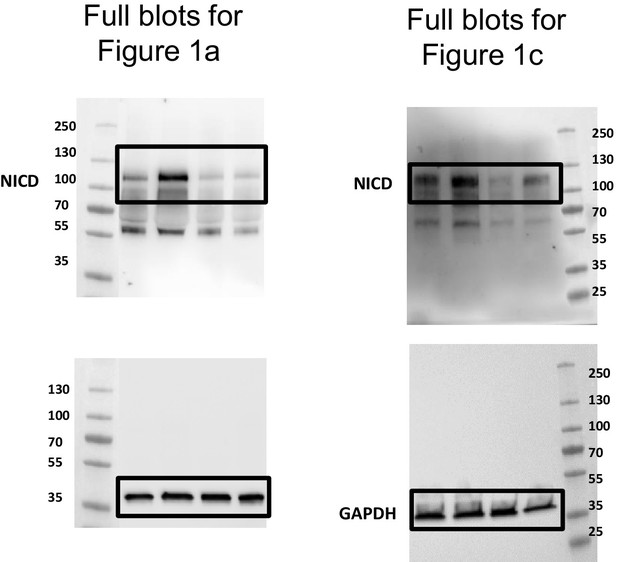
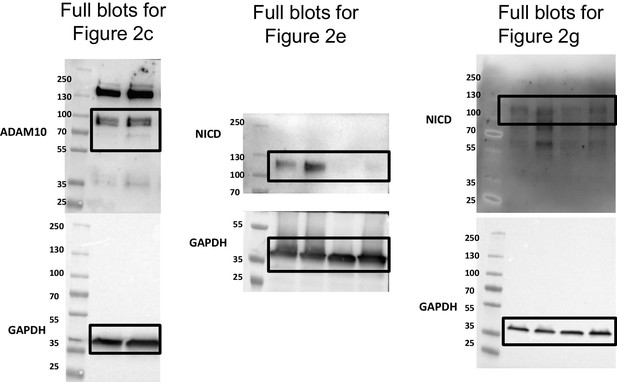
Shear stress activates ADAM10 sheddase to regulate Notch1 via the Piezo1 force sensor in endothelial cells
Figures
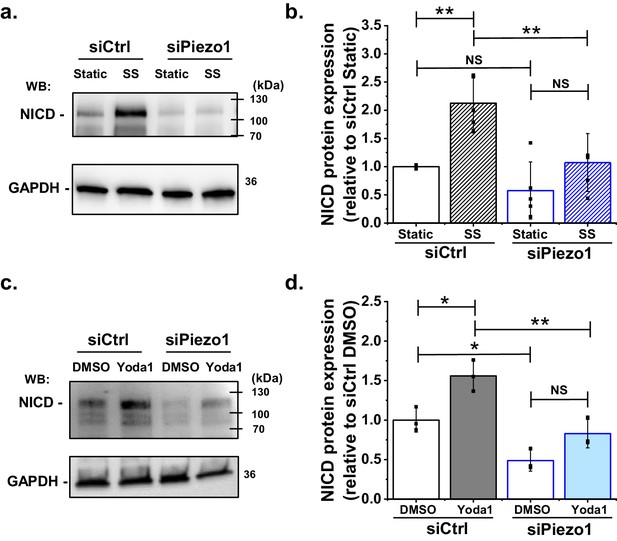
Shear stress induced S3 cleavage of Notch1 is Piezo1 dependent.
(a) Representative Western blot labelled with anti-NICD and anti-GAPDH (loading control) antibodies for HMVEC-Cs exposed to 10 dyn.cm−2 laminar shear stress (SS) for 1 hr. Static was without SS. Cells were transfected with control siRNA (siCtrl) or Piezo1 siRNA (siPiezo1). The expected mass of NICD is 110 kDa. Lower molecular bands were also apparent in some experiments and may have been degraded NICD. (b) Quantification of data of the type exemplified in (a), showing mean ± SD data for abundance of NICD normalized to siCtrl Static (n = 4). (c) Representative Western blot labelled with anti-NICD and anti-GAPDH antibodies for HMVEC-Cs treated for 30 min with 0.2 µM Yoda1 or vehicle (DMSO) after transfection with control siRNA (siCtrl) or Piezo1 siRNA (siPiezo1). (d) Quantification of data of the type exemplified in (c), showing mean ± SD for abundance of NICD normalized to siCtrl DMSO (n = 3). Statistical analysis: Two-way ANOVA test was used, indicating *p<0.05, **p<0.01 or not significantly different (NS).
-
Figure 1—source data 1
Source data for Figure 1.
- https://cdn.elifesciences.org/articles/50684/elife-50684-fig1-data1-v2.xlsx
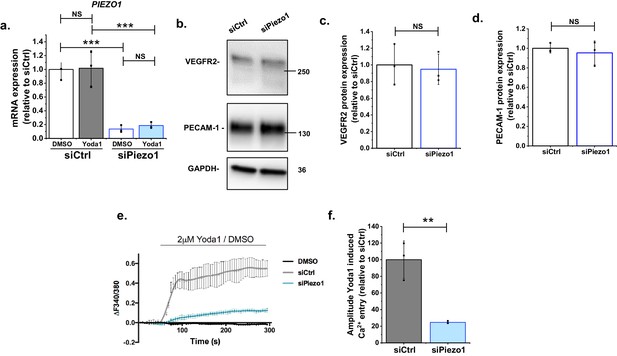
Additional supporting data for Figure 1.
(a) Summarized mean ± SD (n = 3) quantitative PCR data for fold-change in Piezo1 mRNA in HMVEC-Cs treated for 2 hr min with 0.2 mM Yoda1 or vehicle (DMSO) after transfection with control siRNA (siCtrl) or Piezo1 siRNA (siPiezo1). (b) Representative Western blot labelled with anti-VEGRF2, anti-PECAM-1 (anti-CD31) and anti-GAPDH antibodies for HMVEC-Cs after transfection with control siRNA (siCtrl) or Piezo1 siRNA (siPiezo1). (c, d) Quantification of data of the type exemplified in (b), showing mean ± SD data for abundance of VEGFR2 (c) and PECAM-1 (d) normalized to siCtrl (n = 3). (e–f) Representative intracellular Ca2+ measurement traces (e) in HMVEC-Cs during application of 2 μM Yoda1 or its control DMSO, 48 hr after transfection with control siRNA (siCtrl) or Piezo1 siRNA (siPiezo1), shown as mean ± SD of the amplitude for n = 3 (f). Two-way ANOVA test was used for (a) and t-test for (c, d, f), indicating **p<0.01, ***p<0.001 or not significantly different (NS).
-
Figure 1—figure supplement 2—source data 1
Source data for Figure 1—figure supplement 2.
- https://cdn.elifesciences.org/articles/50684/elife-50684-fig1-figsupp2-data1-v2.xlsx
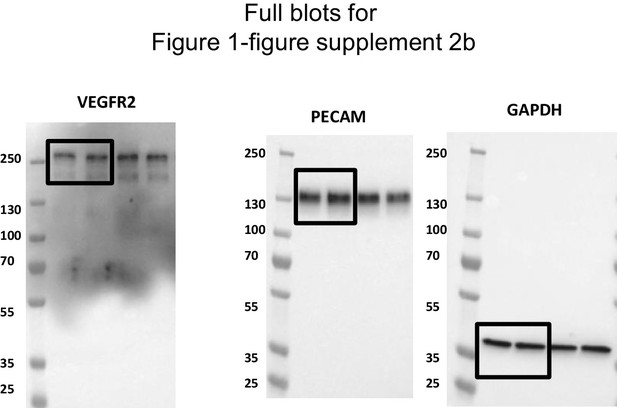
Uncropped western blots for the Figure 1—figure supplement 2b.
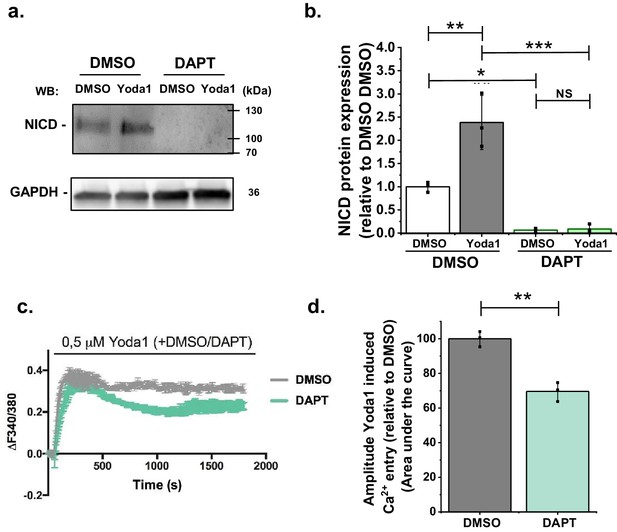
The response depends on γ-secretase.
(a) Representative Western blot labelled with anti-NICD and anti-GAPDH antibodies for HMVEC-Cs treated for 30 min with 0.2 µM Yoda1 or vehicle (DMSO) in the absence or presence of 10 µM DAPT. (b) Quantification of data of the type exemplified in (a), showing mean ± SD data for abundance of NICD normalized to vehicle (DMSO) control (n = 3). (c–d) Representative intracellular Ca2+ measurement traces (c) in HMVEC-Cs during prolonged application of 0.5 μM Yoda1 or its vehicle control (DMSO) simultaneously with 10 mM DAPT or its vehicle control (DMSO), shown as mean ± SD of the area under the curve for n = 3 (d). Two-way ANOVA test was used for (b) and t-test for (d), indicating *p<0.05, **p<0.01, ***p<0.001 or not significantly different (NS).
-
Figure 1—figure supplement 4—source data 1
Source data for Figure 1—figure supplement 4.
- https://cdn.elifesciences.org/articles/50684/elife-50684-fig1-figsupp4-data1-v2.xlsx
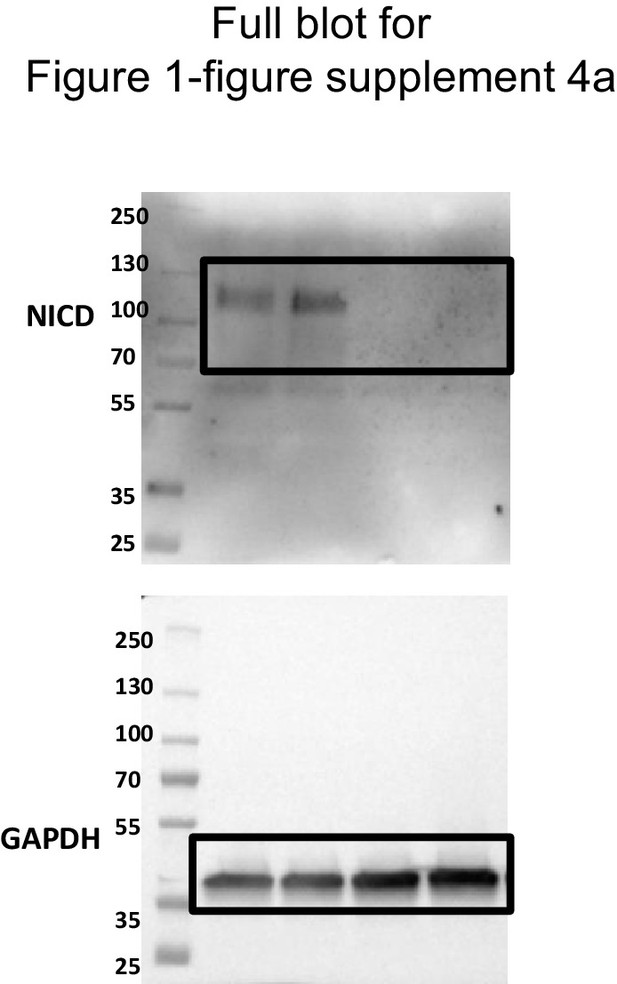
Uncropped western blots for Figure 1—figure supplement 4a.
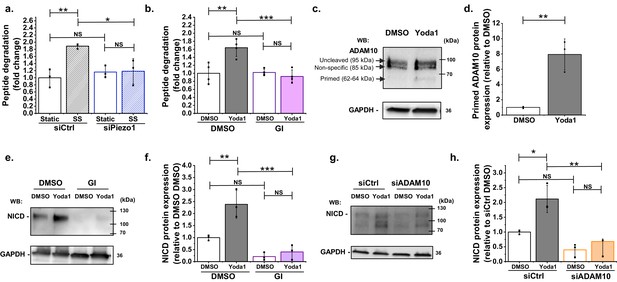
ADAM10 is important for Piezo1 regulation of NICD.
(a) ADAM10 enzyme activity assessed by specific peptide degradation and subsequent fluorescence emission after 30 min exposure of HMVEC-Cs to 10 dyn.cm−2 laminar shear stress (SS). Static was without SS. Cells were transfected with control siRNA (siCtrl) or Piezo1 siRNA (siPiezo1). Data are shown as mean ± SD data (n = 3) relative to static condition. (b) ADAM10 enzyme activity assessed after 30 min treatment of HMVEC-Cs with 0.2 µM Yoda1 in the absence or presence of 5 µM GI254023X (GI). Data are shown as mean ± SD data (n = 4) relative to DMSO condition. (c, d) Quantification of uncleaved (95 kDa) and cleaved (62–64 kDa) ADAM10 in HMVEC-Cs after treatment for 30 min with Yoda1 (0.2 µM). The 85 kDa band between the uncleaved and cleaved ADAM10 was non-specific labelling not related to ADAM10 (Figure 2—figure supplement 2). Data represent mean ± SD (n = 3) and normalization was to the reference protein, GAPDH. (e) Example Western blot labelled with anti-NICD and anti-GAPDH antibodies for HMVEC-Cs treated for 30 min with 0.2 µM Yoda1 or vehicle (DMSO) in the absence or presence of 5 µM GI254023X (GI). (f) Quantification of data of the type exemplified in (e), showing mean ± SD data for abundance of NICD normalized to DMSO (n = 3). (g) Representative Western blot labelled with anti-NICD and anti-GAPDH antibodies for HMVEC-Cs treated for 30 min with 0.2 µM Yoda1 or vehicle (DMSO) after transfection with control siRNA (siCtrl) or ADAM10 siRNA (siADAM10). (h) Quantification of data of the type exemplified in (g), showing mean ± SD data for abundance of NICD normalized to siCtrl DMSO (n = 3). Statistical analysis: Two-way ANOVA test was used for (a, b, f, h), indicating *p<0.05, **p<0.01, ***p<0.001; t-Test was used for (d), indicating **p<0.01; NS, not significantly different.
-
Figure 2—source data 1
Source data for Figure 2.
- https://cdn.elifesciences.org/articles/50684/elife-50684-fig2-data1-v2.xlsx
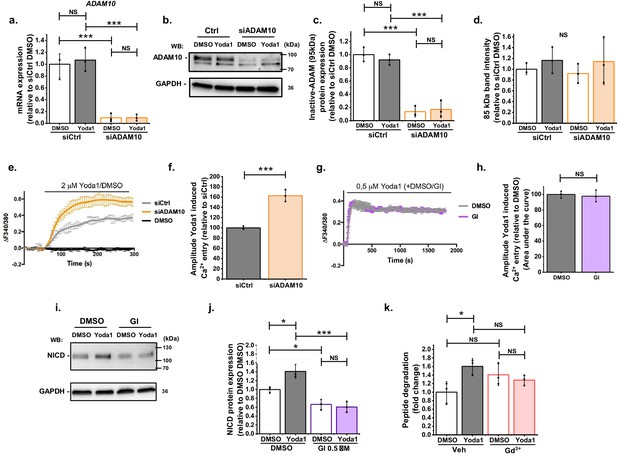
Additional supporting data for Figure 2.
(a) Summarized mean ± SD (n = 3) quantitative PCR data for fold-change in ADAM10 mRNA in HMVEC-Cs treated for 2 hr with 0.2 µM Yoda1 or vehicle (DMSO) after transfection with control siRNA (siCtrl) or ADAM10 siRNA (siADAM10). (b) Representative Western blot labelled with anti-ADAM10 antibody for HMVEC-Cs treated for 30 min with 0.2 µM Yoda1 after transfection with control siRNA (siCtrl) or ADAM10 siRNA (siADAM10). (c, d) Quantification of data of the type exemplified in (b), showing mean ± SD data for abundance of uncleaved ADAM10 (95 kDa) (c) and non-specific labelling of an unknown protein (85 kDa) (d) normalized to siCtrl (n = 3). (e–f) Representative intracellular Ca2+ measurement traces (e) in HMVEC-Cs during application of 2 μM Yoda1 or its vehicle control (DMSO), 48 hr after transfection with control siRNA (siCtrl) or ADAM10 siRNA (siADAM10), shown as mean ± SD of the amplitude for n = 3 (f). (g–h) Representative intracellular Ca2+ measurement traces (g) in HMVEC-Cs during prolonged application of 0.5 μM Yoda1 or its vehicle control (DMSO) simultaneously with 5 µM GI254023X (GI) or its vehicle control (DMSO), shown as mean ± SD of the area under the curve for n = 3 (h). (i) Representative Western blot labelled with anti-NICD and anti-GAPDH antibodies for HMVEC-Cs treated for 30 min with 0.2 µM Yoda1 or vehicle (DMSO) in the absence or presence of 0.5 µM GI254023X (GI) (j) Quantification of data of the type exemplified in (i), showing mean ± SD data for abundance of NICD normalized to siCtrl DMSO (n = 3). (k) ADAM10 enzyme activity assessed by specific peptide degradation and subsequent fluorescence emission after 30 min treatment of HMVEC-Cs with 0.2 µM Yoda1 in the absence or presence of 30 µM Gd3+. Data are shown as mean ± SD data (n = 3) relative to vehicle condition. Statistical analysis: Two-way ANOVA test was used for (a, c, d, j, k), indicating *p<0.05, **p<0.01 and ***p<0.001. T-test was used for (f, h), indicating ***p<0.001. NS indicates no significant difference.
-
Figure 2—figure supplement 2—source data 1
Source data for Figure 2—figure supplement 2.
- https://cdn.elifesciences.org/articles/50684/elife-50684-fig2-figsupp2-data1-v2.xlsx
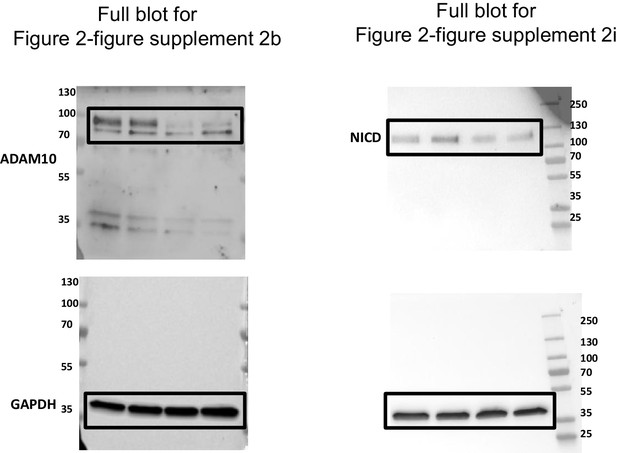
Uncropped western blots for Figure 2—figure supplement 2b and Figure 2—figure supplement 2i.
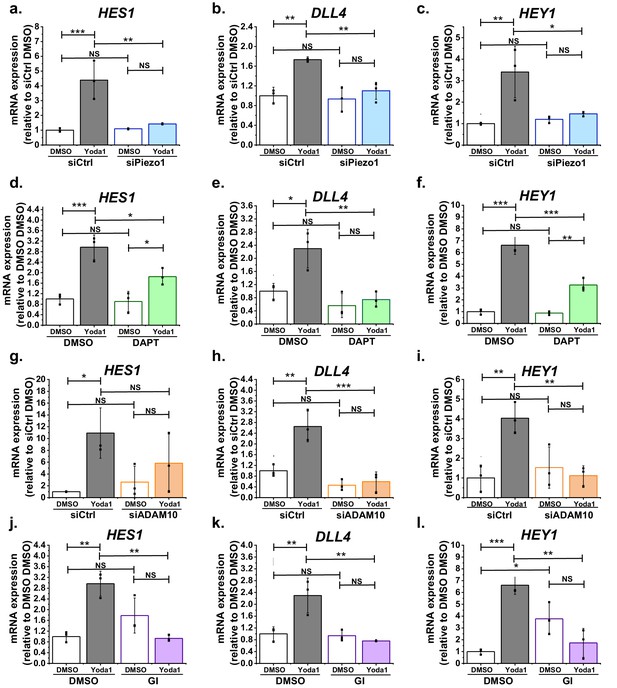
Function significance for downstream gene expression.
(a, b, c) Summarized mean ± SD (n = 3) quantitative PCR data for fold-change in HES1 (a), DLL4 (b) and HEY1 (c) mRNA in HMVEC-Cs treated for 2 hr with 0.2 µM Yoda1 or vehicle (DMSO) after transfection with control siRNA (siCtrl) or Piezo1 siRNA (siPiezo1). (d, e, f) Summarized mean ± SD (n = 3) quantitative PCR data for fold-change in HES1 (d), DLL4 (e) and HEY1 (f) mRNA in HMVEC-Cs treated for 2 hr with 0.2 µM Yoda1 in the absence or presence of 10 µM DAPT. (g, h, i) Summarized mean ± SD (n = 3) quantitative PCR data for fold-change in HES1 (g), DLL4 (h) and HEY1 (i) mRNA in HMVEC-Cs treated for 2 hr with 0.2 µM Yoda1 or vehicle (DMSO) after transfection with control siRNA (siCtrl) or ADAM10 siRNA (siADAM10). (j, k, l) Summarized mean ± SD (n = 3) quantitative PCR data for fold-change in HES1 (j), DLL4 (k) and HEY1 (l) mRNA in HMVEC-Cs treated for 2 hr with 0.2 µM Yoda1 in the absence or presence of 5 µM GI254023X (GI). Normalization and statistical analysis: mRNA expression was normalized to GAPDH mRNA abundance. Two-way ANOVA test was used, indicating *p<0.05, **p<0.01, ***p<0.001 or not significantly different (NS).
-
Figure 3—source data 1
Source data for Figure 3.
- https://cdn.elifesciences.org/articles/50684/elife-50684-fig3-data1-v2.xlsx
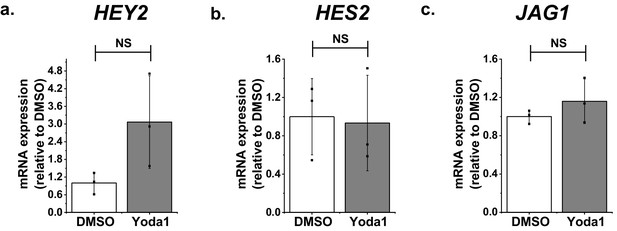
Additional data relating to Figure 3.
Additional gene quantification for HMVEC-Cs treated for 2 hr with 0.2 μM Yoda1 or vehicle (DMSO) (a) HEY2. (b) HES2. (c) JAG1. Statistical analysis: t-test was used.
-
Figure 3—figure supplement 1—source data 1
Source data for Figure 3—figure supplement 1.
- https://cdn.elifesciences.org/articles/50684/elife-50684-fig3-figsupp1-data1-v2.xlsx
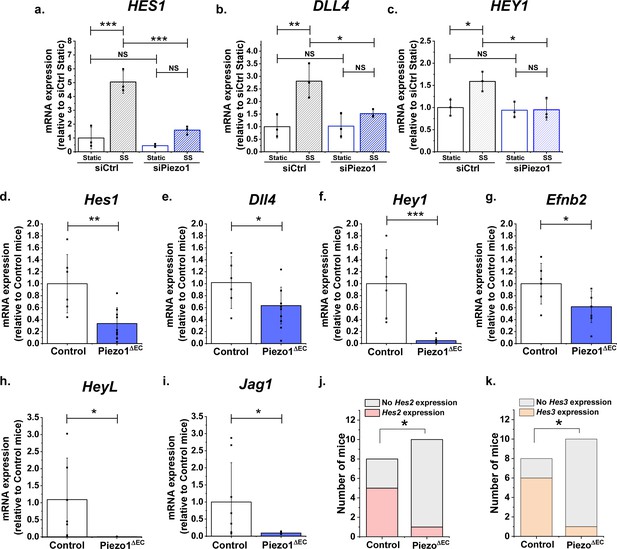
Endothelial Piezo1 is required for the gene expression of Notch1 targets in HMVEC-C exposed to shear stress and in mouse liver endothelial cells.
(a, b, c) Summarized mean ± SD (n = 3) quantitative PCR data for fold-change in HES1 (a), DLL4 (b) and HEY1 (c) mRNA in HMVEC-Cs exposed to 10 dyn.cm−2 laminar shear stress (SS) for 2 hr after transfection with control siRNA (siCtrl) or Piezo1 siRNA (siPiezo1). Hes1 (d), Dll4 (e), Hey1 (f) and Efnb2 (g) mRNA expression in liver endothelial cells freshly-isolated from control mice (Control, n = 6) and endothelial Piezo1 knockout mice (Piezo1ΔEC) (n = 9). HeyL (h) (Control mice, n = 7; Piezo1ΔEC mice, n = 7) and Jag1 (i) (Control mice, n = 8; Piezo1ΔEC mice, n = 9) mRNA expression in liver endothelial cells. Normalization and Statistical analysis: mRNA expression was normalized to abundance of Actb mRNA, which was not different between Control and Piezo1ΔEC (Figure 4—figure supplement 2). Hes2 (j) and Hes3 (k) mRNA expression in liver endothelial cells freshly-isolated from control mice (Control, n = 8) and Piezo1ΔEC mice (n = 10), represented as the number of mice with detectable expression or no detectable expression of the gene. Statistical analysis: Two-way ANOVA test was used for (a, b, c), indicating *p<0.05, **p<0.01, ***p<0.001. t-Test was used for (d, e, f, g, h, i) indicating significant difference of Piezo1ΔEC cf Control *p<0.05, **p<0.01, ***p<0.01. Fisher’s exact test was used for (j, k) indicating significant difference of Piezo1ΔEC cf Control *p<0.05. NS indicates not significantly different.
-
Figure 4—source data 1
Source data for Figure 4.
- https://cdn.elifesciences.org/articles/50684/elife-50684-fig4-data1-v2.xlsx
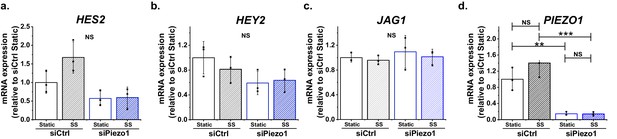
Additional data relating to Figure 4.
Additional gene quantification for HMVEC-Cs exposed to 10 dyn.cm-2 laminar shear stress (SS) for 2 hr after transfection with control siRNA (siCtrl) or Piezo1 siRNA (siPiezo1). Static was without SS. (a) HES2. (b) HEY2. (c) JAG1. (d) PIEZO1. Data are expressed as mean ± SD (n = 3), and are normalized to GAPDH gene expression and expressed as fold change of control condition, Statistical analysis: Two-way ANOVA test was used indicating **p<0.01 and ***p<0.001 or not significantly different (NS).
-
Figure 4—figure supplement 1—source data 1
Source data for Figure 4—figure supplement 1.
- https://cdn.elifesciences.org/articles/50684/elife-50684-fig4-figsupp1-data1-v2.xlsx
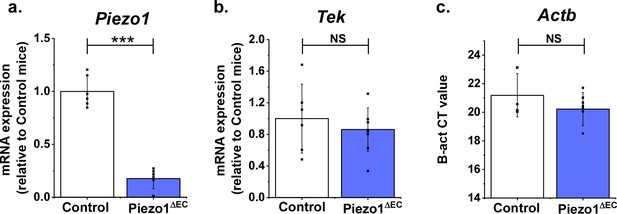
Supporting data for Figure 4.
Additional gene quantification for liver endothelial cells from Piezo1ΔEC and Control mice. (a) Piezo1. (b) Tek. (c) Actb (raw CT values). (a, b) Normalized to abundance of Actb (reference) expression. Statistical analysis: t-test was used for comparisons, indicating ***p<0.001 or not significantly different (NS).
-
Figure 4—figure supplement 2—source data 1
Source data for Figure 4—figure supplement 2.
- https://cdn.elifesciences.org/articles/50684/elife-50684-fig4-figsupp2-data1-v2.xlsx
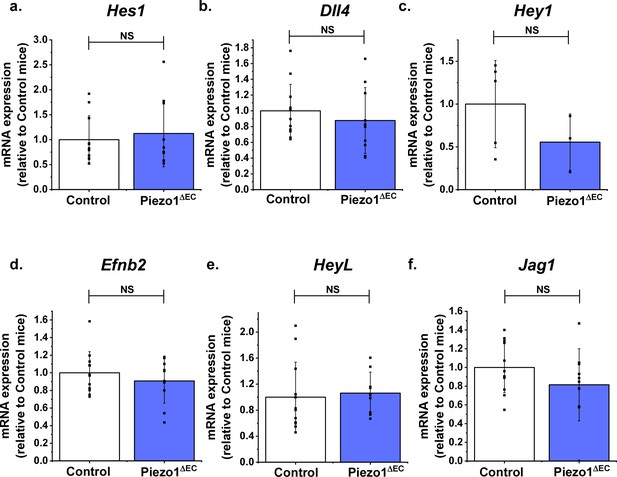
Notch1 target gene expression changes are not evident in whole liver.
Gene quantification for whole liver from Piezo1ΔEC (n = 10) and Control mice (n = 12). (a) Hes1. (b) Dll4. (c) Hey1 (in 7 Control and 7 Piezo1ΔEC experiments Hey1 expression was not convincingly detected, so these data were excluded). (d) Efnb2. (e) HeyL. (f) Jag1. Normalized to abundance of the reference Actb expression. Statistical analysis: t-test was used for comparisons. There were no significant differences between any pairs (NS).
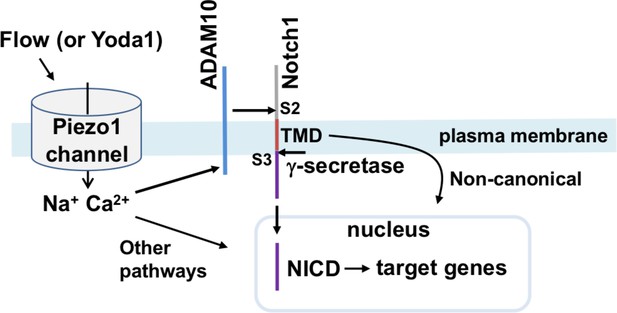
Summary of the proposed pathway.
Activation of Piezo1 channel by mechanical force (e.g. fluid flow) or chemical agonist (Yoda1) causes elevation of the intracellular Ca2+ concentration which stimulates enzymatic activity of ADAM10 to cause S2 and S3 Notch1 cleavage and release of NICD to drive target gene expression that includes increased expression of Hes1, Dll4, Hey1, HeyL, Jag1, Hes2, Hes3 and Efnb2 in hepatic vasculature of the mouse. In the schematic we also include the suggested contributions from ADAM10/Notch1/NICD independent signalling from Piezo1 and non-canonical Notch1 signalling via the Notch1 transmembrane domain (TMD), as described and referenced in the text.
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Strain, strain background (Mus musculus, C57BL6/J, male) | Piezo1flox/flox/Cdh5-Cre | University of Leeds (Rode et al., 2017) | N/A | |
| Cultured cells (Homo sapiens) | HMVEC-Cs | Lonza | Cat# CC-7030 | |
| Antibody | anti-cleaved Notch1 val1744 (D3B8) (Rabbit monoclonal) | Cell Signaling Technology | Cat# 4147, RRID:AB_2153348 | WB (1:1000) |
| Antibody | anti-ADAM10 (Rabbit polyclonal) | Merck Millipore | Cat# AB19026, RRID:AB_2242320 | WB (1:1000) |
| Sequence-based reagent | ON-TARGET plus Control siRNA | Dharmacon | Cat# L-001810 | |
| Sequence-based reagent | ON-TARGET plus SMARTpool Human siRNA ADAM10 | Dharmacon | Cat# L-004503 | |
| Sequence-based reagent | Piezo1 siRNA | Sigma-Aldrich | N/A | GCAAGUUCGUGCGCGGAUU[DT][DT] |
| Commercial assay or kit | SensoLyte 520 ADAM10 Activity Assay Kit | AnaSpec Inc | Cat# AS-72226 | Use kit directions |
| Chemical compound, drug | GI 254023X | Tocris Bioscience | Cat# 3995 | |
| Chemical compound, drug | DAPT | Sigma-Aldrich | Cat# D5942 | |
| Chemical compound, drug | Yoda1 | Tocris Bioscience | Cat# 5586–10 |
Additional files
-
Supplementary file 1
PCR primer sequences.
- https://cdn.elifesciences.org/articles/50684/elife-50684-supp1-v2.docx
-
Transparent reporting form
- https://cdn.elifesciences.org/articles/50684/elife-50684-transrepform-v2.docx