Sucrose intensity coding and decision-making in rat gustatory cortices
Figures
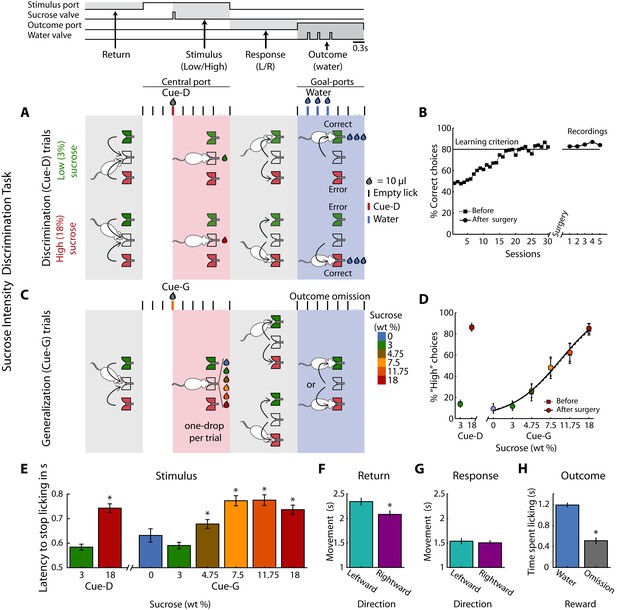
Behavioral report of sucrose’s sweet intensity in a one-drop discrimination task.
(A) Structure of a single trial. The behavioral box was equipped with three spouts each connected to a pressure-controlled solenoid valve that delivered 10 µL drops (not shown, see Materials and methods). One spout was in the central (stimulus) port and the others in the left and right lateral (choice) ports. After the first trial, in the Return epoch, animals after obtaining one of the outcomes in the lateral ports, returned to the central port to begin a new trial. In the Stimulus epoch, after two or three dry licks, the cues (Cue-D -for discrimination) were delivered, and the animals had to make a High/Low decision as to which lateral port to go (Response epoch). If they choose correctly, a water reward was delivered in the Outcome epoch. Errors were unrewarded. Thus, in this task, the perceived intensity of sucrose (i.e., concentration) served as a discriminative cue (Cue-D, see the red tick and drop). (B) Performance (percent correct choices) across training days before (dashed line squares) and after (circles) electrode implantation. (C) Interleaved in sessions, all animals were tested in a variant of the above-described intensity discrimination task -named generalization sessions. These sessions were composed of 80% discrimination trials (3% Low/18 wt% High) that were rewarded as indicated in ‘A,’ and 20% generalization trials; that is, 0, 3, 4.75, 7.5, 11.75, and 18 wt% sucrose cues (named Cue-G). For generalization trials, rats were required to ‘classify’ these sucrose concentrations as either a ‘Low’ or ‘High,’ but these trials were unrewarded. (D) The percent responses to the ‘High’ port during discrimination (Cue-D) and generalization (Cue-G) trials increases as the sucrose concentration increase. Note that the psychometric function was nearly identical both before (dashed line squares) and after surgery (circles). (E) Latency to stop licking after cue delivery. On average, the higher the sucrose concentration, the longer the latency to stop licking. (F–G) Movement time for making a leftward or rightward movement in the Return and Response epochs. (H) Time spent licking, in the Outcome epoch, in Cue-D trials that received water as a reward was longer then in Cue-G trials where the water reward was omitted. * Statistically significant with an alpha level of 0.05.
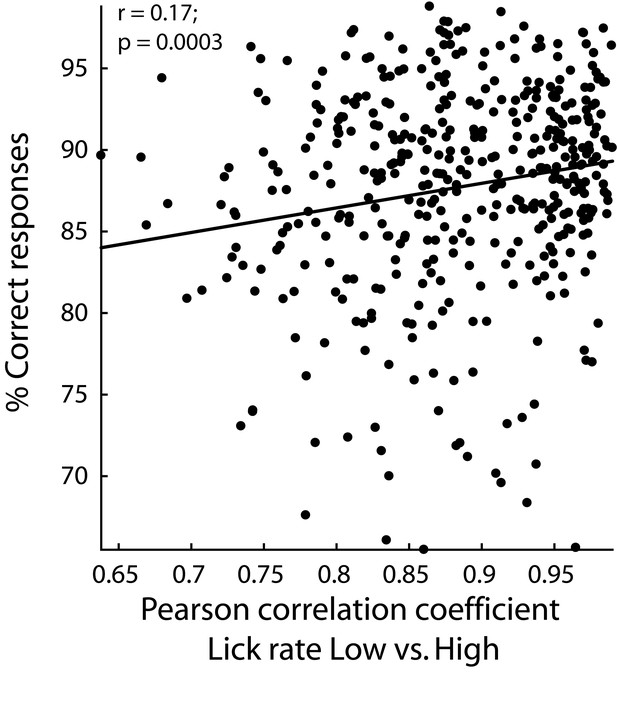
In sessions with rats having better performances, they licked similarly for both cues.
Percentage of correct responses as a function of the increase in the correlation between low and high Cue-D lick rates. Each black dot represents one single session. Behavioral data from the three cortical areas are shown.
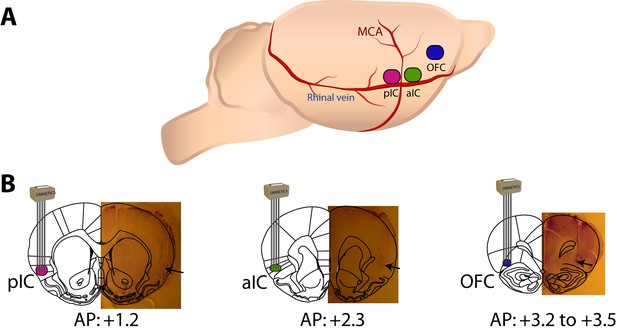
Extracellular recordings were obtained in either the posterior, anterior Insular cortices (pIC or aIC), or orbitofrontal cortex (OFC).
A schematic drawing of the parasagittal view of the brain showing in color the lateral location of each taste cortices recorded, using the medial cerebral artery (MCA) and the caudal rhinal vein as anatomical landmarks. Below is a coronal brain section with a drawing of a 4 × 4, 1 mm2, 16 electrodes array implanted and a Nissl-stained brain slide showing the electrode tracks was overlaid on the Paxinos atlas. Arrows indicate the tip of electrode tracks.
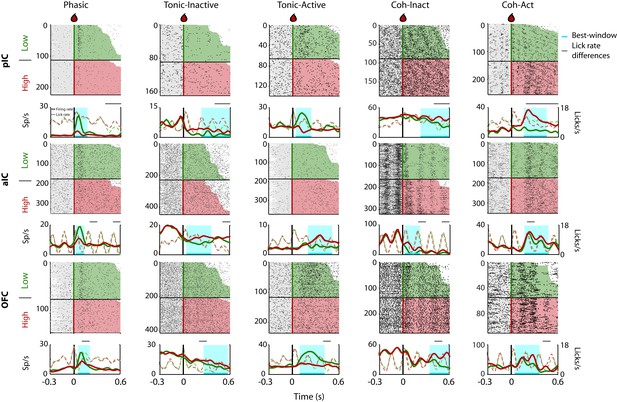
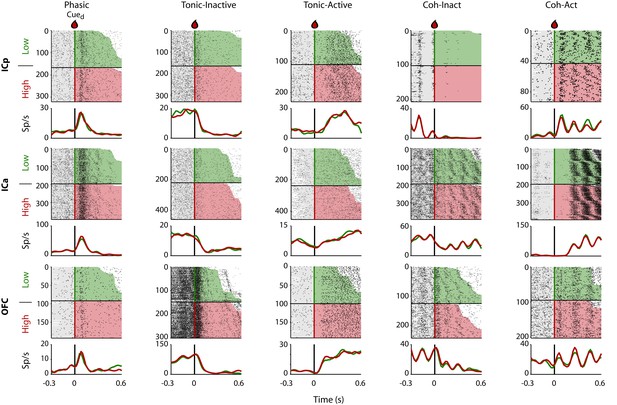
Representative Intensity-selective Cue-evoked responses in the rat pIC, aIC, and OFC.
Representative raster plots and PSTHs (in spikes/s solid lines) of sucrose Intensity-selective neurons belonging to each of the five classes of evoked responses in the pIC (upper), aIC (middle), and OFC (lower) rows: Phasic, Tonic-Inactive, Tonic-Active, Coh-Inactive, and Coh-Active. Coh indicates they are coherent with licking. These exemplar neuronal responses discriminated between 3 and 18 wt% sucrose (Intensity-selective neurons). Most of the Cue-evoked responses were Non-selective to sucrose intensity, and individual examples are presented in Figure 2—figure supplement 1. Action potentials are depicted as black ticks around −0.3 to 0.6 s, from Cue-D delivery (time = 0 s). Only correct trials were included in these plots. The horizontal black line separates the sorted trials according to Cue-D delivery. The licks after 3 and 18 wt% sucrose are indicated by green- and red-shaded area, respectively. The times that animals were licking at the central spout before cue delivery are shown in the shaded gray areas. Also shown are the PSTHs for licking (Licks/s; at right axis) either for Low (green-dashed) or High sucrose (red-dashed line). The rectangle in cyan highlights the best-window where the responses to 3 and 18 wt% sucrose are statistically distinct as determined by a Wilcoxon rank-sum test. The gray horizontal line on top indicates the times where the lick rates were significantly different.
Representative Cue-evoked non-selective responses in the pIC, aIC, and the OFC.
Representative examples of the five classes of evoked responses in the pIC (top), aIC (middle), and the OFC (bottom panels): Phasic, Inactive, Active, Coh-Inactive, and Coh-Active. Most of Cue-evoked responses were Non-selective to sucrose intensity. Raster plot and corresponding peri-stimulus time histogram (PSTH) are shown. Same conventions as in Figure 2.
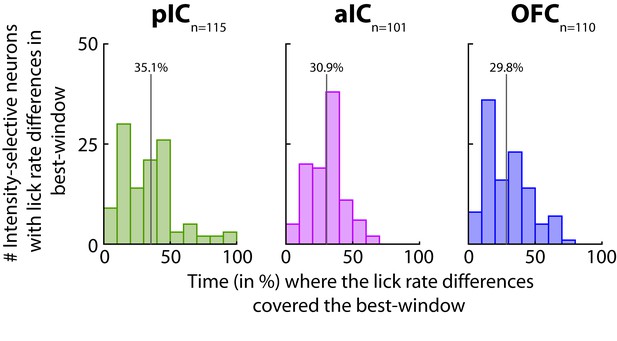
Times in % where the lick rate differences overlapped the best-window.
Percentage of time during which the best-window contained lick rate differences for Intensity-selective neurons with difference in licking. Note that pIC, aIC, and OFC distributions (left, middle and right panels, respectively) are shifted to the left, implying that differences in licking could not entirely account for the neuronal representation of sucrose intensity. A vertical line depicts the mean % of overlap, and the exact value is on top.
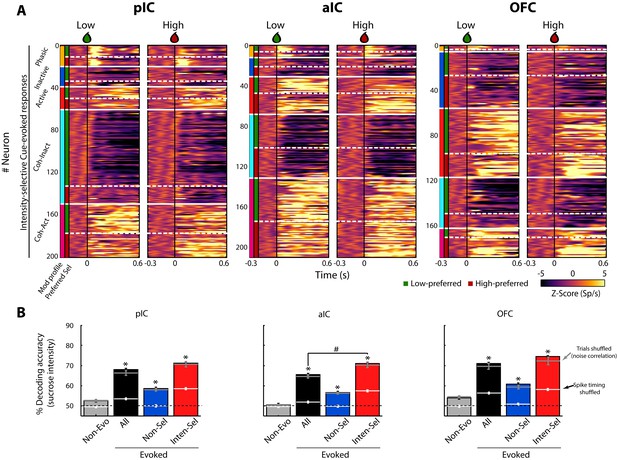
A subpopulation of Intensity-selective neurons decodes sucrose concentrations (3 vs. 18 wt%) better than other neuron classes.
(A) The color-coded PSTHs of the five Cue-evoked responses in pIC (left panel), aIC (middle panel), and OFC (right panel) sorted by modulation profile and Intensity-selectivity (Low/High). Response types were first sorted from top to bottom as follows: Phasic (orange vertical line at the left edge), Inactive (dark blue), Active (red), Lick-coherent Inactive (cyan), and Lick-coherent Active (magenta). The white horizontal dashed lines separate each modulation profile as a function of Low and High selectivity (see green and red vertical lines at the left edge). Each row represents the firing rate normalized in Z-score of a neuronal response aligned to 3 (Low, left panel) and 18 wt% (High, right panel) sucrose delivery (time = 0, black vertical line). (B) Percent decoding accuracy of sucrose intensity achieved by the neurons recorded in pIC, aIC, and OFC. Each colored bar represents a different group of neurons: Non-evoked (gray), All (black), Non-selective (blue), and Intensity-selective (red). A black dashed line indicates the 50% chance level, and the upper dashed line the behavioral performance. * Indicates significant differences against the Non-evoked population, while # indicates significant differences against All group. Only correct Cue-D trials were included for analysis. The white horizontal line in each bar indicates the percent decoding achieved by each population when spike timing information was removed (i.e., shuffled spikes but maintaining same firing rates). The gray horizontal lines depict the contribution of noise correlations for population decoding.
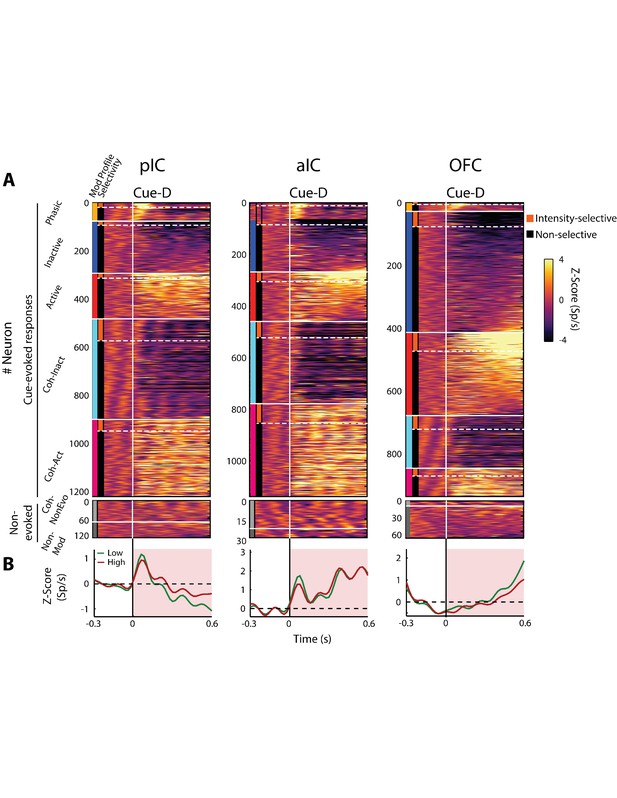
The population temporal activation pattern of all neurons recorded in the posterior IC (n = 1348), anterior IC (n = 1169), and the OFC (n = 1010), elicited in rats by a single10 µL drop of 3 and 18 wt% sucrose.
(A) The top panel displays the color-coded PSTHs of the five Cue-evoked responses in the pIC (left panel), the aIC (middle panel), and the OFC (right panel). Response types were sorted from top to bottom as follows: Phasic (yellow vertical line at the left edge), Inactive (blue), Active (red), Lick-coherent Inactive (cyan) and Lick-coherent Active (magenta). Each row represents the firing rate normalized in Z-score of a neuron aligned to Cue-D delivery (time = 0, black vertical line). The bottom panel shows the color-coded PSTH for Non-evoked response types: Lick-coherent and Non-evoked (light gray) and Non-modulated (dark gray). From the Cue-evoked neurons, the subpopulation of Intensity-selective neurons was always less in percentage (18.2 ± 1.04%) than the Non-selective (81.8 ± 1.04%; Table 2). (B) Population PSTHs of the Cue-evoked responses were aligned to Cue-D delivery (black vertical line) for Low (3 wt% -green) and High (18 wt% -red) sucrose trials. Note that the evoked population activity in the pIC and aIC exhibited a rapid peak (at 80 ± 20 ms), and its responses faithfully covary (oscillates) with licking whereupon the population activity gradually decreases in the pIC and increases in the aIC. In contrast, the population activity in the OFC had less contribution of Phasic and Lick-coherent responses but, after Cue-D delivery, it showed a tonic and gradual increase in activity. Note that the population responses do not discriminate between sucrose concentrations.
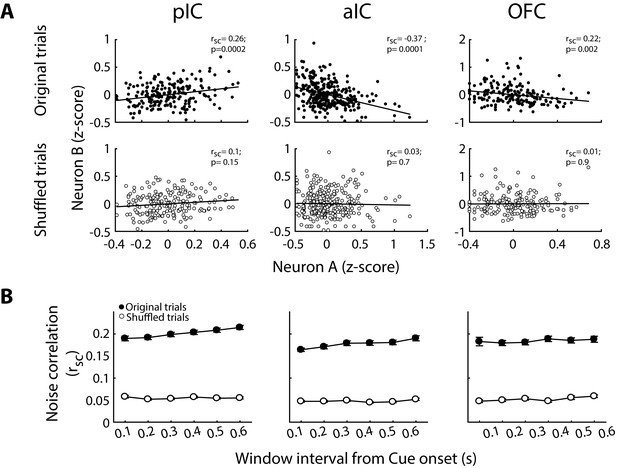
Noise correlation is removed after shuffling the order of trials.
(A) Representative examples of noise correlation from pIC, aIC, and OFC pairs of neurons are depicted in the upper row from left, middle, and right panels, respectively. The normalized (Z-score) activity during each original trial (black dots) for pairs of neurons A-B of each structure is depicted in the upper panel. The lower panel shows the same neuron pair but after shuffled trials (white circles). B. Mean noise-correlation for original (dot circles) and shuffled (white circles) trials measured in different time windows from Stimulus onset.
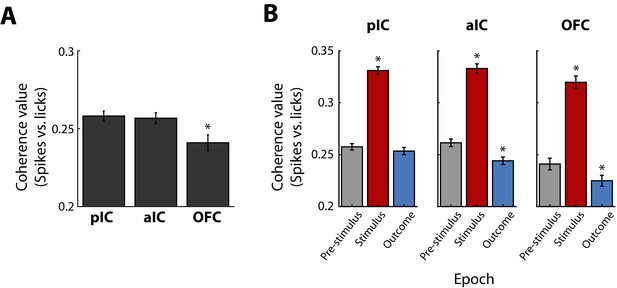
The coherence between licks and spikes is larger after Cue-D delivery in the Stimulus epoch than in pre-Stimulus and Outcome.
(A) Coherence values between licks and spikes in the pIC, aIC, and OFC. * Significantly different relative to pIC and aIC. B. The coherence values of Lick-coherent neurons in the pIC, aIC, and OFC (left, middle, and right panel, respectively) during licks given in the central port during the pre-Stimulus (gray), Stimulus (red), and Outcome (blue) epochs. * Significantly different in comparison to pre-Stimulus epoch.
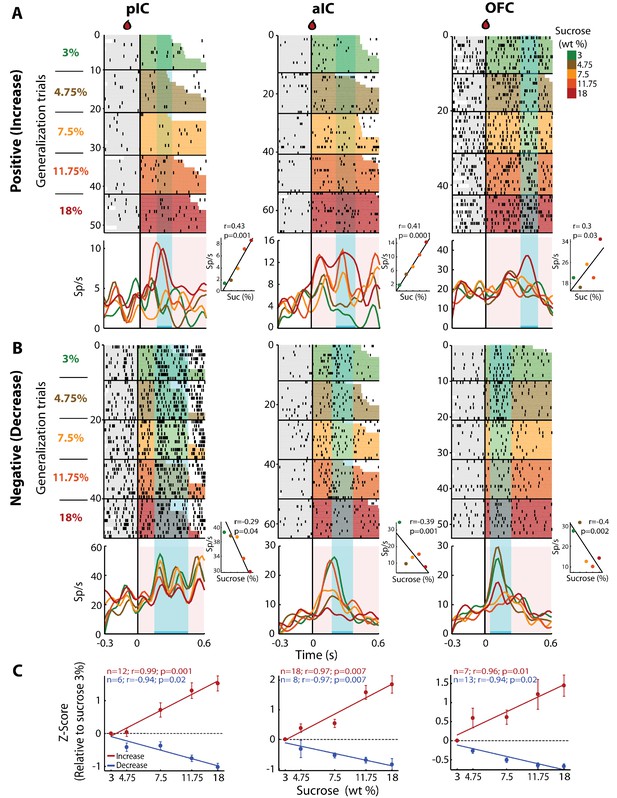
pIC, aIC, and OFC neurons track sucrose concentrations with either increasing or decreasing firing rates.
(A) Raster plots and PSTHs of three representative Sensory neurons, with a positive correlation, recorded in pIC (left), aIC (middle), and OFC (right panel). Responses were aligned to Cue-G delivery (i.e., 3, 4.75, 7.5, 11.75, 18 wt%). The colormap on the right indicates the five sucrose concentrations delivered in the generalization trials. Each row represents a single trial and trials were sorted according to the sucrose concentration. The cyan-shaded rectangles display the ‘best window,’ in which the firing rates best correlated with sucrose concentrations (see Materials and methods for additional details). Same conventions as in Figure 2. The Insets displayed the firing rates in the ‘best window’ where responses had the best Pearson correlation coefficient against sucrose concentrations. (B) Representative examples of chemosensory neurons with negative correlation, recorded in pIC, aIC, and OFC. Same conventions as in A. (C) Normalized activity (relative to the 3 wt% trials) of all Sensory neurons that correlated either positively (red) or negatively (blue) with increasing sucrose’s concentrations. Only generalization trials (Cue-G) were included in the analysis.
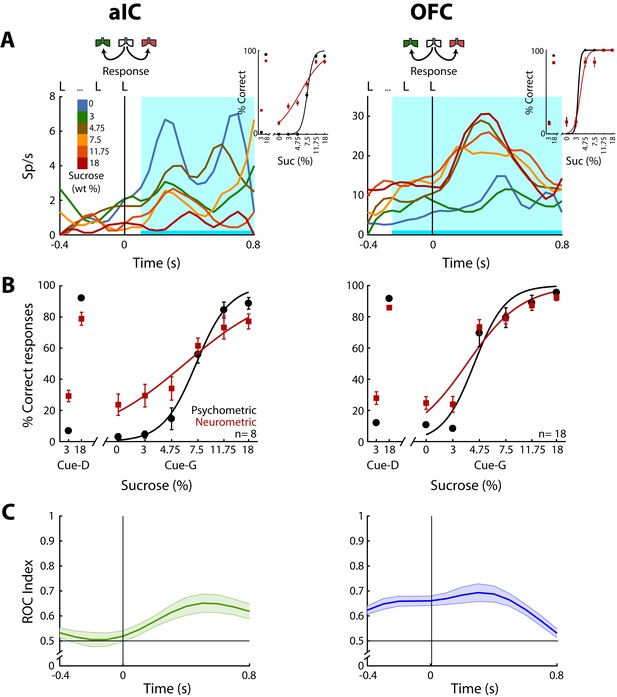
OFC Choice neurons carried information about the subjects’ decisions earlier than in the aIC.
(A) Examples of an aIC (left panel) and an OFC (right panel) neuronal response aligned to the last lick given at the central spout. Firing rates covaried with subject’s Low or High choices. After the last lick in the central spout, subjects must walk to one of the lateral spouts. The colormap indicates the sucrose concentration. The PSTHs show the firing rates before and after rats had initiated the response movement. The cyan-shaded rectangle indicates the ‘best window’ where neural activity tracked subject’s choices, see Inset for psychometric (black) and neurometric (red) functions of each neuron. (B) Mean percentage of correct behavioral responses (psychometric curve, black sigmoid) and neuronal responses (neurometric curve, red sigmoid) of 8 aIC and 18 OFC neurons that tracked choice along the Response epoch. Responses to discrimination (Cue-D) and generalization (Cue-G) trials are depicted on the left and right side of the sigmoid, respectively. Data are expressed as mean ± sem. (C) ROC index across the Response epoch for the aIC (left) and the OFC (right panel) neurons.
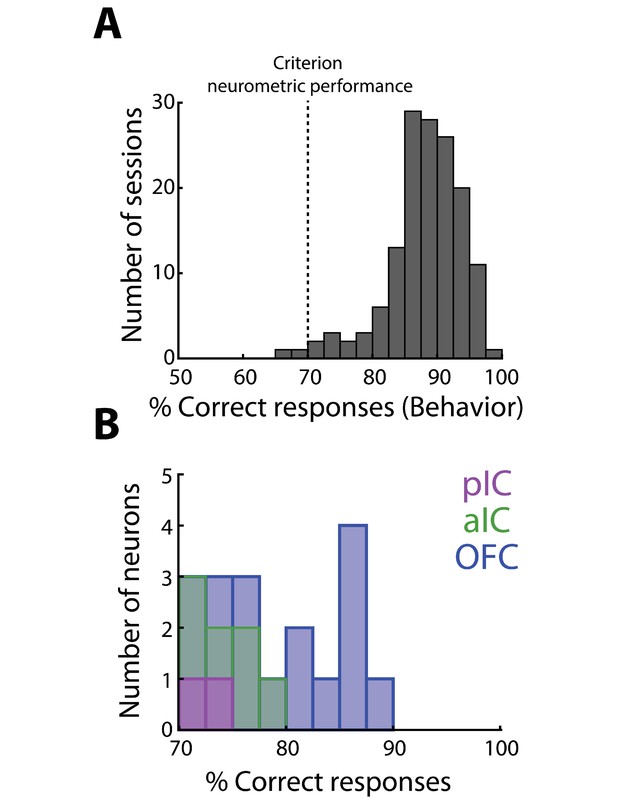
OFC Choice neurons display a better accuracy (% correct responses) than pIC and aIC neurons.
(A) Histogram of the behavioral performance during classification sessions from all animals. Dark grey dashed line indicates the behavioral criterion used to designate a neuron as a Choice neuron. (B) Number of pIC (magenta), aIC (green), and OFC (blue) neurons that crossed criterion, and thus they had a neurometric function that matched behavioral choices.
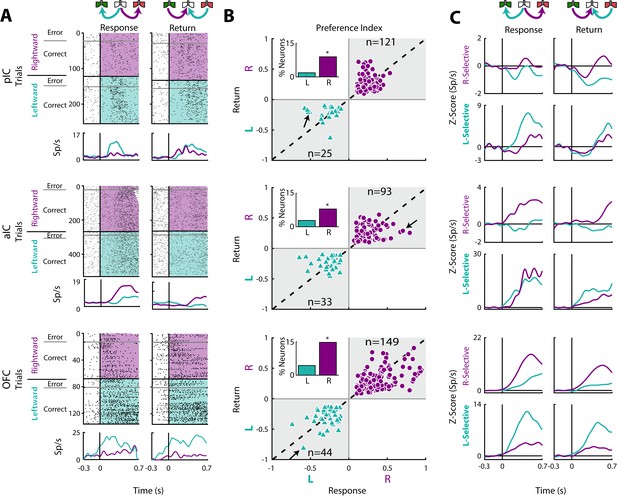
Movement direction coding in pIC, aIC, and OFC.
(A) Three representative examples of movement direction neuronal responses in the pIC, aIC, and the OFC are depicted in the upper, middle and lower panel, respectively. In the rasters, each row is a single trial aligned to the beginning of the Response (left) and Return (right) epochs. Trials were sorted according to the movement direction and task performance: rightward (purple) and leftward (cyan) and error and correct trials. Each black tick represents a single spike. Below the rasters are displayed its corresponding PSTHs. The error trials were omitted for clarity. (B) Preference Indices for left (−1) or right (+1) side during the Return vs. the Response epoch (see Materials and methods). The quadrants in gray indicate where the Rightward- (purple circles) and Leftward-selective (cyan triangles) neurons are expected to be. The Inset depicts the proportion of neurons with Left/Right selectivity. The black arrows signal the examples displayed in panels A. (C) Normalized firing rates for Rightward- (upper) and Leftward-selective (lower panel) neurons, during the Response (left) and Return (right side) epochs. Data are expressed as mean ± sem. It is seen that the OFC exhibits the greatest difference in the Z-scores in comparison to pIC and aIC.
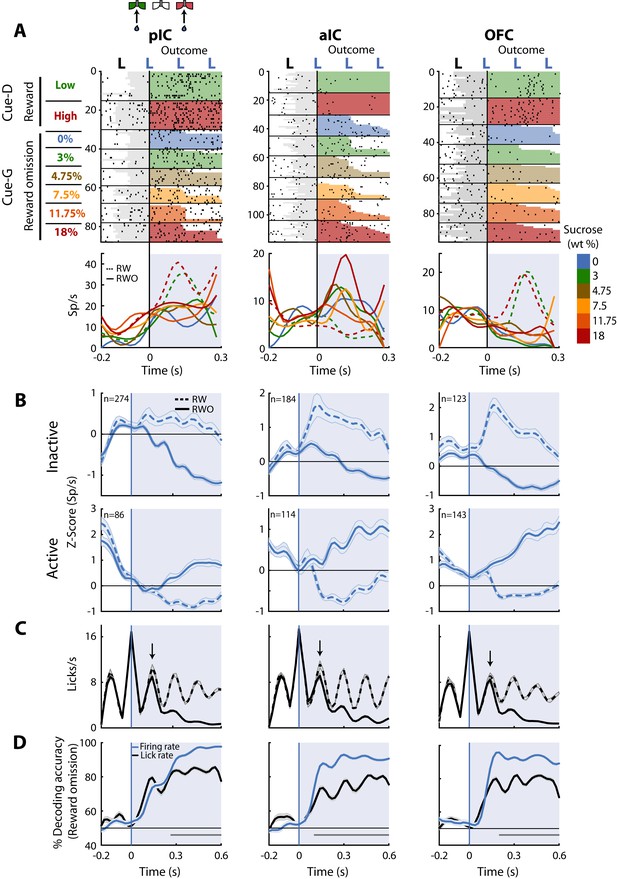
Neurons in pIC, aIC, and OFC are sensitive to reward omission.
(A) Shown are three representative neuronal responses from the pIC (left panel), aIC (middle panel), and OFC (right panel) that encoded reward omission (RWO). The raster plot was aligned to water delivery upon the second lick in a goal-port lateral spout. The first lick was always dry. In the raster plots, the first two rows are for the rewarded (RW) Low (3%) and High (18%) correct discrimination trials. The sessions below were for the six types of generalization trials that were not rewarded (RWO; Cue-G trials). The sucrose concentration is indicated by the color-coded bar on the right side. Below are the PSTHs for Cue-D discrimination trials (dashed lines) and Cue-G generalization trials (solid lines). The blue-shaded rectangle indicates responses in Outcome epoch. (B) Population activity (Z-score) of the Inactive (upper panel) and the Active Reward-omission population (lower panel). These reflect those responses that either decreased or increased their firing rates after reward omission (continuous blue lines) relative to reward delivery (blue dashed line). (C) Lick rates from all generalization sessions. Continuous and dashed black lines indicate lick rates during reward omission and rewarded trials, respectively. The population PSTHs, of the firing and lick rate, were expanded from −0.2 to 0.6 s from the second lick to better appreciate the difference in firing and lick rates elicited by outcome delivery or omission. Note that the subjects required only one additional lick to detect reward absence (see arrows). (D) Decoding accuracy of the population of Outcome neurons discriminating between rewarded and unrewarded trials, using either the firing rates (blue) or the lick rates (black). The horizontal dark-gray line depicts where differences reached statistical significance.
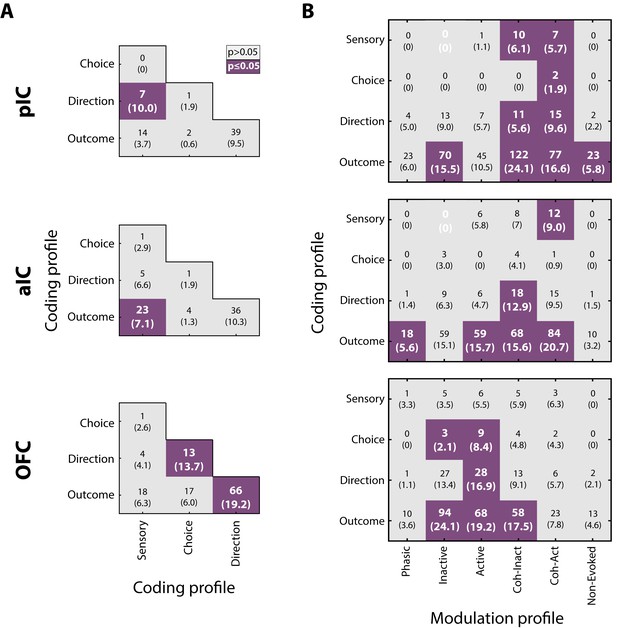
Neurons in pIC, aIC, and OFC encode more than one task-related variable.
(A) Overlap of coding profiles. Contingency matrix indicating the number (and percentage) of neurons that belong to more than one group. Purple squares depict significant overlap as detected by a Fisher’s exact test (p<0.05). (B) Contingency matrix showing the overlap between coding and modulation profiles. Same conventions as in A. Only neurons recorded from generalization sessions were included in this analysis to guarantee that data is drawn from the same distribution. Non-significant associated categories are indicated with a white 0. Data are presented as a number of neurons and percent in parenthesis.
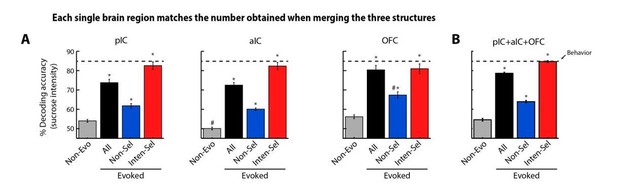
Decoding accuracy when each population (Non-evoked), All, Nonselective, and Int-selective) of each brain region matches the number of neurons sampled across regions collapsed (pIC+aIC+OFC).
(A) Decoding accuracy when the number of neurons in a population (Non-evo, All, Non-sel or Int-sel) matched the number sampled from the three structures combined. That is, the number of neurons in each population of a brain region was the same as in the three regions combined (pIC+aIC+OFC: nNon-evoked = 200, nAll = 3527, nNon-selective = 2701, and nInten-selective = 596). It is seen that decoding accuracy obtained from each population of each region was not significantly different from the result obtained by combining the three structures together (panel B). Note that decoding accuracy obtained by using Non-evoked and Non-selective population never reached that of Intensity-selective neurons. * Significantly different relative to Non-evoked neurons from the same structure. # In comparison to the same population of the three regions combined.
Tables
Cue-Evoked and Non-Evoked neurons.
https://doi.org/10.7554/eLife.41152.006| Brain region | Cue-Evoked responses | Non-Evoked | |||||
|---|---|---|---|---|---|---|---|
| Phasic | Inactive | Active | Coh-Inac | Coh-Act | Non-Mod | Coh-NonEvo | |
| pIC (n=1348) | 75 (5.6) | 217 (16.1) | 193 (14.3) | 414 (30.7) | 322 (23.9) | 53 (3.9) | 74 (5.5) |
| aIC (n=1169) | 67 (5.7) | 202 (17.3) | 192 (16.4) | 317 (27.2) | 361 (30.9)* | 7 (0.6)* | 23 (2)* |
| OFC (n=1010) | 27 (2.7)*# | 386 (38.2)*# | 265 (26.2)*# | 169 (16.7)*# | 90 (8.9)*# | 62 (6.1)*# | 11 (1.1)* |
-
Number of Cue-D responsive neurons (%). Data in bold indicate statistically different against pIC (*) or aIC (#), detected by a chi-squared test. Alpha level set at 0.05.
Cue-Evoked responses: Non-selective and Intensity-selective neurons.
https://doi.org/10.7554/eLife.41152.007| Brain region | Cue-Evoked responses | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Phasic | Inactive | Active | Coh-Inac | Coh-Act | Total | |||||||
| Non-Sel | Int-Sel | Non-Sel | Int-Sel | Non-Sel | Int-Sel | Non-Sel | Int-Sel | Non-Sel | Int-Sel | Non-Sel | Int-Sel | |
| pIC (n=1348) | 55 (4.1) | 20 (1.5) | 198 (14.7) | 19 (1.4) | 171 (12.7) | 22 (1.6) | 325 (24) | 89 (6.6) | 272 (20.2) | 50 (3.7) | 1021 (75.7) | 200 (14.8) |
| aIC (n=1169) | 55 (4.7) | 12 (1) | 183 (15.7) | 19 (1.6) | 155 (13.3) | 37 (3.2)* | 254 (21.6) | 63 (5.4) | 283 (24.2) | 78 (6.7)* | 930 (79.6) | 209 (17.9) |
| OFC (n=1010) | 21 (2.1)*# | 6 (0.6)* | 336 (33.3)*# | 50 (5)*# | 204 (20.2)*# | 61 (6)# | 124 (12.3)*# | 45 (4.5)* | 65 (6.4)*# | 25 (2.5)# | 750 (74.2) | 187 (18.5)* |
-
Number of Non-selective and Intensity-selective neurons (%). Data in bold indicate statistically different against pIC(*) or aIC (#) detected by a chi-squared test. Alpha level set at 0.05.
Percent decoding accuracy of sucrose’s intensity, when only one a single population, at a time, was included in the analysis.
https://doi.org/10.7554/eLife.41152.015| Brain region | Non-Evo | All | Phasic | Inactive | Active | Coh-Inac | Coh-Act | All-Tonic | All-Coh |
|---|---|---|---|---|---|---|---|---|---|
| pIC | 52.7 ± 0.6 | 67.6 ± 1.3 | 60.2 ± 1.7 | 53.1 ± 0.5 | 53.5 ± 0.6 | 62.0 ± 1.2 | 61.6 ± 0.8 | 54.3 ± 0.6 | 66.6 ± 1.2 |
| aIC | 50.4 ± 0.3 | 65.7 ± 0.9 | 54.7 ± 1.1 | 52.1 ± 0.4 | 55.4 ± 0.4 | 60.0 ± 0.7 | 62.1 ± 0.8 | 55.1 ± 0.5 | 65.6 ± 0.9 |
| OFC | 54.8 ± 0.5 | 71.0 ± 1.6 | 56.6 ± 1.4 | 55.3 ± 0.6 | 61.1 ± 1.1 | 62.7 ± 1.1 | 65.9 ± 1.5 | 60.8 ± 1.0 | 69.3 ± 1.6 |
-
Percent decoding accuracy (mean ± sem). Data in bold indicate statistically different relative to Non-Evoked neurons by using a one-way ANOVA and a Dunnett´s post hoc. Alpha level set at 0.05.
One population dropped analysis: Decoding sucrose intensity excluding only one population from All neurons.
https://doi.org/10.7554/eLife.41152.016| Brain region | All | Phasic | Inactive | Active | Coh-Inac | Coh-Act | All-Tonic | All-Coh |
|---|---|---|---|---|---|---|---|---|
| pIC | 67.8 ± 1.3 | 58.5 ± 1.7 | 68.1 ± 1.3 | 68.5 ± 1.3 | 63.4 ± 0.9 | 64.0 ± 1.2 | 68.5 ± 1.2 | 58.0 ± 0.7 |
| aIC | 65.9 ± 0.8 | 63.4 ± 2.0 | 66.3 ± 0.9 | 65.1 ± 0.9 | 62.7 ± 0.7 | 61.2 ± 0.7 | 66.1 ± 0.9 | 55.8 ± 0.5 |
| OFC | 71.2 ± 1.7 | 63.1 ± 3.0 | 72.6 ± 1.7 | 68.8 ± 1.5 | 67.6 ± 1.5 | 66.9 ± 1.3 | 70.5 ± 1.6 | 62.1 ± 1.1 |
-
Percent decoding accuracy (mean ± sem). Data in bold indicate statistically different in comparison with the All subpopulation by using a one-way ANOVA and a Dunnett´s post hoc.
Note that values in the 'All' group are not identical to those in Table 3 due to random sampling in the population decoder.
Additional files
-
Supplementary file 1
Statistical analysis for all figures.
- https://doi.org/10.7554/eLife.41152.023
-
Supplementary file 2
Statistical analysis for all tables.
- https://doi.org/10.7554/eLife.41152.024
-
Transparent reporting form
- https://doi.org/10.7554/eLife.41152.025





