Endothelial PKA activity regulates angiogenesis by limiting autophagy through phosphorylation of ATG16L1
Figures
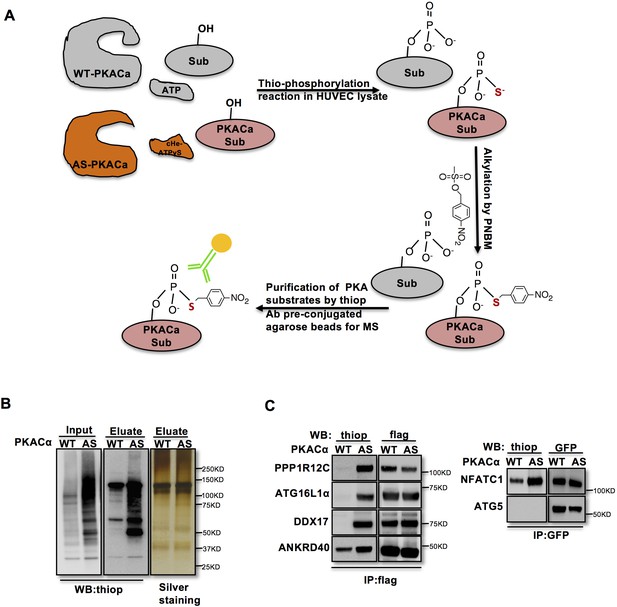
Identification of direct substrates of PKACα.
(A) Strategy for labeling, immunoprecipitation and identifying of PKACα substrates in HUVEC lysates. (B) Thio-phosphorylation of PKA substrates in HUVEC lysates expressing WT-PKACα or AS-PKACα for mass spectrometry analysis. Left panel (input) shows western blot analysis of lysates after alkylation before immunoprecipitation, middle panel (Eluate) shows western blot analysis of the eluted proteins from the immunoprecipitation beads, right panel (Eluate) shows the silver staining of the same samples as middle panel. (C) Validation of the six PKACα substrates identified in the chemical genetic approach screen by overexpressing of potential substrates and WT-PKACα (or AS-PKACα) in 293Tcells, and labeling the substrate in 293 T cell lysates. Western blots are representative of two independent experiments.
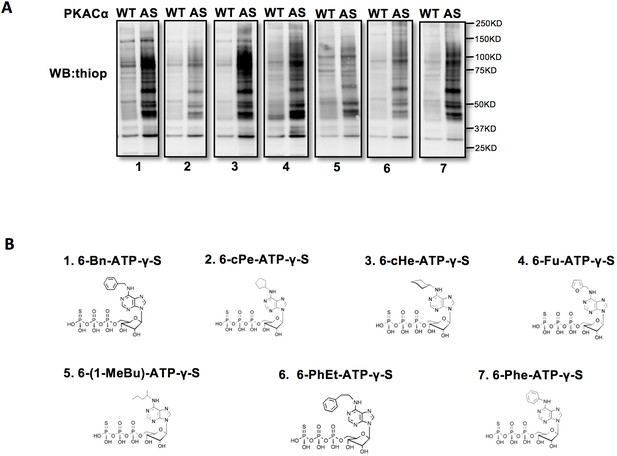
Screen for the best N6-substituted ATPγS analog as a phosphodonor of AS-PKACα.
(A) Kinase assay in HUVEC lysates using WT-PKACα and AS-PKACα. All the western blot membranes were exposed at the same time. (B) Structural formulas of 7 different N6-substituted ATPγS analogs.
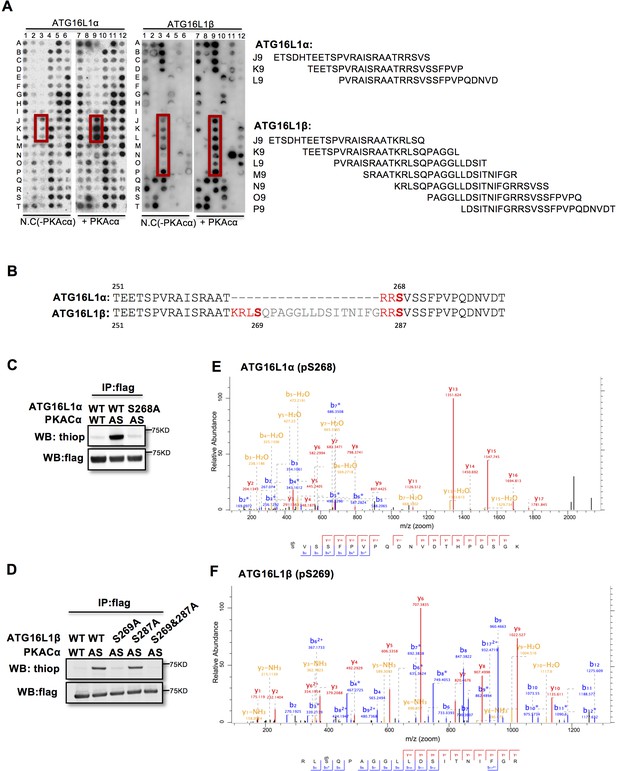
PKACα phosphorylates ATG16L1α at S268 and ATG16L1β at S269.
(A) Peptide SPOT assay of ATG16L1 phosphorylation sites screening. (B) Amino acid sequence including potential PKACα phosphorylation sites in ATG16L1α and ATG16L1β according to the peptide SPOT assay result of Figure 2A. (C–D) Identification of PKACα phosphorylation site in ATG16L1α (C) and ATG16L1β (D). Analysis was performed as in Figure 1C. Western blots are representative of two independent experiments. (E–F) Flag tagged ATG16L1α and ATG16L1β were thio-phosphorylated by AS-PKACα and purified twice using M2 beads and thioP antibody coupled beads, followed by mass spectrometric analysis. LC-MS/MS spectra of the PKA- phosphorylated ATG16L1α tryptic peptide pSVSSFPVPQDNVDTHPGSGK and ATG16L1β tryptic peptide RLpSQPAGGLLDSITNIFGR. The results demonstrate that PKA phosphorylated ATG16L1α at S268 and phosphorylated ATG16L1β at S269.
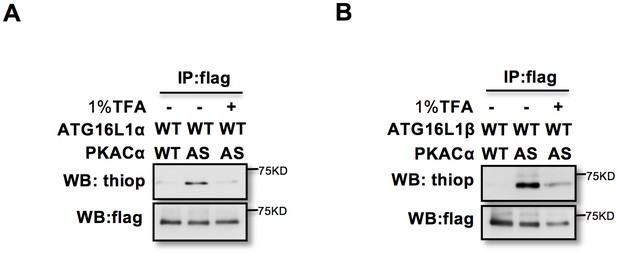
Thiophophorylation is converted to normal phosphoryation by 1% TFA acid–promoted hydrolysis.
(A–B) ATG16L1α (A) and ATG16L1β (B) thiophosphorylated and immunoprecopitated as in Figure 1C, then treated with or without 1% TFA at 37°C for 4 hr.
PKACα mediated phosphorylation of ATG16L1 facilitates its degradation whereas PKA deficiency stabilizes ATG16L1.
(A–A'') HUVECs infected with Flag ATG16L1α (WT or S268A) were treated with 250 µM 6-bnz-cAMP and 20 µg/ml CHX at the time points indicated when they reached confluence (A). Quantifications of Flag ATG16L1α WT (A') and S268A (A'') expression. (B–B'') HUVECs infected with Flag ATG16L1β (WT or S269A) were treated with 250 µM 6-bnz-cAMP and 20 µg/ml CHX at the time points indicated when they reached confluence (B). Quantifications of Flag ATG16L1β WT (B') and S269A (B'') expression. (C–C'') HUVECs infected with GFP ATG16L1α (WT or S268D) were treated with 20 µg/ml CHX at the time points indicated when they reached confluence (C). Quantifications of GFP ATG16L1α WT (C') and S268D (C'') expression. (D–D'') HUVECs infected with GFP ATG16L1β (WT or S269D) were treated with 20 µg/ml CHX at the time points indicated when they reached confluence (D). Quantifications of GFP ATG16L1β WT (D') and S269D (D'') expression. (E–E') HUVECs infected with shRNA (scramble or shPKACα) virus were lysed in RIPA buffer and proteins were analyzed by western blot using indicated antibodies (E). Quantifications of indicated protein expression (E'). (F–F') HUVECs treated with DMSO (control) and 500 µM 6-bnz-cAMP were lysed in RIPA buffer and proteins were analyzed by western blot using indicated antibodies (F). Quantifications of indicated protein expression (F'). (G–G') Endothelial cells isolated from mice (wild type or dnPKAiEC) were lysed in RIPA buffer and ATG16L1 protein was analyzed by western blot (G). Quantifications of indicated ATG16L1 expression (G'). Data present the mean ± SD of 3 independent experiments. *P<0,05; **P<0,01; ***P<0,001; ****P<0,0001.
-
Figure 3—source data 1
Values for quantification of indicated protein expression in Figure 3A, B, C, D, E, F and G.
- https://cdn.elifesciences.org/articles/46380/elife-46380-fig3-data1-v3.xlsx
Autophagy inhibition partially rescues retinal vascular hypersprouting caused by PKA deficiency.
(A–F) Mice were injected with tamoxifen from P1 to P3, then retinas were collected at P6. Isolectin B4 and ESM1 staining of P6 retinas isolated from wtPKA with wtATG5 or ATG5ECKO mice and dnPKAiEC with wtATG5 or ATG5ECKO mice. Representative images are shown (A,D). Quantifications of radial expansion (B), sprouts per 100µm (C), vascular area (E) and ESM1 positive area (F) per field of retinal fronts. 8-10 retinas were measured for each group, *P<0,05; **P<0,01; ***P<0,001; ****P<0,0001.
-
Figure 4—source data 1
Values for quantification of radial expansion (Figure 4B), sprouts per 100 µm (Figure 4C), vascular area (Figure 4E) and ESM1 positive area (Figure 4F) per field of retinal fronts.
- https://cdn.elifesciences.org/articles/46380/elife-46380-fig4-data1-v3.xlsx
Autophagy inhibition partially rescues retinal vascular hypersprouting caused by PKA deficiency.
(A–B) Mice were injected with tamoxifen from P1 to P3, and retinas were collected at P6. Isolectin B4 and ESM1 staining of P6 retinas isolated from wtPKA and dnPKAiEC mice treated with PBS or CQ. Representative images are shown (A,B). (C–D) Quantifications of vascular area (C) and ESM1 positive area (D) per field of retinal fronts. 30–35 fields (images) taken from 6 to 7 retinas were measured for each group, ****p<0,0001.
-
Figure 4—figure supplement 1—source data 1
Values for vascular area (Figure 4—figure supplement 1C) and ESM1 positive area (Figure 4—figure supplement 1D) per field of retinal fronts.
- https://cdn.elifesciences.org/articles/46380/elife-46380-fig4-figsupp1-data1-v3.xlsx
Autophagy inhibition partially rescues retinal vascular hypersprouting caused by PKA deficiency through reducing endothelial cell number but not the ratio of proliferation endothelial cells and apoptosis of endothelial cells.
(A–D) Mice were injected with tamoxifen from P1 to P3, 50 µl 1 mg/ml EdU were I.P injected 2 hr before retinas collecting at P6. CD31 and ERG staining of P6 retinas isolated from wtPKA with wtATG5 or ATG5ECKO mice and dnPKAiEC with wtATG5 or ATG5ECKO mice followed by EdU Click-iT 647 dye labeling. Representative images are shown (A). Quantifications of endothelial cells (ERG positive cells) (B), proliferating endothelial cells (EdU and ERG positive cells) (C) and ratio of proliferating endothelial cells (EdU and ERG positive cells/ERG positive cells) (D). 6–9 retinas were measured for each group, *p<0,05; **p<0,01; ***p<0,001; ****p<0,0001. (E–F) Mice were injected with tamoxifen from P1 to P3, then retinas were collected at P6. CD31 and Cleaved caspase 3 staining of P6 retinas isolated from wtPKA with wtATG5 or ATG5ECKO mice and dnPKAiEC with wtATG5 or ATG5ECKO mice. Representative images are shown (E). Quantifications of endothelial apoptosis (F). 6–8 retinas were measured for each group.
-
Figure 5—source data 1
Values for quantification of endothelial cells (ERG positive cells) (Figure 5B), proliferating endothelial cells (EdU and ERG positive cells) (figure 5C), ratio of proliferating endothelial cells (EdU and ERG positive cells/ERG positive cells) (Figure 5D) and endothelial apoptosis (Figure 5F).
- https://cdn.elifesciences.org/articles/46380/elife-46380-fig5-data1-v3.xlsx
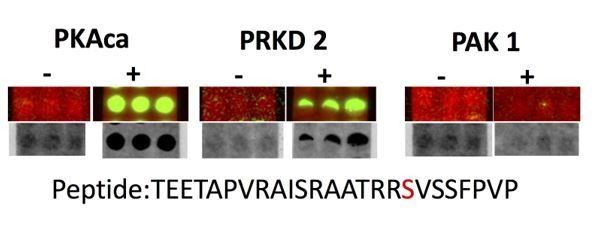
Spot array assay testing additional kinases predicted to phosphorylate the identified PKA sites on ATG16L1.
Tables
List of PKACa substrates.
Proteins are listed according to the log of fold changes of AS-PKACα to WT- PKACα. Two independent experiments have been done to prepare the PKACα substrates samples for mass spectrometric analysis.
| Uniprot | Protein.names | Gene.names | Peptides | Log ratio AS/WT | |
|---|---|---|---|---|---|
| experiment1 | experiment2 | ||||
| Q6AI12 | Ankyrin repeat domain-containing protein 40 | ANKRD40 | 9 | 10 | 10 |
| Q6P6C2 | RNA demethylase ALKBH5 | ALKBH5 | 6 | 10 | 10 |
| Q9NRY4 | Rho GTPase-activating protein 35 | ARHGAP35 | 9 | 10 | 10 |
| E7EVC7 | Autophagy-related protein 16–1 | ATG16L1 | 8 | 10 | 10 |
| J3KPC8 | Serine/threonine-protein kinase SIK3 | SIK3;KIAA0999 | 5 | 10 | 10 |
| A1 × 283 | SH3 and PX domain-containing protein 2B | SH3PXD2B | 4 | 10 | 10 |
| Q8IWZ8 | SURP and G-patch domain-containing protein 1 | SUGP1 | 5 | 10 | 10 |
| Q9UJX5 | Anaphase-promoting complex subunit 4 | ANAPC4 | 5 | 10 | 10 |
| O43719 | HIV Tat-specific factor 1 | HTATSF1 | 4 | 10 | 10 |
| O95644-5 | Nuclear factor of activated T-cells, cytoplasmic 1 | NFATC1 | 5 | 10 | 10 |
| G8JLI6 | Prolyl 3-hydroxylase 3 | LEPREL2 | 3 | 10 | 10 |
| F8W781 | Zinc finger CCCH domain-containing protein 13 | ZC3H13 | 3 | 10 | 10 |
| Q9BZL4 | Protein phosphatase 1 regulatory subunit 12C | PPP1R12C | 21 | 6,04440274 | 7,30701515 |
| O14974 | Protein phosphatase 1 regulatory subunit 12A | PPP1R12A | 26 | 5,72796034 | 7,12654716 |
| Q00537 | Cyclin-dependent kinase 17 | CDK17 | 31 | 6,37867381 | 6,39216838 |
| Q9Y4G8 | Rap guanine nucleotide exchange factor 2 | RAPGEF2 | 21 | 10 | 6,17455504 |
| Q9BYB0 | SH3 and multiple ankyrin repeat domains protein 3 | SHANK3 | 32 | 5,26591421 | 5,6389181 |
| J3KSW8 | Myosin phosphatase Rho-interacting protein | MPRIP | 18 | 4,61398477 | 5,61155414 |
| P31323 | cAMP-dependent protein kinase type II-beta regulatory subunit | PRKAR2B | 19 | 7,05077105 | 5,33509437 |
| P13861 | cAMP-dependent protein kinase type II-alpha regulatory subunit | PRKAR2A | 24 | 5,42841998 | 5,04010629 |
| Q14980-2 | Nuclear mitotic apparatus protein 1 | NUMA1 | 61 | 3,45625969 | 4,47466712 |
| O15056 | Synaptojanin-2 | SYNJ2 | 13 | 4,64022655 | 4,46069701 |
| J3KNX9 | Unconventional myosin-XVIIIa | MYO18A | 10 | 10 | 4,43208178 |
| Q86UU1-2 | Pleckstrin homology-like domain family B member 1 | PHLDB1 | 19 | 5,4105243 | 4,10782285 |
| P28715 | DNA repair protein complementing XP-G cells | ERCC5;BIVM-ERCC5 | 8 | 3,3571826 | 4,09305592 |
| P12270 | Nucleoprotein TPR | TPR | 104 | 3,3333472 | 4,04477536 |
| Q15111 | Inactive phospholipase C-like protein 1;Phosphoinositide phospholipase C | PLCL1 | 10 | 10 | 3,32188704 |
| Q9HD67 | Unconventional myosin-X | MYO10 | 39 | 4,13973415 | 3,21827463 |
| Q14185 | Dedicator of cytokinesis protein 1 | DOCK1 | 34 | 4,50413426 | 3,18515106 |
| O75116 | Rho-associated protein kinase 2 | ROCK2 | 29 | 3,27701864 | 3,08277835 |
-
Table 1—source data 1
The full list of proteins identified in both experiments is provided.
- https://cdn.elifesciences.org/articles/46380/elife-46380-table1-data1-v3.docx
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Strain, strain background (Mus musculus) | Prkar1αtm2Gsm | PMID: 21533282 | ||
| Strain, strain background (Mus musculus) | Tg(Cdh5-cre/ERT2)1Rha | MGI:3848980 | ||
| Genetic reagent (Mus musculus) | ATG5flox/flox | PMID: 16625204 | ||
| Cell line (human) | HUVEC | PromoCell and freshly isolated cells | ||
| Cell line (human) | HEK293T | ATCC | ||
| Transfected construct (Mus) | pRRL.CMV.flag- PKACα | This paper | ||
| Transfected construct (Mus) | pRRL.CMV.flag- PKACαM120G | This paper | ||
| Transfected construct (human) | pECE-M2-PPP1R12A | Addgene:31658 | ||
| Transfected construct (human) | EGFPC1-huNFATc1EE-WT | Addgene: 24219 | ||
| Transfected construct (human) | pDESTmycDDX17, pRRL.CMV.flag-DDX17 | Addgene: 19876 This paper | ||
| Transfected construct (human) | pMRX-IP/SECFP-hATG16L1 pRRL.CMV.flag- ATG16L1α | Addgene: 58994 This paper | ||
| Transfected construct (human) | pRRL.CMV.flag- ATG16L1β | This paper | ||
| Transfected construct (human) | pRRL.CMV.GFP- ATG5 | This paper | ||
| Transfected construct (human) | pRRL.CMV.flag- ATG16L1α S268A | This paper | ||
| Transfected construct (human) | pRRL.CMV.flag- ATG16L1β S269A | This paper | ||
| Transfected construct (human) | pRRL.CMV.flag- ATG16L1β S287A | This paper | ||
| Transfected construct (human) | pRRL.CMV.flag- ATG16L1β S269A and S287A | This paper | ||
| Transfected construct (human) | pRRL.CMV.GFP- ATG16L1α | This paper | ||
| Transfected construct (human) | pRRL.CMV.GFP- ATG16L1α S268D | This paper | ||
| Transfected construct (human) | pRRL.CMV.GFP- ATG16L1β | This paper | ||
| Transfected construct (human) | pRRL.CMV.GFP- ATG16L1β S269D | This paper | ||
| Transfected construct (human) | pLKO.1-TRC cloning shRNA vector | Addgene: 10878 | ||
| Transfected construct (human) | pLKO.1-TRC shPKACα | This paper | ||
| Antibody | Anti-Thiophosphate ester antibody [51-8] | Abcam: ab92570 | WB 1:5000 | |
| Antibody | Anti-Thiophosphate ester antibody [51-8] | Abcam: ab133473 | IP | |
| Antibody | GAPDH (14C10) Rabbit mAb | cell signaling: #2118 | WB 1:1000 | |
| Antibody | Atg16L1 (D6D5) Rabbit mAb | cell signaling: #8089 | WB 1:1000 | |
| Antibody | LC3B (D11) XP Rabbit mAb | cell signaling: #3868 | WB 1:1000 | |
| Antibody | PKA C-α Antibody | cell signaling: #4782 | WB 1:1000 | |
| Antibody | SQSTM1/p62 (D5E2) Rabbit mAb | cell signaling: #8025 | WB 1:1000 | |
| Antibody | Phospho-PKA Substrate (RRXS*/T*) (100G7E) Rabbit mAb | cell signaling: #9624 | WB 1:1000 | |
| Antibody | GFP Tag Polyclonal Antibody | Invitrogen: A11122 | WB 1:1000 | |
| Antibody | goat anti-Actin(c-11) | Santa Cruz Biotechnology: sc-1615 | WB 1:2000 | |
| Antibody | ANTI-FLAG antibody produced in rabbit | Sigma: F7425 | WB 1:1000 | |
| Antibody | Monoclonal ANTI-FLAG M2 antibody produced in mouse | Sigma: F3165 | WB 1:1000 | |
| Antibody | Anti-rabbit IgG, HRP-linked Antibody | cell signaling: #7074 | WB 1:2000 | |
| Antibody | chicken anti-goat IgG-HRP | Santa Cruz Biotechnology: sc-516086 | WB 1:5000 | |
| Antibody | Peroxidase AffiniPure Donkey Anti-Mouse IgG (H+L) | Jackson Immuno Research: 715-035-151 | WB 1:2000 | |
| Antibody | Isolectin GS-IB4 From Griffonia simplicifolia, Alexa Fluor 488 Conjugate | Thermo Fisher: I21411 | IF 1:100 | |
| Antibody | Mouse Endocan/ESM-1 Antibody | R and D: AF1999 | IF 1:100 | |
| Antibody | Donkey anti-Goat IgG (H+L) Cross-Adsorbed Secondary Antibody, Alexa Fluor 555 | Thermo Fisher: A21432 | IF 1:500 | |
| Antibody | Rabbit anti-ERG | Abcam: ab92513 | IF 1:500 | |
| Antibody | rabbit anti-cleaved caspase 3 | R and D: AF835 | IF 1:200 | |
| Antibody | Rat anti-CD31 | BD Pharmingen: BD553370 | IF 1:200 IP | |
| Antibody | Donkey anti-Rat IgG (H+L) Cross-Adsorbed Secondary Antibody, Alexa Fluor 488 | Thermo Fisher: A21208 | IF 1:500 | |
| Antibody | Donkey anti-Rabbit IgG (H+L) Cross-Adsorbed Secondary Antibody, Alexa Fluor 555 | Thermo Fisher: A31572 | IF 1:500 | |
| Commercial assay or kit | Q5 Site-Directed Mutagenesis Kit | NEB: E0554S | ||
| Commercial assay or kit | Lenti-X p24 Rapid Titer Kit | Clontech:632200 | ||
| Commercial assay or kit | Pierce Silver Stain Kit | Thermo Fisher:24600 | ||
| Commercial assay or kit | Click-iT EdU Alexa Fluor 647 Imaging Kit | Thermo fishier:C10340 | ||
| Chemical compound, drug | X-tremeGENE HP DNA transfection reagent | Roche | ||
| Chemical compound, drug | 6-cHe-ATPγS | Biolog:C127 | ||
| Chemical compound, drug | Sp-8-CPT-cAMPS | Biolog:C012 | ||
| Chemical compound, drug | 6-bnz-cAMP | Biolog:C009 | ||
| Software, algorithm | image J | image J | ||
| Software, algorithm | GraphPad Prism 7 | GraphPad Prism 7 | ||
| Other | Recombinant Protein G Agarose | Invitrogen | ||
| Other | sheep anti-Rat IgG-coupled Dynabeads | Invitrogen |
Additional files
-
Supplementary file 1
Array map of spot-synthesized 25-mer overlapping peptides covering the entire ATG16L1 protein.
- https://cdn.elifesciences.org/articles/46380/elife-46380-supp1-v3.docx
-
Transparent reporting form
- https://cdn.elifesciences.org/articles/46380/elife-46380-transrepform-v3.docx






