Coordinated hedgehog signaling induces new hair follicles in adult skin
Figures
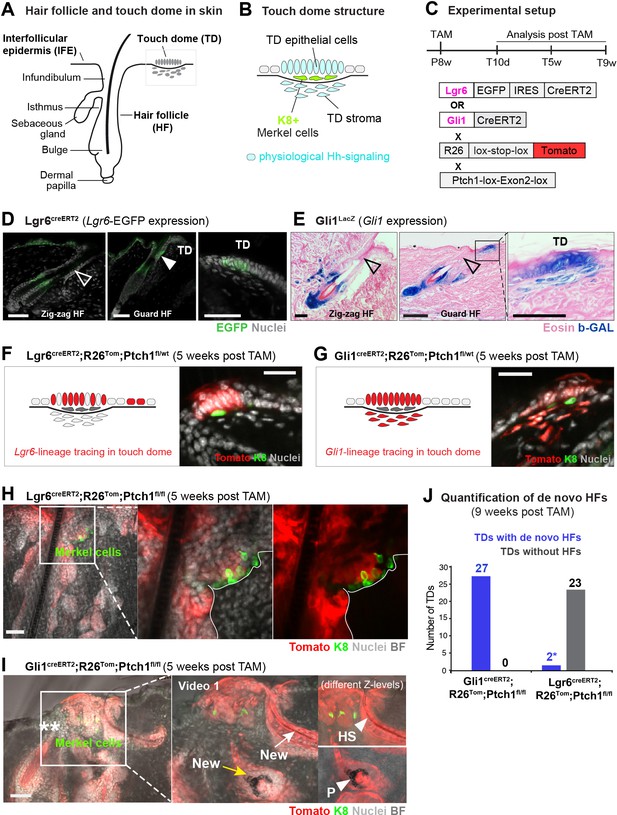
Formation of hair follicle (HF)-like structures in touch domes (TDs) of the Gli1 mouse model.
(A–B) Illustrative cartoon of HF and TD structures in wild type skin. Physiological Hh signaling is present in both TD epithelium and TD stroma. The presence of K8+ Merkel cells is characteristic for TDs. (C) Schematic representation of the experimental timeline and the Lgr6creERT2;R26Tom;Ptch1fl/fl and Gli1creERT2;R26Tom;Ptch1fl/fl mouse models. (D–E) Lgr6EGFP and Gli1LacZ expression in dorsal telogen skin. Filled arrowhead: indicates Lgr6-expression in the HF infundibulum. Empty arrowheads: indicate lack of Lgr6- or Gli1-expression in HF infundibula (n = 3 mice per genotype). (F–I) Mice were treated with tamoxifen (TAM) at 8 weeks of age and dorsal skin was analyzed 5 weeks post TAM. For each genotype ≥3 mice and numerous TDs were analyzed (Supplementary file 1). (F) Illustrative cartoon and experimental Tomato-tracing of Lgr6-expressing cells. TDs of Lgr6creERT2;R26Tom;Ptch1fl/wt control skin were phenotypically normal. (G) Illustrative cartoon and experimental Tomato-tracing of Gli1-expressing cells. TDs of Gli1creERT2;R26Tom;Ptch1fl/wt control skin were phenotypically normal. (H) Tomato-traced TD of Lgr6creERT2;R26Tom;Ptch1fl/fl skin displaying basal cell carcinoma (BCC)-like tumor growth. (I) Tomato-traced TD of Gli1creERT2;R26Tom;Ptch1fl/fl skin with BCC-like tumor growth and several de novo HFs. Asterisks: mark a non-traced infundibulum in the pre-existing Guard HF adjacent to the TD. Inset: different z-levels better depicting specific de novo HF structures. A video containing all z-levels is provided (Figure 1—video 1). Arrows: de novo HFs with continuous tracing into the IFE. Yellow arrow: anagen bulb of a de novo HF. Arrowheads: hair shaft (HS) or pigment (P). (J) Quantification of de novo HFs in the TDs of Gli1creERT2;R26Tom;Ptch1fl/fl and Lgr6creERT2;R26Tom;Ptch1fl/fl mice (9 weeks post TAM; n = 3 mice for each genotype). Asterisk: the Lgr6 mouse model cannot inform on de novo HFs trough lineage tracing; based on morphology we observed in two TDs a single potentially new hair shaft, respectively. TD: touch dome. HS: hair shaft. P: pigment. TAM: tamoxifen. BF: bright field. Scale bars: 50 μm (D–I).
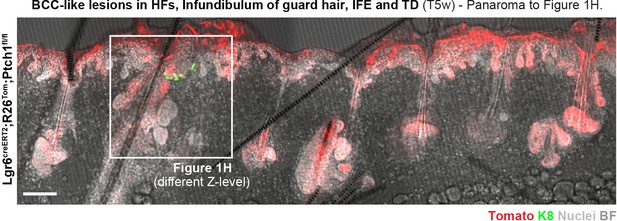
Panorama to Figure 1H.
Lgr6creERT2;R26Tom;Ptch1fl/fl mice were treated with TAM at 8 weeks of age, and dorsal skin was analyzed 5 weeks post TAM (n = 3 mice). BCC-like lesions were observed in the HF isthmus, the infundibulum of guard HFs, the IFE, and in TDs. This panorama shows a different z-level than Figure 1H to better depict HF- and IFE-associated BCC-like lesions. Scale bar: 100 μm.

Phenotype and lineage-tracing pattern in Lgr6creERT2;R26Tom;Ptch1fl/wt and Lgr6creERT2;R26Tom;Ptch1fl/fl skin.
(A–B) Mice were treated with TAM at 8 weeks of age and dorsal skin was analyzed post TAM as indicated in the figure panels (n = 3 mice for each genotype and time point). (A) Lgr6creERT2;R26Tom;Ptch1fl/wt control mice. At all time points, the skin phenotype was normal. IFE and HFs were Tomato-traced as reported (Füllgrabe et al., 2015). The infundibula of Guard HFs were often partially traced (filled arrowheads) and the infundibula of TD-adjacent pre-existing Zig-zag HFs were not traced (empty arrowheads). (B) Lgr6creERT2;R26Tom;Ptch1fl/fl mice. At 5 and 9 weeks post TAM, BCC-like lesions are present in HFs, IFE and TDs; traced and non-traced lesions are present as the Ptch1fl/fl alleles are ‘floxed’ with higher efficiency than LSL-Tomato alleles (data not shown). The infundibula of Guard HFs were partially traced (filled arrowheads) and the infundibula of TD-adjacent pre-existing Zig-zag HFs retained non-traced (empty arrowheads) even when BCC-like lesions formed. Please note that some images are used in the main figures, however with different emphasis and display ((a) extended area view of Figure 1F; (b) same image as in Figure 1H). TD: touch dome. Gu: Guard HF. Zz: Zig-zag HF. BF: bright field. Scale bars: 50 μm.
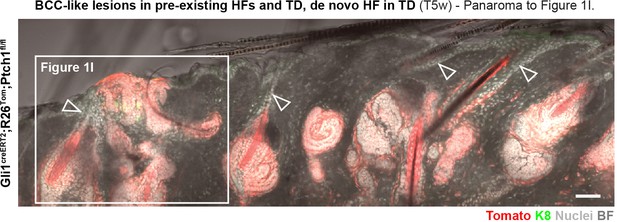
Panorama to Figure 1I.
Gli1creERT2;R26Tom;Ptch1fl/fl mice were treated with TAM at 8 weeks of age, and dorsal skin was analyzed 5 weeks post TAM (n = 3 mice). BCC-like lesions were observed in pre-existing HFs and TDs. Additionally, new HF-like structures were found in the TDs of Gli1creERT2;R26Tom;Ptch1fl/fl skin. Note the untraced infundibulum of pre-existing HFs (arrowheads). Scale bar: 50 μm.
Video containing all recorded z-levels of the touch dome area presented in Figure 1I.
K8 in green. Tomato in red. Nuclear stain in blue. Bright field in gray.
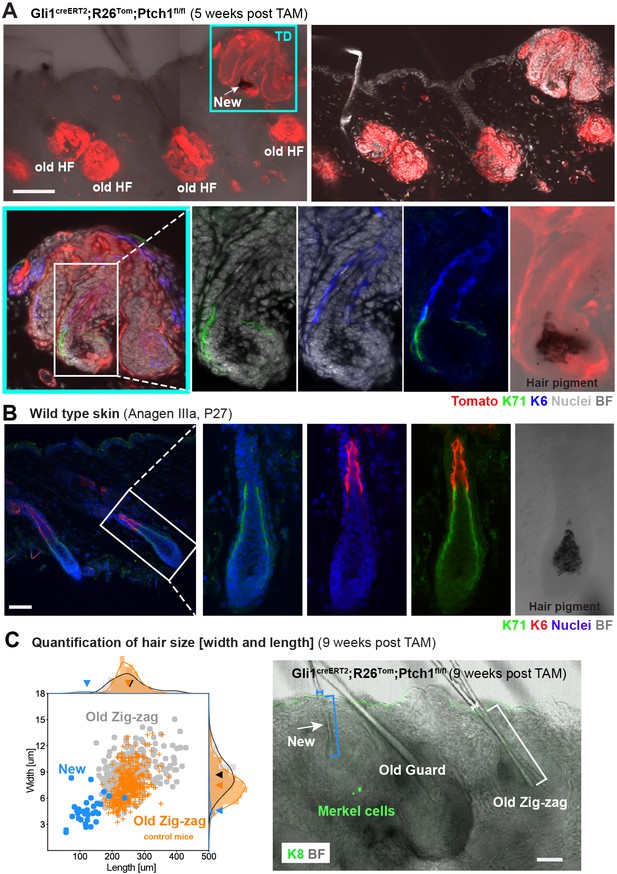
Characterization of de novo hair follicles (HFs) in Gli1creERT2;R26Tom;Ptch1fl/fl touch domes (TDs).
(A) Gli1creERT2;R26Tom;Ptch1fl/fl mice were treated with tamoxifen (TAM) at 8 weeks and dorsal skin was analyzed 5 weeks post TAM treatment (n = 3 mice). The TD area shows a de novo anagen HF (turquois frame). Additionally, traced pre-existing (old) telogen HFs with basal cell carcinoma (BCC)-like growth are present. Inset (white frame): anagen hair bulb of de novo HF showing K71-positive Henle’s layer, K6-positive companion layer, hair pigment and continuous Tomato-tracing from the hair bulb into the TD. (B) Immunofluorescent co-staining of K71 (Henle’s layer) and K6 (companion layer) in a wild type HF of a similar hair cycle stage (Anagen IIIa, P27) (n = 2 mice). (C) Quantification of hair size in Gli1creERT2;R26Tom;Ptch1fl/fl and Gli1creERT2;R26Tom;Ptch1fl/wt mice that were treated with TAM at 8 weeks and dorsal skin was analyzed 9 weeks post TAM treatment. Right panel: De novo telogen HFs with a thin hair shaft formed in the TDs of Gli1creERT2;R26Tom;Ptch1fl/fl skin (white arrow). For the quantification, we analyzed hair shafts of de novo HFs from Gli1creERT2;R26Tom;Ptch1fl/fl mice (blue bracket), old/pre-existing Zig-zag HFs from the same mice (white bracket), and Zig-zag HFs from wild-type-phenotype control mice (Gli1creERT2;R26Tom;Ptch1fl/wt) (n = 3 mice for each genotype; 34 de novo, 314 old/pre-existing, and 437 control HFs; Figure 2—source data 1). Hair shaft length was measured in telogen stage hair shafts from the hair club to the HF opening (as indicated by the blue and white brackets). Left panel: Each dot represents a hair shaft and the dots are colored according to the HF type (blue for de novo HFs, gray for pre-existing Zig-zag HFs, orange for control Zig-zag HFs). De novo hair shafts were significantly smaller (p-value<1.10−6) and thinner (p-value<1.10−6) compared to old/pre-existing and control Zig-zag hairs (Mann-Whitney U test). Arrowheads on the x- and y-axis indicate the mean values of hair shaft length and width of de novo, old/pre-existing or control Zig-zag HFs, respectively. TD: touch dome. HF: hair follcile. TAM: tamoxifen. BF: bright field. Scale bars: 100 μm (A), 50 μm (B, C).
-
Figure 2—source data 1
Quantification of hair width and length.
Data are grouped according to hair type and mouse genotype.
- https://cdn.elifesciences.org/articles/46756/elife-46756-fig2-data1-v1.xlsx
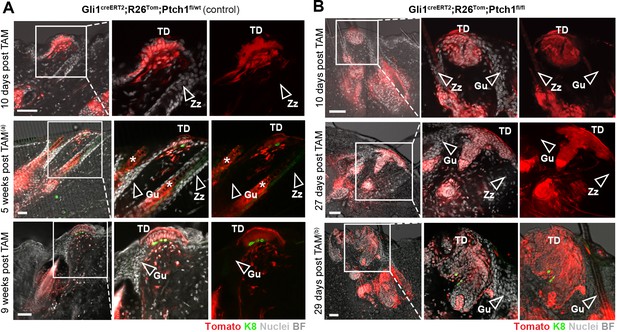
Lineage-tracing pattern of TD and surrounding area in Gli1creERT2;R26Tom;Ptch1fl/wt control and Gli1creERT2;R26Tom;Ptch1fl/fl skin during de novo HF formation.
(A–B) Mice were treated with TAM at 8 weeks of age and dorsal skin was analyzed post TAM as indicated in the figure panels (n = 3 mice for each genotype and time point; except n = 1 for 29 days post TAM). The infundibula of pre-existing TD-adjacent HFs (Guard HFs as well as Zig-zag HFs) in both Gli1creERT2;R26Tom;Ptch1fl/wt control mice (A) and Gli1creERT2;R26Tom;Ptch1fl/fl mice (B) were never traced (empty arrowheads), even when BCC-like lesions and/or de novo HFs formed. In contrast, de novo HFs always showed continuous HF-to-TD tracing. Please note that some images are used in the main figures with different emphasis and display ((a) extended area view of Figure 1G; (b) same image as in Figure 3D). Asterisks: mark hair shafts that are naturally traced during anagen in these mice. TD: touch dome. Gu: Guard HF. Zz: Zig-zag HF. BF: bright field. Scale bars: 50 μm.
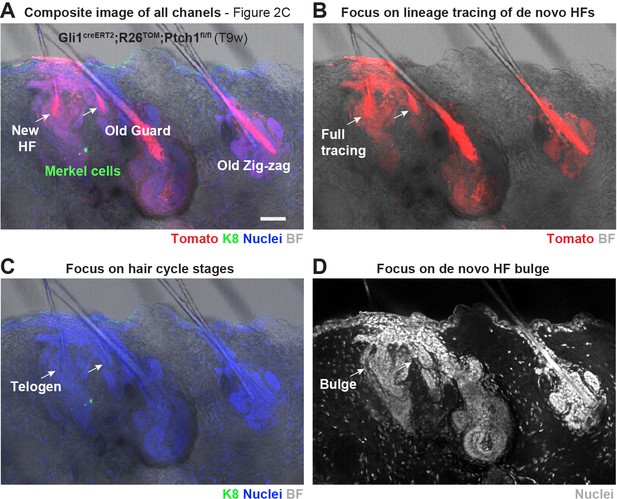
De novo HFs in TDs of Gli1creERT2;R26Tom;Ptch1fl/fl mice 9 weeks post TAM were in telogen and displayed a HF bulge.
(A–D) Gli1creERT2;R26Tom;Ptch1fl/fl mice were treated with TAM at 8 weeks and dorsal skin was analyzed 9 weeks post TAM treatment (n = 3 mice). Note that the BF and K8 channel of this picture are shown in Figure 2C. The de novo HFs originated from the TD next to the guard hair (A). They are fully traced while there is a clearly non-traced gap in the infundibulum of guard HF and pre-existing Zig-zag HFs (B). Both de novo HFs were in telogen stage (C) with a noticeable telogen bulge (D). TAM: tamoxifen. BF: bright field. Scale bars: 50 μm.
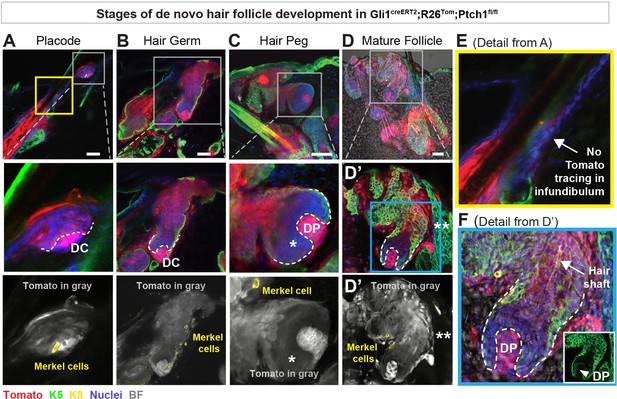
Developmental stages of de novo hair follicles (HFs) in Gli1creERT2;R26Tom;Ptch1fl/fl touch domes (TDs).
(A–F) Gli1creERT2;R26Tom;Ptch1fl/fl mice were treated with tamoxifen (TAM) at 8 weeks and dorsal skin was analyzed 10–36 days post TAM to characterize de novo HFs originating from TDs in the following developmental stages (n = 6 mice): early placode (A), hair germ (B), hair peg (C), and mature follicle (D). These stages recapitulate embryonic HF development (Rendl et al., 2005). Dermal condensates (A, B) or dermal papillae (C, D) are clearly visible, and the de novo HFs – both early and mature – are continuously traced into the TD. Note: HF-matrix cells are Tomato-traced with reduced intensity (* in C), while the infundibulum of pre-existing HFs was not Tomato- traced (** in D’ and arrow in E). Furthermore, mature HFs contain a hair shaft (arrow in F). Inset in F shows K5-positive staining of the HF epithelial cells and a K5-negative dermal papilla. DC: dermal condensate. DP: dermal papilla. BF: bright field. Scale bars: 50 μm (A-D).
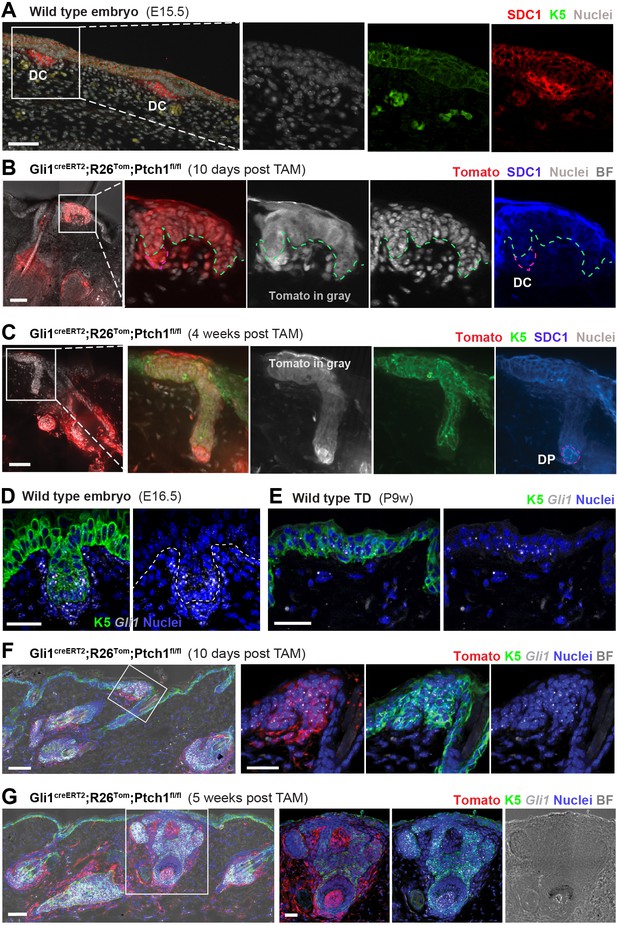
SDC1 protein and Gli1 mRNA expression in developing hair follicles (HFs).
(A) In wild type embryonic skin (E15.5), Syndecan-1 (SDC1) staining highlights dermal condensates (DCs) (n = 2 mice). (B–C) Gli1creERT2;R26Tom;Ptch1fl/fl mice were treated with tamoxifen (TAM) at 8 weeks and dorsal skin was analyzed for SDC1 expression in the newly formed HF buds (n = 3 mice). (B) Early placode stage (image from a TD 10 days post TAM) displaying faint SDC1 staining in dermal condensate cells. Comparable to epithelium of wild type embryonic HF buds, early de novo placodes also express some SDC1. (C) Positive SDC1 staining of dermal papilla (image from a TD 4 weeks post TAM). (D–E) Gli1 RNA-FISH. Both the epithelial placode and dermal condensate have active Hh/Gli signaling in wild type embryonic skin (E16.5) (D) and at lower levels in wild type TDs of adult skin (E). (F–G) Gli1creERT2;R26Tom;Ptch1fl/fl mice were treated with TAM at 8 weeks and dorsal skin was analyzed for Gli1 mRNA expression in the placode stage (10 days post TAM) as well as in an intermediate developmental stage (5 weeks post TAM). Active canonical Hh/Gli signaling was present in all analyzed de novo HF stages in epithelial and dermal papilla cells. Note that Tomato-tracing was visualized using an RFP-antibody. Green and white dashed lines: epithelial-stromal border. Purple dashed line: outlines the dermal condensate. DC: dermal condensate. DP: dermal papilla. TD: touch dome. BF: bright field. For RNA-FISH stainings, n = 2 mice (D, E and G) and n = 1 mouse (F). Scale bars: 25 μm (D–G), 50 μm (A-C, F-G panoramas).
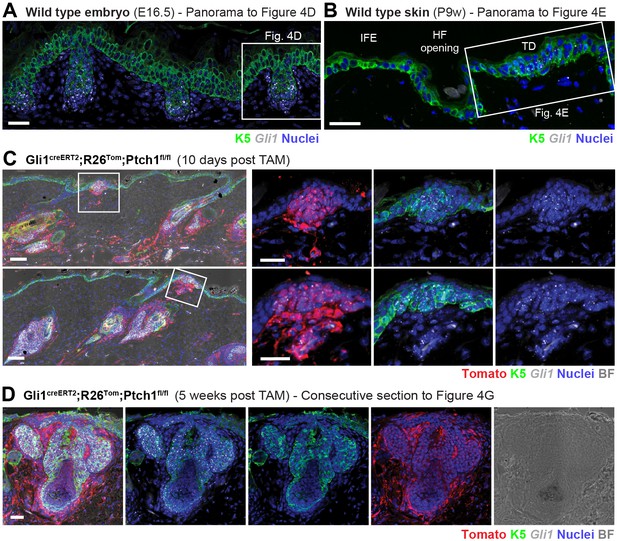
Gli1 mRNA staining reveals cells with active canonical Hh/Gli signaling during de novo HF development in the TDs of Gli1creERT2;R26Tom;Ptch1fl/fl mice.
(A) Panorama to Figure 4D. In wild type embryonic skin (E16.5), the epithelial placodes as well as dermal condensates show clear Gli1 mRNA expression, while the IFE is mostly devoid of Gli1 mRNA. (B) Panorama to Figure 4E. In wild type skin from postnatal 9-week-old mice, Gli1 mRNA is present in the epithelial and stromal components of the TD (white frame) but absent in the rest of the IFE. (C) Gli1creERT2;R26Tom;Ptch1fl/fl mice were treated with TAM at 8 weeks and dorsal skin was analyzed for Gli1 mRNA expression 10 days post TAM with a focus on TDs. Left panels: Panoramas showing TDs in their tissue context. Right panels: Early placodes emerging from TDs clearly display active, canonical Hh signaling in the epithelium and stroma. (D) Gli1creERT2;R26Tom;Ptch1fl/fl mice were treated with TAM at 8 weeks and dorsal skin was analyzed for Gli1 mRNA expression 5 weeks post TAM. Shown is a consecutive section of the HF in Figure 4G. Note that Tomato-tracing was visualized using an RFP-antibody (C-D). For stainings n = 2 mice (A, B, D); n = 1 mouse (C). TAM: tamoxifen. IFE: interfollicular epidermis. BF: bright field. Scale bars: 50 μm (C panaroma), 25 μm (A-B, C insets and D).
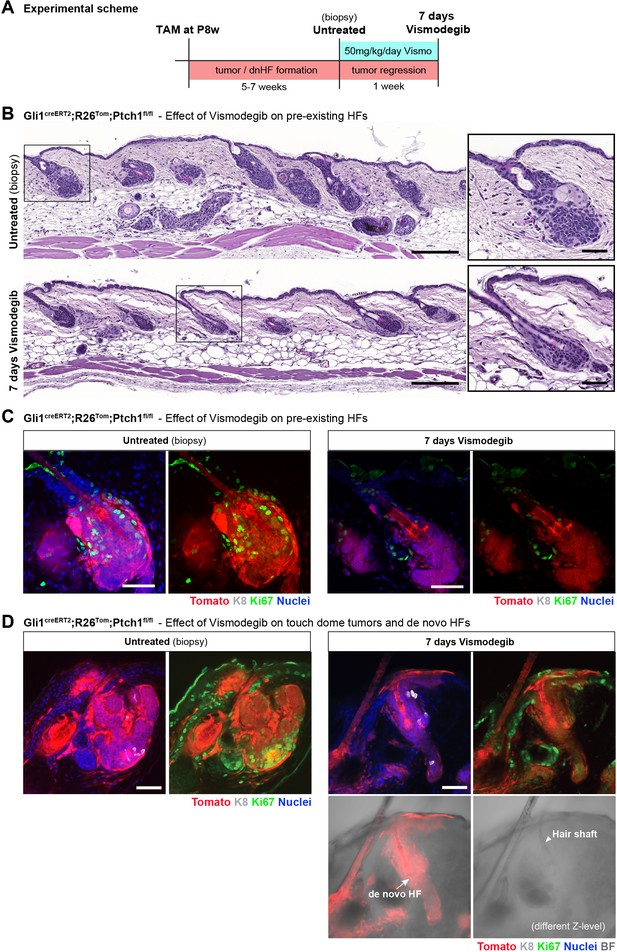
Established de novo hair follicles (HFs) in touch domes (TDs) persist upon short-term vismodegib treatment.
(A) Experimental scheme of vismodegib treatment. Gli1creERT2;R26Tom;Ptch1fl/fl mice were treated with tamoxifen (TAM) at 8 weeks of age. Five to seven weeks post TAM treatment, when de novo HFs were clearly established in the TDs, vismodegib was given daily for a week and dorsal skin was analyzed (n = 3 mice for Gli1creERT2;R26Tom;Ptch1fl/fl, n = 1 mouse for Gli1creERT2;Ptch1fl/fl). (B) Hematoxylin and eosin stainings showing that basal cell carcinoma (BCC)-like lesions were considerably reduced in response to a week of daily vismodegib treatment at a dose of 50 mg/kg body weight. (C) Tumor-cell proliferation in bulge area of pre-existing HFs assessed by Ki67 immunostaining. Bulge areas of untreated control biopsies showed high proliferation, which was almost entirely stalled in 7-day vismodegib samples; in the HF, only the sebaceous glands retained Ki67 expression. (D) BCC-like lesions were present in the TDs of dorsal biopsies taken prior to vismodegib treatment (left panel). In response to vismodegib, the tumor-growth area was considerably reduced while de novo HFs presisted (arrow). The de novo HFs are fully Tomato-traced and have a clearly visible hair shaft (arrowhead). HF: hair follicle. K8: marking TD area. Ki67: marking proliferating cells. TAM: tamoxifen. BF: bright field. Scale bars: 200 μm (B panaroma), 50 μm (B inset, C, D).
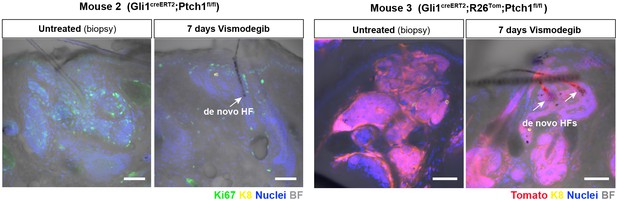
Effect of vismodegib on BCC-like lesions and de novo HFs in TDs.
Shown are two additional examples to Figure 5D. Arrowheads mark de novo HFs. Scale bars: 50 μm.
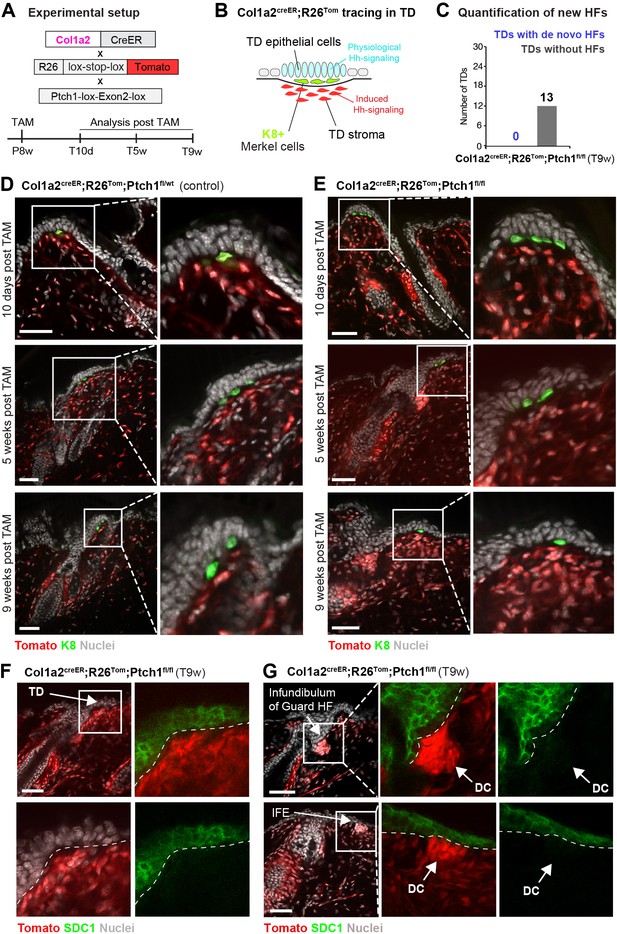
Stromal Hh pathway activation alone is not sufficient to induce hair follicle (HF) neogenesis in the touch domes (TDs) of Col1a2creER;R26Tom;Ptch1fl/flskin.
(A) Schematic representation of the Col1a2creER;R26Tom;Ptch1fl/fl mouse model and the experimental timeline. (B) Illustrative cartoon of Tomato-tracing of Col1a2-expressing cells and Hh-signaling levels in TD epithelium and TD stroma in this mouse model. (C) Quantification of de novo HFs in the TDs of Col1a2creER;R26Tom;Ptch1fl/fl mice treated with tamoxifen (TAM) at 8 weeks. Dorsal skin was analyzed 9 weeks post TAM treatment (n = 3 mice). No de novo HFs were observed. (D–G) Col1a2creER;R26Tom;Ptch1fl/fl and Col1a2creER;R26Tom;Ptch1fl/wt control mice were treated with TAM at 8 weeks and dorsal skin was analyzed 10 days, 5 weeks, and 9 weeks after TAM treatment (n = 3 mice per genotype and time point). (D) TDs of mice with heterozygous Ptch1 deletion were phenotypically normal. (E) TDs of mice with homozygous Ptch1 inactivation did not develop de novo HFs. Frequently, a higher cell density in stroma was observed. (F–G) Syndecan-1 (SDC1) staining was negative in the condensed stroma of TDs (F) as well as in dermal cell condensations (arrows) underneath the IFE and HF infundibula (G) in Col1a2creER;R26Tom;Ptch1fl/fl mice. TD: touch dome. HF: hair follcile. IFE: interfollicular epidermis. DC: dermal cell condensation. Scale bars: 50 μm (D–G).

Leakiness in Col1a2creER;R26Tom;Ptch1fl/fl skin.
A small number of Tomato-traced cells was present in non-tamoxifen treated Col1a2creER;R26Tom;Ptch1fl/fl mice at 8 weeks of age (i.e. start of tamoxifen treatment) (n = 2 mice). BF: bright field. Scale bars: 100 μm.
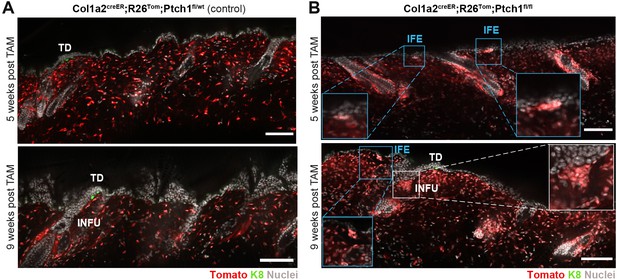
Stromal Hh-pathway activation in Col1a2creER;R26Tom;Ptch1fl/fl skin leads to dermal cell condensations.
(A–B) Col1a2creER;R26Tom;Ptch1fl/fl mice and Col1a2creER;R26Tom;Ptch1fl/wt control mice were treated with TAM at 8 weeks of age and dorsal skin was analyzed 5 or 9 weeks after TAM treatment. Dermal cell condensations were not found in Col1a2creER;R26Tom;Ptch1fl/wt control skin (A) (n = 2 mice), but were frequently detected underneath the interfollicular epidermis (IFE) and HF-infundibulum (INFU) areas of Guard HFs in the skin of Col1a2creER;R26Tom;Ptch1fl/fl mice (B) (n = 3 mice). TD: touch dome. Scale bars: 100 μm.
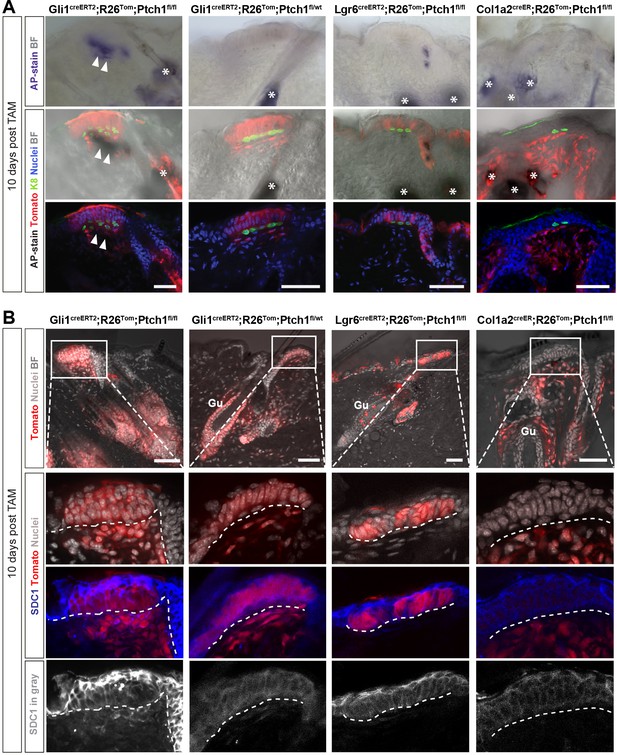
Expression of markers indicative for hair follicle (HF)-induction competent stroma in Gli1creERT2;R26Tom;Ptch1fl/fl touch domes (TDs).
(A–B) Mice were treated with tamoxifen (TAM) at 8 weeks and dorsal skin was analyzed 10 days later using the alkaline phosphatase (AP) enzymatic assay (A; n = 2–3 mice per genotype) or Syndecan-1 (SDC1) immunofluorescence staining (B; n = 3 mice per genotype). (A) The TD stroma of Gli1creERT2;R26Tom;Ptch1fl/fl skin stained AP-positive, indicating pre-dermal condensate formation. Please note that the nuclear staining (DAPI) in the areas of AP-positive signal is present but very dim (quenched). The TD stroma in skin of Gli1creERT2;R26Tom;Ptch1fl/wt, Lgr6creERT2;R26Tom;Ptch1fl/fl and Col1a2creER;R26Tom;Ptch1fl/fl stained AP-negative. (B) TD stroma of Gli1creERT2;R26Tom;Ptch1fl/fl skin was clearly positive for SDC1 staining, while the TD stroma of Gli1creERT2;R26Tom;Ptch1fl/wt skin showed very weak to negative SDC1 staining. In the TDs of Lgr6creERT2;R26Tom;Ptch1fl/fl and Col1a2creER;R26Tom;Ptch1fl/fl skin, SDC1 staining was absent. Gu: Guard hair. TAM: tamoxifen. BF: bright field. Asterisks: sebaceous glands stain positive for AP. Arrowheads: positive AP-staining in TD stroma. Dashed line: epithelial-stromal border. Scale bars: 50 μm (A–B).

Capturing very early epithelial bud formation in TDs using BrdU incorporation.
BrdU staining in TD areas. Mice were treated with TAM at 8 weeks of age and dorsal skin was collected 10 days after TAM treatment. Two hours prior to animal sacrifice, BrdU was given i.p. Clustered BrdU-positive cells were present in a potential developing TD-associated epithelial bud of Gli1creERT2;R26Tom;Ptch1fl/fl skin (arrowhead). Fewer and non-clustered BrdU-positive cells were detected in the TDs of the Gli1creERT2;R26Tom;Ptch1fl/wt control mice, Lgr6creERT2;R26Tom;Ptch1fl/fl, and Col1a2creER;R26Tom;Ptch1fl/fl mice. Note: HCl treatment for BrdU-staining results in more ‘blurry’ nuclei and loss of endogenous Tomato fluorescence. Tomato-tracing was visualized using an RFP antibody. n = 1 mouse per genotype, except n = 2 mice for Lgr6creERT2;R26Tom;Ptch1fl/fl. BrdU: Bromodeoxyuridine; labels proliferating cells. Scale bars: 50 μm.
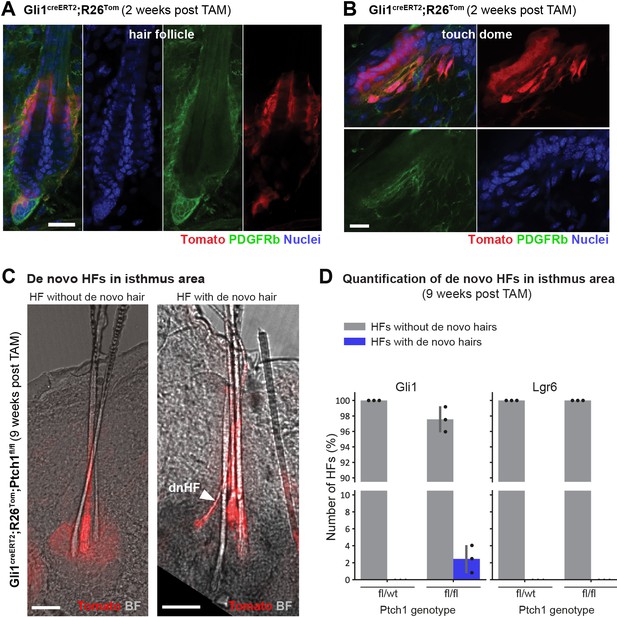
De novo hair follicle (HF) formation in isthmus area of pre-existing HFs.
(A–B) PDGFRb (CD140b antibody) staining of Gli1creERT2;R26Tom mice, treated with tamoxifen (TAM) at 8 weeks of age and traced for 2 weeks. Tomato-traced PDGFRb+ stromal cells are present in the HF isthmus area (A) and in the touch dome (TD) (B). (C–D) Mice were treated with TAM at 8 weeks of age and dorsal skin was analyzed 9 weeks post TAM. (C) Gli1creERT2;R26Tom;Ptch1fl/fl dorsal skin. Left panel: HFs without de novo HF contain three hair shafts (3 rounds of anagen). Right panel: HFs with de novo HF often contain four hair shafts including one thinner hair shaft with bent shape (arrowhead). (D) Quantification of de novo HFs in the isthmus area of Gli1creERT2;R26Tom;Ptch1fl/fl (n = 668 HFs from n = 3 mice), Lgr6creERT2;R26Tom;Ptch1fl/fl (n = 207 HFs from n = 3 mice), Gli1creERT2;R26Tom;Ptch1fl/wt (n = 335 HFs from n = 3 mice) and Lgr6creERT2;R26Tom;Ptch1fl/wt (n = 83 HFs from n = 3 mice) (Figure 8—source data 1). De novo HFs were only detected in isthmus areas of Gli1creERT2;R26Tom;Ptch1fl/fl mice. p-value=0.09 (t-test comparing Gli1creERT2;R26Tom;Ptch1fl/fl and Gli1creERT2;R26Tom;Ptch1fl/wt). n = 3 mice (A–C). dnHF: de novo hair follicle. TAM: tamoxifen. BF: bright field. Scale bars: 20 μm (A), 10 μm (B), 50 μm (C).
-
Figure 8—source data 1
Quantification of de novo hairs from isthmus.
Data are grouped according to mouse genotype.
- https://cdn.elifesciences.org/articles/46756/elife-46756-fig8-data1-v1.xlsx
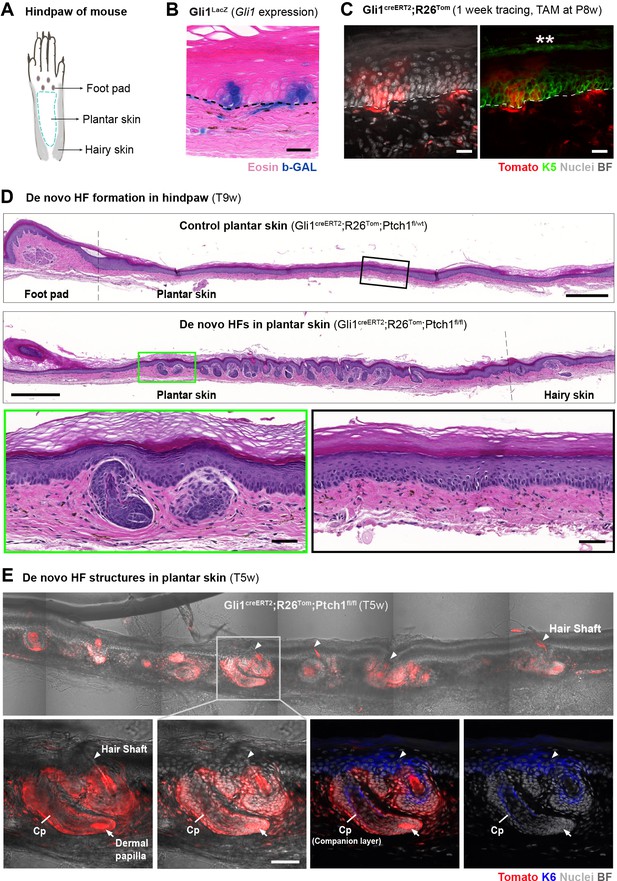
Formation of de novo hair follicles (HFs) in the plantar skin of Gli1creERT2;R26Tom;Ptch1fl/fl mice.
(A) Illustrative cartoon of a mouse hindpaw. (B) Gli1LacZ expression in the plantar skin (n = 3 mice). (C) Gli1creERT2;R26Tom mice were treated with tamoxifen (TAM) at 8 weeks of age. The hindpaws were collected 1 week post TAM and immuno-stained with K5 antibody (n = 3 mice). Asterisks mark autofluorescence on the outermost keratinized layer. (D–E) Gli1creERT2;R26Tom;Ptch1fl/fl and control Gli1creERT2;R26Tom;Ptch1fl/wt mice were treated with TAM at 8 weeks of age. Hindpaws were collected 5 or 9 weeks post TAM (n = 3 mice for each genotype; except n = 2 for Gli1creERT2;R26Tom;Ptch1fl/wt 5 weeks post TAM ). (D) Numerous de novo HFs formed in the plantar skin of Gli1creERT2;R26Tom;Ptch1fl/fl mice, while the same skin region in the control mice remained phenotypically normal. Green and black frames: zoom-in of plantar epidermis. Hematoxylin and eosin stained. (E) De novo HF structure in plantar skin of Gli1creERT2;R26Tom;Ptch1fl/fl mice are fully Tomato-traced and contain a K6+ companion layer as well as hair shafts (n = 2 mice). Arrowheads: de novo hair shafts. Arrows: dermal papilla. Cp: companion layer. Dashed line: epithelial-stromal border. TAM: tamoxifen. BF: bright field. Scale bars: 20 μm (B–C), 500 μm (D panaromas), 50 μm (D, E insets).

Physiological Hh/Gli signaling in the hindpaw plantar skin.
(A) Gli1LacZ expression in the plantar skin epidermis and dermis (n = 3 mice). Insets show Gli1-expressing cells at higher magnification. Dashed lines indicate the border between epidermis and dermis. (B) Gli1creERT2;R26Tom mice were treated with TAM at 8 weeks and traced for a week (n = 3 mice). Hindpaw skin was collected and immunostained with K5 antibody. Upper panel: maximum intensity projection of Tomato-tracing pattern in plantar skin. Lower panel: details of frame B’ (a single z-level is shown). Asterisks mark auto-fluorescence of the outermost keratinized layer. TAM: tamoxifen. BF: bright field. Scale bars: 200 μm (Panaroma in A and B), 20 μm (Insets A’,A’’,B’).
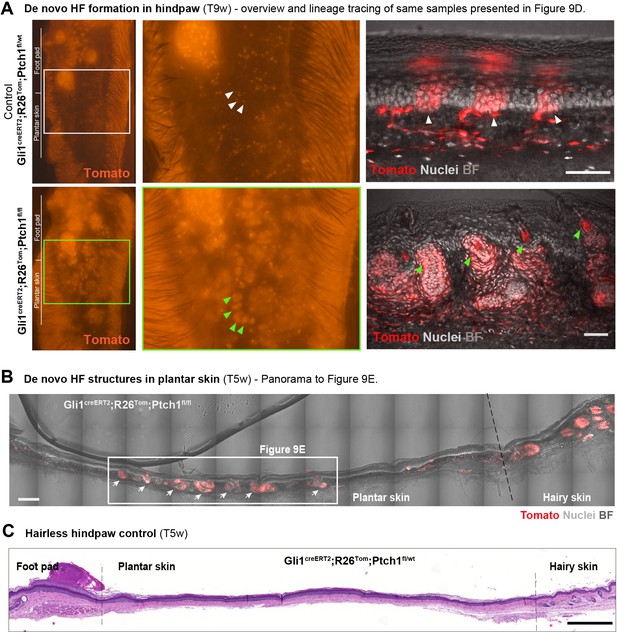
De novo HF formation in hindpaw of Gli1creERT2;R26Tom;Ptch1fl/fl mice.
(A–C) Gli1creERT2;R26Tom;Ptch1fl/fl and control Gli1creERT2;R26Tom;Ptch1fl/wt mice were treated with TAM at 8 weeks of age. The hindpaws were collected 5 or 9 weeks post TAM (n = 3 mice for each genotype). (A) Overview images of Tomato fluorescence in paws aquired with a stereo microscope (left panels) and lineage tracing pattern (right panels) of the same samples that were presented in Figure 9D. While in plantar skin of the control mice only Gli1-traced IFE cell clusters were present (white arrowheads), numerous de novo HFs with hair shafts formed in the plantar skin of Gli1creERT2;R26Tom;Ptch1fl/fl mice 9 weeks post TAM (green arrowheads). (B) Panorama to Figure 9E: de novo HF structures in plantar skin. Arrows: de novo HFs. (C) The control plantar skin shows no signs of de novo HF formation 5 weeks after TAM treatment. TAM: tamoxifen. BF: bright field. Scale bars: 50 μm (A), 200 μm (B), 500 μm (C).

Formation of HF-like structures in touch domes of the Lgr6/Col1a2 mouse model.
(A-D) Lgr6creERT2;Col1a2creER;Ptch1fl/fl mice were treated with Tamoxifen (TAM) at 8 weeks of age and dorsal skin was analyzed post TAM as indicated in the figure panels. Time points were chosen when de novo HF buds or HFs were clearly visible in the TDs of Gli1creERT2;R26Tom;Ptch1fl/fl mice. (A) Formation of epithelial buds outside of the touch dome areas at 17 days post TAM treatment, predominantly located in the upper part of the infundibulum connecting to the IFE (blue and green frame) and occasionally in the IFE (yellow frame) (n = 4 mice; biopsies). (B) Immunofluorescent co-staining of Keratin 5 (K5) and Syndecan-1 (SDC1) of 17 days post TAM dorsal skin, showing SDC1 positive dermal cells surrounding an epithelial bud from the infundibulum region (red arrowheads). (C) 36 days post TAM, a small hair shaft in the IFE/infundibulum area can be observed that was likely newly formed based on its morphology, size and thickness. However, it doesn’t seem properly anchored. Inset shows a zoom-in on the indicated area, with added K5 co-staining. (D) In 2 touch domes de novo hair follicles or hair shafts were observed based on morphology, size and thickness (white arrowheads) at 31 or 42 days post TAM treatment, respectively (n=3 for 31-35 days post TAM, n=2 for 42-49 days post TAM). BF: bright field. Scale bars: 200um (A), 20um (B, C inset), 100um (C panorama), 50um (D).

H&E stained dorsal skin of Lgr5creERT2/Ptch1fl/fl mice, treated with tamoxifen at P20.
Samples were taken at P16w, when HFs normally have produced 3 hair shafts (i.e. morphogenesis and 2 times anagen). Inset shows a magnified area of left panel with black arrows indicating HFs with 3 hair shafts.
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Genetic reagent (M. musculus) | Lgr6-EGFP-IRES-CreERT2 | Snippert et al., 2010 | Jackson Labs stock no: 016934 RRID:IMSR_JAX:016934 | Received from H Clevers |
| Genetic reagent (M. musculus) | Gli1-LacZ | Bai et al., 2002 | Jackson Labs stock no: 008211 RRID:IMSR_JAX:008211 | Received from F Aberger |
| Genetic reagent (M. musculus) | Gli1-CreERT2 | Ahn and Joyner, 2004 | Jackson Labs stock no: 007913 RRID:IMSR_JAX:007913 | Received from F Aberger |
| Genetic reagent (M. musculus) | Col1a2-CreER | Zheng et al., 2002 | Jackson Labs stock no: 029567 RRID:IMSR_JAX:029567 | Received from B Crombrugghe |
| Genetic reagent (M. musculus) | R26-tdTomato | Madisen et al., 2010 | Jackson Labs stock no: 007908 RRID:IMSR_JAX:007908 | Obtained from Jackson Laboratory |
| Genetic reagent (M. musculus) | Ptch1neo(fl)Ex2(fl) | Kasper et al., 2011 | Received from S. Teglund | |
| Genetic reagent (M. musculus) | C57BL/6J | Jackson Labs stock no: 000664 RRID:IMSR_JAX:000664 | Received from Preclinical Laboratory Karolinska Institutet | |
| Antibody | Rabbit polyclonal anti-K5 | BioLegend | Cat# PRB-160P RRID:AB_291581 | (1:1000) |
| Antibody | Guinea pig polyclonal anti-K5 | US Biological | Cat# C9097-37 RRID:AB_2134285 | (1:50) (1:200 for RNA-FISH) |
| Antibody | Rat monoclonal anti-SDC1 | BD Biosciences | Cat# 553712 RRID: AB_394998 | (1:500) |
| Antibody | Rat monoclonal anti-K8 | DSHB (Developmental Studies Hybridoma Bank) | Cat# TROMA-I RRID:AB_531826 | (1:1000) |
| Antibody | Rabbit polyclonal anti-K6 | BioLegend | Cat# PRB-169P RRID:AB_10063923 | (1:2000) |
| Antibody | Guinea pig polyclonal anti-K71 | Progen | Cat# GP-K6irs1 RRID:AB_2716781 | (1:100) |
| Antibody | Rabbit polyclonal anti-RFP | Rockland | Cat# 600-401-379 RRID:AB_2209751 | (1:100) |
| Antibody | Rat monoclonal anti-BrdU | Serotec | Cat# OBT0030G RRID: AB_609567 | (1:400) |
| Antibody | Rabbit polyclonal anti-GFP | Thermo Fisher Scientific | Cat# A-11122 RRID:AB_221569 | (1:500) |
| Antibody | Rabbit polyclonal anti-Ki67 | Abcam | Cat# ab15580 RRID:AB_443209 | (1:200) |
| Antibody | Rat monoclonal anti-CD140b | BioLegend | Cat# 136008 RRID:AB_2268091 | (1:100) |
| Chemical compound, drug | TO-PRO-3 | Invitrogen | Cat# T3605 | (1:1000) |
| Chemical compound, drug | Hoechst 33342 | Invitrogen | Cat# H3570 | (1 µg/mL) |
| Chemical compound, drug | DAPI | Invitrogen | Cat# D1306 | (1 µg/mL) |
| Chemical compound, drug | Tamoxifen | Sigma | Cat# T5648 | |
| Chemical compound, drug | 4-OH Tamoxifen | Sigma | Cat# H6278 | |
| Chemical compound, drug | NBT/BCIP stock solution | Roche | Cat# 11681451001 | |
| Chemical compound, drug | Vismodegib, Free Base | LC Laboratories | Cat# V-4050 | |
| Commercial assay or kit | RNAscope Multiplex Fluorescent Kit v2 | ACDBio/Bio-Techne | Cat# 323100 | |
| Commercial assay or kit | TSA Cy 3, Cy 5, TMR, Fluorescein Evaluation Kit | Perkin Elmer | Cat# NEL760001KT | |
| Sequence-based reagent | 3-plex Positive Control Probe | ACDBio/Bio-Techne | Cat# 320881 | RNA-FISH Probe |
| Sequence-based reagent | 3-plex Negative Control Probe | ACDBio/Bio-Techne | Cat# 320871 | RNA-FISH Probe |
| Sequence-based reagent | Mm-Gli1 | ACDBio/Bio-Techne | Cat# 311001 | RNA-FISH Probe |
Additional files
-
Supplementary file 1
List of mouse experiments.
Given are the following details per mouse: mouse genotype, treatment, analysis time point, tissue sampling (biopsy or final material), comments/observations, mouse ID.
- https://cdn.elifesciences.org/articles/46756/elife-46756-supp1-v1.xlsx
-
Transparent reporting form
- https://cdn.elifesciences.org/articles/46756/elife-46756-transrepform-v1.docx





