Cell-type specific innervation of cortical pyramidal cells at their apical dendrites
Figures
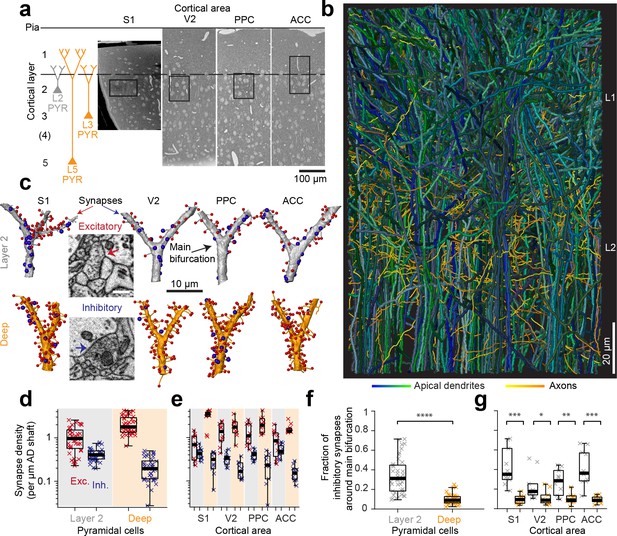
Complete synaptic input mapping of pyramidal cell apical dendrites (ADs) around their main bifurcation.
(a) Overview EM images indicating the location of 3D EM datasets in primary somatosensory (S1), secondary visual (V2), posterior parietal (PPC) and anterior cingulate (ACC) cortices relative to pia surface (solid line) and layer 1 / 2 border (dashed line). Schematic location of layer 2 (gray) and deep layer (orange) pyramidal neurons. (b) Reconstruction of all ADs contained in the ACC dataset (blue-green, n = 61 layer 2 and n = 152 deep layer ADs, respectively) and a subset of axons innervating them (n = 62, yellow-orange). Note that 80–90% of all pyramidal cells in cortex extend their apical dendrites into the L1/2 border region, allowing for massive synaptic convergence (https://wklink.org/8300). (c) Complete synaptic input maps of apical dendrite main bifurcations for deep layer (orange, S1: https://wklink.org/5413, V2: https://wklink.org/6059, PPC: https://wklink.org/9261, ACC: https://wklink.org/8408) and layer 2 (gray, S1: https://wklink.org/4049, V2: https://wklink.org/8965, PPC: https://wklink.org/8307, ACC: https://wklink.org/8695) pyramidal cells (example excitatory (spine, red spheres, https://wklink.org/9524) and putative inhibitory (shaft, blue spheres, https://wklink.org/2603) synapses, inset). (d) Box plot of putative inhibitory (blue crosses) and excitatory (red crosses) synapse densities per µm of AD shaft path length for L2 (n = 41, left) and DL ADs (n = 41, right) in S1, V2, PPC and ACC datasets. Wilcoxon rank-sum test, p < 10−6, 10−7 for excitatory and putative inhibitory densities, respectively. (e) Same as (d) reported separately per cortical region (n = 20 for S1, V2 and PPC, n = 22 for ACC). (f) Box plot of fraction of putative inhibitory synapses at the main bifurcation of deep (orange) and layer 2 (gray) ADs; individual ADs shown (crosses). (g) As (f) reported separately per cortical region. Asterisks indicate significance level of the Wilcoxon rank-sum test (*p < 0.05, **p < 0.01, ***p < 10−3, ****p < 10−4). Scale bars: 0.5 µm (inset in c).
-
Figure 1—source data 1
The density of excitatory and putative inhibitory synapses (Figure 1d, e).
- https://cdn.elifesciences.org/articles/46876/elife-46876-fig1-data1-v2.xlsx
-
Figure 1—source data 2
The fraction of excitatory and putative inhibitory synapses (Figure 1f, g).
- https://cdn.elifesciences.org/articles/46876/elife-46876-fig1-data2-v2.xlsx
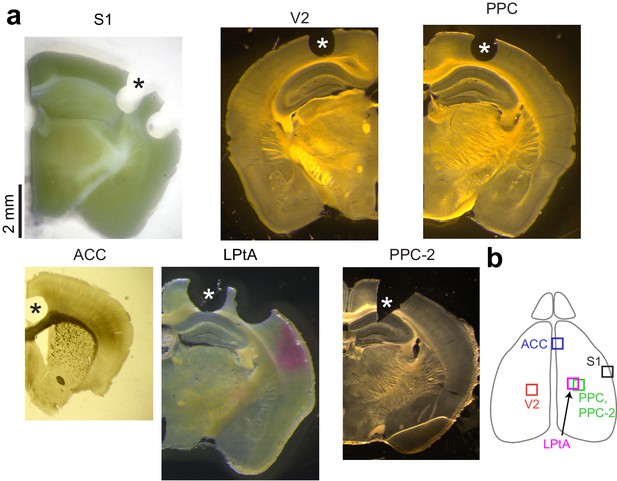
Location of cortical tissue used for EM volume imaging.
(a) Coronal sections (thickness: 100–1000 µm) demonstrating the cortical regions (asterisks) used for 3D electron microscopy. (b) Schematic of mouse brain illustrating approximate location of all six cortical regions used for 3D-EM.
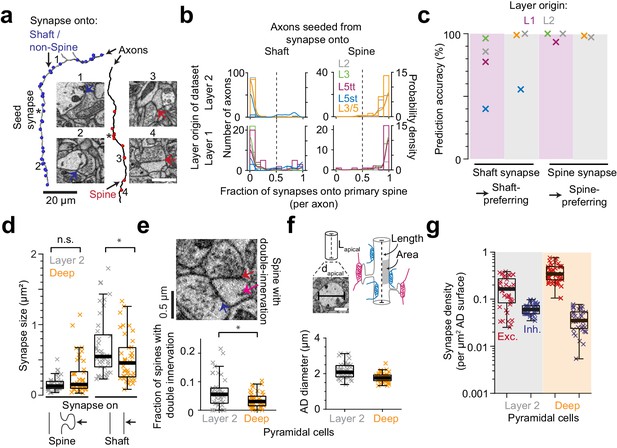
Effect of synapse identity, size, dendrite diameter and double-innervated spines on synaptic composition of apical dendrites.
(a) Reconstruction of two axons seeded from synapses onto ADs (asterisks) and all the output synapses within the volume (red: spine, blue: shaft/non-spine). Note the stark difference in their innervation preference based on their seed location. Example spine (red arrows, 3: https://wklink.org/5224, 4: https://wklink.org/9411) and shaft (blue arrows, 1: https://wklink.org/8807, 2: https://wklink.org/3270) synapse EM micrographs (insets). (b) Histogram for the fraction of synapses made onto single-innervated spines. Axons seeded from synapses on deep (orange), L2 (gray), L3 (green), L5tt (magenta) and L5st (blue) ADs. Annotations are from layer 1 (n = 142, LPtA dataset) and layer 2 (n = 288, S1, V2, PPC and ACC datasets); probability density estimations and synapse identity threshold as solid and dashed lines, respectively. Note highly bimodal distribution allowing the clear distinction of axons into spine-preferring (excitatory) and shaft-preferring (putative inhibitory), with exception of axons innervating shafts of L5st ADs (these were corrected in the following, see c). (c) Scatter plot for accuracy in predicting presynaptic axon type (shaft- vs. spine-prefering) based on the location of synapse on AD (shaft vs. spine). (d) Size of synapses onto deep (n = 41, orange crosses) and layer 2 (n = 41, gray crosses) ADs. (e) Fraction of double-innervated spines in the main bifurcation annotations of deep (orange, n = 41) and layer 2 (gray, n = 41) neurons. Spine with putative inhibitory (blue arrow) and excitatory (red arrow) inputs (inset). (f) Sketches illustrating the synapse density normalized to surface area and length of AD and their measurement (upper panel). Box plot of the average diameter of layer 2 (gray, n = 41) and deep (orange, n = 41) AD around the main bifurcation in S1, V2, PPC and ACC datasets (lower panel, p < 10−4, Wilcoxon rank-sum test). (g) Box plot of putative inhibitory (blue crosses) and excitatory (red crosses) synapse densities per µm2 of AD surface area for L2 (n = 41, left) and DL ADs (n = 41, right). Wilcoxon rank-sum test, p < 10−6 for both densities. Asterisks indicate significance level of the Wilcoxon rank-sum test (not significant (n.s.): p > 0.05,*p < 0.05). Scale bars: 1 µm (inset in a,f).
-
Figure 2—source data 1
Dendritic spine vs. shaft innervation preference for AD-targeting axons (Figure 2b).
- https://cdn.elifesciences.org/articles/46876/elife-46876-fig2-data1-v2.xlsx
-
Figure 2—source data 2
Prediction accuracy for dendritic shaft vs. spine innervation preference (Figure 2c).
- https://cdn.elifesciences.org/articles/46876/elife-46876-fig2-data2-v2.xlsx
-
Figure 2—source data 3
Size of synapses onto ADs (Figure 2d).
- https://cdn.elifesciences.org/articles/46876/elife-46876-fig2-data3-v2.xlsx
-
Figure 2—source data 4
Fraction of dendritic spines with double-innervation (Figure 2e).
- https://cdn.elifesciences.org/articles/46876/elife-46876-fig2-data4-v2.xlsx
-
Figure 2—source data 5
Apical dendrite diameter and the synapse density normalized to surface area of ADs (Figure 2f, g).
- https://cdn.elifesciences.org/articles/46876/elife-46876-fig2-data5-v2.xlsx
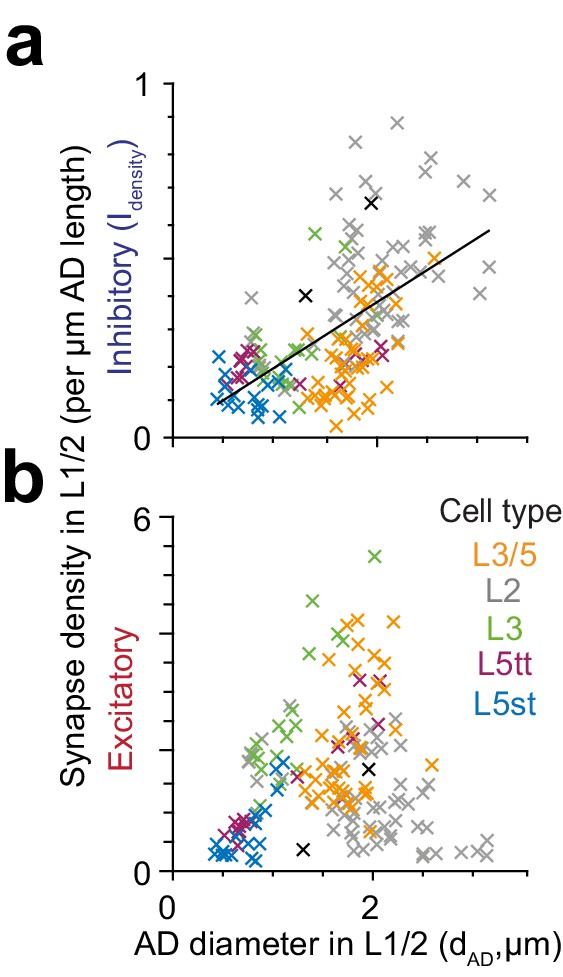
Relationship of synapse density to apical dendrite diameter in layers 1 and 2.
(a–b) Scatter plot of the relationship between putative inhibitory (a) and excitatory (b) synapse density (normalized to AD shaft path length) and the diameter of apical dendrite in layers 1 and 2 (n = 153). Black line indicates linear fit to putative inhibitory density (Idensity = 0.18 x dAD + 0.01, R2 = 0.37, p < 10−15). The linear relationship between excitatory synapse density and AD diameter was not significant (p = 0.096).
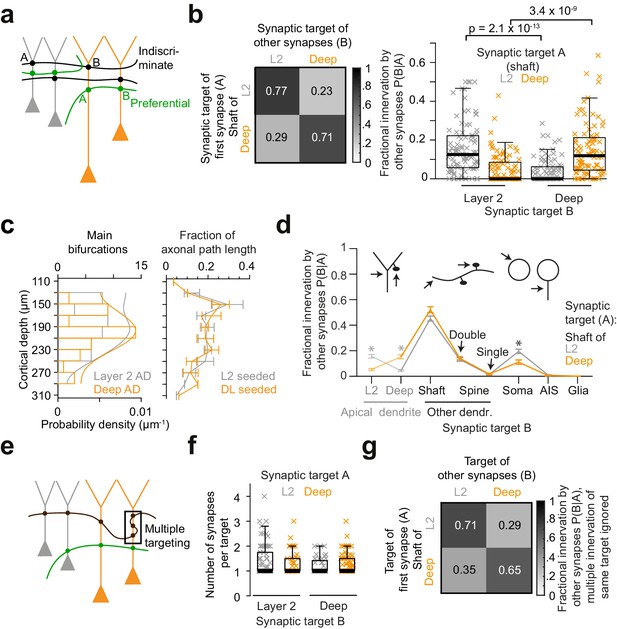
Preference of inhibitory axons for the type of postsynaptic pyramidal cell.
(a) Sketch illustrating two extreme innervation models for AD-targeting inhibitory axons: innervation could be selective for the type of AD (L2 vs L3/5 pyramidal cells, green), or indiscriminate for the type of AD (black). In the latter case, conditional dependence of targeting p(B|A) would be expected to be indistinguishable from overall probability of innervation p(B). (b) Conditional dependence of synaptic innervation p(B|A) shown as the mean probability of deep and layer 2 AD targeting (target ‘B’) given the target of the first synapse of an axon (target ‘A’, left panel). Probabilities reported as the average fraction of AD synapses onto each AD type. Box plot of AD fractional innervation for axons seeded from layer 2 (n = 92, gray crosses) and deep (n = 91, orange crosses) ADs (right panel), corresponding to the entries in the innervation matrix (left panel). P-values: Wilcoxon rank-sum test. Note that indiscriminate innervation can be revoked. (c) Distribution of main bifurcations along cortical depth (n = 41 per AD type; probability density estimates, lines), and distribution of axonal path length along cortical depth (n = 183 axons, of these n = 92 seeded at L2 (gray) and n = 91 seeded at DL ADs (orange)). Note that neither the pre- nor the postsynaptic targets are sorted along the cortical axis, excluding simple layering effects for the conditional innervation (b,c). Error bars indicate mean ± SEM over cortical region (S1, V2, PPC and ACC). (d) Mapping of axonal output onto subcellular targets. Error bars indicate mean ± SEM; asterisks: significance of bootstrapping test at p = 0.05 with Bonferroni correction. (e) Sketch illustrating innervation of an AD with multiple synapses (box). (f) Box plot for comparison of the average number of synapses per individual AD target for axons seeded from L2 (gray crosses) and deep (orange crosses) ADs. Wilcoxon rank-sum test, p = 0.49, 0.23 for L2 and deep ADs, respectively. (g) Conditional dependence of AD innervation p(B|A) where multiple innervation of same AD target were ignored (Compare to b). Probabilities indicate fraction of individual ADs targeted by axons seeded from L2 (first row) and deep (second row) ADs.
-
Figure 3—source data 1
Conditional innervation probability of sub-cellular structures by AD-targeting putative inhibitory axons (Figure 3b, d and Figure 4b, c).
- https://cdn.elifesciences.org/articles/46876/elife-46876-fig3-data1-v2.xlsx
-
Figure 3—source data 2
The coordinate of analyzed AD main bifurcations (y-axis corresponds to the depth relative to pia, Figure 3c).
- https://cdn.elifesciences.org/articles/46876/elife-46876-fig3-data2-v2.xlsx
-
Figure 3—source data 3
Distribution of AD-targeting axonal path length along the cortical depth in S1, V2, PPC and ACC datasets (Figure 3c).
- https://cdn.elifesciences.org/articles/46876/elife-46876-fig3-data3-v2.xlsx
-
Figure 3—source data 4
Average number of synapses on individual AD targets by putative inhibitory axons (Figure 3f).
- https://cdn.elifesciences.org/articles/46876/elife-46876-fig3-data4-v2.xlsx
-
Figure 3—source data 5
The distribution for the number of synapses on individual AD targets by putative inhibitory axons (Figure 3f, g).
- https://cdn.elifesciences.org/articles/46876/elife-46876-fig3-data5-v2.xlsx
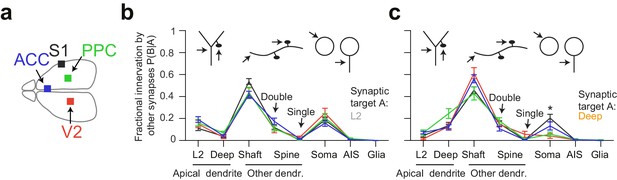
Quantitative consistency in the innervation profile of inhibitory axons across four cortical regions.
(a) Sketch of the horizontal view of the mouse brain with location of cortical datasets used for comparison. See also Figure 1a. (b,c) Comparative analysis across cortical regions. Postsynaptic target specificity for axons seeded from (b) layer 2 ADs (n = 21, 20, 21, 30 for S1, V2, PPC and ACC, respectively) and (c) for axons seeded from deep layer ADs (n = 19, 20, 20, 32 for S1, V2, PPC and ACC, respectively). Note the high level of quantitative consistency of synaptic target fractions across cortices with the exception of somatic innervation in axons seeded from DL ADs. Error bars indicate mean ± SEM. *p < 0.05 MANOVA test followed by multiple One-way ANOVA with Bonferroni correction.
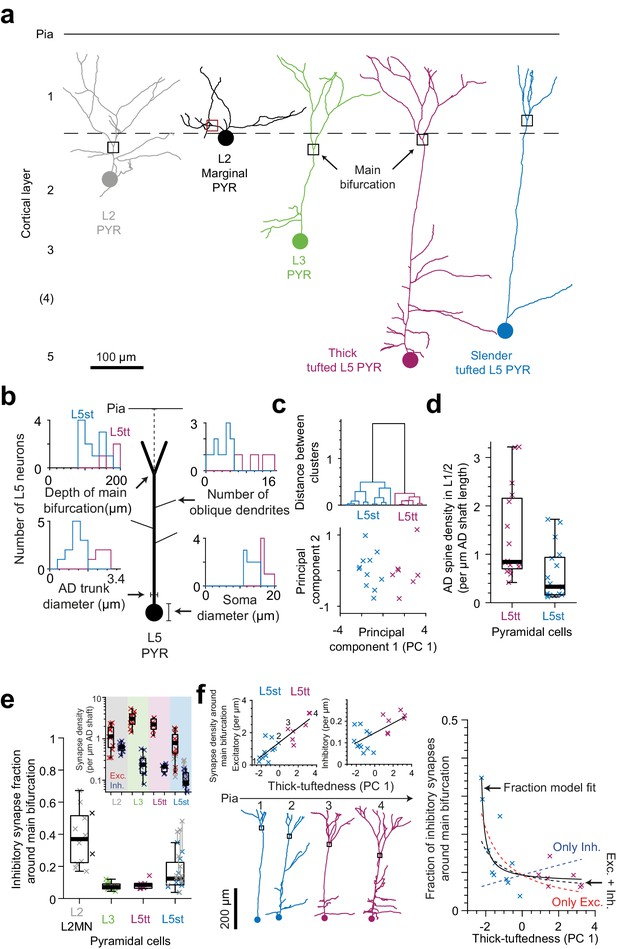
Synaptic input composition for L2, L3, and subtypes of L5 pyramidal cells at their main bifurcations.
(a) Complete skeleton reconstruction of apical dendrites contained within the high- and low-resolution volume of the PPC cortex dataset (PPC-2). Main bifurcation (black/red rectangles) and soma of origin (circles) is marked for L2 (gray, https://wklink.org/7762), L2MN (black, https://wklink.org/5352), L3 (green, https://wklink.org/7652), L5tt (magenta, https://wklink.org/3415) and L5st (blue, https://wklink.org/1435) neurons. (b) Sketch illustrating four morphological features of L5 neurons used for subtype classification. Insets show histograms of each feature for the L5tt (magenta, n = 7) and L5st (blue, n = 11) neurons. Note the differences between the two cell types. (c) Agglomerative hierarchical linkage tree (upper panel) and scatter plot of the first two principal components (lower panel) for (b). (d) Box plot for comparison of spine density between L5tt (magenta, n = 16) and L5st (blue, n = 17) neurons at their main bifurcation and distal apical tuft. (e) Fraction of putative inhibitory input synapses around the main bifurcation of apical dendrites from layer 2 (gray, n = 10), 2MN (black, n = 2, box plot shared with L2 group), 3 (green, n = 10), 5tt (magenta, n = 7), 5st (blue, n = 11) pyramidal cells (box plots) and density of excitatory (red crosses) and putative inhibitory (blue crosses) synapses at the main bifurcation (inset). Note clear distinction of synaptic input composition for L2 vs L3 and L5tt pyramidal cells at their main bifurcation. Kruskal-Wallis test, p < 10−4. (f) Scatter plot of synapse densities (left panels) and putative inhibitory fraction (right panel) of L5 neurons in relation to their thick-tuftedness (PC 1). Skeleton reconstruction of neurons indicated by numbers in the left panel demonstrate the relationship of PC 1 to AD tuft size (https://wklink.org/1293). Excitatory synapse density and putative inhibitory fraction have linear (, R2 = 0.74, p < 10−5) and non-linear relationships (, R2 = 0.70, p < 10−7) to thick-tuftedness, respectively. Dashed lines in right panel indicate models using only linear excitatory (red), linear putative inhibitory (blue) or combined excitatory and putative inhibitory linear relationships (black).
-
Figure 5—source data 1
Morphological properties of L5 pyramidal neurons (Figure 5b, c, f).
- https://cdn.elifesciences.org/articles/46876/elife-46876-fig5-data1-v2.xlsx
-
Figure 5—source data 2
Dendritic spine density for subtypes of L5 pyramidal neurons (Figure 5d).
- https://cdn.elifesciences.org/articles/46876/elife-46876-fig5-data2-v2.xlsx
-
Figure 5—source data 3
More detailed cell-type comparison of the synapse density and fraction at the main bifurcation of ADs (Figure 5e).
- https://cdn.elifesciences.org/articles/46876/elife-46876-fig5-data3-v2.xlsx
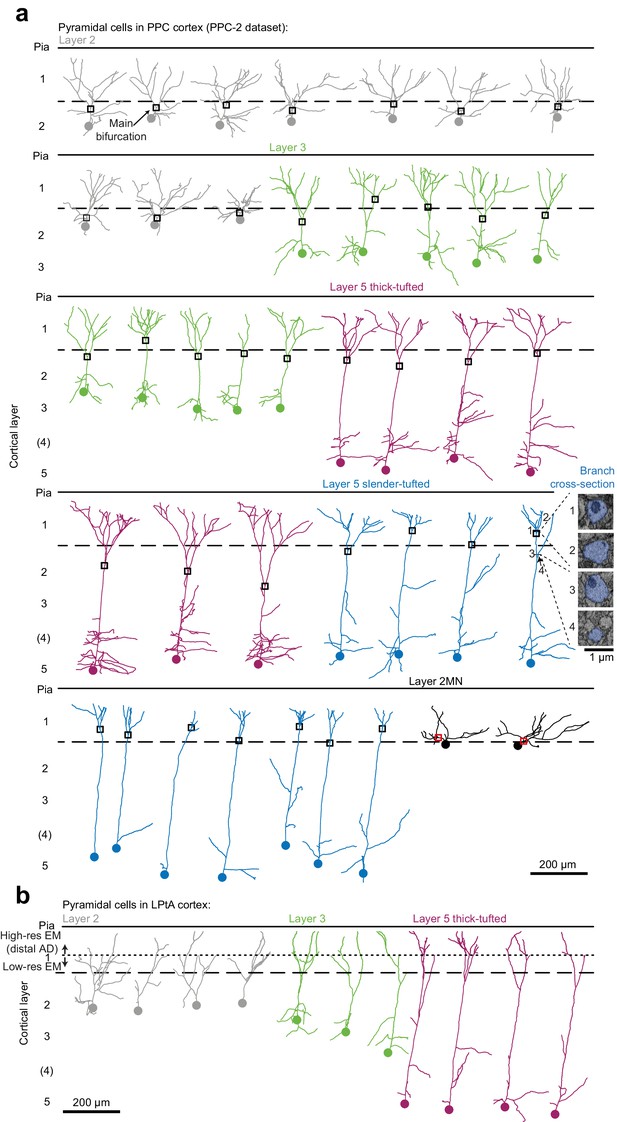
Skeleton reconstruction of apical dendrites of pyramidal neurons in PPC and LPtA cortical regions.
(a) Skeleton reconstruction of apical dendrites of layer 2 (gray traces, n = 10), 2MN (black, n = 2), 3 (green, n = 10), 5tt (magenta, n = 7), 5st (blue, n = 11) pyramidal cells in PPC-2 dataset (PPC cortex). Black/red squares indicate the main bifurcation area used for synaptic input mapping in each annotation. Branch diameter was used to help determine the main bifurcation (EM micrographs, inset). (b) Same as in (a) for neurons with dense synaptic input mapping of their distal apical dendrite tuft in LPtA cortex. The border between the high- (used for dense synaptic input mapping) and low-resolution EM datasets (used for AD trunk annotation towards cell body of origin) is indicated by a dotted line. Also see Figure 7. The dashed and solid lines indicate pia and layer 1 / 2 border, respectively. The cell body location of each neuron is indicated by a circle.
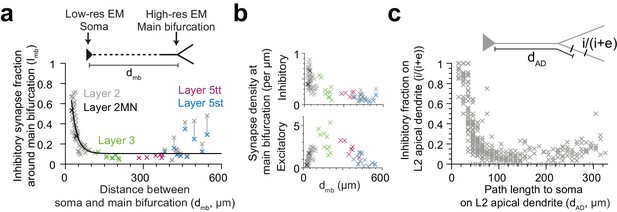
Dependence of inhibitory input fraction at the main bifurcation of apical dendrites to the distance to soma.
(a) Relationship between distance of main bifurcation to soma and putative inhibitory fraction at the main bifurcation for L2 (n = 51, gray), L2MN (n = 2, black), L3 (n = 10, green), L5tt (n = 7, magenta) and L5st (n = 11, blue) ADs. Black line indicates single exponential regression (, R2 = 0.73, p < 10−22). (b) Same as in (a) for putative inhibitory and excitatory synapse densities, respectively. Note tight distance dependence for the putative inhibitory input fraction (a), but not the absolute synapse densities (b). (c) Relationship between path distance to soma on the apical dendrite and the fraction of putative inhibitory synapses on L2 apical dendrites (n = 66, S1, V2, PPC, ACC, PPC-2 and LPtA datasets, n = 12,532 synapses). Each cross represents the putative inhibitory fraction (i/(i+e)) for a single apical dendrite within a 10 µm path length range (dAD) to cell body of origin (inset).
-
Figure 6—source data 1
Path distance between soma and main bifurcation (MB) with the corresponding synapse densities and fractions at MB (Figure 6a, b).
- https://cdn.elifesciences.org/articles/46876/elife-46876-fig6-data1-v2.xlsx
-
Figure 6—source data 2
The fraction of putative inhibitory synapses along the apical dendrite of L2 neurons (Figure 6c).
- https://cdn.elifesciences.org/articles/46876/elife-46876-fig6-data2-v2.xlsx
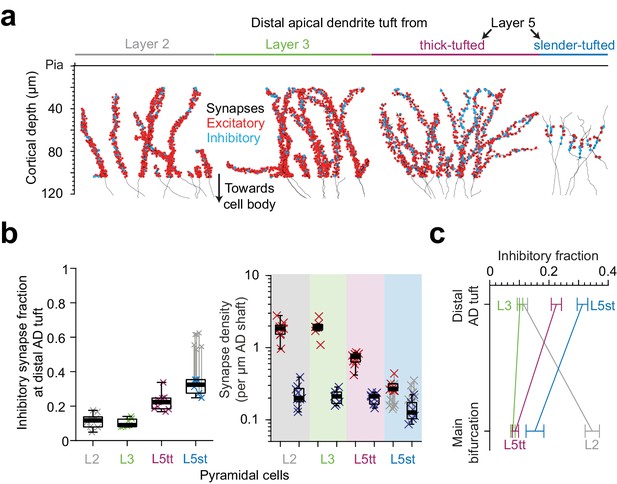
Inhibitory input fraction at distal apical tuft dendrites.
(a) Skeleton reconstruction of 14 L2, L3, L5tt and L5st ADs with all synapses mapped within the high-resolution EM image volume in layer 1 of LPtA (L2, L3, L5tt, https://wklink.org/9047) and PPC (L5st, https://wklink.org/8828) cortex (red: excitatory synapses, n = 3,812, cyan: putative inhibitory synapses n = 694). Note the difference in synapse densities (red/cyan sphere density) for layer 2, 3, 5tt, 5st ADs. (b) Box plot for fraction of putative inhibitory synapses at the distal apical dendritic site from L2 (n = 9 individual branches, gray crosses), L3 (n = 7, green crosses), L5tt (n = 9, magenta crosses) and L5st (n = 6, blue crosses) pyramidal cells and box plot for density of excitatory (red crosses) and putative inhibitory (blue crosses) synapses at the distal tuft (right panel). Note the clear distinction of synaptic input composition in the distal tuft for L2 vs. L3 vs. L5tt vs. L5st pyramidal cells (Kruskal-Wallis test, p < 10−4) mainly due to differences in excitatory synapse density. (c) Summary of distinct putative inhibitory input fraction at the main bifurcation and distal tufts of apical dendrites for four main classes of pyramidal cells in the cerebral cortex. Note that only L3 pyramidal cells show approximately homogeneous ratio of putative inhibitory and excitatory synaptic inputs along their apical dendrites. Error bars indicate mean ± SEM.
-
Figure 7—source data 1
The synapse density and fraction in the distal part of apical dendrites (Figure 7b).
- https://cdn.elifesciences.org/articles/46876/elife-46876-fig7-data1-v2.xlsx
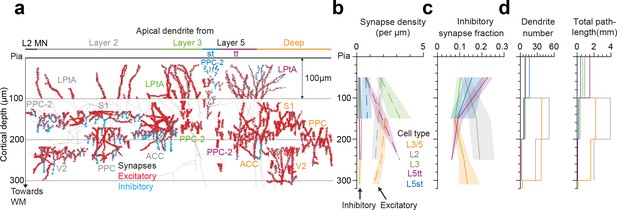
Synaptic composition for layer 2, 3, 5tt and 5st apical dendrites (ADs) along the upper cortex.
(a) Partial or complete skeleton reconstruction of 141 layer 2, 3, 5tt and 5st ADs with all synapses mapped (red: excitatory, n = 22,825, cyan: putative inhibitory n = 3,723, total AD shaft path length = 14.1 mm). Four datasets containing layer 2 (n = 20, 20, 22, 28 for S1, V2, PPC, ACC, respectively), one dataset from layer 1 (n = 11, LPtA) and one containing both layers (n = 40, PPC-2) were used. Note the difference in synapse densities (red/cyan sphere density) for layer 2, 3, 5tt, 5st and deep (L3/5) ADs at different cortical depths relative to pia. Horizontal location of the skeleton tracings is adjusted for illustration. (b) Spatial distribution of excitatory (dashed line) and putative inhibitory synapse (solid line) densities of L2 (gray), 3 (green), 5tt (magenta), 5st (blue) and 3/5 (deep, orange) ADs in upper cortex (bin size = 100 µm). Lines and shades indicate the pooled average and 95% bootstrap confidence interval (n = 10,000 resamples) for data combined across all datasets, respectively. (c) Same as in (b) for fraction of putative inhibitory synapses. (d) Histogram of the number of dendrites and total AD shaft path length within each cortical depth bin.
Additional files
-
Supplementary file 1
Supplementary tables 1-5 reporting parameters for experiments and analysis, axonal preference for shaft and spine innervation, and location of pyramidal cell somata.
- https://cdn.elifesciences.org/articles/46876/elife-46876-supp1-v2.docx
-
Transparent reporting form
- https://cdn.elifesciences.org/articles/46876/elife-46876-transrepform-v2.docx





