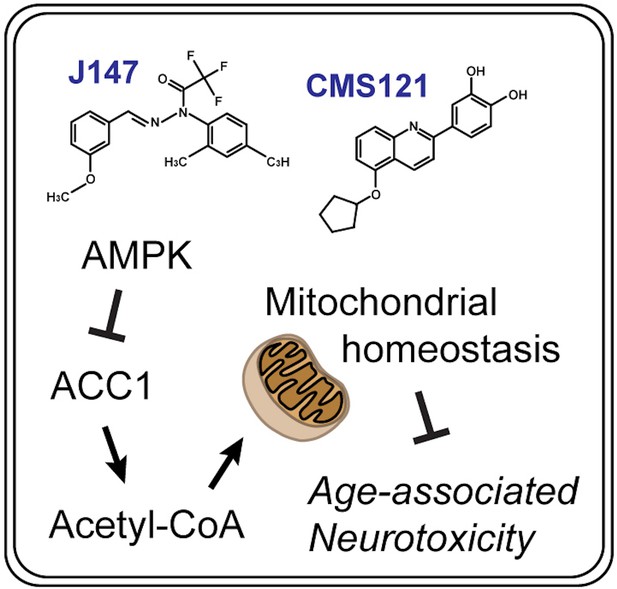
Elevating acetyl-CoA levels reduces aspects of brain aging
Figures
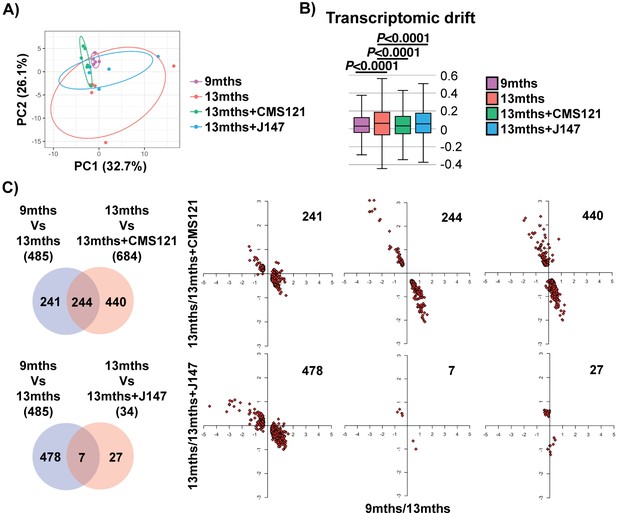
Aging of the hippocampal transcriptome in SAMP8 mice is significantly prevented by CMS121 and J147.
(A) PCA of the top 10% most highly expressed genes in the hippocampus of 9 months, 13 months, 13 months+CMS121 and 13 months+J147 SAMP8 mice. Cutoff was sum(log2(FPKM+5))>=110.41 (2458 genes selected; n = 5–6/group). Ellipses show the 70% confidence interval. (B) Drift analysis of the whole transcriptome. Values are expressed as box-and-whisker plots. Brown-Forsythe test. n = 5–6/group. (C) Venn diagrams illustrating shared and uniquely affected genes between the 9 months vs 13 months old mice and 13 months vs each of the 13 months+compounds. n = 5–6/group. Please see Supplemental Materials and methods for detailed statistical analysis. Correlation of gene expression for each group of genes is represented on the right side of the panel (units are -log(fold change)).
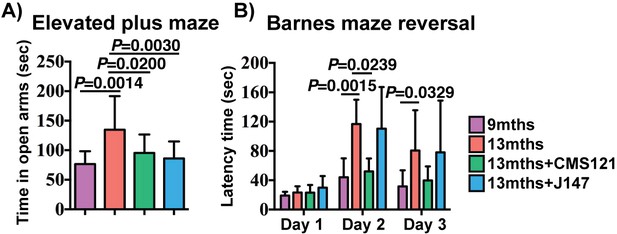
CMS121 and J147 preserve cognitive parameters in SAMP8 mice when administered at advanced stages of the phenotype.
(A) The elevated plus maze was used to measure disinhibition behavior in 9 months, 13 months, 13 months+CMS121 and 13 months+J147 SAMP8 mice. One-way ANOVA followed by Tukey-Kramer post-hoc test (n = 11–18/group). (B) Spatial learning/memory was evaluated in the same mice by the Barnes maze assay. All data are mean ± SD. Two-way repeated measures ANOVA and post hoc Bonferroni corrected t-test (n = 5–9/group).
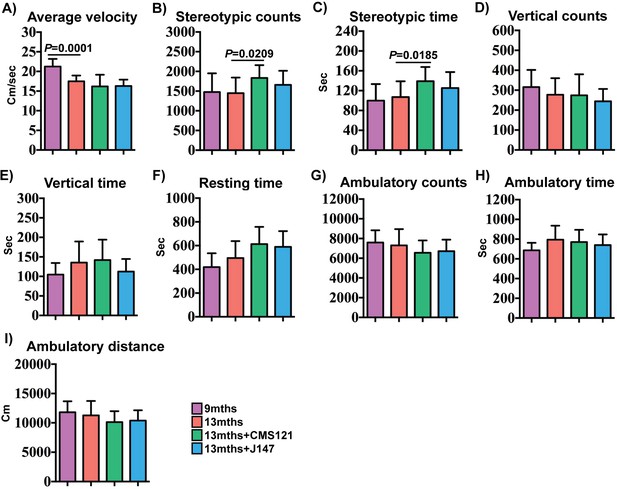
Effect of CMS121 and J147 on activity parameters.
Average velocity (A), stereotypic counts (B), stereotypic time (C), vertical counts (D), vertical time (E), resting time (F), ambulatory counts (G), ambulatory time (H), and ambulatory distance (I) were assessed in 9 months, 13 months, 13 months+CMS121 and 13 months+J147 SAMP8 mice with the open field test. All data are mean ± SD. One-way ANOVA followed by Tukey-Kramer post-hoc test (n = 11–18/group).
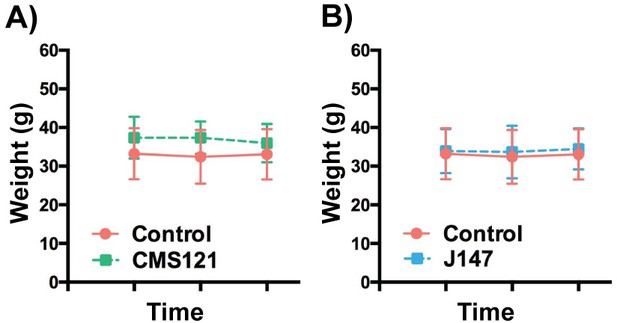
Body weights of SAMP8 mice fed with vehicle, CMS121 and J147 diets.
9 months old female SAMP8 mice were fed with vehicle (n = 23), CMS121 (n = 22) or J147 (n = 22) diets until 13 months. Six SAMP8 mice fed with control diet, four SAMP8 mice fed with CMS121 diet and four SAMP8 mice fed with J147 diet died throughout the course of this study. No significant differences were found between the body weights of the two groups at three different time points (9 months, 11 months and 13 months). Two-way repeated measures ANOVA and post hoc Bonferroni corrected t-test. All data are mean ± SD.
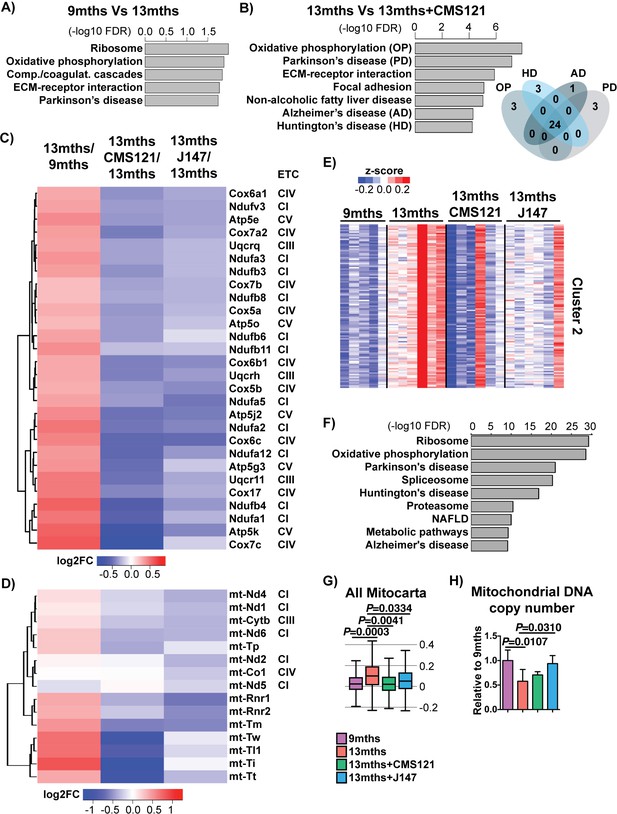
CMS121 and J147 specifically maintain the expression of genes associated with the mitochondria that are altered with aging in SAMP8 mice.
Top KEGG pathways and respective enrichment scores associated with the 485 genes with altered expression in 9 months vs 13 months SAMP8 mice (A) and with the 684 genes with altered expression in 13 months vs 13 months+CAD121 (B) SAMP8 mice. Gene overlap for the KEGG pathways oxidative phosphorylation (OP), Parkinson's disease (PD), Huntington's disease (HD) and Alzheimer's disease (AD) in 13 months vs 13 months+CMS121 SAMP8 mice is shown. n = 5–6/group. (C) Heatmap of the fold gene expression for 9 months/13 months, 13 months/13 months+CMS121 and 13 months/13 months+J147, regarding the mitochondrial genes present in the pathways OP, PD, HD and AD. The log2 fold-change (FC) is plotted in red–blue color scale with red indicating up-regulation and blue indicating down-regulation. ETC: Electron transport chain complex; CI = Complex I; CIII = Complex III; CIV = Complex IV; CV = Complex V. (D) Heatmap of the fold gene expression for 9 months/13 months, 13 months/13 months+CMS121 and 13 months/13 months+J147, regarding all genes from the mitochondrial DNA with detectable expression levels. Color key = log2 FC. (E) Cluster two from the clustering heatmap (K-means, Euclidean distance on normalized log2(FPKM+5) for 20077 genes) of the whole hippocampal transcriptome of 9 months, 13 months, 13 months+CMS121 and 13 months+J147 SAMP8 mice (For full heatmap see Figure 3—figure supplement 1). (F) Top KEGG pathways associated with cluster 2. (G) Transcriptomic drift of genes that encode for all proteins known to be associated with the mitochondria (Mitocarta). Values are expressed as box-and-whisker plots. Brown-Forsythe test. n = 5–6/group. (H) Mitochondrial DNA copy number in the hippocampus (normalized to 9 months). Data are mean ± SD. One-way ANOVA followed by Tukey-Kramer post-hoc test (n = 5/group).
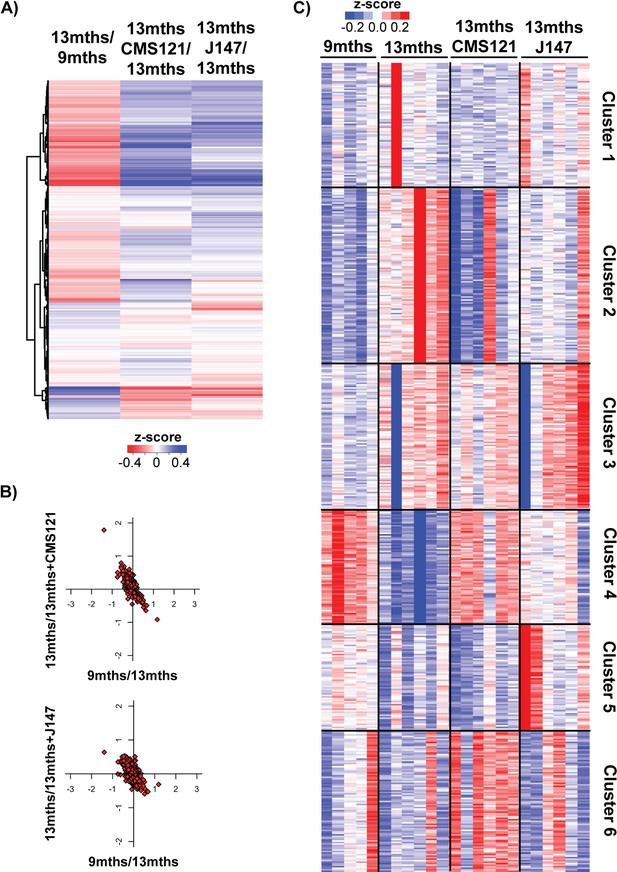
Additional analysis of transcriptomic data.
(A) Heatmap of the fold gene expression for 9 months/13 months, 13 months/13 months+CMS121 and 13 months/13 months+J147, regarding all genes that encode for proteins known to be associated with the mitochondria (Mitocarta). The log2 fold-change is plotted in red–blue color scale with red indicating up-regulation and blue indicating down-regulation. (B) Correlation of expression of all Mitocarta genes for each comparison (units are -log(fold change)). (C) Full clustering heatmap (K-means, Euclidean distance on z-scaled log2(FPKM+5) of expressed 20077 genes) of the whole hippocampal transcriptome of 9 months, 13 months, 13 months+CMS121 and 13 months+J147 SAMP8 mice.
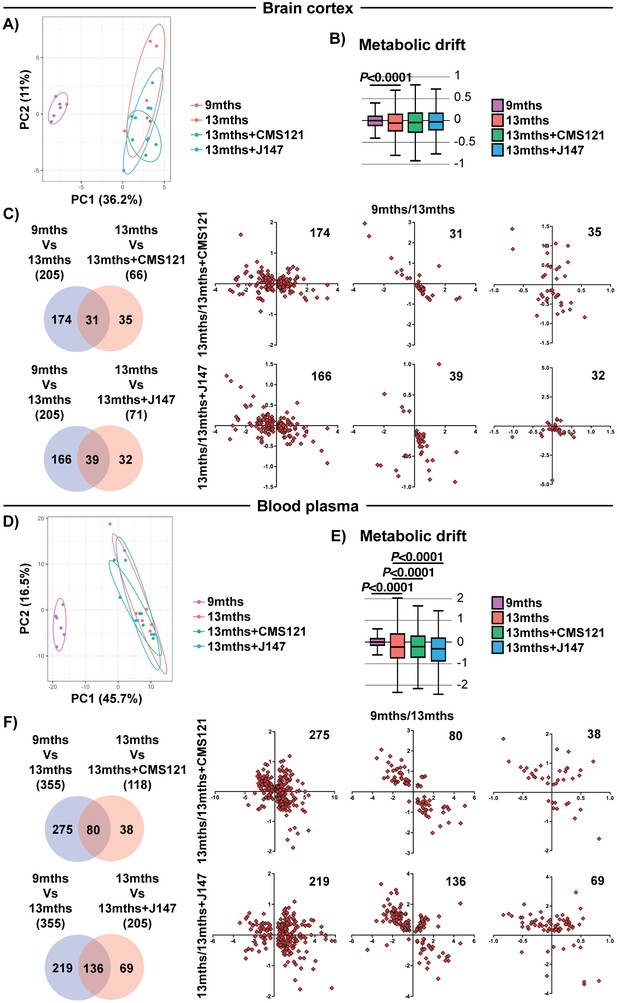
Global metabolic analysis of brain cortex and blood plasma.
PCA of the 496 and 604 metabolites measured in the brain cortex (A) and blood plasma (D), respectively, of 9 months, 13 months, 13 months+CMS121 and 13 months+J147 SAMP8 mice. (n = 6/group). Drift analysis of cortex (B) and plasma (E) metabolites. Values are expressed as box-and-whisker plots. Brown-Forsythe test. n = 6/group. Venn diagrams illustrating shared and uniquely affected cortex (C) and plasma (F) metabolites between the 9 months vs 13 months old mice and 13 months vs each of the 13 months+compounds. n = 6/group. Correlation of metabolite levels for each group of genes is represented on the right side of the panel (units are -log(fold change)).
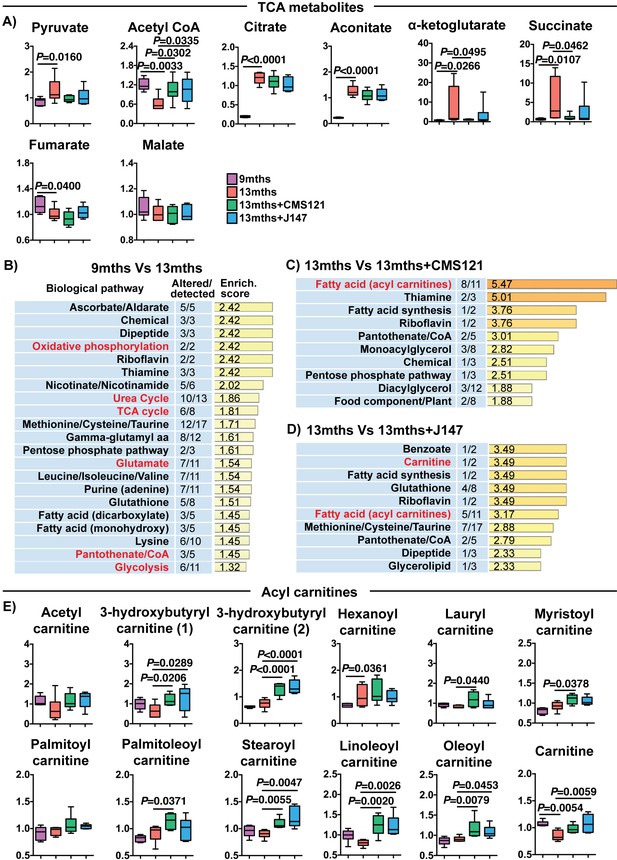
CMS121 and J147 modulate key metabolites associated with mitochondria in the brain of SAMP8 mice.
(A) Levels of TCA metabolites detected in the cortex of 9 months, 13 months, 13 months+CMS121 and 13 months+J147 SAMP8 mice. Values are expressed as box-and-whisker plots. One-way ANOVA followed by Tukey-Kramer post-hoc test (n = 6/group). Pathway enrichment analysis with cortex metabolites found altered in 9 months vs 13 months (B), 13 months vs 13 months+CMS121 (C), and 13 months vs 13 months+J147 (D). Enrichment scores were calculated as described in the Supplemental Materials and methods. Number of metabolites altered in a biological pathway and the total number of metabolites measured in that pathway (Altered/detected) are shown as a complement to the enrichment score. (E) Levels of all acyl carnitine metabolites measured in the cortex of 9 months, 13 months, 13 months+CMS121 and 13 months+J147 SAMP8 mice. Values are expressed as box-and-whisker plots. One-way ANOVA followed by Tukey-Kramer post-hoc test (n = 6/group).
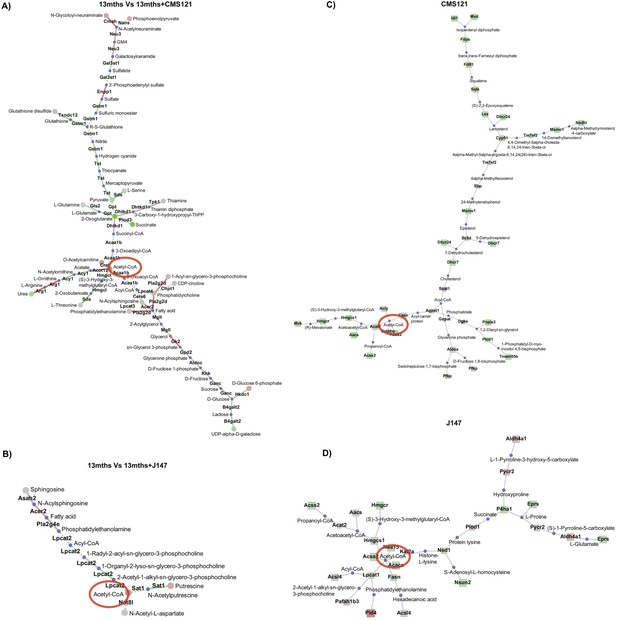
Network analysis.
Network analysis was carried out integrating both the transcriptomic and the metabolomic data obtained from the brains of 13 months vs 13 months+CMS121 (A) and 13 months vs 13 months+J147 (B) SAMP8 mice. Nodes represent the metabolites; links represent the genes that encode the corresponding enzymes of the reactions (solid line) or trans-reaction (dashed line). Larger nodes represent lower p-values. Red color indicates up-regulation; green color indicates down-regulation; blue color indicates missing data. Network analysis was also carried out with the whole transcriptomic data obtained from 7 DIV rat primary neurons treated with 1 μM of CMS121 (C) and J147 (D) for 24 hr. Circles represent the metabolites and squares represent the genes that encode the corresponding enzymes of the reactions. Acetyl-CoA is identified in all graphs with a red circle.
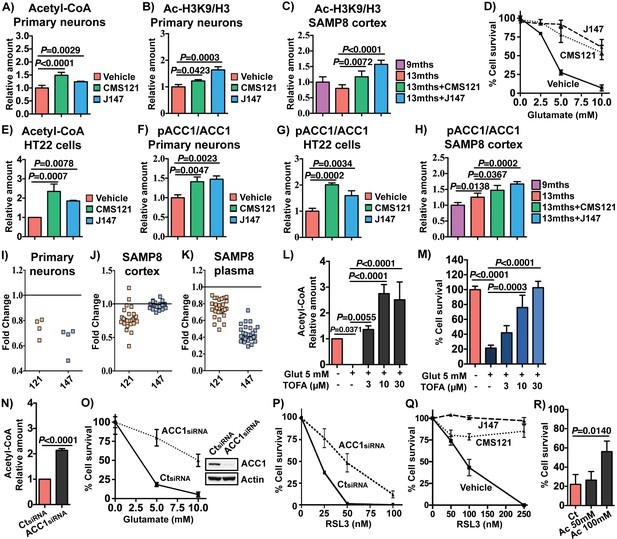
CMS121 and J147 increase acetyl-CoA levels associated with inhibition of ACC1.
Increases in acetyl-CoA levels are protective and associated with inhibition of ACC1 by CMS121 and J147. (A) Total levels of acetyl-CoA in rat primary neurons (n = 5/group), after treatment with 1 μM of each compound for 24 hr. Acetylation of H3 at Lys 9 (Ac-H3K9) was assessed by Western blotting and normalized to levels of total H3 in rat primary neurons (n = 3/group) (B) and mouse cortical tissue (n = 5/group) (C). (D) Protection during oxytosis in HT22 cells with CMS121 (500 nM) and J147 (50 nM) (n = 3/group). (E) Total levels of acetyl-CoA in HT22 cells (n = 3/group). Phosphorylation of ACC1 was assessed by Western blotting and normalized to levels of respective total protein in rat primary neurons (n = 3/group) (F), HT22 cells (n = 3/group) (G) and SAMP8 mouse cortical tissue (n = 5/group) (H). Scans of the blots are shown in Figure 6—figure supplement 1. (I) Fold change in the levels of different polyunsaturated fatty acids in primary neurons treated with 1 μM of each compound for 24 hr. Fold change in the levels of multiple long-chain fatty acids including major polyunsaturated fatty acids in the brains (J) and plasma (K) of old SAMP8 treated with CMS121 and J147, relative to old SAMP8. (L) Total levels of acetyl-CoA in HT22 cells treated with 5 mM glutamate alone or in the presence of 3, 10 or 30 μM TOFA, determined 8 hr after treatment (n = 3/group). (M) Protection during oxytosis in HT22 cells after inhibition of ACC1 with 3, 10 or 30 μM TOFA (n = 3/group). (N) Total levels of acetyl-CoA in HT22 cells after knock down with ACC1 siRNA, measured 48 hr after knock down (n = 3/group). Protection during oxytosis (O) and ferroptosis (P) in HT22 cells after knock down with ACC1 siRNA (n = 3/group). (Q) Protection during ferroptosis in HT22 cells with CMS121 (250 nM) and J147 (100 nM) (n = 3/group). (R) Protection during oxytosis in HT22 cells treated with sodium acetate (Ac), with survival normalized to control treatment with sodium chloride. All data are mean ± SD. One-way ANOVA followed by Tukey-Kramer post-hoc test.
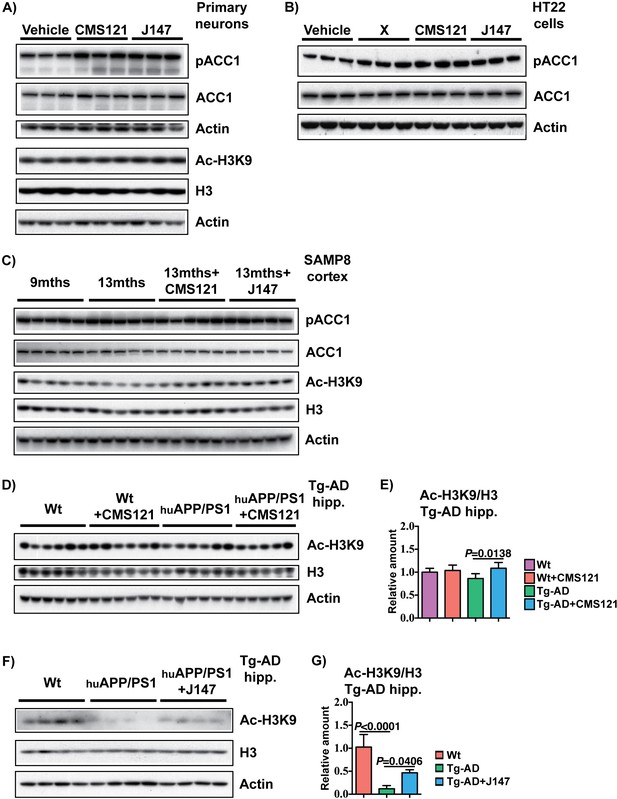
Phosphorylation of ACC1 and acetylation of H3 in primary neuronal cultures, HT22 cells and brain cortex of SAMP8 mice by CMS121 and J147.
RIPA-soluble fractions from 7 DIV rat primary neurons (A) and HT22 cells (B) treated with 1 μM of each compound for 4 hr, cortical tissue of 9 months, 13 months, 13 months+CMS121 and 13 months+J147 SAMP8 mice (C), cortical tissue of 18 months Wt, Wt+CMS121, APPswe/PSEN1dE9 and APPswe/PSEN1dE9+CMS121 mice (D and E) and cortical tissue of 23 months Wt, APPswe/PSEN1dE9 and APPswe/PSEN1dE9+J147 (F and G) were analyzed by Western blotting for ACC1 phosphorylated at serine 79 (pACC1), total ACC1, acetylated histone H3 at Lys 9 (Ac-H3K9), total histone H3 and actin. Samples from Tg-AD mice treated with CMS121 and J147 are from two separate studies. Quantifications represented are Ac-H3K9 normalized to total H3. All data are mean ± SD. One-way ANOVA followed by Tukey-Kramer post-hoc test (n = 3/group for rat primary neurons and HT22 cells, n = 5/group for SAMP8, n = 5–6/group for APPswe/PSEN1dE9 CMS121 study, n = 4/group for APPswe/PSEN1dE9 J147 study).
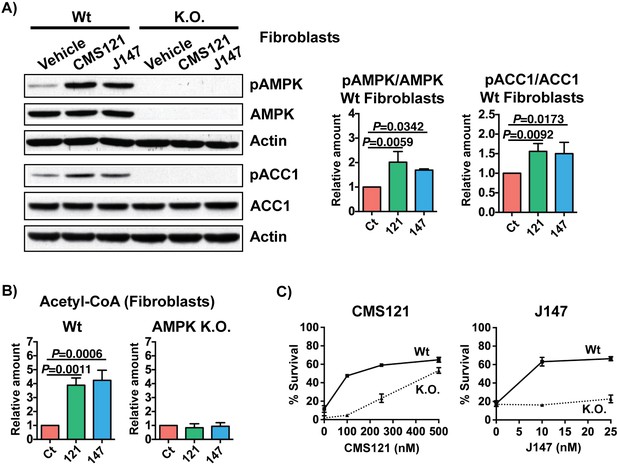
Inhibition of ACC1 by CMS121 and J147 is dependent on AMPK.
Increases in acetyl-CoA levels and protection against oxytosis via inhibition of ACC1 by CMS121 and J147 rely on AMPK activation. (A) Phosphorylation of AMPK and ACC1 after treatment with 1 μM of each compound was assessed by Western blotting and normalized to levels of respective total protein in wildtype (Wt) and AMPK K.O. fibroblasts (n = 3–4/group). No basal phosphorylation of ACC1 was detected in the absence of AMPK. (B) Total levels of acetyl-CoA in fibroblasts (n = 3/group) after compound treatment. (C) Protection against oxytosis with CMS121 and J147 (n = 3/group). All data are mean ± SD. One-way ANOVA followed by Tukey-Kramer post-hoc test.
Additional files
-
Supplementary file 1
List of the DE genes found in 9 vs 13 months old SAMP8 mice and 13 months vs 13 months+compounds SAMP8 mice.
- https://cdn.elifesciences.org/articles/47866/elife-47866-supp1-v2.xlsx
-
Supplementary file 2
List of all 496 metabolites quantified in the brain cortex of 9 months, 13 months, 13 months+CMS121 and 13 months+J147 SAMP8 mice.
Fold changes and specific P values are indicated. One-way ANOVA (n = 6/group).
- https://cdn.elifesciences.org/articles/47866/elife-47866-supp2-v2.xlsx
-
Supplementary file 3
List of all 604 metabolites quantified in the blood plasma of 9 months, 13 months, 13 months+CMS121 and 13 months+J147 SAMP8 mice.
Fold changes and specific P values are indicated. One-way ANOVA (n = 6/group).
- https://cdn.elifesciences.org/articles/47866/elife-47866-supp3-v2.xlsx
-
Transparent reporting form
- https://cdn.elifesciences.org/articles/47866/elife-47866-transrepform-v2.docx
-
Reporting standard 1
The ARRIVE Guidelines Checklist.
- https://cdn.elifesciences.org/articles/47866/elife-47866-repstand1-v2.pdf