The human gut chemical landscape predicts microbe-mediated biotransformation of foods and drugs
Figures
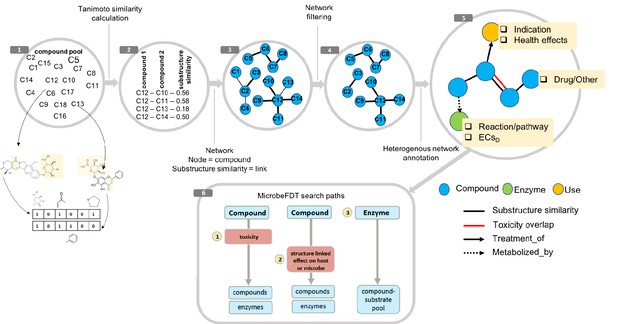
MicrobeFDT is a searchable resource of gut microbiome food and drug metabolism with associated toxicities.
(1) Diet-derived, xenobiotic-derived and endogenous compounds were clustered based on the PubChem fingerprint system (Kim et al., 2016) and the Tanimoto coefficient (Bajusz et al., 2015). (2) The pairwise similarity matrix forms the basis of the (3) substructure similarity network in which nodes are compounds and links are weighted by substructure similarity. (4) A Z-score based threshold method was used to identify significant chemical similarity relationships between nodes (Baldi and Nasr, 2010). (5) The property graph model of nodes and relationships in the network highlights node-relationship pairs that can be queried. Node entities include compounds (blue), uses (orange) and enzymes (green). A compound node can have up to four types of directional relationships: compound pairwise substructure similarity, compound pairwise toxicity similarity, compound treatment use descriptor and compound microbial mediated metabolism descriptor.
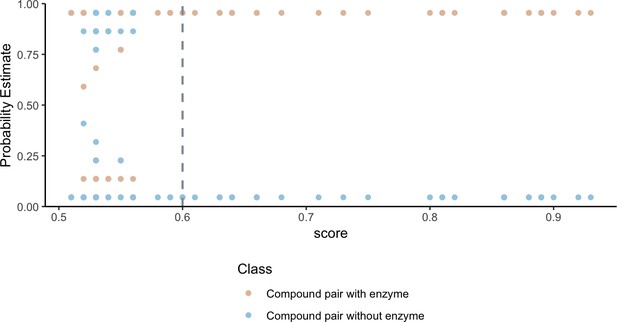
Higher substructure similarity scores between pairs of compounds are associated with higher probability of sharing an enzyme.
Potential enzyme mediated metabolism of compound pairs is compared with substructure similarity to determine the probability that compounds have an experimentally determined shared enzyme (pink) or no known shared enzyme (blue). The gray vertical dashed line indicates the average cutoff for significance in substructure similarity neighborhood construction. Probability estimates are based on a Bayesian approach for support vector machines implemented in R using the probsvm package (Zhang et al., 2013).
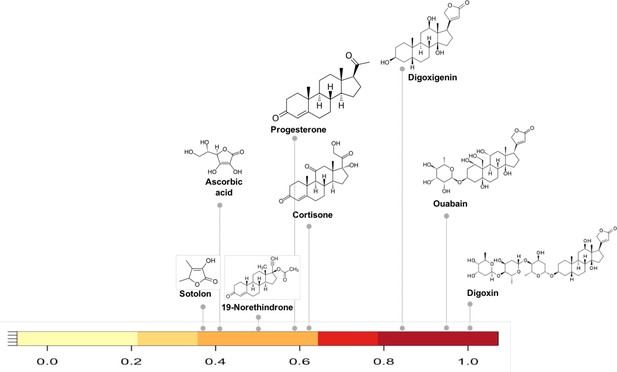
Substructure similarity range of Cgr2 enzyme susceptible compounds.
Substructure based clustering distinguishes experimentally characterized substrates from non-substrates of the Cgr2 enzyme. Digoxin clusters with other cardenolides that are experimentally characterized substrates (Koppel et al., 2018) for Cgr2 at substructure similarity values greater than 0.8. Compounds that are not substrates of Cgr2 have lower substructure similarity with digoxin; compounds with minimal reduction (Koppel et al., 2018) include progesterone and cortisone (substructure similarity <= 0.63). Color bar intensity increases with compound overlap with digoxin.
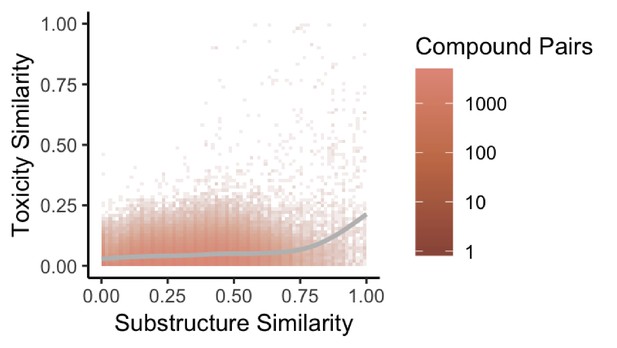
Substructure similarity is predictive of toxicity similarity.
We evaluated the predictive power of substructure similarity to identify compounds with shared toxicity using a measure of pairwise toxicity defined by Campillos et al. (2008) and used a linear regression to determine the strength of the association. We find a modest positive correlation between substructure similarity and toxicity similarity that is stronger for more structurally similar compounds.
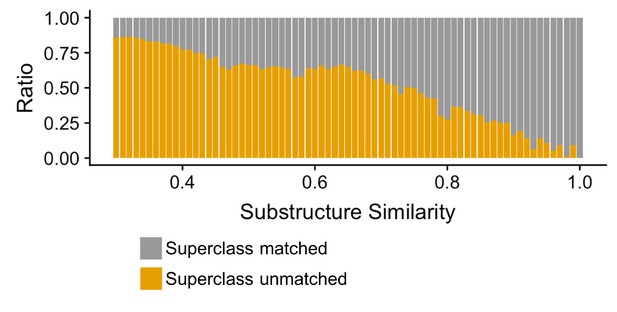
Compound-pairs share superclass annotation at a greater frequency as substructure similarity scores increase.
Ratio of compound-pairs substructure similarity with matched and unmatched superclass annotation for all compound pairs represented in MicrobeFDT. Within the hierarchical ClassyFire classification schema, the superclass level annotation represents the second level and includes 31 different structure-based categories (Djoumbou Feunang et al., 2016).
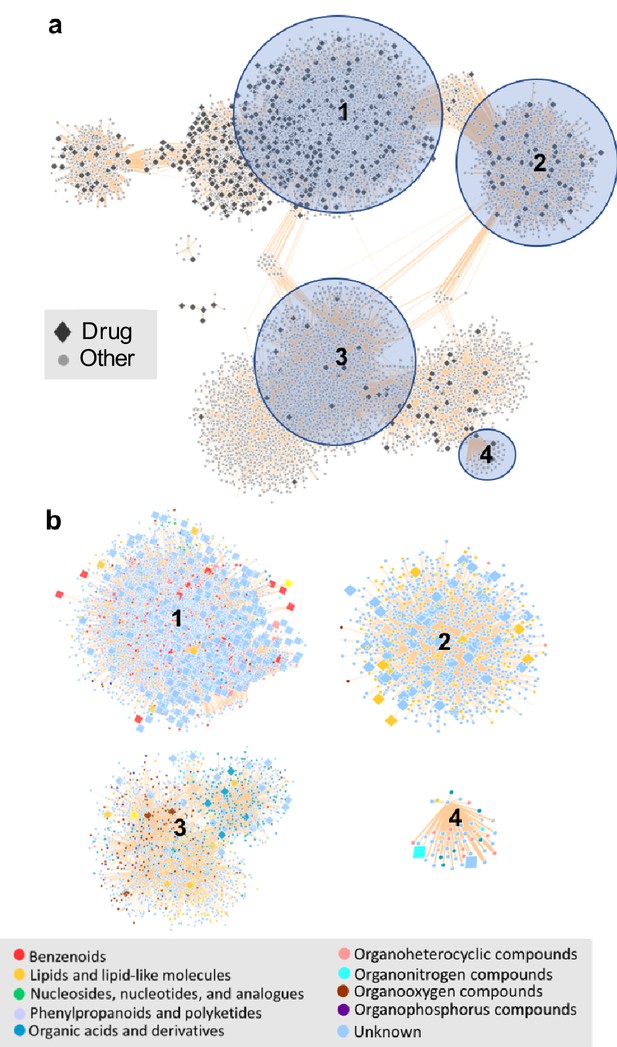
The chemical space of the gut microbiome.
(a) Chemical similarity network of food-derived or endogenous compounds (gray circles, "Other") and therapeutic drugs (black diamonds, "Drug"). Tan edges are weighted by substructure similarity where thicker edges indicate higher substructure similarity. The distribution of compounds in chemical similarity space illuminates regions of low and high chemical substructure overlap between drugs and other compounds. (b) Compounds from selected regions of the network are colored by their superclass level taxonomy based on the FooDB chemical structure classification (Wishart, 2012). Food-derived or endogenously produced compounds are identified with blue circles, therapeutic drugs with red diamonds. Within high-drug density, highlighted regions 1 and 2, drugs share substructure similarity with food-derived benzenoids, lipids, phenylpropanoids and polyketides. In the low-drug density highlighted region 3, drugs overlap with organonitrogen compounds and nucleosides. Region 4 includes organonitrogen compounds and nucleosides in addition to lipid-like molecules which have minimal overlap with therapeutic drugs.
-
Figure 6—source data 1
Chemical similarity scores for drug and non-drug compounds.
- https://doi.org/10.7554/eLife.42866.010

Distribution of Tanimoto scores between drug-drug (blue) and other-other (red) substructure similarity pairs. "Other" refers to foods and endogenous compounds.
https://doi.org/10.7554/eLife.42866.009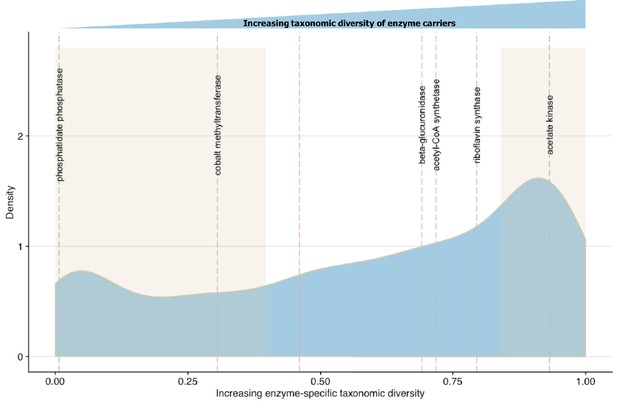
Linking enzymatic functions with taxonomic diversity.
The Simpson index was adapted to describe enzyme-specific taxonomic dominance and diversity based on enzyme abundance in taxonomy-linked gene counts across healthy individuals in the Integrative Human Microbiome Project (Proctor et al., 2014). We define a microbial enzyme as high dominance and low taxonomic diversity if its Simpson index value falls below 0.46 (red dotted line), the mean value across all enzymes. Dominance-diversity values for gut microbiota functions that fall above or below the mean are highlighted by gray dashed lines and include the following enzymes and pathways: phosphatidate phosphatase (0.007353), cobalt-precorrin-2 C(20)-methyltransferase (0.305502) from the Vitamin B12 synthesis pathway, β-glucuronidase (0.691618), Acetyl-CoA synthase (0.718163) which is involved in the production of propionate from complex carbohydrates, riboflavin synthase (0.794781) from the riboflavin synthesis pathway and acetate kinase (0.931892) which is involved in acetate production. The shaded regions indicate the range of EDsD values that are one standard deviation above and below the mean and reflect the most broadly distributed functions and most specialized functions.
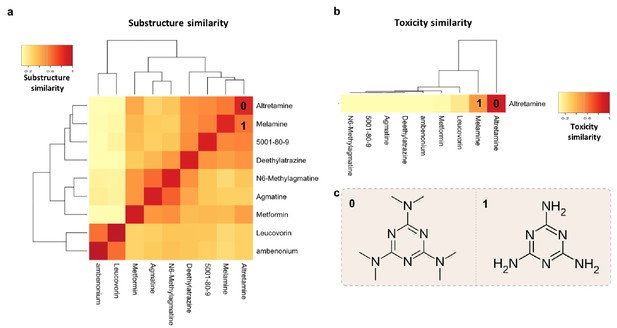
Structure-toxicity relationship between melamine and altretamine suggests a role for microbial N-demethylases in altretamine toxicity.
(a) Substructure overlap between altretamine and its nearest neighbors in MicrobeFDT. A Z-score based threshold of significant overlap indicates that altretamine has both high substructure and (b) toxicity overlap with melamine. (c) The two compounds are distinguishable by the presence of N-methyl groups.
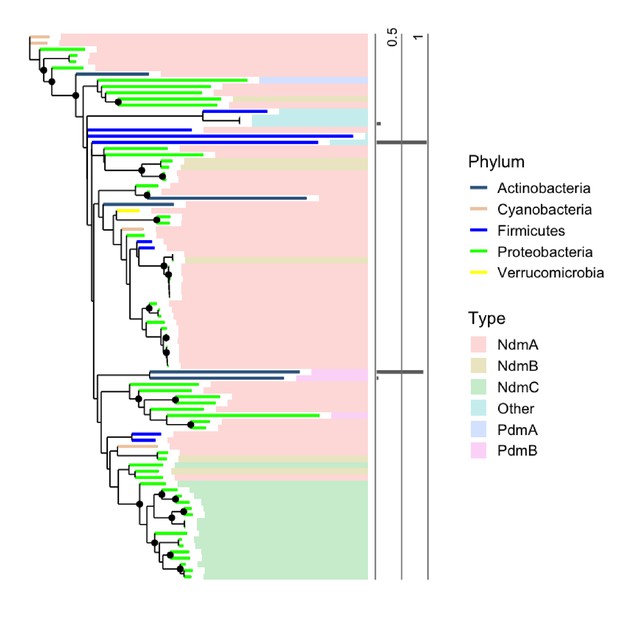
Phylogenetic distribution of N-demethylases in healthy human guts.
Phylogenetic distribution and abundance of N-demethylase enzymes in healthy individuals from the HMP (Human et al., 2012). Phylum-level taxonomy is indicated by branch color. Colored bars specify the type of N-demethylase based on protein sequence similarity between represented proteins and purified and structurally characterized N-demethylases. The adjoining bar plot displays the relative abundance of N-demethylase sequences represented in the tree with values normalized on a scale from 0, being least abundant to 1, being most abundant.
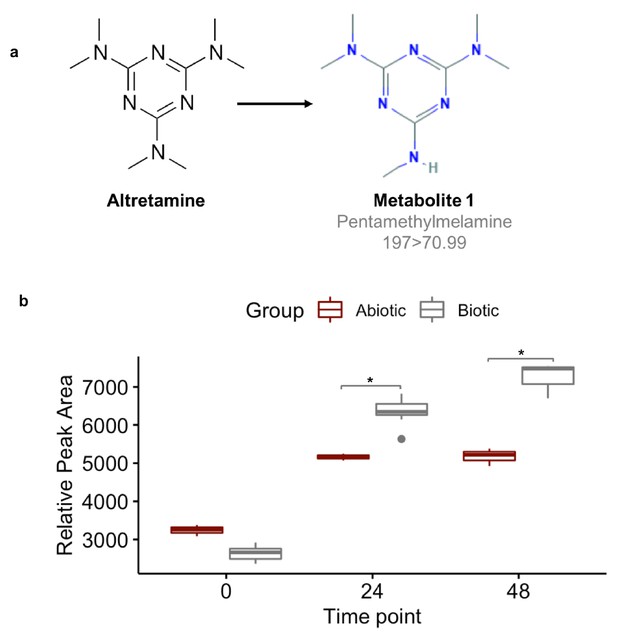
Fecal microbiomes actively demethylate altretamine.
Liquid chromatography with tandem mass spectrometry (LC-MS) was used to quantify the formation of (a) pentamethylmelamine, an N-demethylated metabolite of altretamine identified in the pooled fecal microbiomes of three healthy unrelated individuals. (b) The formation of metabolite 1 at 24 and 48 hr was significantly increased under the experimental condition in comparison to the contribution of spontaneous N-demethylation by an unpaired two-sample Wilcoxon test (*=P < 0.05).
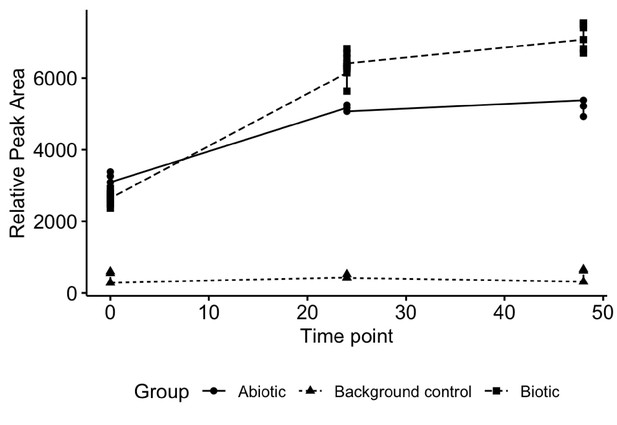
Experimental design and controls used to quantify fecal microbiome turnover of altretamine.
No altretamine or metabolite were detected in background fecal slurry control.
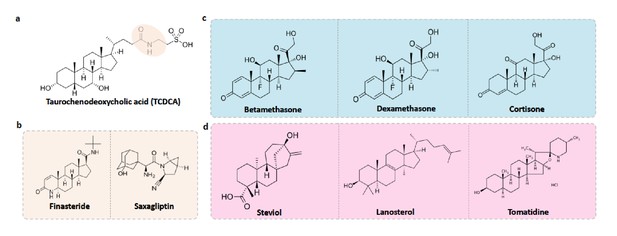
Food-drug compounds chemically similar to TCDCA are putative antimicrobials.
(a) Chemical structure of taurochenodeoxycholic acid (TCDCA). (b) TCDCA-like therapeutic drugs that are susceptible to bile salt hydrolases include finasteride and saxagliptin. (c) Non-susceptible TCDCA-like therapeutic drugs include betamethasone, dexamethasone and cortisone. (d) TCDCA-like food derived compounds include steviol, lanosterol and tomatidine.

Phylogenetic distribution of bile salt hydrolases in healthy human guts.
Phylogenetic distribution and abundance of bile salt hydrolase enzymes in healthy individuals from the HMP (Human et al., 2012). Phylum-level taxonomy is indicated by branch color. Colored bars specify whether bile salt hydrolase activity has been determined in the literature for a specific protein. The adjoining bar plot displays the relative abundance of bile salt hydrolase sequences represented in the tree with values normalized on a scale from 0, being least abundant to 1, being most abundant.
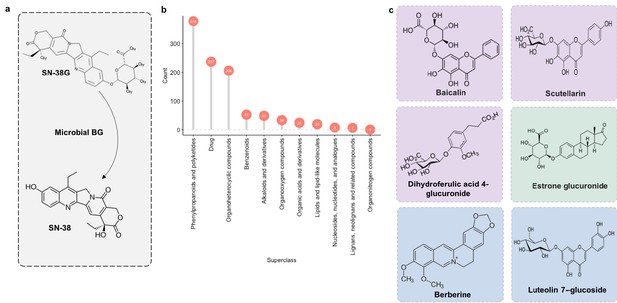
Microbial β-glucuronidase potential substrate pool of compounds structurally similar to SN-38G.
(a) SN-38G conversion to SN-38 in the gut is mediated by microbial β-glucuronidases. (b) The substrate pool for β-glucuronidases with above threshold substructure overlap with SN-38G are members of a diverse range of chemical structure superclasses as defined by FooDB chemical ontology (Wishart, 2018). (c) These compounds include glucuronidated food-derived compounds (purple), endogenous glucuronides (tan) and other non-glucuronides (blue).
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Biological sample (community microbiota, feces) | fecal sample | other | fecal sample obtained from three healthy adults | |
| Chemical compound, drug (altretamine) | altretamine | Sigma | Pubchem_ID:329748966; CAS_No:645-05-6 | prepared in DMSO, 0.1 mM final concentration in fecal slurry |
| Other | Brain Heart Infusion broth | Himedia | Himedia:M210I | |
| Chemical compound (Dimethyl sulfodixe) | DMSO | MP Biomedicals | MP:191418; CAS_No:67-68-5 | |
| Chemical compound (Melamine-triamine-(15N3)) | Melamine-triamine-(15N3) | Sigma | Pubchem:329758619; CAS_No:287476-11-3 | prepared in DMSO, 400 nM final concentration in analytical sample |
Additional files
-
Transparent reporting form
- https://doi.org/10.7554/eLife.42866.019





