Dopamine neuron ensembles signal the content of sensory prediction errors
Figures
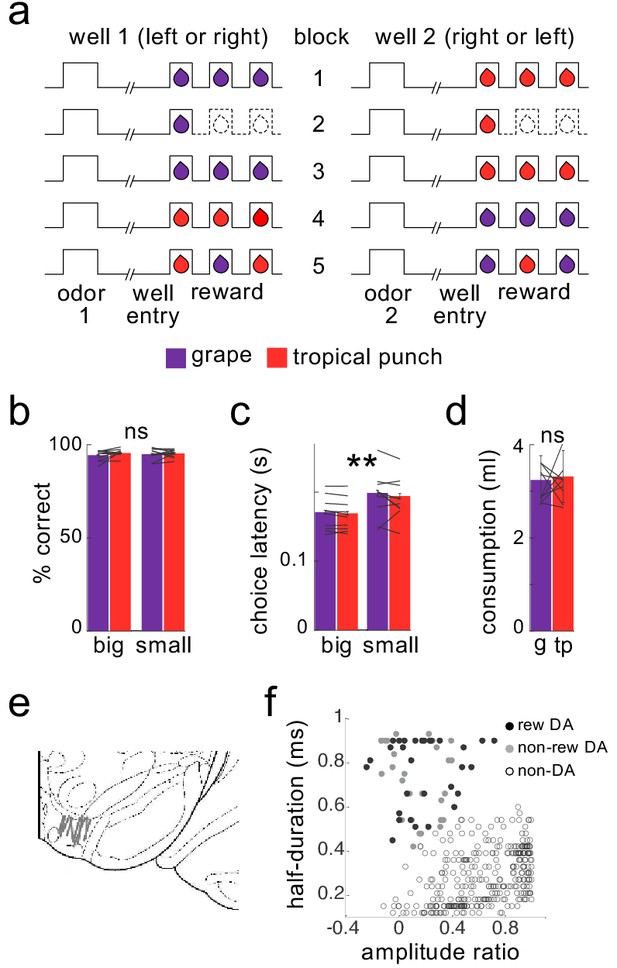
Task design and behavior during recording.
Schematic (a) illustrates the order of events in trials at each well and the number and type of reward delivered at each well in the five trial-blocks performed in all recording sessions. Dashed lines indicate the omission of drops previously delivered. Rats were highly accurate in choosing the rewarded well during recording (b), and accuracy was unaffected by the flavor or number of drops at a particular well, either for the group or for individual subjects (flavor: F1,193=1.3, p=0.26; number: F1,193=1.0, p=0.32; interactions with subject: F’s <= 1.0, p’s > 0.47). Rats were faster to respond for the 3-drop rewards (c), and this effect was again unaffected by the flavor of reward, either for the group or for individual subjects (main effect of number: F1,193=190, p<10−6; main effect of flavor: F1,193=1.75, p=0.19; flavor X subject interaction: F9,193=0.86, p=0.56). A two-bottle preference test run at the end of the sessions (d) also revealed no effect of flavor (F1,9=0.17, p=0.69). Data for individual subjects is illustrated by lines; error bars represent standard errors across sessions for percent correct and latency and across rats for the consumption test. Recordings were made in ventral tegmental area (e), and dopaminergic neurons (n = 30) were identified by waveform cluster analysis (f). **p<0.01. g = grape, tp = tropical punch.
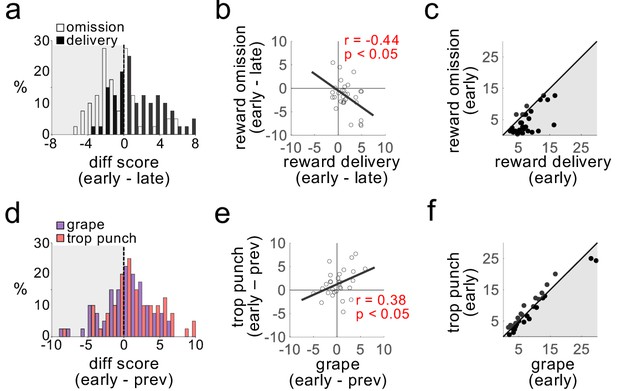
Dopamine neurons do not distinguish the identity of sensory prediction errors.
Plots show firing rates of dopamine neurons in response to transitions in number of reward drops (omission or delivery; a–c) and flavor (grape or tropical punch; d–f). Changes in firing rate in response to omission (negative errors) and delivery (positive errors) were readily distinguishable (a; t29 = 4.0, p<10−3), inversely correlated across neurons (b), and firing rates were markedly different after the transition (c; t29 = 5.2, p<10−4). The same neurons exhibited increased firing rates in response to transitions in the expected flavor of reward (d); t29 = 2.1, p<0.05), but the increases to the two flavors were indistinguishable (t29 = −1.95, ns), positively correlated across neurons (e), and firing rates after the transition also did not distinguish the two flavor errors (f; t29 = 0.13, ns).
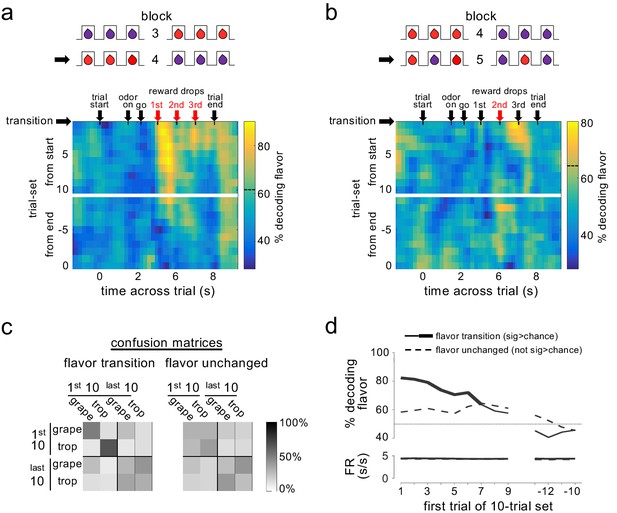
Dopamine ensembles distinguish the identity of sensory prediction errors.
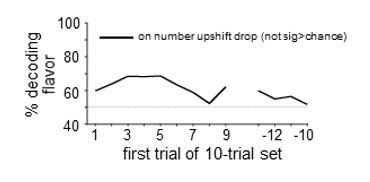
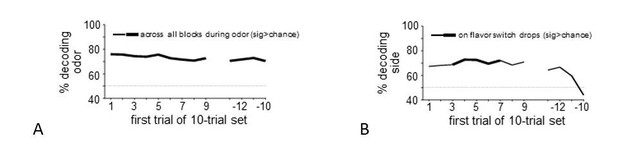
Heat plots show decoding of flavor by dopamine neuron ensembles, using data from a sliding window during trials after all three drops changed flavor (a) or when only the second drop changed flavor (b). Red arrows indicate the time of the new flavor drop delivery. In each case, decoding was significantly above chance at the changed drops, but only early in the block (dotted lines on scale bars show one-tailed 95% confidence interval upper bounds for chance, by permutation tests). This effect was also evident when we collapsed data from the two blocks and compared decoding in epochs capturing firing to the drops where flavor changed versus control epochs capturing firing where flavors had not changed (c); flavor could be decoded accurately by dopamine ensembles only immediately after changes in flavor (patterns in confusion matrices were significantly different at p<10−4 by permutation test). A more detailed analysis using sliding sets of 10-trials (d) showed the decay of flavor decoding as the block progressed (upper plot, solid line), while control decoding of flavor (dotted line) and baseline firing rates in both conditions (lower plot) were unchanged across the block. Thick line in the upper plot shows significance compared to chance (p<0.05 for at least five significant trial sets by permutation test). Thin dotted line in upper plot shows chance decoding level.
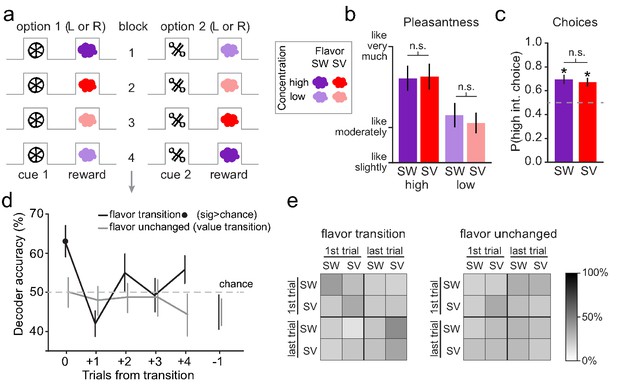
Human midbrain distinguishes the identity of sensory prediction errors.
(a) The reversal learning task involved binary choices between two visual cues to receive either a high or low concentration of one of two food odor rewards (one sweet [SW] and one savory [SV]). The associations were covertly changed throughout the task to induce either sensory prediction errors (e.g. transition from block 1 to block 2) or value prediction errors (e.g. transition from block 2 to block 3). (b) Sweet and savory food odors were matched for pleasantness within each odor concentration (SW high vs. SV high: t22 = 0.18, p=0.86; SW low vs. SV low: t22 = 1.16, p=0.26). Error bars depict within-subject s.e.m. (c) On free choice trials, the cue associated with the high-concentration odor was chosen significantly above chance (50%) for both odor identities (SW: t22 = 4.03, p=2.83×10–4; SV: t22 = 4.20, p=1.83×10−4) and these choice proportions did not differ significantly from each other (t22 = 0.71, p=0.48). Error bars depict within-subject s.e.m. (d) Decoding accuracy of SW vs. SV was significantly above chance on the error trial of flavor transitions (black line) (t22 = 3.22, p=0.004), but not for subsequent trials or the trial preceding error trials (p’s > 0.12). Decoding accuracy of SW vs. SV was at chance for the error trial on value transitions (gray line), as well as subsequent trials, and the trial preceding the value transitions (p’s > 0.15). Error bars depict within-subject s.e.m. (e) Confusion matrices show the decoding accuracy for individual conditions within the decoding analyses (there was a trend that patterns in confusion matrices were different at p=0.08 by permutation test). Within the top left quadrant of the flavor transition matrix (i.e. training and testing the classifier on the error trial of flavor transitions), across all subjects and iterations, accuracy was at 63.3% for SW predictions and 63.8% for SV predictions. All other comparisons for flavor transitions and all comparisons for value transitions were at chance.
Tables
Numbers of putative dopamine neurons recorded in each subject (subjects without dopamine neurons are not listed).
https://doi.org/10.7554/eLife.49315.004| Rat ID | # Dopamine Neurons |
|---|---|
| AA01 | 6 |
| AA05 | 9 |
| AA06 | 1 |
| AA07 | 4 |
| AA09 | 3 |
| AA10 | 1 |
| AA12 | 6 |
Additional files
-
Transparent reporting form
- https://doi.org/10.7554/eLife.49315.007