Physiological constraint on acrobatic courtship behavior underlies rapid sympatric speciation in bearded manakins
Figures
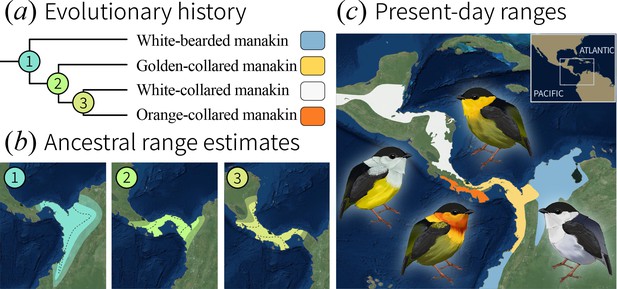
Evolutionary history of the bearded manakins.
(a) From a molecular phylogeny that represents our most up-to-date understanding of the group’s evolutionary history (see Materials and methods). (b) Ancestral state reconstruction of range polygons. 95% credible intervals are denoted by transparent outer edges (maximum extent) and the dotted lines (minimum extent). Golden-collared, white-collared, and orange-collared manakins arose from ancestors (2 and 3) that overlapped across 49% to 73% [95 CI] of their range. (c) Present-day species ranges are geographically isolated from one another.
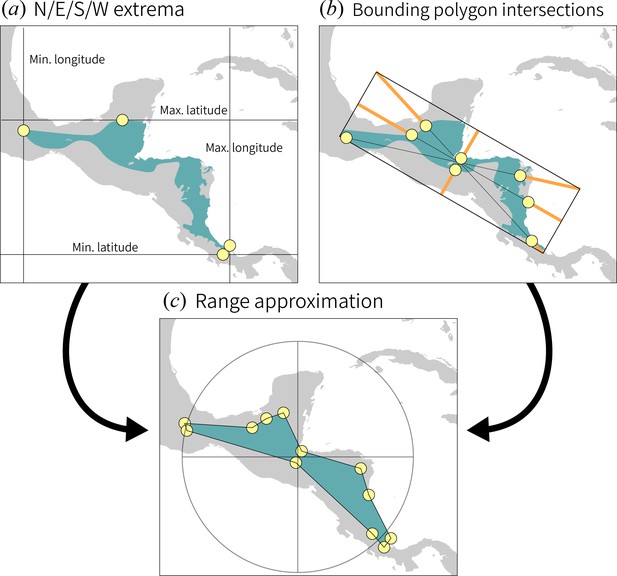
We reconstructed ancestral ranges by computing the coordinates for 12 index points on the range maps of each manakin species on our phylogeny.
The first four points (a) are simply those at which the minimum and maximum longitude/latitude occur. We then fit the minimum bounding polygon to each shapefile in ArcGIS and drew the diagonals and midlines of that rectangle (b), and the remaining eight points were drawn at the outermost intersection point between each line and the original polygon. Together, this collection of points provides an approximation of the original range (c) that can be reconstructed using phylogeographic modeling. To regenerate the range approximation from a set of points, connect them by proceeding radially (we connected counterclockwise, but the direction does not matter) from the cluster’s center.
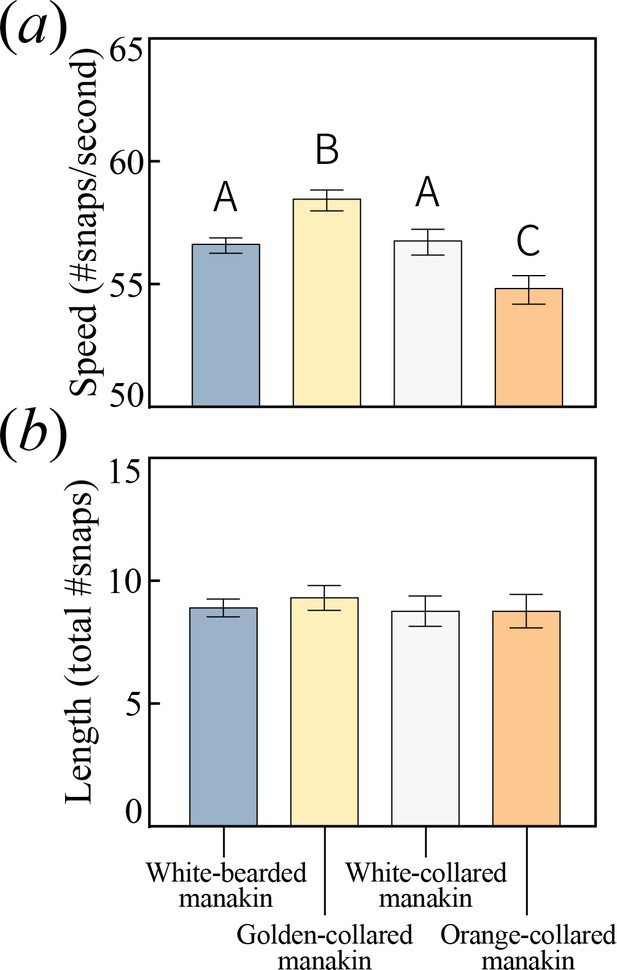
All bearded manakins use a unique wing-snapping display, called the roll-snap, for courtship.
This signal has phenotypically diverged in terms of (a) speed, or the rate (snaps sec−1) at which an individual repeatedly hits its wings together above its back (p<0.05, with statistically significant differences between groups denoted by different letters atop the bars). (b) There is no apparent divergence in the display’s duration in terms of the total number of snap events within a single roll. Values plotted are estimated marginal means ± 1 SEM.
-
Figure 2—source data 1
Data_acoustics.txt: Measurements of roll-snap speed and length obtained from audio recordings of displaying birds.
Includes accession numbers for locating the original recordings on internet archives.
- https://doi.org/10.7554/eLife.40630.006
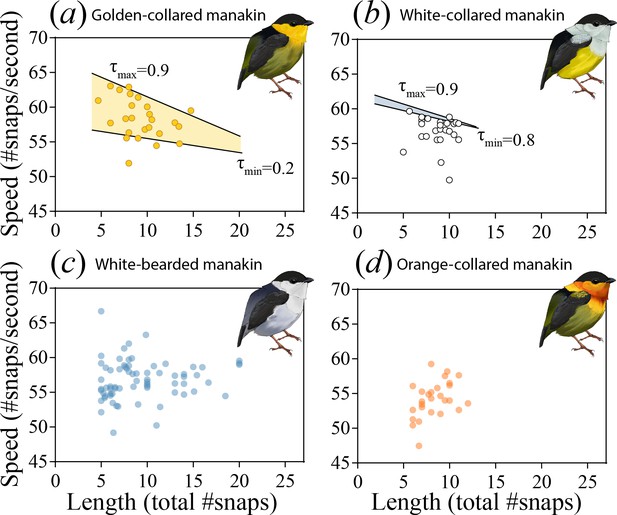
The roll-snap display phenotype can be characterized as a bivariate distribution by plotting speed (# of snaps s−1) as a function of length (total snaps in a roll) for (a) golden-collared, (b) white-collared, (c) white-bearded, and (d) orange-collared manakins.
Values are individual means computed from multiple roll-snap displays. (a) Golden-collared manakins and (b) white-collared manakins both have a significantly negative upper-bound (τ = 0.9, p<0.05) to the distribution, which is consistent with a performance constraint on speed. (a) For the golden-collared manakin, the negative bound extends continuously into the 20th quantile, thus impacting 80% of the population (at τ = 0.2: t = -2.95, p=0.004). (b) By contrast, in white-collared manakins, this constraint is only present at the uppermost end of the distribution (80th quantile and above; τ = 0.8, t = -2.33, p=0.028).
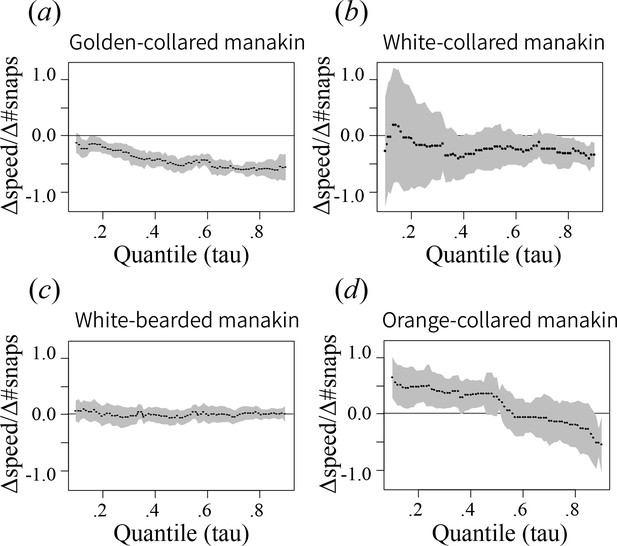
Roll-snap display length (# snaps) as it predicts speed (#snaps/second) when evaluated at every quantile (τ) between 0.1 and 0.9 at intervals of 0.01.
Each point plotted represents the best-fit slope (±SEM) estimate at a given quantile, where values near 0 indicate no relationship between length and speed. For quantiles where the slope is significantly less than 0 (p<0.05), there is a trade-off between display speed and length.
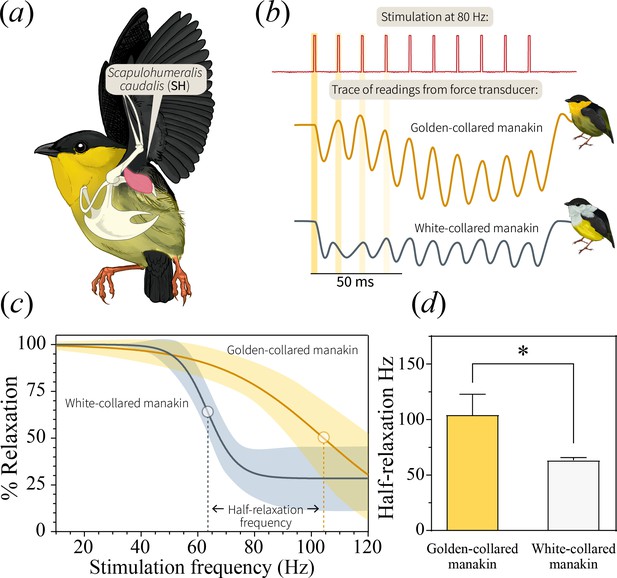
In bearded manakins, (a) the main humeral retractor, the scapulohumeralis caudalis (SH), actuates roll-snap display behavior.
Panel (b) is a representative in situ recording of twitch speed from the SH when stimulated with an 80 Hz pulse train. Representative recordings from both golden-collared and white-collared manakins are shown. Note that the first four individual stimulation pulses within the entire stimulation train are highlighted yellow, as these data are used for later analyses (See Figure 5). (c) We subjected the SH to pulse trains of varying frequencies and measured percent relaxation for each contraction in this series. We then averaged these values within each train to generate a plot of percent relaxation as it changes with stimulation frequency. From there, we fit a four-parameter logistic curve to the data that illustrates the twitch dynamics of each species’ SH muscle. In these models, the dark solid line reflects the best-fit model ±95% confidence bands (shaded area). (d) Our models also allow us to extract an inflection point, which corresponds to the tissue’s half-relaxation frequency. This is an index of twitch speed that we can use to compare between species. Bars represent mean ±1 SEM, with the asterisk (*) denoting a significant different between species (F1,49=79.8, p<0.001).
-
Figure 4—source data 1
Data_fatigue.txt: Contractile phase measurements collected from each twitch cycle during stimulation of golden-collared and white-collared manakin SH muscles.
- https://doi.org/10.7554/eLife.40630.012
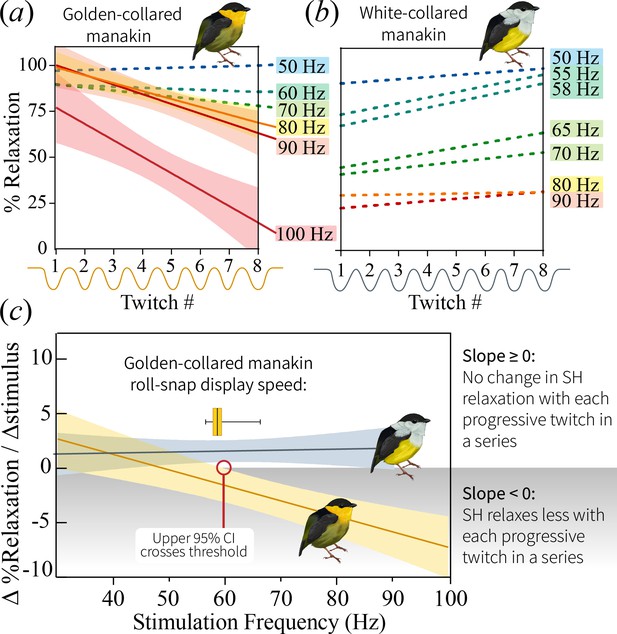
Change in percent relaxation of the SH muscle across a single stimulation train at the different stimulation frequencies.
(a) SH performance in the golden-collared manakin, whereby SH percent relaxation declines across stimulation trains that are greater than 80 Hz. This is just above the species maximum observed roll-snap speed of 68 Hz. Solid lines represent represent significant regression slopes (p<0.05, β <0), with corresponding shaded areas denoting 95% confidence bands. Non-significant slopes (p>0.05) are indicated by dotted lines. (b) SH performance in the white-collared manakin. Note that all slopes are non-significant (p>0.05), as denoted by the dotted lines; however, the y-intercept appears to progressively decrease as the stimulation frequency increases, suggesting that the muscle fuses at the onset of stimulation and stays that way across the entire stimulation train. (c) Regression lines of the slope of the lines in (a) and (b) plotted as a function of stimulation train frequency. Note that SH performance—as measured by the SH’s ability to resist ‘rapid fatigue’ during high frequency stimulations—declines in golden-collared manakins (F1,6=27.7, p=0.004, R2 = 0.82), but not in white-collared manakins (F1,7=0.15, p=0.711). Solid lines associated with each species represent the mean change in percent relaxation at a given frequency,±95% confidence bands (shaded areas). The red line indicates the stimulation frequency at which the 95% CI of the SH performance line in golden-collared manakins intersects with the slope = 0 point. In theory, this represents that maximum twitch speed that the muscle can attain without incurring an endurance cost, and it notably corresponds to the species’ average roll-snap speed (denoted by horizontal box and whisker plot, where the vertical line is at the mean, shaded box indicates ±1 SEM, and whiskers extend to the species range).
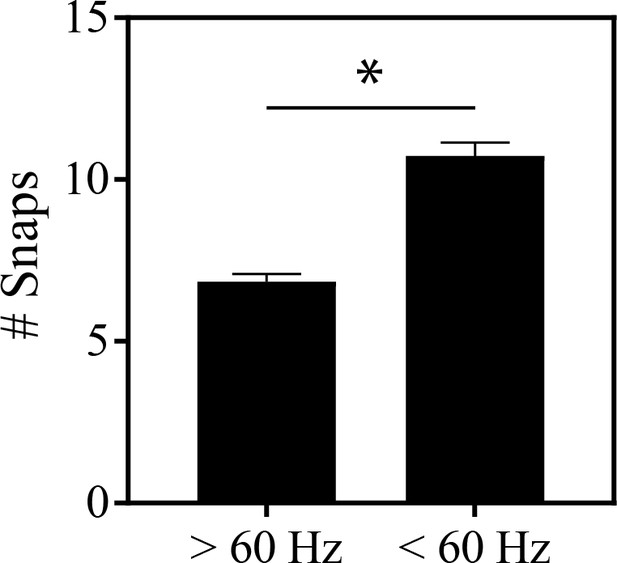
In the golden-collared manakin, roll-snap displays performed >60 Hz are significantly shorter (t = 6.5, df = 152, p<0.0001) than those ≤60 Hz.
Value plotted is mean ±SEM computed from individual roll-snap displays.
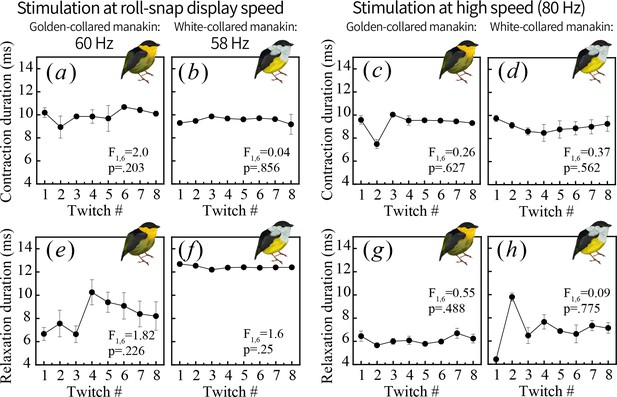
One muscle twitch consists of two phases: contraction, or shortening (a–d) and relaxation, or lengthening (e–h).
To better pinpoint the physiological mechanisms generating endurance costs in the golden-collared manakin SH (see Figure 5), we measured the duration of each phase and tested whether contractile timing also changes over repeated stimulations administered near the species’ roll-snap display speed (a, b, e, f) or at a high speed of 80 Hz (c, d, g, h). We found no change throughout the stimulation series for any measure, which means the observed decline in percent relaxation shown in Figure 5 occurs independently of shifts in contractile timing.
-
Figure 6—source data 1
Data_twitchspeed.txt: Twitch speed measurements collected from muscle recordings of golden-collared and white-collared manakin SH muscles.
- https://doi.org/10.7554/eLife.40630.016
Videos
Animated reconstruction of ancestral range movements in Manacus species.
The video was created by first importing the vector images of reconstructed range approximations as vector graphics into Adobe Animate CC. We then used the shape tween function to model how range approximations shifted between their reconstructed node states.
A slowed-down version of Video 1, with additional written information contextualizing how the roll-snap display evolved alongside ongoing range shifts and species divergence.
https://doi.org/10.7554/eLife.40630.018Tables
Sample size breakdown from citizen-scientist audio recordings of wild bearded manakins performing the roll-snap display.
All recordings were obtained from Xeno Canto (XC) or the Macaulay Library of Natural Sound (LNS). For accession information and metadata, please see source data file (Figure 2—source data 1). For each species, we were able to collect data from more individuals than there are audio recordings. This is because most high-quality recordings captured displays from two or more males displaying at different distances from the recordist, which were clearly distinguished by amplitude.
| Species | # Recordings | # Individuals | # Displays (±1 SEM) per individual |
|---|---|---|---|
| Golden-collared manakin | 21 | 34 | 4.5 ± 0.53 |
| White-collared manakin | 16 | 24 | 3.1 ± 0.51 |
| Orange-collared manakin | 9 | 25 | 2.6 ± 0.46 |
| White-bearded manakin | 40 | 76 | 3.4 ± 0.42 |
Quantile regression results summary for the roll-snap performance distribution of golden-collared and white-collared manakins, where the display length is evaluated as a predictor of display speed at different quantiles (τ) of the distribution.
A significantly negative slope (p<0.05; values adjusted to control the false discovery rate) suggests that there is a trade-off between speed and endurance at a given quantile. Here we present representative models from every 10th quantile (τ interval = 0.1), but we also characterized the performance distribution at a finer-grained scale (see Figure 3—figure supplement 1) at every whole quantile (τ interval = 0.01) between 0.1 and 0.9. We did not extend the analysis below the 10th or above the 90th quantile because quantile regression performs poorly at extreme portions of the distribution for smaller datasets (n < 1000).
| Quantile (τ) | Golden-collared manakin | White-collared manakin | ||||||
|---|---|---|---|---|---|---|---|---|
| Slope | s.e. | t-value | p-value | Slope | s.e. | t-value | p-value | |
| 0.1 | −0.13 | 0.11 | −1.18 | .239 | −0.02 | 0.58 | −0.03 | .976 |
| 0.2 | −0.20 | 0.07 | −2.95 | .004* | 0.03 | 0.51 | 0.06 | .976 |
| 0.3 | −0.31 | 0.07 | −4.13 | .0001* | −0.04 | 0.31 | −0.14 | .976 |
| 0.4 | −0.44 | 0.08 | −5.27 | <0.0001* | −0.29 | 0.24 | −1.24 | .305 |
| 0.5 | −0.53 | 0.08 | −6.51 | <0.0001* | −0.20 | 0.23 | −0.85 | .512 |
| 0.6 | −0.44 | 0.07 | −6.39 | <0.0001* | −0.19 | 0.15 | −1.27 | .305 |
| 0.7 | −0.58 | 0.03 | −21.69 | <0.0001* | −0.11 | 0.16 | −0.67 | .606 |
| 0.8 | −0.60 | 0.08 | −7.91 | <0.0001* | −0.31 | 0.13 | −2.33 | .028* |
| 0.9 | −0.56 | 0.14 | −4.03 | .0001* | −0.41 | 0.15 | −2.76 | .020* |
Statistical summary for linear models assessing how percent relaxation changes over each successive twitch in stimulation trains of different frequencies.
After evaluating whether each species’ SH exhibited a change in performance with repeated stimulations, we also compared the two slope estimates with an F-test. All p-values reported have been adjusted to control for the false discovery rate.
| Stimulation hz | Golden-collared manakin | White-collared manakin | Species comparison | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Slope | F (1,6) | p-value | Slope | F (1,6) | p-value | F (1,12) | p-value | ||
| 50 | 0.4002 ± 0.3983 | 1.01 | .708 | 1.154 ± 0.6165 | 3.50 | .586 | 1.942 | .189 | |
| 55 | 3.098 ± 1.301 | 5.67 | .237 | ||||||
| 58 | 3.287 ± 1.759 | 3.49 | .586 | ||||||
| 60 | −1.682 ± 0.8066 | 4.35 | .246 | ||||||
| 65 | 2.7 ± 1.885 | 2.05 | .767 | ||||||
| 70 | −0.5629 ± 1.87 | 0.09 | .774 | 1.705 ± 2.215 | 0.59 | 1.00 | 1.44 | .254 | |
| 80 | −5.268 ± 1.185 | 19.8 | .017* | 0.2476 ± 1.25 | 0.04 | 1.00 | 18.7 | .002** | |
| 90 | −4.298 ± 0.8332 | 26.6 | .013* | 1.268 ± 1.382 | 0.84 | 1.00 | 33.9 | <0.0001*** | |
| 100 | −8.949 ± 1.874 | 22.8 | .016* | ||||||
Additional files
-
Transparent reporting form
- https://doi.org/10.7554/eLife.40630.019





