |
| Genetic reagent (D. melangoaster) | SOP-flp (#42) | other | (Matsubara et al., 2011) | |
| Genetic reagent (D. melangoaster) | ppk-Gal4 on II and III chromosome | other | (Grueber et al., 2003) | |
| Genetic reagent (D. melangoaster) | UAS-Kin-β-gal | other | (Clark et al., 1997) | |
| Genetic reagent (D. melangoaster) | UAS-EB1-GFP | other | (Stone et al., 2008) | |
| Genetic reagent (D. melangoaster) | UASp-mCherry-Patronin | other | (Nashchekin et al., 2016) | |
| Genetic reagent (D. melangoaster) | patroninc9-c5 | other | (Nashchekin et al., 2016) | |
| Genetic reagent (D. melangoaster) | UAS-GFP-Patronin | other | (Derivery et al., 2015) | |
| Genetic reagent (D. melangoaster) | UASp-Arf79F-EGFP | other | (Shao et al., 2010) | |
| Genetic reagent (D. melangoaster) | UAS-Klp10A | this paper | ||
| Genetic reagent (D. melangoaster) | UAS-GFP-Klp10A | this paper | ||
| Genetic reagent (D. melangoaster) | UAS-Patronin | this paper | ||
| Genetic reagent (D. melangoaster) | UAS-Venus-Patronin | this paper | ||
| Genetic reagent (D. melangoaster) | UASp-PatroninΔCH | this paper | ||
| Genetic reagent (D. melangoaster) | UASp-PatroninΔCKK | this paper | ||
| Genetic reagent (D. melangoaster) | UASp-PatroninCKK | this paper | ||
| Genetic reagent (D. melangoaster) | UAS-Venus-PatroninΔCH | this paper | ||
| Genetic reagent (D. melangoaster) | UAS-Venus-PatroninΔCKK | this paper | ||
| Genetic reagent (D. melangoaster) | UAS-Venus-PatroninCC1-3 | this paper | ||
| Genetic reagent (D. melangoaster) | UAS-Venus-PatroninCKK | this paper | ||
| Genetic reagent (D. melangoaster) | UAS-Venus-PatroninCH | this paper | ||
| Genetic reagent (D. melangoaster) | Gal4109(2)80 | Bloomington Stock Center | BDSC: 8769 | |
| Genetic reagent (D. melangoaster) | ppk-CD4-tdGFP | Bloomington Stock Center | BDSC: 35843 | |
| Genetic reagent (D. melangoaster) | GSG2295-Gal4 | Bloomington Stock Center | BDSC: 40266 | |
| Genetic reagent (D. melangoaster) | Gal44-77 | Bloomington Stock Center | BDSC: 8737 | |
| Genetic reagent (D. melangoaster) | UAS-Nod-β-gal | Bloomington Stock Center | BDSC: 9912 | |
| Genetic reagent (D. melangoaster) | patronin RNAi #2 | Bloomington Stock Center | BDSC: 36659 | |
| Genetic reagent (D. melangoaster) | klp10A RNAi #2 | Bloomington Stock Center | BDSC: 33963 | |
| Genetic reagent (D. melangoaster) | Gal42-21 | Bloomington Stock Center | FBal0328157 | |
| Genetic reagent (D. melangoaster) | patronin RNAi # 1 | Vienna Drosophila RNAi Centre | VDRC: v108927 | |
| Genetic reagent (D. melangoaster) | klp10A RNAi # 1 | Vienna Drosophila RNAi Centre | VDRC: v41534 | |
| Genetic reagent (D. melangoaster) | control RNAi | Vienna Drosophila RNAi Centre | VDRC: v36355 | |
| Genetic reagent (D. melangoaster) | control RNAi | Vienna Drosophila RNAi Centre | VDRC: v37288 | |
| Genetic reagent (D. melangoaster) | patronink07433 | Drosophila Genetic Resource Center | #111217 | |
| Genetic reagent (D. melangoaster) | patronin RNAi #3 | National Institute of Genetics, Japan | #18462 Ra-1 | |
| Genetic reagent (D. melangoaster) | kat-60 RNAi # 1 | Vienna Drosophila RNAi Centre | VDRC: v38368 | |
| Genetic reagent (D. melangoaster) | kat-60 RNAi # 2 | Vienna Drosophila RNAi Centre | VDRC: v106487 | |
| Genetic reagent (D. melangoaster) | kat-60L1 RNAi # 1 | Vienna Drosophila RNAi Centre | VDRC: v31599 | |
| Genetic reagent (D. melangoaster) | kat60-L1 RNAi # 2 | Vienna Drosophila RNAi Centre | VDRC: v108168 | |
| Genetic reagent (D. melangoaster) | tau RNAi # 1 | Bloomington Stock Center | BDSC: 28891 | |
| Genetic reagent (D. melangoaster) | tau RNAi # 2 | Bloomington Stock Center | BDSC: 40875 | |
| Genetic reagent (D. melangoaster) | cnnhk21 | Bloomington Stock Center | BDSC: 5039 | |
| Genetic reagent (D. melangoaster) | FRT 82B, APC2N175K, APC1Q8 | Bloomington Stock Center | BDSC: 7211 | |
| Genetic reagent (D. melangoaster) | futschN94 | Bloomington Stock Center | BDSC: 8805 | |
| Antibody | anti-β-galactosidase | Promega | Cat#: Z3783 | 1:1000 |
| Antibody | anti-Patronin | M Gonzalez-Gaitan | 1:500 | |
| Antibody | Cy3-conjugated goat anti-Rabbit antibody | Jackson | Cat#111-165-003 | 1:500 |
| Antibody | 647-conjugated goat anti-Rabbit antibody | Jackson | Cat#111-605-144 | 1:500 |
| Antibody | anti-Futsch | DSHB | 22c10 | 1:50 |
Additional files
-
Supplementary file 1
Supplementary list of fly strains.
- https://doi.org/10.7554/eLife.39964.037
-
Transparent reporting form
- https://doi.org/10.7554/eLife.39964.038





