A protein quality control pathway at the mitochondrial outer membrane
Figures
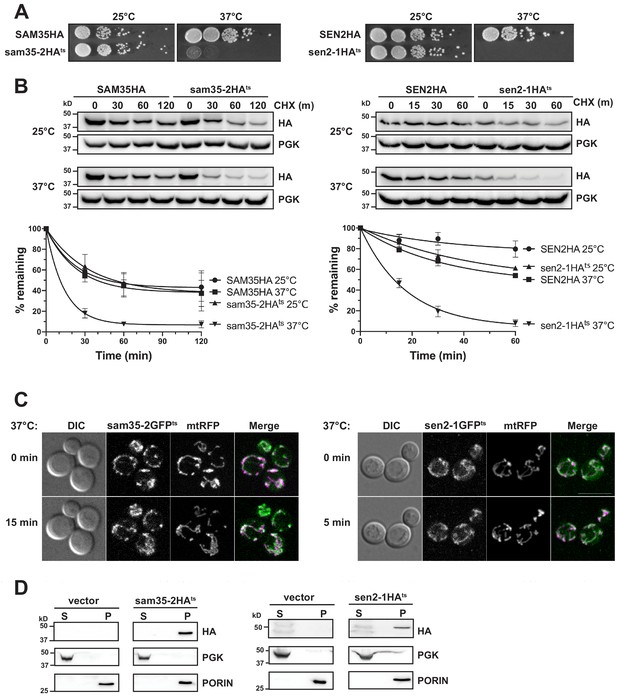
The temperature sensitive proteins sam35-2HAts and sen2-1HAts are novel thermosensitive substrates for mitochondrial quality control.
(A) Spot growth assay of cells expressing chromosomal SAM35HA, sam35-2HAts, SEN2HA, or sen2-1HAts (yMM36, 37, 40, and 41, respectively) at permissive (25°) or non-permissive (37°) temperatures. (B) Wild type (WT; WCG4a) yeast were treated with cycloheximide (CHX) at 25°C or 37°C and analyzed at the indicated times to assess the degradation of centromeric (CEN) plasmid-expressed SAM35HA, sam35-2HAts, SEN2HA, or sen2-1HAts (pMM158, 157, 159, 160, respectively). The ts- proteins were detected by immunoblotting with HA antibody. Phosphoglycerate kinase (PGK) served as a protein loading control. Graphed below is the mean and standard deviation (SD) of the PGK-normalized HA signal at each time point for three biological replicates. (C) Live-cell microscopy analysis of agarose-embedded WT cells (WCG4a) co-expressing a mitochondrial-matrix targeted RFP (mtRFP; pMD12) and either sam35-2GFPts (pMD1) or sen2-1GFPts (pMD4) at the indicated times after temperature shift to 37°C. CHX was also added at 0 min, although CHX diffusion through agarose is likely problematic. ‘Merge’ of GFP (green) and RFP (magenta) channels and differential interference contrast (DIC) are shown; Scale bar = 10 μm. (D) Lysates of spheroplasted yeast from the strains used in B were fractionated at 12,000xg at 37°C into mitochondrial pellet (P) and post-mitochondrial supernatant (S). Fractions were subject to immunoblotting with antibodies to HA, PGK (cytosolic protein control), and PORIN (mitochondrial protein control).
-
Figure 1—source data 1
Quantifications of cycloheximide chases.
- https://cdn.elifesciences.org/articles/51065/elife-51065-fig1-data1-v2.xlsx
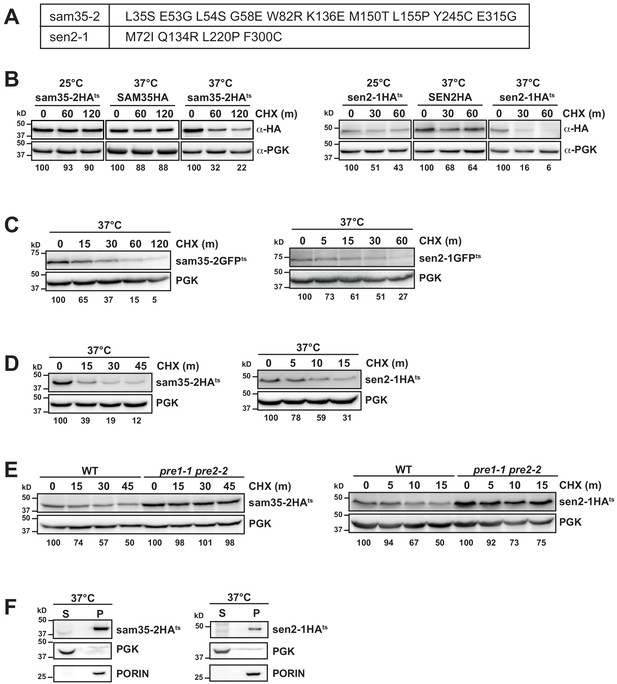
The temperature sensitive proteins sam35-2HAts and sen2-1HAts are novel thermosensitive substrates for mitochondrial quality control.
(A) Amino acid substitutions in the sam35-2ts and sen2-1ts proteins compared to the WT proteins. (B) Cells with chromosomal SAM35HA, sam35-2HAts, SEN2HA, or sen2-1HAts (yMM36, 37, 40, and 41, respectively) were treated with cycloheximide (CHX) at 25°C or 37°C and analyzed at the indicated times to assess the degradation of genomic SAM35HA, sam35-2HAts, SEN2HA, or sen2-1HAts. Ts- proteins were detected by immunoblotting with HA antibody. Phosphoglycerate kinase (PGK) served as a protein loading control. Percentage of each ts- substrate remaining is labeled for each time point under blots. (C) CHX chase for the indicated times at 37°C assessing the turnover of sam35-2GFPts or sen2-1GFPts (pMD1 or 4, respectively) in WT yeast cells (WCG4a). (D) CHX chase for the indicated times at 37°C assessing the turnover of sam35-2HAts or sen2-1HAts (pMM157 or 160, respectively) in WT (WCG4a) cells. (E) CHX chase for the indicated times at 37°C assessing the turnover of sam35-2HAts or sen2-1HAts (pMM157 or 160, respectively) in spheroplasts generated from WT (WCG4a) and pre1-1 pre2-2 (WCG4-11/21a) yeast cells. (F) Lysates from strains expressing genomic sam35-2HAts or sen2-1HAts (yMM37 and 41, respectively) were fractionated at 12,000xg at 37°C into mitochondrial pellets (P) and post-mitochondrial supernatants (S). Fractions were subject to immunoblotting with antibodies to HA, PGK, and PORIN.
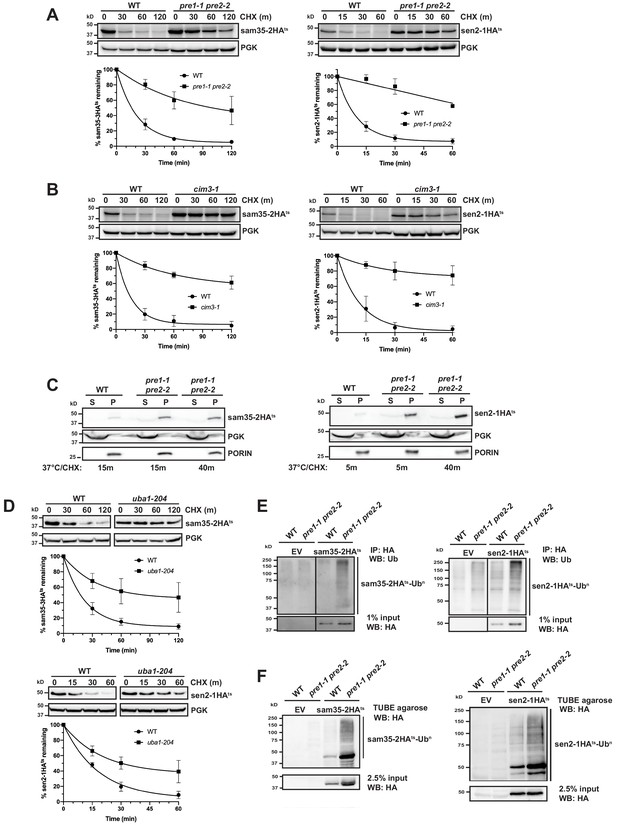
The degradation of MAD QC substrates requires the ubiquitin-proteasome system.
(A, B) CHX chase for the indicated times at 37°C assessing the turnover of sam35-2HAts or sen2-1HAts (pMM157 or 160, respectively) in WT (WCG4a) and pre1-1 pre2-2 proteasome mutant (WCG4-11/21a) cells (A) or WT (CIM) and cim3-1 proteasome mutant cells (B). Proteins were detected by immunoblotting. Graphed below is the mean and SD of the PGK-normalized HA signal at each time point for three biological replicates. (C) Lysates from the strains used in A were fractionated at 12,000xg into mitochondrial pellets (P) and post-mitochondrial supernatants (S) after incubation at 37°C for the indicated times. Fractions were subject to immunoblotting with antibodies to HA, PGK, and PORIN. (D) CHX chase for the indicated times at 37°C assessing the turnover of sam35-2HAts or sen2-1HAts (pMM157 or 160, respectively) in a uba1-204 strain relative to its isogenic WT strain. (E) Ubiquitination of sam35-2HAts and sen2-1HAts was assessed by immunoprecipitation (IP) from lysates of the strains used in A with anti-HA agarose, followed by immunoblotting with ubiquitin antibodies. 1% of IP input lysate was reserved and also analyzed by immunoblotting. (F) Ubiquitination of sam35-2HAts and sen2-1HAts was assessed by IP from lysates of the strains used in A using tandem ubiquitin-binding entities (TUBE) agarose, followed by immunoblotting with HA antibody. 2.5% of the TUBE input lysate was reserved and analyzed by immunoblotting.
-
Figure 2—source data 1
Quantifications of cycloheximide chases.
- https://cdn.elifesciences.org/articles/51065/elife-51065-fig2-data1-v2.xlsx
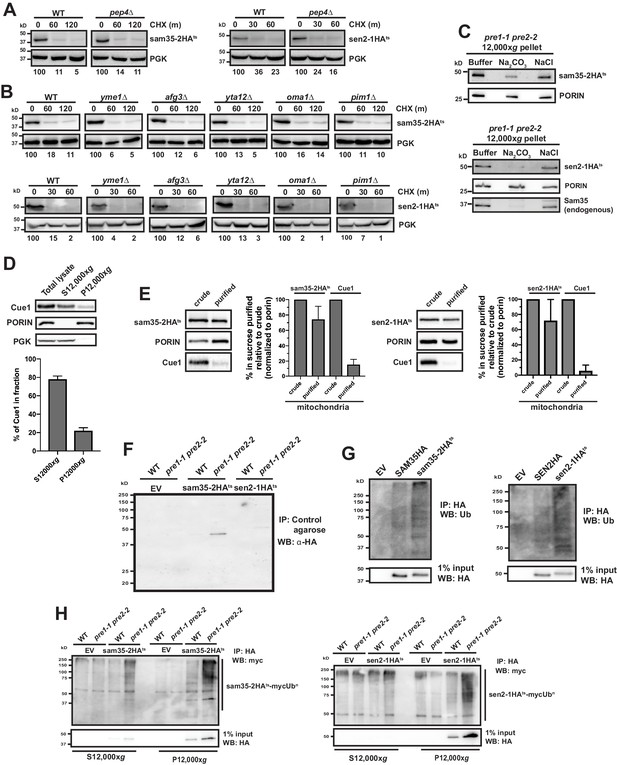
The degradation of MAD QC substrates requires the ubiquitin-proteasome system.
(A, B) CHX chase for the indicated times at 37°C assessing the turnover of sam35-2HAts or sen2-1HAts (pMM157 or 160, respectively) in WT (BY4741) and pep4Δ (A) or yme1Δ, afg3Δ, yta12Δ, oma1Δ, or pim1Δ (B) cells. Proteins were detected by immunoblotting. Percentage of each ts- substrate remaining is labeled for each time point under blots. (C) Sodium carbonate (Na2CO3) extraction of sam35-2HAts or sen2-1HAts (pMM157 and 160, respectively) from the 12,000xg mitochondrial pellet of pre1-1 pre2-2 (WCG4-11/21a) cells. Control treatments of sodium chloride (NaCl) and buffer are shown for comparison. Proteins were detected with immunoblotting for HA, PORIN, and Sam35 (in analysis of sen2-1HAts only). (D) Total lysate from pre1-1 pre2-2 (WCG4-11/21a) cells expressing sam35-2HAts (pMM157) were fractionated at 37°C into mitochondrial pellets (P12,000xg) and post-mitochondrial supernatants (S12,000xg). Fractions were subject to immunoblotting with antibodies to Cue1, PGK, and PORIN. The Cue1 signal in each fraction was quantified and the mean plus standard deviation (SD) of the percentage of the total Cue1 signal is graphed below (N = 3). (E) P12,000xg mitochondrial fractions (‘crude’) isolated from pre1-1 pre2-2 (WCG4-11/21a) cells expressing sam35-2HAts or sen2-1HAts (pMM157 and 160, respectively) were further purified by sucrose gradient fractionation (‘purified’). The HA (sam35-2HAts or sen2-1HAts) and Cue1 signals in crude and purified mitochondrial fractions were quantified and normalized to the PORIN signal. The crude mitochondrial signal was set to 100% and the mean plus SD of the percentage in purified mitochondria relative to crude is graphed (N = 3). (F) Negative control experiment for Figure 2E. Cell lysates from pre1-1 pre2-2 (WCG4-11/21a) cells expressing sam35-2HAts, sen2-1HAts, or empty vector (EV; pMM157, pMM160, or pRS315, respectively) were precipitated with unconjugated TUBE control agarose (containing no ubiquitin-binding domains), followed by immunoblotting with HA antibody. (G) Ubiquitination was assessed by IP with anti-HA agarose from lysates of WT (BY4741) cells expressing empty vector (EV; pRS315), SEN2HA (pMM159), sen2-1HAts (pMM160), SAM35HA (pMM158), or sam35-2HAts (pMM157), followed by immunoblotting with ubiquitin antibody. 1% of the IP was reserved and analyzed by immunoblotting for the unmodified proteins. (H) Ubiquitination in the 12,000xg mitochondrial pellet (P12,000xg) or post-mitochondrial supernatant (S12,000xg) was assessed by IP from each fraction with anti-HA agarose from WT (WCG4a) and pre1-1 pre2-2 (WCG4-11/21a) cells expressing myc-Ub (pSM3666) and either empty vector (EV; pRS315), sam35-2HAts (pMM157), or sen2-1HA (pMM160), followed by immunoblotting with c-myc antibody. 1% of IP input lysate was reserved and analyzed by immunoblotting.
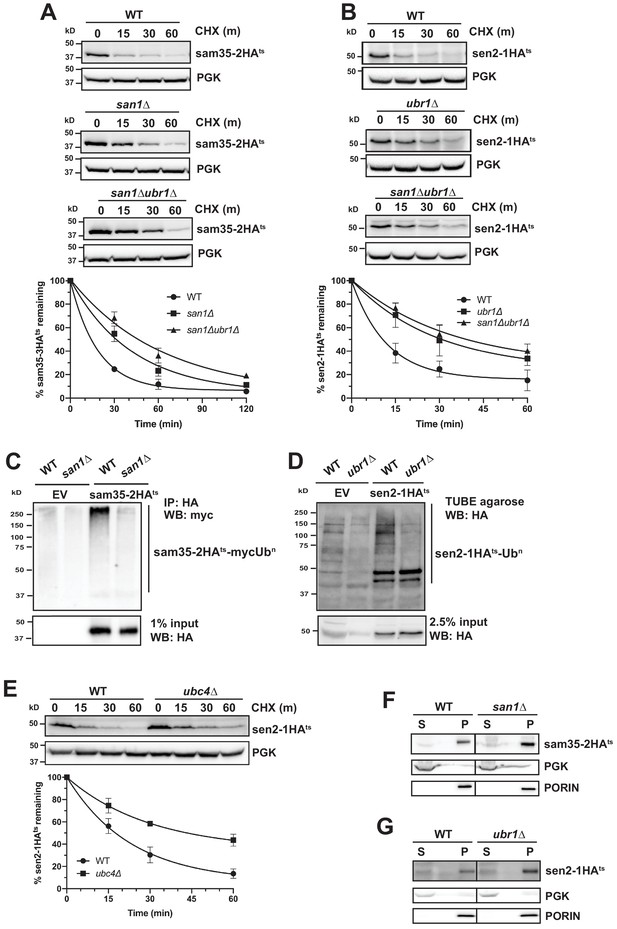
Distinct E3 ubiquitin ligases act on sam35-2HAts and sen2-1HAts.
(A) CHX chase for the indicated times at 37°C assessing the turnover of sam35-2HAts (pMM157) in WT (BY4741), san1Δ, and san1Δ ubr1Δ (SM5770) cells. Proteins were detected by immunoblotting. Graphed below is the mean and SD of the PGK-normalized HA signal at each time point for three biological replicates. (B) CHX chase for the indicated times at 37°C assessing the turnover of sen2-1HAts (pMM160) in WT (BY4741), ubr1Δ (yMM149), and san1Δ ubr1Δ (SM5770) cells, as in A. (C) Ubiquitination of sam35-2HAts was assessed by IP with anti-HA agarose of lysates from WT (BY4741) and san1Δ cells expressing myc-Ub (pSM3666) and either empty vector (EV; pRS315) or sam35-2HAts (pMM157), followed by immunoblotting with c-myc antibody. 1% of IP input lysate was reserved and analyzed for sam35-2HAts by immunoblotting. (D) Ubiquitination of sen2-1HAts was assessed by IP of lysates from WT and ubr1Δ strains expressing either EV (pRS315) or sen2-1HAts (pMM160) using TUBE agarose, followed by immunoblotting with HA antibody. 2.5% of the TUBE input lysate was reserved and analyzed by immunoblotting for sen2-1HAts. (E) CHX chase for the indicated times at 37°C assessing the turnover of sen2-1HAts (pMM160) in ubc4Δ cells compared to isogenic WT (BY4741). (F) Lysates from the WT and san1Δ strains used in A expressing sam35-2HAts (pMM157) were fractionated at 12,000xg at 37°C into mitochondrial pellets (P) and post-mitochondrial supernatants (S). Fractions were subject to immunoblotting with antibodies to HA, PGK, and PORIN. (G) Lysates from the WT and ubr1Δ strains used in B expressing sen2-1HAts (pMM160) were fractionated at 12,000xg at 37°C into mitochondrial pellets (P) and post-mitochondrial supernatants (S). Fractions were subject to immunoblotting with antibodies to HA, PGK, and PORIN.
-
Figure 3—source data 1
Quantifications of cycloheximide chases.
- https://cdn.elifesciences.org/articles/51065/elife-51065-fig3-data1-v2.xlsx
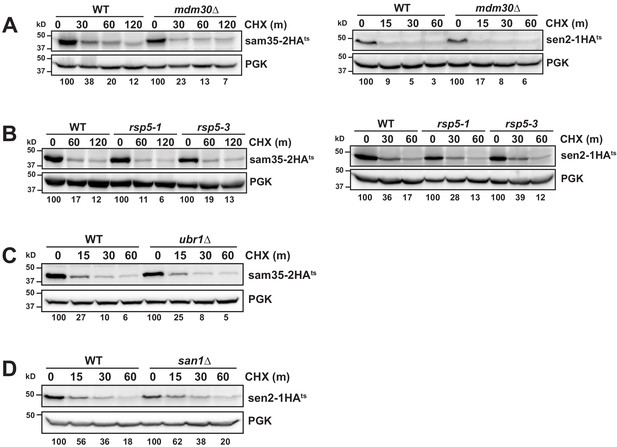
Distinct E3 ubiquitin ligases act on sam35-2HAts and sen2-1HAts.
(A–D) CHX chase for the indicated times at 37°C assessing the turnover of sam35-2HAts or sen2-1HAts (pMM157 or 160, respectively) in mdm30Δ (A); rsp5-1 and rsp5-3 (B); ubr1Δ (yMM149; C); or san1Δ (D) strains relative to isogenic WT strains. Proteins were detected by immunoblotting. Percentage of each ts- substrate remaining is labeled for each time point under blots.
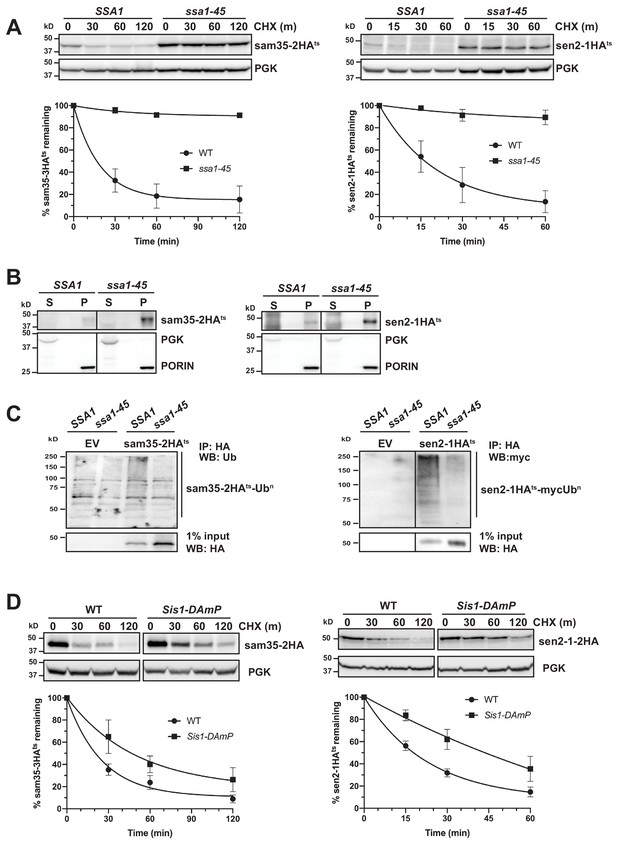
Cytosolic chaperones are required for the degradation of sen2-1HAts and sam35-2HAts.
(A) CHX chase for the indicated times at 37°C assessing the turnover of sam35-2HAts (pMM231) and sen2-1HAts (pMM234) in WT (SSA1) and ssa1-45ts cells. Proteins were detected by immunoblotting. Graphed below is the mean and SD of the PGK-normalized HA signal at each time point for three biological replicates. (B) Lysates from WT and ssa1-45ts strains expressing sam35-2HAts (pMM231) or sen2-1HAts (pMM234) were fractionated at 12,000xg at 37°C into mitochondrial pellets (P) and post-mitochondrial supernatants (S). Fractions were subject to immunoblotting with antibodies to HA, PGK, and PORIN. (C) Ubiquitination of sam35-2HAts and sen2-1HAts was assessed by IP with anti-HA agarose from lysates from WT and ssa1-45ts cells expressing either empty vector (EV; pRS316), sam35-2HAts (pMM231), or sen2-1HAts (pMM234), followed by immunoblotting with either ubiquitin or c-myc antibody. 1% of IP input lysate was reserved and analyzed by immunoblotting. (D) CHX chase for the indicated times at 37°C assessing the turnover of sam35-2HAts (pMM157) and sen2-1HAts (pMM160) in WT (yTHC) and Sis1-DAmP cells treated with 10 μg/mL doxycycline for 18 hr at 25°C to decrease Sis1 mRNA abundance prior to the addition of CHX.
-
Figure 4—source data 1
Quantifications of cycloheximide chases.
- https://cdn.elifesciences.org/articles/51065/elife-51065-fig4-data1-v2.xlsx
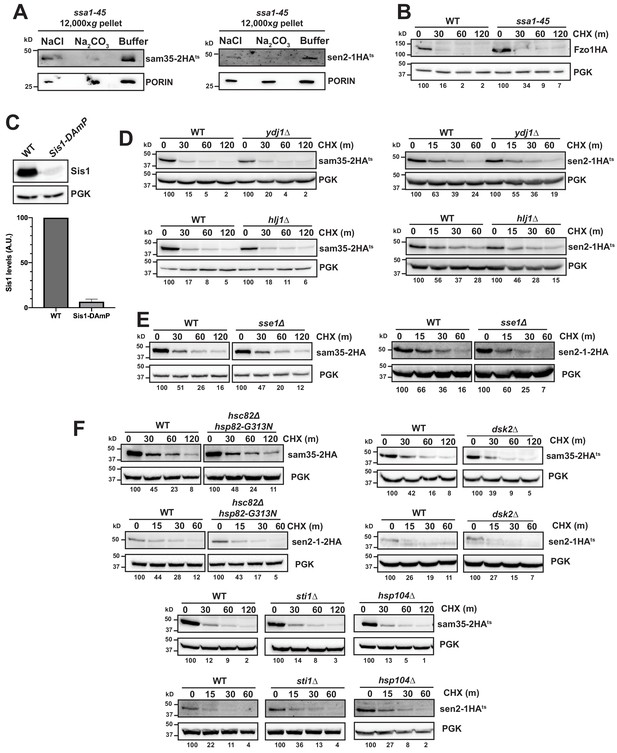
Cytosolic chaperones are required for the degradation of sen2-1HAts and sam35-2HAts.
(A) Sodium carbonate (Na2CO3) extraction of sam35-2HAts or sen2-1HAts (pMM157 and 160, respectively) from the 12,000xg mitochondrial pellet of ssa1-45 cells. Control treatments of sodium chloride (NaCl) and buffer are shown for comparison. Proteins were detected with immunoblotting with HA and PORIN antibodies. (B) CHX chase for the indicated times at 37°C assessing the turnover of Fzo1HA (pADH1-Fzo1pHA) in SSA1 WT and ssa1-45 mutant strains. Proteins were detected by immunoblotting. Percentage of each ts- substrate remaining is labeled for each time point under blots. (C) Sis1 protein levels in WT (yTHC) and Sis1-DAmP cells treated with 10 μg/mL doxycycline for 18 hr at 25°C were quantified and normalized to PGK levels. The mean and SD were graphed (N = 3). (D) CHX chase for the indicated times at 37°C assessing the turnover of sam35-2HAts or sen2-1HAts (pMM157 or 160, respectively) in ydj1Δ and hlj1Δ. (E, F) CHX chase for the indicated times at 37°C assessing the turnover of sam35-2HAts or sen2-1HAts (pMM157 or 160, respectively) in sse1Δ (E) or chaperone mutant strains (hsc82Δ hsp82-G313N, sti1Δ, hsp104Δ, dsk2Δ; F), relative to their isogenic WT strains.
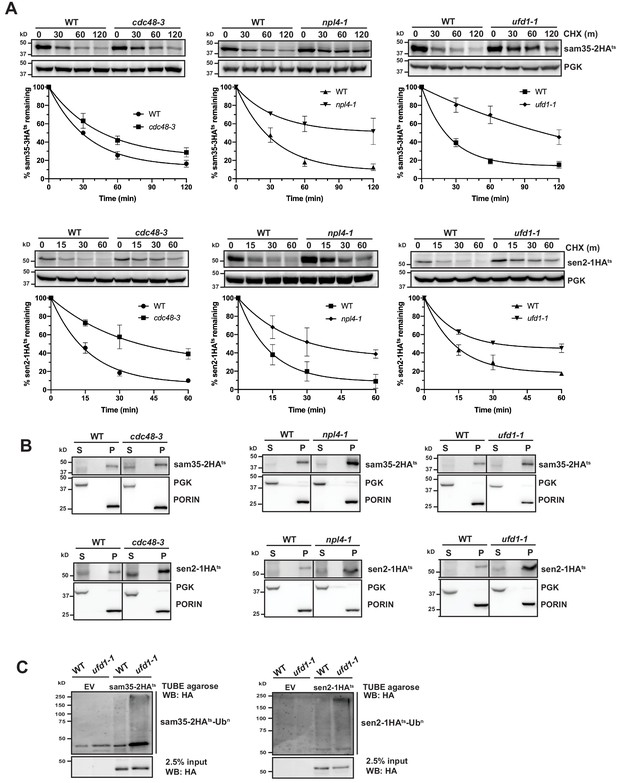
The Cdc48-Npl4-Ufd1 complex is required for degradation of MAD substrates.
(A) CHX chase for the indicated times at 37°C assessing the turnover of sam35-2HAts (pMM157) or sen2-1HAts (pMM160) in cdc48-3, npl4-1, and ufd1-1 mutant strains compared to isogenic WT strains. Proteins were detected by immunoblotting. Graphed below is the mean and SD of the PGK-normalized HA signal at each time point for three biological replicates. (B) Lysates from the strains used in A expressing sam35-2HAts (pMM157) or sen2-1HAts (pMM160) were fractionated at 12,000xg at 37°C into mitochondrial pellets (P) and post-mitochondrial supernatants (S). Fractions were subject to immunoblotting with antibodies to HA, PGK, and PORIN. (C) Ubiquitination of sam35-2HAts and sen2-1HAts was assessed by IP from lysates of the ufd1-1 mutant and isogenic WT strain used in A using TUBE agarose, followed by immunoblotting with HA antibody. 2.5% of the TUBE input lysate was reserved and analyzed by immunoblotting.
-
Figure 5—source data 1
Quantifications of cycloheximide chases.
- https://cdn.elifesciences.org/articles/51065/elife-51065-fig5-data1-v2.xlsx
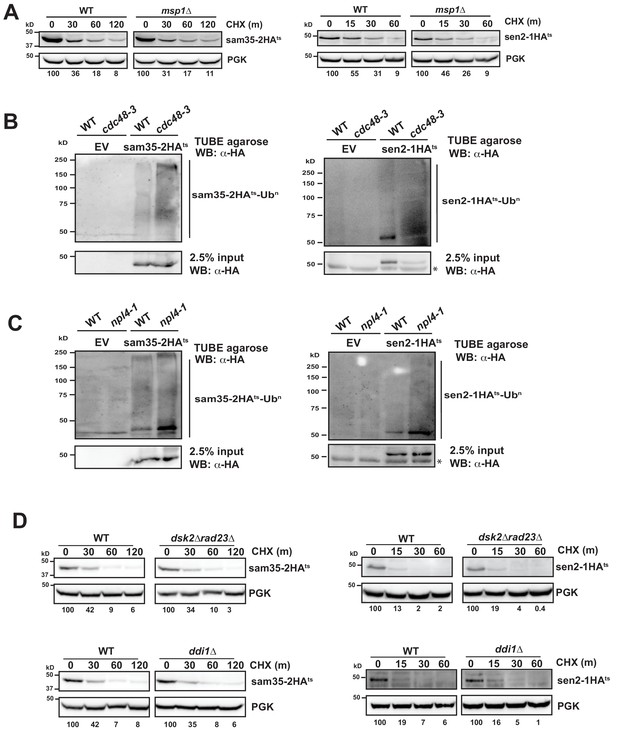
The Cdc48-Npl4-Ufd1 complex is required for degradation of MAD substrates.
(A) CHX chase for the indicated times at 37°C assessing the turnover of sam35-2HAts or sen2-1HAts (pMM157 or 160, respectively) in WT (BY4741) and msp1Δ cells. Proteins were detected by immunoblotting. Percentage of each ts- substrate remaining is labeled for each time point under blots. (B, C) Ubiquitination of sam35-2HAts and sen2-1HAts was assessed by immunoprecipitation (IP) using tandem ubiquitin-binding entities (TUBE) agarose from cdc48-3 (B) and npl4-1 (C) mutant and isogenic WT lysates expressing EV (pRS315), sam35-2HAts (pMM157), or sen2-1HAts (pMM160), followed by immunoblotting with HA antibody. 2.5% of the TUBE input lysate was reserved and analyzed by immunoblotting. Asterisk indicates a cross-reactive band seen in some exposures. (D) CHX chase for the indicated times at 37°C assessing the turnover of sam35-2HAts or sen2-1HAts (pMM157 or 160, respectively) in WT (BY4741), dsk2Δ rad23Δ (SM5186), or ddi1Δ cells.
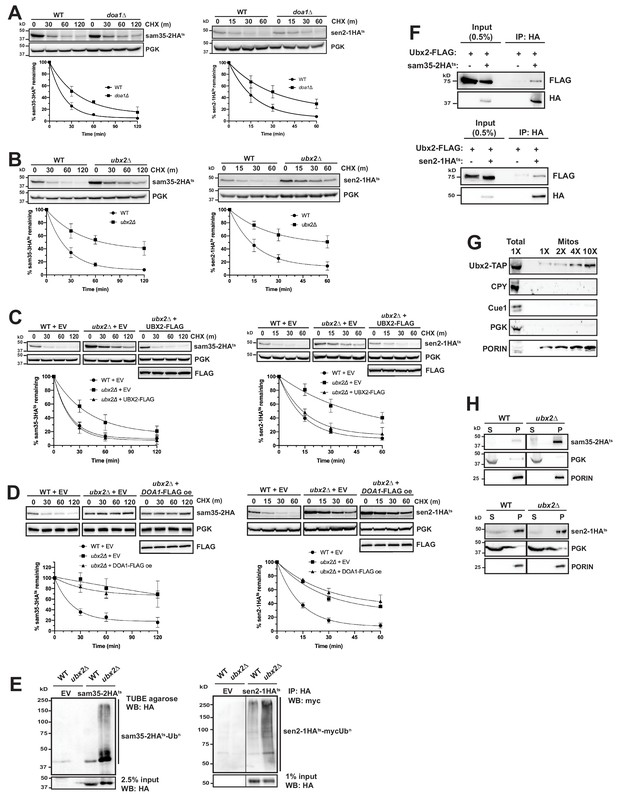
The Cdc48 co-factors Ubx2 and Doa1 are implicated in MAD.
(A) CHX chase for the indicated times at 37°C assessing the turnover of sam35-2HAts (pMM157) and sen2-1HAts (pMM160) in WT (BY4741) and doa1Δ cells (yJS208). Proteins were detected by immunoblotting. Graphed below is the mean and SD of the PGK-normalized HA signal at each time point for three biological replicates. (B) CHX chase as in A for the indicated times at 37°C assessing the turnover of sam35-2HAts (pMM157) and sen2-1HAts (pMM160) in WT (BY4741) and ubx2Δ cells (yJS155). (C) CHX chase as in A for the indicated times at 37°C assessing the turnover of sam35-2HAts (pMM157) and sen2-1HAts (pMM160) in WT (BY4741) and ubx2Δ (yJS155) cells co-expressing either empty vector (EV; pRS315) or CEN Ubx2-FLAG (pMM242). (D) CHX chase as in A for the indicated times at 37°C assessing the turnover of sam35-2HAts (pMM231) or sen2-1HAts (pMM234) in WT (BY4741) cells or ubx2Δ (yJS155) cells expressing either EV (pRS315) or Doa1-FLAG (pMM254) from a high copy 2μ plasmid. (E) Ubiquitination of sam35-2HAts and sen2-1HAts was assessed by IP using TUBE agarose or anti-HA agarose from ubx2Δ (yJS155) and WT (BY4741) lysates expressing EV (pRS315), sam35-2HAts (pMM157), or sen2-1HAts (pMM160), followed by immunoblotting with HA or c-myc antibody. 2.5% or 1% of the IP input lysate was reserved and analyzed by immunoblotting. (F) Co-IP of Ubx2-FLAG (pMM242) with sam35-2HAts or sen2-1HAts (pMM231 and 234, respectively) from pre1-1 pre2-2 (WCG4-11/21a) cells was assessed by immunoblotting with the indicated antibodies. IP of Ubx2-FLAG from cells co-expressing EV (pRS316) in place of HA-tagged substrates and 0.5% of the input lysate are shown for comparison. (G) Lysate (‘Total’) and increasing amounts of mitochondria purified by 12,000xg and sucrose gradient fractionation (‘Mitos’) from Ubx2-TAP-expressing cells were examined by immunoblotting with the indicated antibodies. (H) Lysates from WT (BY4741) and ubx2Δ (yJS155) cells expressing sam35-2HAts or sen2-1HAts (pMM157 and 160, respectively) were fractionated at 12,000xg at 37°C into mitochondrial pellets (P) and post-mitochondrial supernatants (S). Fractions were subject to immunoblotting with antibodies to HA, PGK, and PORIN.
-
Figure 6—source data 1
Quantifications of cycloheximide chases.
- https://cdn.elifesciences.org/articles/51065/elife-51065-fig6-data1-v2.xlsx

The Cdc48 co-factors Ubx2 and Doa1 are required for MAD.
(A) CHX chase for the indicated times at 37°C assessing the turnover of sam35-2HAts or sen2-1HAts (pMM157 or 160, respectively) in vms1Δ compared to isogenic WT (BY4741). Proteins were detected by immunoblotting. Percentage of each ts- substrate remaining is labeled for each time point under blots. (B) CHX chase for the indicated times at 37°C assessing the turnover of sam35-2HAts or sen2-1HAts (pMM157 or 160, respectively) in the indicated UBX protein deletion strains compared to isogenic WT (BY4741). (C) CHX chase for the indicated times at 37°C assessing the turnover of SAM35HA or SEN2HA (pMM158 or 159, respectively) in ubx2Δ (yJS155) compared to isogenic WT (BY4741). (D) Co-IP of Ubx2-FLAG (pMM242) with SAM35HA, sam35-2HAts, SEN2HA, or sen2-1HAts (pMM232, pMM231, pMM233, and 234, respectively) from pre1-1 pre2-2 (WCG4-11/21a) cells was assessed by immunoblotting with the indicated antibodies. IP of Ubx2-FLAG from cells co-expressing EV (pRS316) in place of HA-tagged substrates and 0.5% of the input lysate are shown for comparison. (E) CHX chase for the indicated times at 30°C assessing the turnover of Fzo1HA (pADH1-Fzo1pHA) in WT (BY4741) and doa1Δ (yJS208) strains. (F) CHX chase for the indicated times at 30°C assessing the turnover of Fzo1HA (pADH1-Fzo1pHA) in WT (BY4741) and ubx2Δ (yJS155) cells co-expressing either EV (pRS315) or Ubx2-FLAG (pMM242). (G) Co-IP of Ubx2-FLAG (pMM242) with Fzo1HA (pADH1-Fzo1pHA) from pre1-1 pre2-2 (WCG4-11/21a) cells was assessed by immunoblotting with the indicated antibodies. Negative control of IP of Ubx2-FLAG from cells co-expressing empty vector (pRS316) in place of HA-tagged substrates is shown, as is 0.5% of the input lysate. (H) Ubiquitination of Fzo1HA was assessed by IP using TUBE agarose from ubx2Δ (yJS155) and WT (BY4741) lysates expressing EV (pRS316) or Fzo1HA (pADH1-Fzo1pHA) followed by immunoblotting with HA antibody. 2.5% of the TUBE input lysate was reserved and analyzed by immunoblotting. (I) Microscopy analysis of immobilized Ubx2-GFP cells co-expressing either mtRFP (pMD12) to label mitochondria or SEC63-RFP (pSM1959) to label the ER. Some regions of co-localization are highlighted with yellow arrows. ‘Merge’ of GFP (green) and RFP (magenta) channels and differential interference contrast (DIC) are shown; scale bar = 10 um. (J) CHX chase for the indicated times at 37°C assessing the turnover of sam35-2HAts or sen2-1HAts (pMM157 or 160, respectively) in WT (BY4741) and mmm1Δ, mdm12Δ, mdm34Δ, and mdm10Δ strains. (K, L) CHX chase for the indicated times at 37°C assessing the turnover of sam35-2HAts or sen2-1HAts (pMM157 or 160, respectively) in a ubc6Δubc7Δ (K; SM5364) or doa10Δhrd1Δ (L, SM5360) strains relative to their isogenic WT strain (BY4741). (M) CHX chase for the indicated times at 30°C assessing the turnover of Fzo1HA (pADH1-Fzo1pHA) in WT (BY4741) and mmm1Δ, mdm12Δ, mdm34Δ, and mdm10Δ strains. (N) Lysates from WT (BY4741) and ubx2Δ (yJS155) cells expressing Fzo1HA (pMM190) were fractionated at 12,000xg at 37°C into mitochondrial pellets (P) and post-mitochondrial supernatants (S). Fractions were subject to immunoblotting with antibodies to HA, PGK, and PORIN.
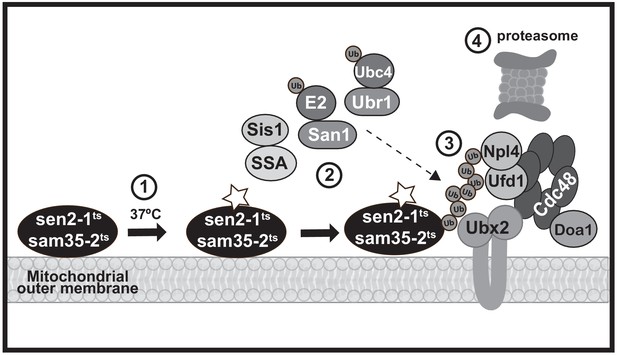
A model MAD QC pathway based on the present study.
When the temperature is increased to 37°C, the peripheral MOM ts- proteins sam35-2HAts and sen2-1HAts become non-functional (denoted by a star) yet remain at the mitochondrial outer membrane (step 1). They are recognized as quality control substrates and ubiquitinated (step 2), which requires cytosolic chaperones (Ssa1 and Sis1) and the ubiquitin ligase San1 (for sam35-2HAts) or Ubr1 and the ubiquitin conjugating enzyme Ubc4 (for sen2-1HAts). Once ubiquitinated, the Cdc48-Npl4-Ufd1 unfoldase, along with its co-factor Doa1 and a mitochondria-localized pool of its co-factor Ubx2 (step 3), act to direct them to the 26S proteasome for degradation (step 4).
Additional files
-
Supplementary file 1
Key resources table.
- https://cdn.elifesciences.org/articles/51065/elife-51065-supp1-v2.docx
-
Transparent reporting form
- https://cdn.elifesciences.org/articles/51065/elife-51065-transrepform-v2.docx





