Large-scale cell-type-specific imaging of protein synthesis in a vertebrate brain
Figures
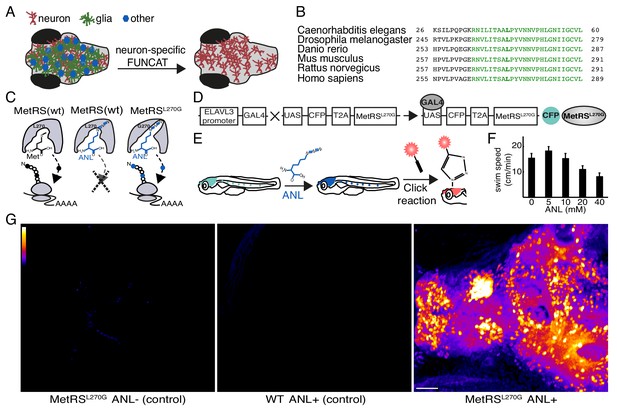
Cell-type-specific labeling of newly synthesized proteins.
(A) Schematic demonstrating the method to visualize newly synthesized proteins in the brain in a cell-type-specific manner. (B) The protein sequence of the catalytic core domain of the MetRS in a number of species including Danio Rerio shows strong conservation (green). Leucine 270 (bold green) was mutated to Glycine to develop cell-type-specific metabolic labeling in zebrafish. (C) Schematic of the binding pocket of the MetRS and the ribosome during translation. The wt MetRS allows the charging of Met (black) that can be incorporated during translation initiation and elongation (left). The non-canonical amino acid ANL (blue), which contains an azide group, does not fit into the binding pocket of the wt MetRS, and thus is notincorporated into nascent protein (center). The mutant MetRSL270G can charge ANL, which is then incorporated into newly synthesized proteins (in cells expressing the MetRSL270G). (D) Schematic of the UAS-CFP-MetRSL270G line transgene. Crossing the line with any Gal4-expressing line allows for the metabolic labeling of newly synthesized proteins in any accessible cell type. (E) A scheme demonstrating the use of the ELAVL3-Gal4:UAS-CFP-MetRSL270G line. Left: a zebrafish larva expressing the transgene in neurons (cyan). Following addition of ANL to the water bath, newly synthesized proteins in neurons incorporate ANL (blue). Right: a whole mount click reaction with a fluorescent alkyne reveals the newly synthesized proteins (red). (F) The effect of different ANL concentrations on swim speed after 24 hr of ANL exposure (measurement was done in the presence of ANL). 10 mM ANL, which had no significant effect on larvae swimming, was used in further experiments. N = 5 to 6 larvae for each concentration. (G) Projections of confocal images of zebrafish larval brains after click reactions demonstrating the specificity of fluorescently labeled nascent protein in the MetRSL270G larva treated with ANL, but not in controls. Scale bar = 50 μm.
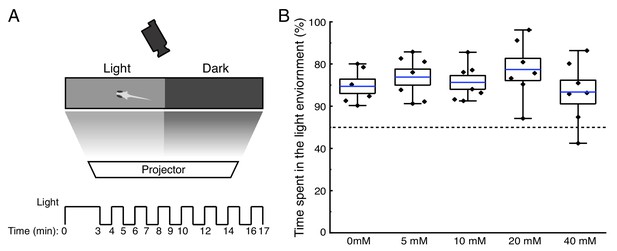
ELAVL3-MetRSL270G Zebrafish larvae maintain light preference following exposure to ANL.
(A) Schematic of the experimental setup. The chamber was placed in a custom-built, enclosed behavioral box that isolated the larvae from outside visual or acoustic stimuli. The behavioral box had a semitransparent bottom, onto which different light environments were projected to each half of the swimming lanes, using a computer-controlled monitor, while the position of the larvae was captured every second using a camera mounted above. The larvae swam for 3 min, then the light and dark regions were shifted every minute in an alternating manner (light region became dark and dark region became light) as indicated by the scheme. (B) Percentage of time spent in light environment by larval zebrafish during the entire experiment. Positions were recorded after 5 s from the time of the light shift and each dot represents the percentage of time for a single larva. Blue line: average, box range: standard error, whiskers show min and max. n = 6 for 0 mM ANL, and 7 larvae for every other ANL concentration. This experiment shows that the larvae maintain the ability to sense the light/dark environment, react to it and keep their natural preferences when exposed to ANL concentrations between 5 and 20 mM.
Shown are dorsal view confocal planes (top to bottom and back) of an ELAVL3-MetRSL270G zebrafish larva brain following 24 hr of ANL labeling followed by click chemistry with a fluorescent alkyne tag.
Left: bright-field, middle: nascent proteins, right: merge. Red: nascent proteins, gray: bright-field.
Shown are dorsal view confocal planes (top to bottom and back) of one ELAVL3-MetRSL270G zebrafish larva brain not incubated with ANL (ANL-) and two larvae incubated with ANL for 24 hr, one WT and one ELAVL3-MetRSL270G.
All samples went through the complete click chemistry procedure. Left: bright-field, middle: nascent protein labelling, right: merge. Red: nascent protein, gray: bright-field.
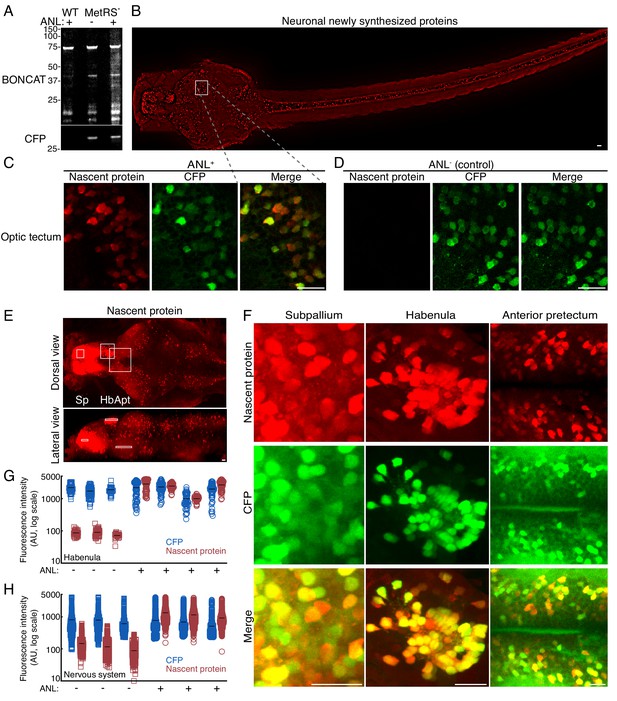
High resolution imaging newly synthesized proteins in different neuronal populations.
3 dpf larvae were incubated with 10 mM ANL for 24 hr in vivo, fixed (at 4 dpf) and clicked to a biotin tag for BONCAT or a fluorescent tag to visualize nascent proteins in situ. (A) Immunoblot detecting newly synthesized proteins in WT larvae treated with ANL (WT ANL+, control), MetRSL270G (MerRS*) larvae treated with ANL (ANL +) or not (ANL-, control). (B) Dorsal view collage projection of confocal images showing fluorescently labeled newly synthesized neuronal proteins (red). (C-F) High magnification view of different brain regions. 4–6 confocal planes are shown (~10 microns in depth). Note the overlap between the CFP channel (Ab staining, green) and the nascent protein channel (click labeling, red), indicating that the signal is specific to cells expressing the MetRSL270G. (C-D) Optic tectum (Ot). (C) Shown are 4 planes of the region indicated by the square in B. (D) The same region in a larva not incubated with ANL demonstrating the CFP but not nascent protein labeling. (E) Maximal projection of labelled newly synthesized proteins in an entire brain (dorsal view and lateral view) (see Figure 2—figure supplement 2A–B for lower brightness). White frames indicate the subpallium (Sp), habenula (Hb) and anterior pretectum (Apt), regions zoomed in (in F). (F) CFP Ab staining and nascent protein labeling in 4–6 confocal images indicated in the white frames in E. See Figure 2—figure supplement 2 for more brain regions. (G-H) Quantification of the average nascent protein levels in the habenula (G) and the entire nervous system (H). Neurons were segmented in 3D using the CFP channel (see supplementary material and Figure 2—figure supplements 6–7) and the average voxel fluorescence intensity for the CFP and fluorescently labeled nascent protein was measured in each cell. Plotted are the average fluorescence intensities in single cells. 30 to 60 neurons were segmented for each habenula of 4 ANL-treated and 3 control larvae (G). (H) Quantification of the average CFP and nascent protein fluorescence intensity in neurons across the entire nervous system. More than 1000 neurons were segmented in 3D using the CFP channel (similar to G). Plotted are the mean fluorescence intensities in single cells from 3 larvae treated (ANL+, squares) or not treated (ANL-, circles) with ANL. See Figure 2—figure supplement 5 for statistical differences between the groups. Squares: ANL- (control), circles: ANL+, blue: CFP, red: Nascent protein, black line – mean fluorescence intensity within a single larva. One cell had a nascent protein intensity value below ten and is shown on the x-axis. Scale bars = 20 μm.
-
Figure 2—source data 1
Source data for Figure 2G–H.
- https://cdn.elifesciences.org/articles/50564/elife-50564-fig2-data1-v1.xlsx
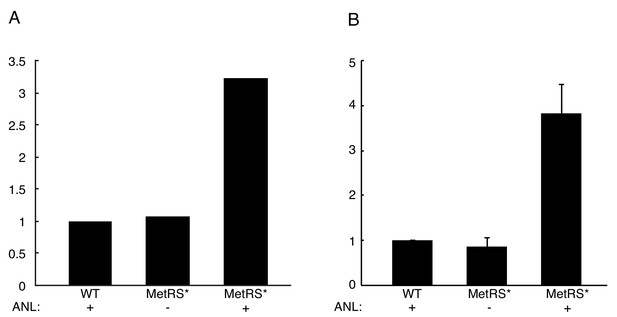
Quantification of Immunoblot following BONCAT for Figure 2A.
(A) Average intensity was measured for each of the three lanes and normalized to the WT, ANL+ sample. (B) Average intensity was measured for each of the three lanes and normalized to the WT, ANL+ sample as in A. Shown is an average of two independent experiments. Bars indicate standard error of the mean.
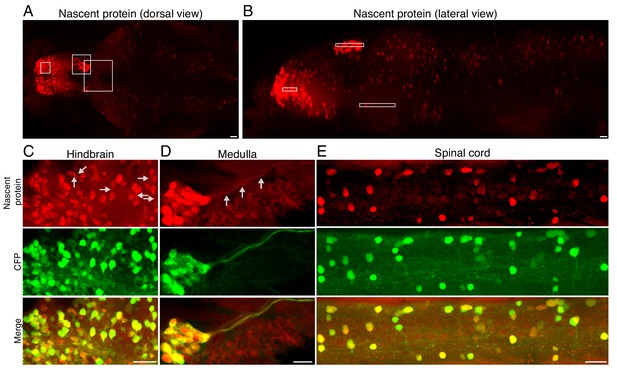
Fluorescently labeled nascent proteins in neurons across the zebrafish larva brain.
(A-B) Maximum intensity projection of newly synthesized protein labeling in an entire brain (A) - dorsal view, B - lateral view) (same image as in Figure 2E,F with lower brightness to demonstrate the variability of nascent protein intensity in regions that appear saturated in Figure 2). White frames indicate the subpallium, habenula and anterior pretectum as in Figure 2E,F. (C-E) Maximum intensity projections of nascent protein labeling (red, top), and CFP Ab staining (green, middle) in indicated brain areas: hindbrain (C), the medulla (D), and the spinal cord (E). Note the nascent protein labeling in neuronal processes indicated in some examples (gray arrows in C, D). Scale bar = 20 μm.
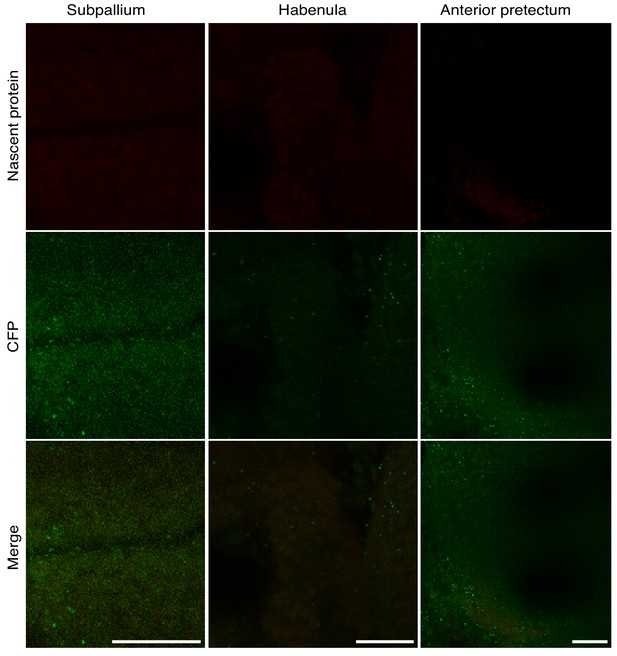
High resolution imaging of neuron-specific newly synthesized proteins in different brain regions – WT larvae exposed to 10 mM ANL for 24 hr (controls).
CFP Ab staining and ANL labeling were performed on WT zebrafish larvae. Shown are confocal images of the similar regions of the same dimensions (X, Y, Z) of the confocal images shown in Figure 2F. The same procedure indicated in Figure 2 was performed and the same microscopy settings were used for the imaging.
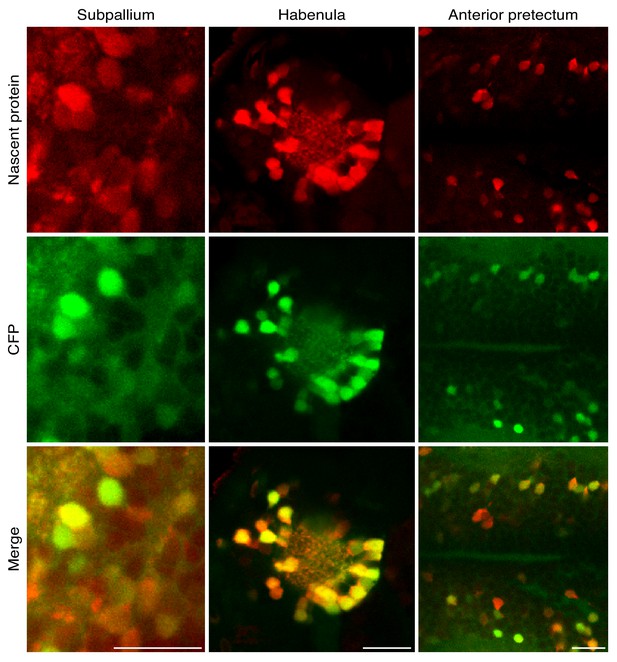
Neuron-specific high resolution imaging of newly synthesized proteins in different brain regions.
3 dpf larvae were incubated with 10 mM ANL for 24 hr in vivo, fixed (at 4 dpf) and clicked to a fluorescent alkyne tag to label nascent proteins in situ. Shown are single planes of CFP Ab staining and nascent protein labeling of the regions in the images in Figure 2F. Scale bars = 20 μm.
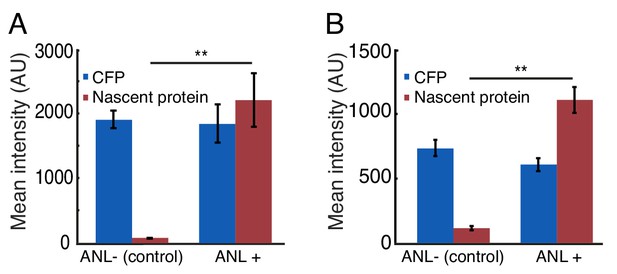
Quantification of neuronal nascent protein levels following 24 hr incubation with ANL.
(A) Quantification of the average levels of nascent proteins in the habenula (data from Figure 2G). Neurons were segmented in 3D using the CFP channel. 30 to 60 neurons were segmented for each habenula and the average voxel intensity for the CFP and nascent protein synthesis channels was measured in each cell. The average neuronal fluorescence intensity in the CFP channel or the nascent protein was calculated for each larva (average data in Figure 2G). Plotted is the average of 4 ANL-treated and 3 control larvae. Bars indicate SEM. (B) Quantification of the averaged fluorescent nascent protein intensity in neurons across the entire nervous system (data from Figure 2H). More than 1000 neurons were segmented in 3D using the CFP channel (similar to Figure 2H). The average fluorescence intensity of CFP or nascent proteins was calculated for each larva (averaged data in Figure 2H). Plotted are the mean intensities of 3 larvae treated (ANL+) or not treated (ANL-) before click chemistry. **p<0.01 (0.0071 and 0.0061 for A and B respectively).
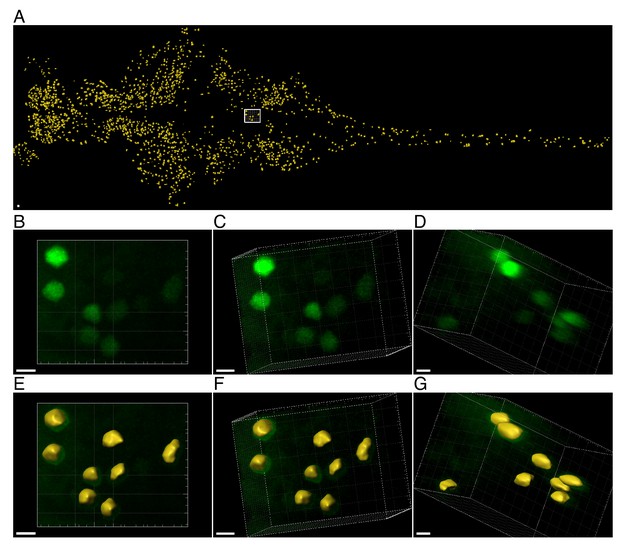
Three-dimensional segmentation of CFP positive cells.
Click chemistry was performed to label nascent proteins. Antibody staining for CFP was performed to detect cells expressing CFP (and MetRSL270G) across the larvae. Individual neurons were segmented in 3D according to the CFP signal, before measuring nascent proteins (in the segmented ROIs). (A) Projection of the surface of segmented neurons across a larva following semi-automatic segmentation using Zen and Imaris 9.2 software (see Materials and methods for details). (B-G) Zoom-in on a representative ROI (white frame in A showing the raw data and segmentation. Projections of ~20 microns in depth, of the same ROI from different viewpoints (demonstrated by rotating the image (B-D)) are used to show the overlay with the surface after segmentation (E-G respectively). A ROI with a relatively low density of neurons was chosen for visualization purposes. 1805 cells were segmented in this larva (A). Yellow – surfaces (segmentation), Scale bars: A = 50 μm, B-G = 5 μm.
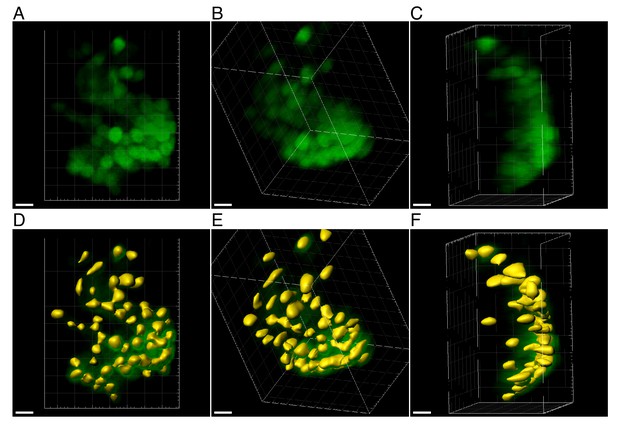
Three-dimensional segmentation of CFP positive cells in the habenula region.
Zoom-in on a more challenging densely labeled ROI in the habenula. (A-C) Projections of the CFP channel from different viewpoints and their respective surfaces after segmentation (D-F respectively). The watershed algorithm (in Imaris) was used to separate adjacent cells. Cells that were not detected by Imaris were not included. Scale bars = 5 μm.
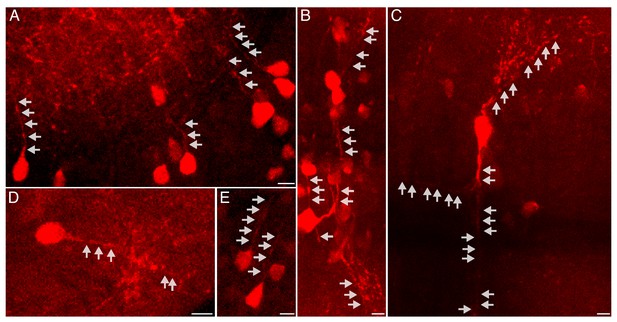
Detection of nascent protein in neuronal processes.
Representative images of cells exhibiting labeling of nascent proteins in neurites. Maximum intensity projections of 3–6 confocal planes (5.7–12 microns) are shown. Freely swimming larvae were incubated with ANL for 24 hr (same larvae as in Figures 2 and 3). Click chemistry was performed to label nascent proteins prior to imaging. Nascent proteins in neuronal processes were detected in different brain regions including optic tectum (A, note the staining in the neuropil in the top left corner), Mauthner cells (B, C), hindbrain (D) and forebrain (E). Gray arrows indicate neurites. Scale bars = 5 μm.
Shown are 3D projections of a representative region-of-interest (tegmentum or anterior hindbrain, see Figure 2—figure supplement 6) from the brain of an ELAVL3-MetRSL270G larva, demonstrating the segmentation of neurons using the CFP channel.
Green: CFP, yellow: cell area following segmentation.
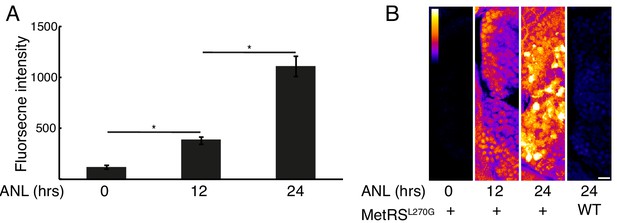
Newly synthesized proteins following different durations of metabolic labeling.
Larvae were incubated with 10 mM ANL in their water bath for the indicated durations before fixation and click reaction. (A) Neurons in the habenula of MetRSL270G larvae were segmented in 3D using the CFP channel and the fnascent proteinintensity was measured in each neuron. Plotted are the average intensities of 3 larvae for each treatment (N = 3 for each treatment). Error bars indicate the SEM (N = 3). *p<0.05 (0.005 for 12 hr and 0.009 for 24 hr). (B) Representative images. Shown are maximal projections of 10 planes (~4 μm) of confocal images in the nascent proteins channel of the forebrain focusing on the olfactory bulb. Calibration bar – top left, scale bar = 10 μm.
-
Figure 3—source data 1
Source data for Figure 3A.
- https://cdn.elifesciences.org/articles/50564/elife-50564-fig3-data1-v1.xlsx
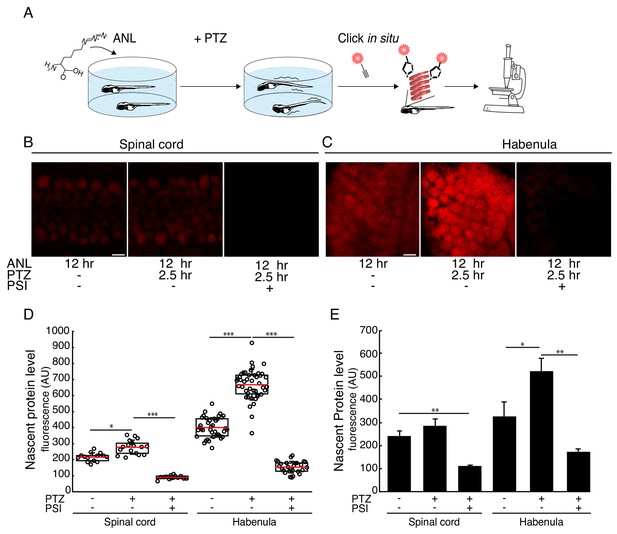
Seizure-induced neuron-specific protein synthesis.
(A) Schematic of the experiment. Freely swimming larvae were incubated with ANL. After 10 hr incubation, PTZ was added for 2.5 hr, inducing seizures. Following fixation, whole-mount click with a fluorescent alkyne and confocal imaging were performed. (B-C) Representative images of the ANL signal in the spinal cord (B) or habenula (C). Shown are maximum projections of 4 confocal planes of the specific regions of larvae treated or non-treated with the protein synthesis inhibitor puromycin (PSI) and treated or non-treated with PTZ. (D) Quantification of the images shown in (B-C). Cells were segmented in 3D using the CFP channel, and the mean nascent protein labeling was measured in each cell using the fluorescent-alkyne channel. The dots represent the mean intensity in cells in the corresponding image (of B-C respectively). Red line – mean, whiskers - STDEV. (E) Bar plot showing the average nascent protein intensity in 3 to 5 larvae PTZ- and PTZ+, respectively. CFP positive cells in the spinal cord or the habenula were segmented in 3D using the CFP antibody staining. The levels of newly synthesized proteins were measured using the fluorescent alkyne (similar to D). More than 100 neurons in the spinal cord and 30 neurons in the habenula were analyzed. The average intensity was calculated for each larva. Plotted are the averaged intensities for each treatment (N = 3 to 5 larvae for each treatment). Error bars indicate SEM. *p<0.05, **p<0.01***p<0.001. Scale bars = 5 μm (B, C).
-
Figure 4—source data 1
Source data for Figure 4E.
- https://cdn.elifesciences.org/articles/50564/elife-50564-fig4-data1-v1.xlsx
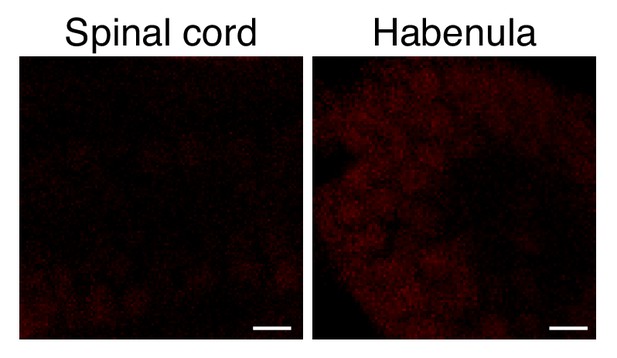
Seizure-induced neuron-specific protein noise levels in WT.
Larvae were incubated with ANL and PTZ and were processed as described in Figure 4, for the ELAVL3-MetRSL270G line. Scale bar = 5 μm.
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Antibody | Rabbit polyclonal anti GFP | Invitrogen | Cat# A11122 | IF(1:600) |
| Antibody | Chicken polyclonal anti GFP | Aves | Cat# GFP 1010 | WB(1:1000) |
| Antibody | Donkey polyclonal anti-chicken IR680 | Licor | Cat# 926–68075 | WB(1:10000) |
| Antibody | Goat anti-rabbit IR800 | Licor | Cat# 925–32211 | WB(1:10000) |
| Zebrafish line | HuC-Gal4 | Stevenson, T. J. et al | Schuman lab, See Materials and methods section | |
| Zebrafish line | UAS-MetRSL270 | Tefor-Amagen/ This paper | Schuman lab, See Materials and methods section | |
| Zebrafish line | ELAVL3-MetRSL270G | Tefor-Amagen/ This paper | Schuman lab, See Materials and methods section |





