Glial Ca2+signaling links endocytosis to K+ buffering around neuronal somas to regulate excitability
Figures
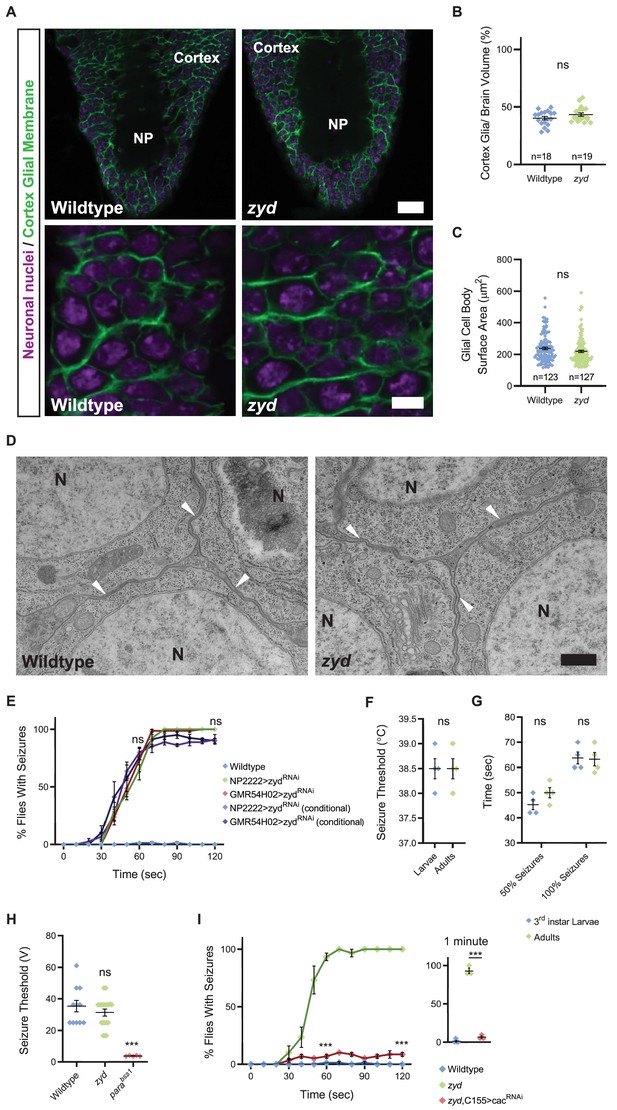
Mutations in a cortex glial NCKX generate stress-induced seizures.
(A) Immunofluorescence imaging reveals no apparent morphological changes in cortex glial wrapping of neuronal soma (3rd instar larval brains, magenta: anti-Elav, neuronal nuclei; green: anti-GFP, mCD8:GFP, cortex glial membrane). Upper panels show a section through the VNC cortex and neuropil (NP), Scale bar = 20 μm. Lower panels show enlarged cortical regions. Scale bar = 5 μm. (B) Quantification of cortex volume occupied by cortex glial processes shows no difference between wildtype and zyd (n > 15 brains for each genotype, p=0.138). (C) Quantification of cortex glial cell body surface area shows no difference between wildtype and zyd (n > 120 cells/ N = 4 animals for each genotype, p=0.0892). (D) Electron microscopy images of cortex glial contacts (arrowheads) with neuronal somas (N). Cortex glial processes between neuronal cell bodies are as thin as 50 nm in both wildtype and zyd. Scale bar = 500 nm. (E) Time course of Heat-shock induced seizures (38.5°C, HS) following chronic or conditional knockdown of zyd with two different cortex glial drivers (NP2222 and GMR54H02) is shown. Rearing adult flies at the restrictive temperature (>30°C) for gal80ts (a temperature-sensitive form of the gal4 inhibitor, gal80, see Materials and methods) removes gal80 inhibition of gal4 and allows expression of zydRNAi only at the adult stage. These manipulations reproduce the zyd mutant seizure phenotype (N = 4 groups of 20 flies/genotype). (F–G) Behavioral analysis of HS-induced seizures at 38.5°C shows that larval and adult seizures have similar temperature threshold (F) and kinetics (G) (N = 4 groups of 10–20 animals/condition/treatment). (H) Recordings of the giant fiber system muscle output. Seizure thresholds in wildtype, zyd and Parabss1 (positive control) are shown. The voltage required to induce seizures in zyd is not significantly different from wildtype (35.32 ± 3.65V and 31.33 ± 2.12V, p=0.3191, n ≥ 7 flies/genotype). (I) Behavioral analysis of the time course of HS-induced seizures indicates neuronal knockdown of cac (C155>cacRNAi) rescues the zyd seizure phenotype. Inset shows results after 1 minute of HS (p=0.0004, N = 4 groups of 20 flies/genotype). Error bars are SEM, ***=P < 0.001, Student’s t-test.
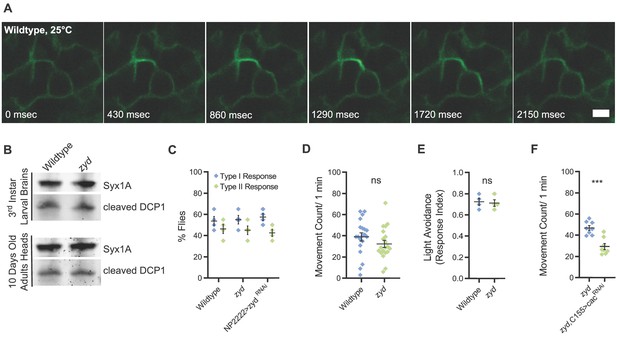
Mutations in a cortex glial NCKX generate stress-induced seizures without affecting brain structure or baseline neuronal function.
Related to Figure 1, Supplementary files 1, 2. (A) Representative images of a single cortex glial Ca2+ oscillation (myrGCamP6s) in wildtype at 25°C. Scale bar = 2 μm. (B) Western blot analysis of cleaved DCP1 (cell death marker) in larval brains and adult heads of wildtype and zyd. (C) Behavioral analysis of the gentle touch response in wildtype and zyd larvae (N = 4 groups of 20 larvae/genotype). (D) Analysis of the activity level of wildtype and zyd adult flies (N = 20 flies/genotype). (E) Light avoidance response of wildtype and zyd 3rd instar larvae (N = 4 groups of 20 larvae/genotype). (F) Activity level analysis shows a significant reduction in the activity of zyd/C155>cacRNAi adult flies relative to zyd (p=0.0002) (n = 8 flies/genotype).
The response of wildtype flies to a 38.5°C heat-shock is shown, following by the response of zyd flies to the same condition.
https://doi.org/10.7554/eLife.44186.004Representative Ca2+ imaging in wildtype cortex glia.
GCaMP6s was expressed specifically in cortex glia using the GMR54H02-gal4 driver.
Representative Ca2+ imaging in zyd cortex glia.
GCaMP6s was expressed specifically in cortex glia using the GMR54H02-gal4 driver.
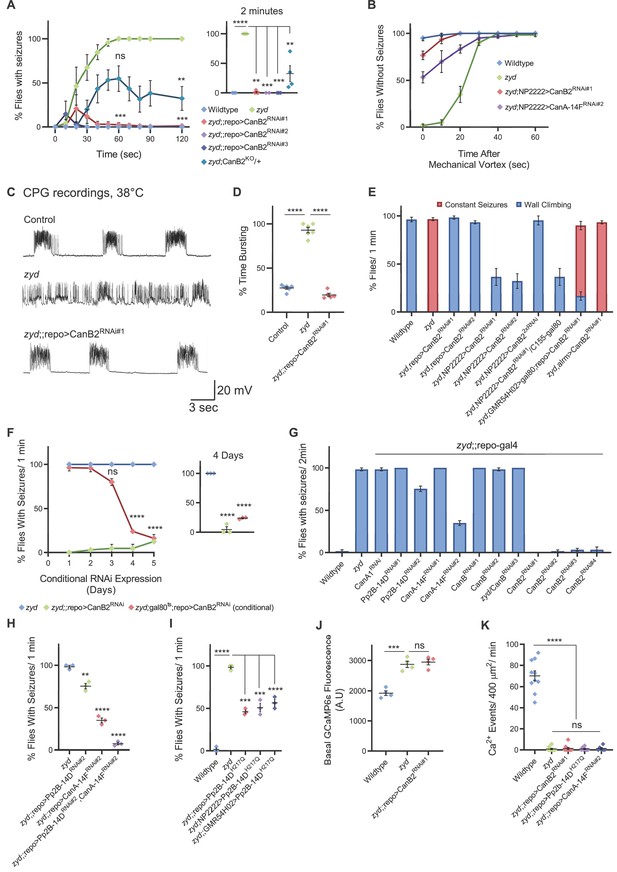
Cortex glial knockdown of calcineurin rescues zyd seizures without affecting intracellular Ca2+.
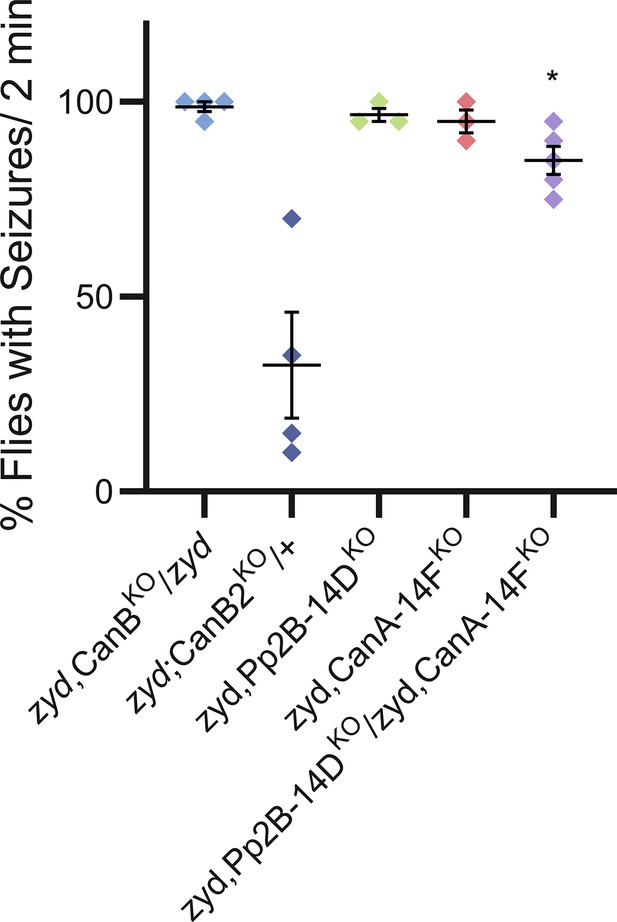
(A) Behavioral analysis of HS-induced seizures. Pan-glial knockdown of the CN regulatory subunit, CanB2, with three partially overlapping hairpins (#1, #2 and #3, see Materials and methods) completely rescues the zyd seizure phenotype, while a single copy of CanB2 knockout allele (CanB2KO/+) rescues ~60% of seizures (N = 4 groups of ≥15 flies/genotype). Inset shows analysis after 2 minutes of HS (p=0.0001). (B) Behavioral analysis of the recovery from vortex-induced seizures. Pan-glial knockdown of CanB2 and CanA-14F rescues zyd vortex-induced seizures (N = 3 groups of 20 flies/genotype). (C) Representative voltage traces of spontaneous CPG activity at larval 3rd instar muscle 6 at 38°C in wildtype, zyd and zyd;;repo >CanB2RNAi#1 animals (n ≥ 5 preparations/genotype). (D) Quantification of average bursting duration for CPG recordings of the indicated genotypes at 38°C (n ≥ 5 preparations/genotype). (E) Detailed analysis (see Materials and methods) of HS induced behaviors of zyd/CanB2RNAi flies. Cortex glial knockdown of CanB2 leads to seizure rescue in ~30% of zyd;NP2222>CanB2RNAi flies, with the remaining ~70% displaying partial rescue. Cortex glial CanB2 knockdown with two copies of the RNAi (zyd;NP2222>CanB22xRNAi) recapitulates the full rescue seen with pan-glial knockdown. Inhibiting gal4 expression of the RNAi in neurons with gal80 (C155-gal80) does not alter the rescue observed with cortex glial knockdown, and astrocyte specific (alrm-gal4) CanB2 knockdown does not rescue zyd seizures (N = 3 groups of >15 flies/genotype, see Figure 2—figure supplement 1D for complete dataset). (F) Cortex glial conditional knockdown of CanB2 using gal4/gal80ts. Rearing adult flies at the restrictive temperature (>30°C) for gal80ts allows expression of CanB2RNAi only at the adult stage. A significant reduction in seizures (p<0.0001) was seen after four days of rearing flies at the restrictive temperature for gal80ts (31°C), with only ~25% of adults showing seizures. The reduction in seizures was enhanced when adults were incubated at 31°C for longer periods (N = 3 groups of >10 flies/genotype). Inset shows analysis after 4 days of incubation at 31°C (p=0.0001). (G) Pan-glial knockdown of the Drosophila calcineurin (CN) family (CanA1, CanA-14D/Pp2B-14D, CanA-14F, CanB and CanB2) indicate CanB2 knockdown completely rescues zyd seizures, CanA-14D and CanA-14F knockdowns partially reduce seizures (N = 4 groups of >10 flies/genotype). (H) Pan-glial knockdown of Pp2B-14D and CanA-14F partially rescues the zyd HS seizures phenotype (~25% rescue for Pp2B-14D, p=0.0032; and ~60% rescue for CanA14F, p<0.0001). Knocking down the two genes simultaneously rescues zyd seizures, with only ~10% of flies showing seizures (~90% rescue, p<0.0001, N = 3 groups of >10 flies/genotype). (I) Overexpressing a dominant-negative form on Pp2B-14D (CanAH217Q) rescues ~50% of zyd seizures regardless of the driver used (repo: p<0.0001; NP2222: p=0.0006; GMR54H02- p=0.0004. N = 3 groups of >10 flies/genotype). (J) Larval Ca2+ imaging in cortex glia expressing myrGCaMP6s indicates the elevated basal Ca2+ fluorescence at 25°C observed in zyd mutants relative to wildtype cortex glia (p=0.0003) is not altered following CanB2 knockdown (zyd;;repo>CanB2RNAi, p=0.6096. n ≥ 5 animals/genotype). (K) Microdomain Ca2+ oscillations observed in wildtype cortex glia expressing myrGCaMP6s are abolished in zyd cortex glia and are not restored following either CanB2 or CanA14F knockdown (n ≥ 5 animals/genotype). Error bars are SEM, **=P < 0.01, ***=P < 0.001, ****=P < 0.0001, Student’s t-test.
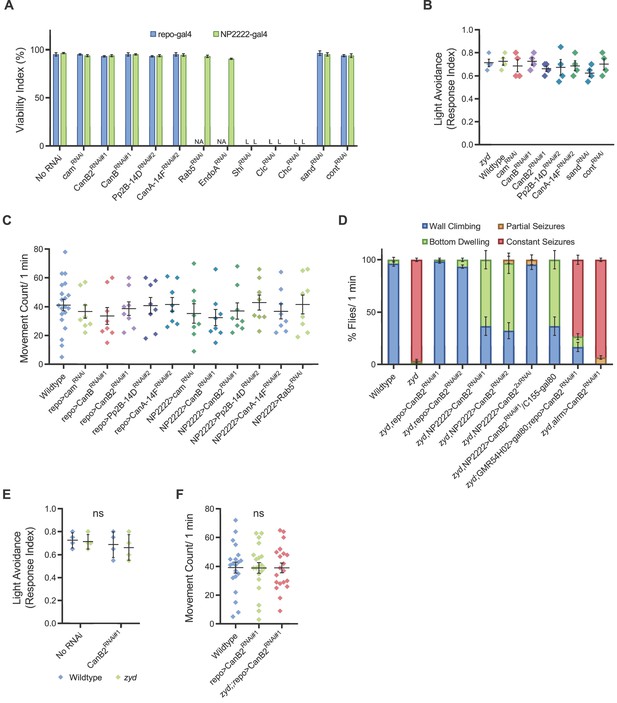
Cortex glial knockdown of calcineurin rescues zyd seizures without affecting intracellular Ca2+.
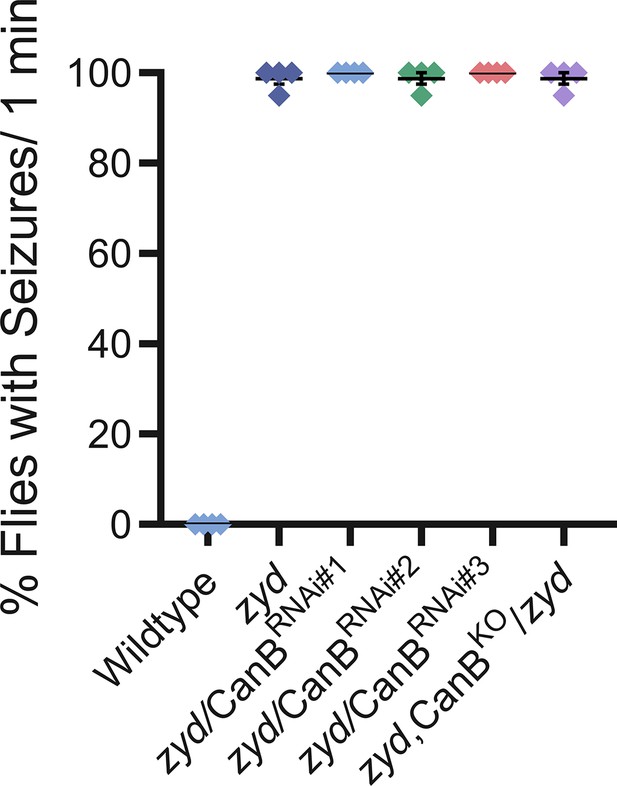
Related to Figures 2 and 7. (A) Viability analysis of the different RNAi used in this study. RNAi was expressed using pan-glial (repo-gal4) or cortex glial specific (NP-2222) drivers (N = 3 groups of 50 embryos/genotype). Control was GFPRNAi. (B) Light avoidance response of 3rd instar larvae expressing the different RNAi hairpins used in this study (N = 4 groups of 20 larvae/genotype). (C) Analysis of the activity level of flies expressing the different RNAi hairpins used in this study (N = 8 flies/genotype). (D) Analysis of HS-induced behaviors of zyd/CanB2RNAi flies (for full details, see Materials and methods). Cortex-glial knockdown of CanB2 leads to complete seizure rescue in ~30% of zyd/CanB2RNAi flies, while the remaining ~70% show an intermediate phenotype. CanB2 knockdown with two copies of the RNAi recapitulates the more robust pan-glial knockdown effect. Inhibiting gal4 expression of the RNAi in neurons with gal80 (C155-gal80) does not alter the rescue effect seen in a single copy cortex glial knockdown (N = 3 groups of >15 flies/genotype). (E) Light avoidance response of wildtype, zyd, repo>CanB2RNAi#1 and zyd;;repo>CanB2RNAi#1 3rd instar larvae (N = 4 groups of 20 larvae/genotype) (F) Activity level analysis of wildtype, zyd and zyd;;repo>CanB2RNAi#1 adult flies (p=0.9843, n = 20 flies/genotype).
The response of zyd/repo>CanB2RNAi flies to a 38.5°C heat-shock is shown.
https://doi.org/10.7554/eLife.44186.009The response of zyd/NP2222>CanB2RNAi flies to a 38.5°C heat-shock is shown.
https://doi.org/10.7554/eLife.44186.010The response of zyd/NP2222>CanB22xRNAi flies to a 38.5°C heat-shock is shown.
https://doi.org/10.7554/eLife.44186.011Representative Ca2+ imaging in repo>CanB2RNAi cortex glia.
GCaMP6s was expressed specifically in cortex glia using the GMR54H02-gal4 driver.
Representative Ca2+ imaging in zyd/repo>CanB2RNAi cortex glia.
GCaMP6s was expressed specifically in cortex glia using the GMR54H02-gal4 driver.
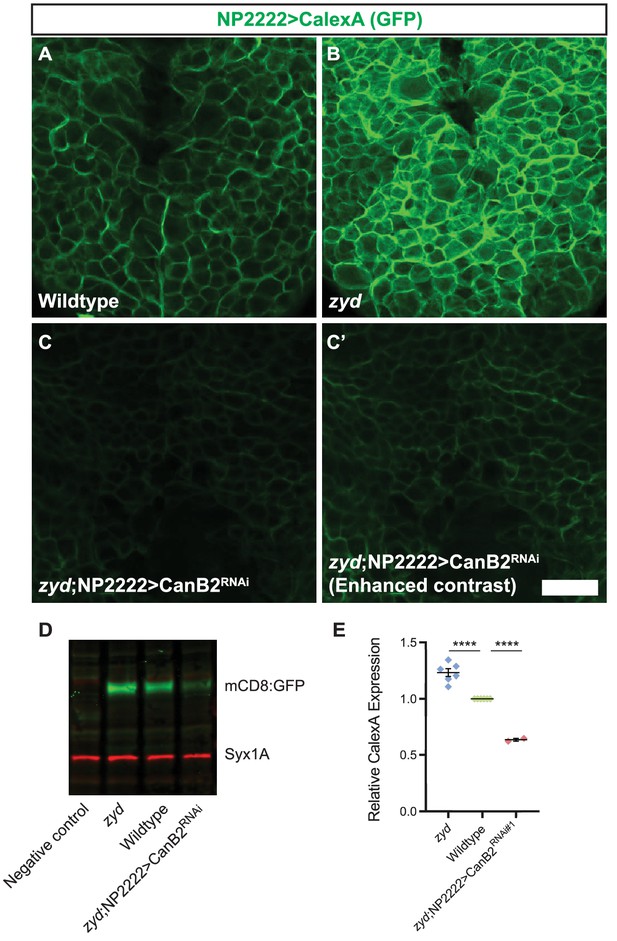
Calcineurin activity is enhanced in zyd cortex glia and can be efficiently suppressed by CanB2 knockdown.
(A-C) Fluorescence microscopy imaging of cortex glial CalexA-derived GFP expression in wildtype (A), zyd (B) and zyd;NP2222>CanB2RNAi (C-C’) larvae. Green: anti-GFP = cortex glial CN activity (animals were reared at 25°C, Scale bar = 20 μm, N ≥ 5 animals/genotype). (D-E) Western blot analysis of cortex glial CalexA derived GFP expression (NP2222>CalexA) in zyd, wildtype and zyd;NP2222>CanB2RNAi adult heads. CN activity is enhanced by ~25% (p<0.0001) in zyd cortex glia and reduced by ~35% (p<0.0001) in CanB2RNAi animals (N ≥ 2 experiment, five heads/sample). GFP signals in each experiment were normalized to wildtype. Error bars are SEM, ****=P < 0.0001, Student’s t-test.
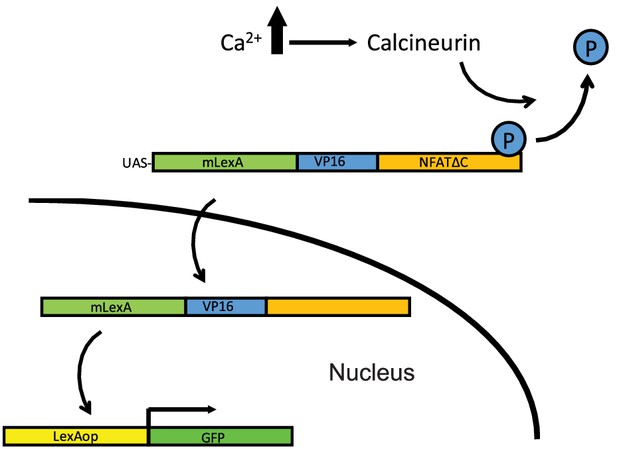
Schematic representation of the CalexA system.
Related to Figure 3. Schematic representation of the CalexA system. Sustained neural activity induces CN activation and dephosphorylation of a chimeric transcription factor LexA-VP16-NFAT (termed CalexA) which is then transported into the nucleus. The imported dephosphorylated CalexA drives GFP reporter expression in Ca2+-active cells.
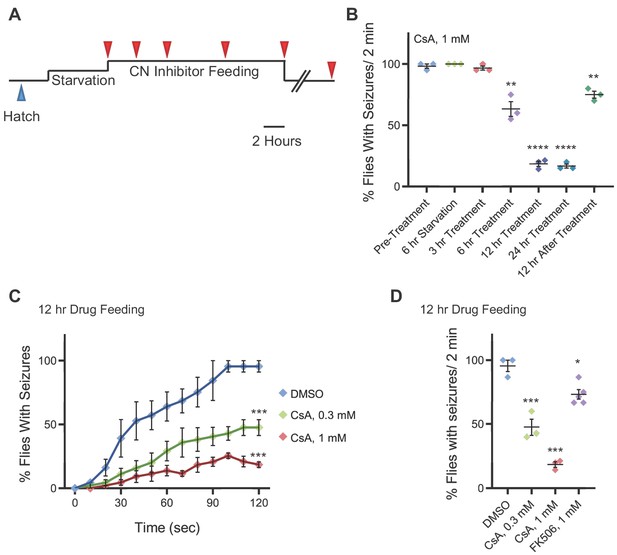
Pharmacologically targeting calcineurin activity suppresses zyd heat shock-induced seizures.
(A) Schematic representation of the experimental design. Adult male flies (<1 day old) were starved for 6 hours and fed with liquid medium containing CN inhibitors for 3, 6, 12 or 24 hours (red arrowheads), before testing for HS induced seizures. Flies were also tested 12 and 24 hours after drug withdrawal. (B-D) Behavioral analysis of HS induced seizures. (B) Summary of all time points for CsA treatment (N = 3 groups of 15–20 flies/treatment. 6 hours feeding: p=0.005; 12/24 hours feeding: p<0.0001; 12 hours drug withdrawal: p=0.0022). (C) Flies were fed with 0.3 mM or 1 mM of CsA for 12 hours. Feeding with 1 mM CsA reduces seizures by ~75% (p<0.0001). The effect of CsA treatment on HS-induced seizures shows a significant dose-dependent reduction in seizure occurrence (N = 3 groups of >15 flies/treatment). (D) After 2 minutes of heat-shock, seizures were reduced by ~50% (p=0.041) in flies that were fed with 0.3 mM CsA, and by ~80% (p=0.0062) in flies that were fed with 1 mM CsA. A ~ 20% (p=0.043) reduction in seizures was observed when flies were fed with 1 mM FK506 (N = 3 groups of >15 flies/treatment). Error bars are SEM, *=P < 0.05, **=P < 0.01, ****=P < 0.0001, Student’s t-test.
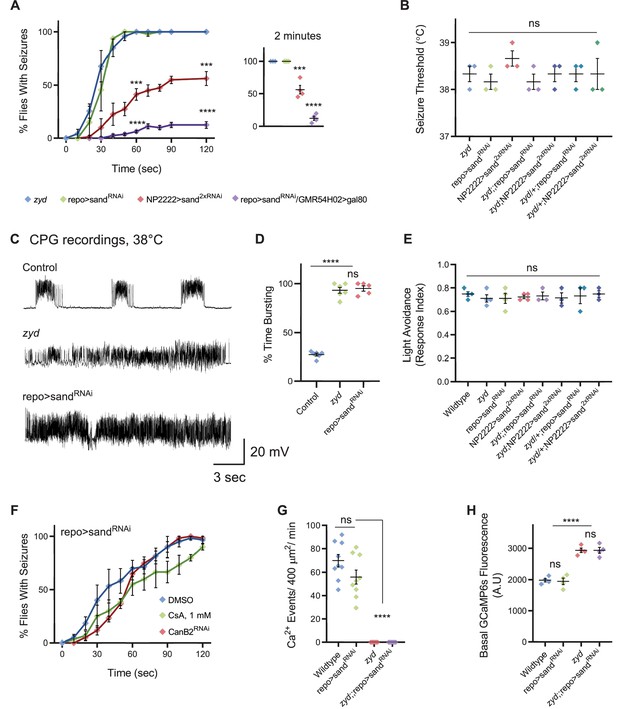
Cortex glial knock-down of sandman, a K2P channel, recapitulates zyd phenotypes.
(A-B) Behavioral analysis of HS induced seizures. (A) Knockdown of sandman (sand) in different glial subtypes: pan-glial (repo), cortex glial (NP2222) and in all glia other than cortex glia (repo>sandRNAi/GMR54H02>gal80 in which gal80 is constitutively inhibiting gal4 activity and sandRNAiexpression only in cortex glia). Inset shows analysis after 2 minutes of HS (p=0.0006 for NP2222>sand2xRNAi, p<0.0001 for repo>sandRNAi/GMR54H02>gal80, N = 4 groups of >10 flies/genotype). (B) Temperature threshold of repo>sandRNAi (p=0.5185) and NP2222>sand2xRNAi (p=0.2302) seizures in comparison to zyd (N = 3 groups of 10/temperature/genotype). (C) Representative voltage traces of spontaneous CPG activity at larval 3rd instar muscle 6 at 38°C in wildtype, zyd and repo>sandRNAi (n ≥ 5 preparations/genotype). (D) Quantification of average bursting duration for CPG recordings of the indicated genotypes at 38°C. (E) Light avoidance assay reveals no defect in this behavior at 25°C (N = 3 groups of 20 flies/genotype). (F) Behavioral analysis of HS-induced seizures. Seizures in repo>sandRNAi animals were not suppressed with either CanB2RNAi#1 or by feeding flies with 1 mM CsA (N = 3 groups of 20 flies/genotype/treatment). (G-H) Ca2+ imaging in larval cortex glial cells using myrGCaMP6s. (G) The average rate of microdomain Ca2+ events was reduced in repo>sandRNAi cortex glia relative to wildtype (20.36 ± 5.5 and 69.83 ± 5.3, p<0.0001). Knockdown of sand on the zyd background did not restore zyd Ca2+ microdomain events (n ≥ 5 animals/genotype). (H) Average myrGCaMP6s fluorescence in cortex glia at 25°C. Elevated basal fluorescence of GCaMP6s in zyd relative to wildtype cortex glia (p=0.0003) is not altered following sand knockdown (zyd;;repo>sandRNAi, N = 4 animals/genotype). Error bars are SEM, ***=P < 0.001, ****=P < 0.0001, Student’s t-test.
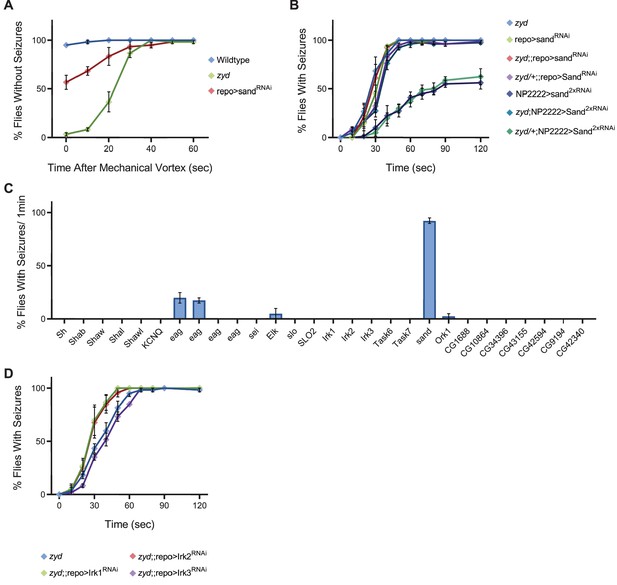
Cortex glial knockdown of sandman, a K2P channel, reproduces zyd phenotypes.
Related to Figure 5. (A) Quantification of vortex-induced seizures in wildtype, zyd and repo>sandRNAi adults. (B-C) Behavioral analysis of HS-induced seizures. (B) Different genetic combinations of the zyd mutation and sandRNAi were analyzed. Combining a single copy of the zyd mutation does not change the partial seizures observed in cortex-glial knockdown of sand (N = 4 groups of >10 flies/genotype). (C) Pan-glial knockdown of other members of the Drosophila K+ channel family besides sand do not cause seizures (N = 2 groups of ≥10 flies/genotype). (D) Pan-glial knockdown of members of the Drosophila Kir family does not enhance zyd seizures (N = 4 groups of >10 flies/genotype).
The response of repo>sandRNAi flies to a 38.5°C heat-shock is shown, following by the response of NP2222>sand2xRNAi flies to the same condition.
https://doi.org/10.7554/eLife.44186.019Representative Ca2+ imaging in repo>sandRNAi cortex glia.
GCaMP6s was expressed specifically in cortex glia using the GMR54H02-gal4 driver.
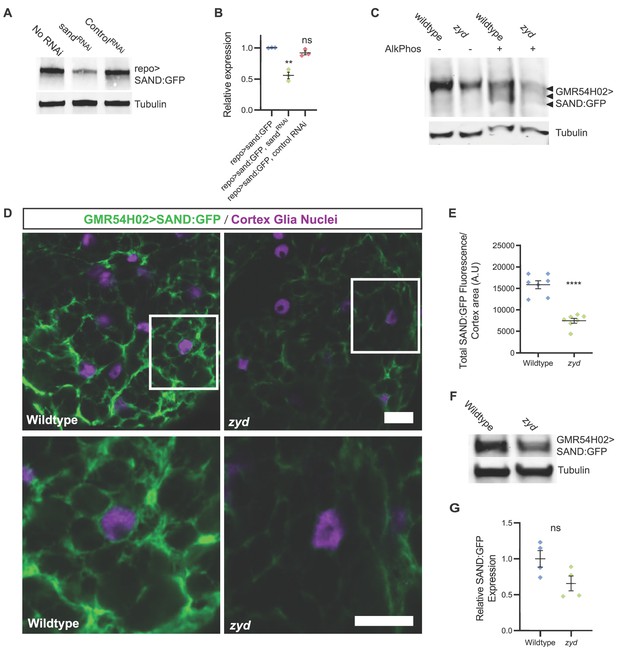
Sand protein levels and plasma membrane localization are reduced in zyd mutants.
(A-B) Western blot analysis of sandRNAi knockdown of SAND:GFP (both sand:GFP and sandRNAi are driven with the pan glial driver, repo). SAND:GFP expression in sandRNAi knockdown is reduced by ~45% (p=0.0011), while the expression of a control RNAi does not significantly change SAND:GFP expression (N ≥ 2 experiments, three head extracts per sample). GFP signals in each experiment were normalized to control. (C) Mn2+ phosphate binding tag (Phos-tag) gel electrophoresis analysis shows that SAND:GFP is not differentially phosphorylated in zyd relative to wildtype cortex glia, indicated by a single SAND:GFP band. Multiple bands were detected when samples were pre-treated with alkaline phosphatase, indicating SAND:GFP is phosphorylated on multiple phosphorylation sites in cortex glia (N = 3, five heads/sample). (D) Immunofluorescence of 3rd instar larval ventral nerve cords (VNCs). Cortex glial levels of SAND:GFP are reduced in zyd relative to wildtype (magenta: anti-repo, glial nuclei; green: anti-GFP, SAND:GFP; scale bars: 10 μm in upper panels, 5 μm in lower panels). (E) Average sand:GFP fluorescence in cortex glia of wildtype and zyd. SAND:GFP fluorescence is reduced by 53 ± 6.85% in zyd cortex glia. (n = 8 larvae/genotype). (F-G) Western blot analysis of SAND:GFP expression level in wildtype and zyd cortex glia (both driven with the cortex glial driver, GMR54H02-gal4). SAND:GFP expression in zyd cortex glia is reduced by ~34 ± 15% relative to wildtype (N ≥ 4 experiments, three heads/sample). Error bars are SEM, **=P < 0.01, ****=P < 0.0001 Student’s t-test.
Sand protein levels and plasma membrane localization are reduced in zyd mutants.
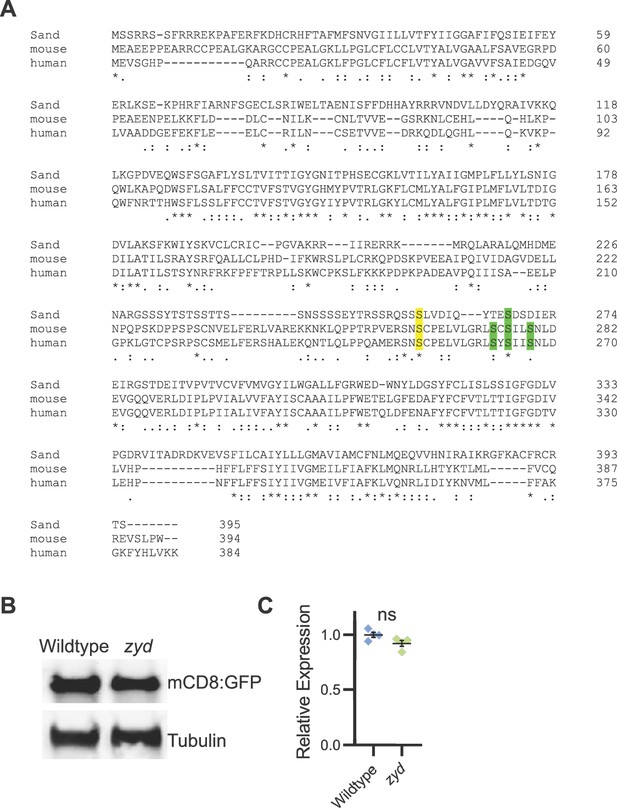
Related to Figure 6. (A) Alignment of the protein sequences of mammalian KCNK18 (human and mouse) and Drosophila SAND. Consensus phosphorylation sites for PKA (yellow) and MARK1 (green) are shown. (B-C) Western blot analysis of mCD8:GFP expression level in wildtype and zyd cortex glia (both driven with the cortex glial driver, GMR54H02-gal4). mCD8:GFP expression in zyd cortex glia is similar to wildtype (N = experiments, three heads/sample). Error bars are SEM.
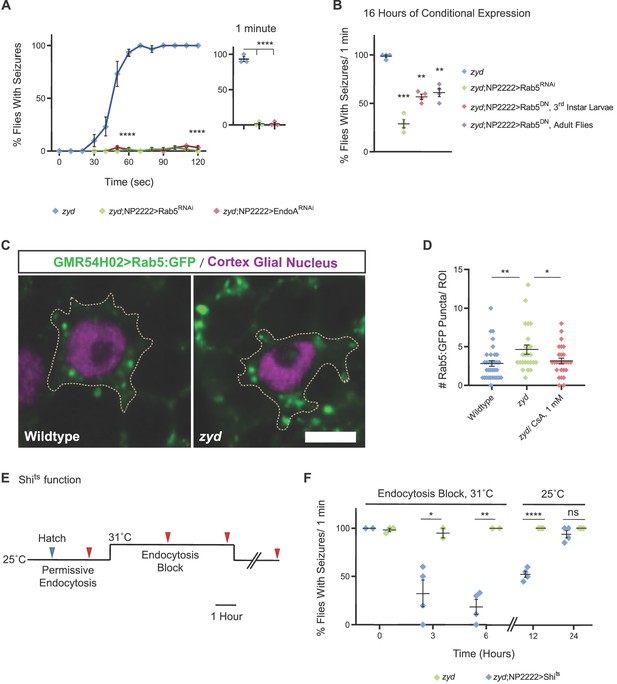
Cortex glial inhibition of endocytosis rescues zyd seizures.
(A-B) Behavioral analysis of HS induced seizures. (A) Cortex glial knockdown of Rab5 and EndoA rescues seizures in the zyd mutant. Inset shows effects after 1 minute of HS (N = 3 groups of 20 flies/genotype, p<0.0001 at 1 minute and 2 minutes). (B) Cortex glial conditional overexpression of Rab5RNAi and dominant-negative Rab5 (Rab5DN) using UAS/gal4/gal80ts. Rearing adult flies at the restrictive temperature (>30°C) for gal80ts allows expression of Rab5RNAi or Rab5DN only in adults. A significant reduction in seizures was seen after 16 hr of incubation at the restrictive temperature for gal80ts (31°C), with only ~25% zyd/Rab5RNAi flies showing seizures (p=0.0006), and ~40% zyd/Rab5DN larvae (p=0.0015)/adult flies (p=0.0055) not showing seizures. (N = 3 groups of >10 flies/genotype). (C) Fluorescence images showing accumulation of Rab5::GFP puncta in zyd cortex glia relative to wildtype cortex glia. Rab5::GFP was expressed using a cortex glial-specific driver (GMR54H02-gal4; scale bar = 5 μm. n ≥ 5 animals/genotype). (D) Analysis of the number of large (>0.1 μm2) Rab5::GFP puncta in wildtype and zyd cortex glia. The number of Rab5::GFP puncta in zyd cortex glia was increased relative to wildtype (average of 4.64 ± 0.58 and 2.85 ± 0.37 puncta/ROI respectively, p=0.0088). The number of large Rab5::GFP puncta in zyd treated with 1 mM CsA for 24 hr was decreased relative to zyd (average of 3.19 ± 0.37 puncta/ROI, p=0.0378; n ≥ 25 ROIs/3 animals/genotype/treatment). (E-F) Conditional inhibition of endocytosis by cortex glial overexpression of shits. (E) Schematic representation of the experimental design. Adult zyd; NP2222 >shits male flies (>1 day old) were incubated at the shits restrictive temperature (31°C) for 3 or 6 hr and then tested for HS-induced seizures (red arrowheads, N = 3 groups of >15 flies/time point). (F) Behavioral analysis of HS induced seizures. Left: A significant reduction in seizures is observed in flies that were incubated at 31°C for 3 hr (p=0.0283) or 6 hr (p=0.013). (N = 3 groups of >15 flies/time point). Right: zyd;NP2222 >Shits flies seizures re-occur after removal from the Shits restrictive temperature (25°C, N = 4 groups of 10–15 animals/time point; 12 hr: p<0.0001). Error bars are SEM, *=P < 0.05, **=P < 0.01, ***=P < 0.001, ****=P < 0.0001, Student’s t-test.
The response of zyd/NP2222>Rab5RNAi flies to a 38.5°C heat-shock is shown.
https://doi.org/10.7554/eLife.44186.024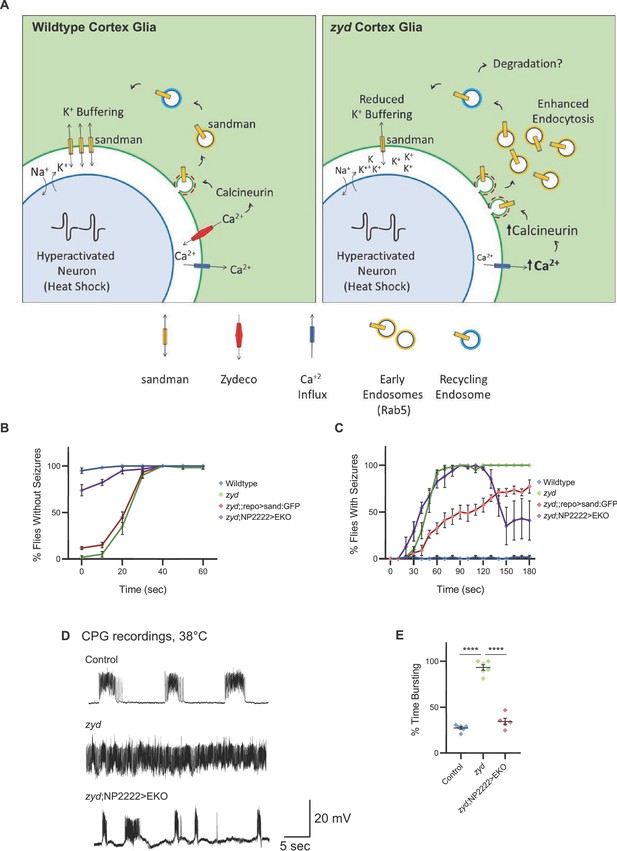
Enhancing glial K+ buffering by overexpressing a leak K+ channel rescues zyd seizures.
(A) A model for zyd function in seizure susceptibility is depicted. In wildtype cortex glia (left), oscillatory Ca2+ signaling maintains normal cortex glia-to-neuron communication and a balanced extracellular ionic environment. In zyd cortex glia (right), the basal elevation of Ca2+ leads to hyperactivation of CN and enhanced endocytosis with accumulation of early endosomes. This disrupts the endo-exocytosis balance of the K2P leak channel sandman (and potentially other cortex glial membrane proteins) and impaired glial K+ buffering. (B) Behavioral analysis of the recovery from vortex-induced seizures. Pan-glial over expression of SAND:GFP partially rescues zyd seizures (~15%), while cortex-glial overexpression of a genetically modified constitutively-open Shaker K+ channel (termed EKO) rescues ~75% of zyd seizures. (N = 3 groups of 20 flies/genotype). (C) Behavioral analysis of HS induced seizures. Cortex glial overexpression of EKO lead to a dramatic change in the behavior of ~60% of zyd animals, showing partial recovery from the seizure phenotype to bottom dwelling and hypoactivity. (D-E) CPG recordings revealed that zyd;NP2222>EKO larvae regain rhythmic muscle activity. (D) Representative voltage traces of spontaneous CPG activity at 3rd instar larval muscle 6 at 38°C in wildtype, zyd and zyd;NP2222>EKO (n ≥ 3 preparations/genotype). Cortex-glial expression of EKO eliminates the continuous CPG seizures observed in zyd mutants. (E) Quantification of average bursting duration for CPG recordings of the indicated genotypes at 38°C. Error bars are SEM, ****=P < 0.0001, Student’s t-test.
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Genetic reagent(D. melanogaster) | w1118 | |||
| Genetic reagent(D. melanogaster) | zyd1 | Melom and Littleton, 2013 | zyd | |
| Genetic reagent(D. melanogaster) | repo-gal4 | Lee and Jones, 2005 | ||
| Genetic reagent(D. melanogaster) | NP2222-gal4 | Hayashi et al., 2002 | RRID:DGGR_112830 | |
| Genetic reagent(D. melanogaster) | GMR5H02-gal4 | RRID:BDSC_45784 | ||
| Genetic reagent(D. melanogaster) | GMRH02-lexA | Gift of Gerald M Rubin | ||
| Genetic reagent (D. melanogaster) | UAS-cam-RNAi | RRID:BDSC_34609 | CamRNAi | |
| Genetic reagent (D. melanogaster) | UAS-CanB-RNAi | RRID:BDSC_27307 | CanBRNAi#1 | |
| Genetic reagent (D. melanogaster) | UAS-CanB-RNAi | VDRC 21611 RRID:FlyBase_FBst0454139 | CanBRNAi#2 | |
| Genetic reagent (D. melanogaster) | UAS-CanB-RNAi | VDRC 52390 RRID:FlyBase_FBst0469806 | CanBRNAi#3 | |
| Genetic reagent (D. melanogaster) | UAS-CanB2-RNAi | RRID:BDSC_27270 | CanB2RNAi#1 | |
| Genetic reagent (D. melanogaster) | UAS-CanB2-RNAi | VDRC 104370 | CanB2RNAi#2 | |
| Genetic reagent (D. melanogaster) | UAS-CanB2-RNAi | VDRC 28764 RRID:FlyBase_FBst0457632 | CanB2RNAi#3 | |
| Genetic reagent (D. melanogaster) | UAS-CanB2-RNAi | RRID:BDSC_38971 | CanB2RNAi#4 | |
| Genetic reagent (D. melanogaster) | UAS-Pp2B-14D-RNAi | RRID:BDSC_25929 | Pp2B-14DRNAi#1 | |
| Genetic reagent (D. melanogaster) | UAS-Pp2B-14D-RNAi | RRID:BDSC_40872 | Pp2B-14DRNAi#2 | |
| Genetic reagent (D. melanogaster) | UAS-CanA-14F-RNAi | RRID:BDSC_38966 | CanA-14FRNAi#1 | |
| Genetic reagent (D. melanogaster) | UAS-CanA-14F-RNAi | VDRC 30105 RRID:FlyBase_FBst0458337 | CanA-14FRNAi#2 | |
| Genetic reagent (D. melanogaster) | UAS-cac-RNAi | VDRC 104168 | cacRNAi | |
| Genetic reagent (D. melanogaster) | UAS-sand-RNAi | VDRC 47977 RRID:FlyBase_FBst0467653 | sandRNAi#1 | |
| Genetic reagent (D. melanogaster) | UAS-sand-RNAi | RRID:BDSC_25853 | sandRNAi#2 | |
| Genetic reagent (D. melanogaster) | UAS-Rab5-RNAi | RRID:BDSC_34832 | Rab5RNAi | |
| Genetic reagent (D. melanogaster) | UAS-Rab5.S43N | RRID:BDSC_42703 RRID:BDSC_42704 | Rab5DN | |
| Genetic reagent (D. melanogaster) | UAS-zyd-RNAi | VDRC 40987 RRID:FlyBase_FBst0463881 | zydRNAi | |
| Genetic reagent (D. melanogaster) | UAS-EKO[+] | White et al., 2001 | RRID:BDSC_40973 | |
| Genetic reagent (D. melanogaster) | CalexA | Masuyama et al., 2012 | RRID:BDSC_66542 | |
| Genetic reagent (D. melanogaster) | Tub-gal80ts | RRID:BDSC_7018 RRID:BDSC_7019 | ||
| Genetic reagent (D. melanogaster) | UAS- Pp2B-14FH217Q | Takeo et al., 2012 | RRID:DGGR_109869 | |
| Genetic reagent (D. melanogaster) | CanB2KO | Nakai et al., 2011 | Gift of Toshiro Aigaki | |
| Genetic reagent (D. melanogaster) | UASc-sand:GFP | Generated in this study | ||
| Antibody | Rabbit polyclonal anti-GFP | ThermoFisher | #A11122 RRID:AB_221569 | Western blot, 1:5000 |
| Antibody | Mouse monoclonal anti-syx1A | DSHB | #8C3 RRID:AB_528484 | Western blot, 1:500 |
| Antibody | Mouse monoclonal anti-Tubulin | Sigma Aldrich | #T5168 RRID:AB_477579 | Western blot, 1:1,000,000 |
| Antibody | Rabbit polyclonal anti-cleaved-Drosophila DCP1 | Cell Signaling | #9578 RRID:AB_2721060 | Western blot, 1:250 |
| Antibody | Mouse monoclonal anti-repo | DSHB | #8D12 RRID:AB_528448 | IF, 1:25 |
| Antibody | Rat monoclonal anti-elav | DSHB | #7E8A RRID:AB_2800446 | IF, 1:50 |
| Antibody | Rabbit polyclonal antiGFP-488 | Invitrogen | #A21311 RRID:AB_221477 | IF, 1:500 |
| Antibody | Goat polyclonal anti-Mouse405 | Life technologies | #A31553 RRID:AB_221604 | IF, 1:3000 |
| Antibody | Goat polyclonal anti-Rat555 | Invitrogen | #A21434 RRID:AB_2535855 | IF, 1:3000 |
| Antibody | IRDye680LT Goat anti-Mouse IgG Secondary Antibody | LI-COR | #926–68020 RRID:AB_10706161 | Western blot, 1:3000 |
| Antibody | IRDye800CW Goat anti-Rabbit IgG Secondary Antibody | LI-COR | #926–32211 RRID:AB_621843 | Western blot, 1:3000 |
| Chemical compound, drug | CyclosporinA | Sigma Aldrich | #30024 | 1 mM |
| Chemical compound, drug | FK506 | InvivoGen | tlrl-fk5 | 1 mM |
| Enzyme | Alkaline Phospatase | Promega | #M282A |
-
*For a complete list of all RNAi stocks used in this study, see Supplementary file 2.
Additional files
-
Supplementary file 1
Summary of zyd suppressor/enhancer RNAi screen.
A table summarizing the results of the zyd suppressor/enhancer RNAi screen performed in this study.
- https://doi.org/10.7554/eLife.44186.026
-
Supplementary file 2
Summary of all RNAi hairpins used in this study.
A table summarizing all the RNAi hairpins used for the zyd suppressor/enhancer and CN target RNAi screens.
- https://doi.org/10.7554/eLife.44186.027
-
Supplementary file 3
Summary of RNAi hairpins used in the CN target screen.
A table summarizing all the RNAi hairpins used for the CN target RNAi screens.
- https://doi.org/10.7554/eLife.44186.028
-
Transparent reporting form
- https://doi.org/10.7554/eLife.44186.029