A gut bacterial amyloid promotes α-synuclein aggregation and motor impairment in mice
Figures
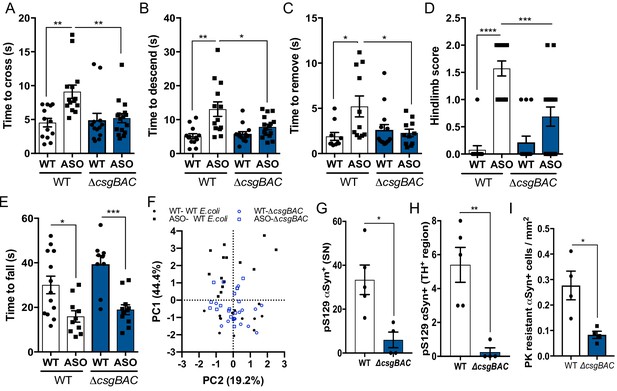
Mono-colonization with curli-producing gut bacteria enhances αSyn pathophysiology germ-free wild-type BDF1 (WT) and Thy1-αSyn (ASO) mice were mono-colonized with wild-type E. coli MC4100 (WT) or an isogenic curli-deficient strain (ΔcsgBAC) at 5–6 weeks of age.
Motor function was assessed at 12–13 weeks of age by quantifying (A), beam traversal time, (B), pole descent time, (C), nasal adhesive removal time, (D), hindlimb clasping score, (E), wirehang time. (F), Principal component analysis of compiled motor scores from tests in (A–E). Quantification of (G, H) pS129αSyn and (I) proteinase K–resistant αSyn by immunofluorescence microscopy in the substantia nigra and midbrain. n = 13–16 (A–F), n = 4.5 (G–I). Points represent individuals, bars represent the mean and standard error. Data analyzed by one-way ANOVA with Tukey post-hoc test (A–E), and two-tailed t-test for (G–I). *p≤0.05; **p≤0.01; ***p≤0.001; ****p≤0.0001. Motor data are compiled from three independent cohorts.
-
Figure 1—source data 1
Source data and statistical analysis.
- https://cdn.elifesciences.org/articles/53111/elife-53111-fig1-data1-v1.xlsx
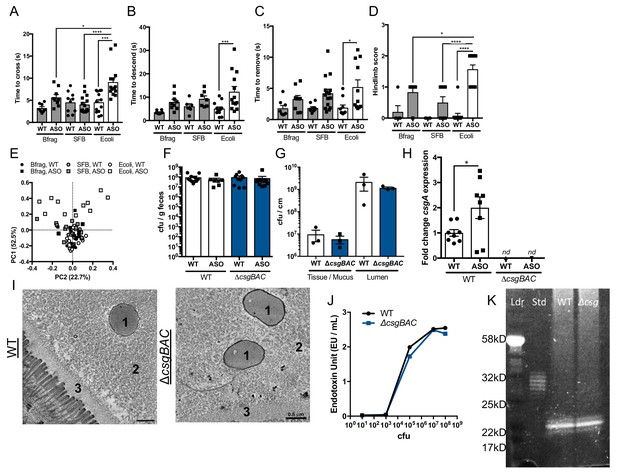
E. coli alters motor deficits, and CsgA does not influence colonization, inflammatory capacity, or dopamine production Germ-free (GF) wild-type (WT) or Thy1- αSyn (ASO) mice were mono-colonized with either Bacteroides fragilis (Bfrag), segmented filamentous bacteria (SFB), or Escherichia coli (Ecoli).
Motor function assessed by (A) beam crossing time, (B) pole descent time, (C) adhesive removal time, or D) hindlimb reflex score.( E,) PCA analysis of scores in (A–D). (F–N) GF WT and ASO mice were mono-colonized with curli-sufficient E. coli (WT) or curli-deficient E. coli (ΔcsgBAC). (F) Fecal bacterial burden determined by plate culture. (G) Bacterial burden of tissue/mucus fractions and luminal fractions of proximal colon. (H) csgA expression within fecal contents determined by qRT-PCR. (I) Representative TEM of bacterial localization in the proximal colon. 1- Bacteria, 2-mucus layer, 3-glycocalyx. (J) Quantification of endotoxin as determined by LAL assay. (K) Pro-Q Emerald stain (LPS) of SDS-PAGE separated cell lysates, Ldr- ladder, Std- LPS standard, WT- wild-type, Δcsg- ΔcsgBAC. n = 7–14 (A–E); n = 7–9 (F, H); n = 3 (G). Points represent individuals, bars represent the mean and standard error. Data analyzed by one-way ANOVA with Tukey post-hoc test (A–D). *p≤0.05; **p≤0.01; ***p≤0.001; ****p≤0.0001. In (A–D), data are compiled from two independent cohorts for B. fragilis and SFB, compared to E. coli mono-colonized cohorts used in Figure 1.
-
Figure 1—figure supplement 1—source data 1
Source data and statistical analysis.
- https://cdn.elifesciences.org/articles/53111/elife-53111-fig1-figsupp1-data1-v1.xlsx
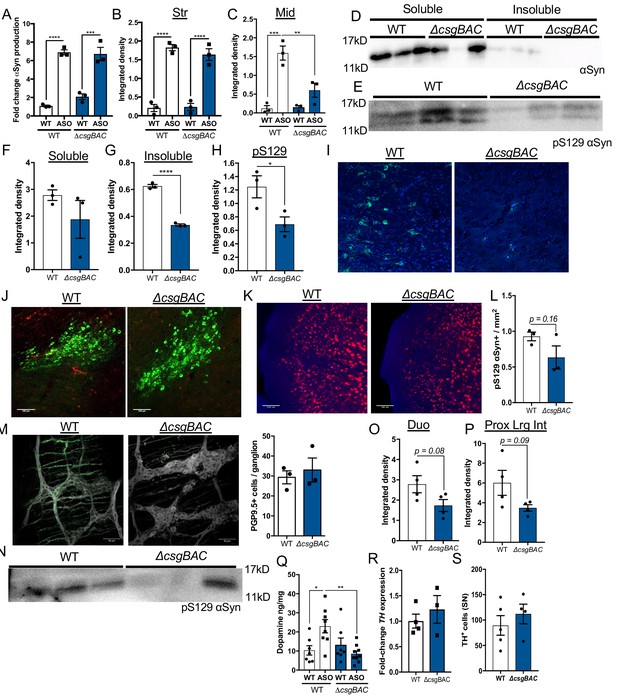
Mono-colonization with curli-sufficient bacteria induce increased αSyn-dependent pathology Germ-free (GF) wild-type (WT) or Thy1-αSyn (ASO) animals were mono-colonized with curli-sufficient E. coli (WT) or curli-deficient E. coli (ΔcsgBAC).
(A) Total αSyn in whole brain lysates quantified by ELISA. Quantification of insoluble αSyn fibrils in the (B) striatum and (C) midbrain by dot blot assay. Midbrain homogenates of ASO animals were (D) fractionated with Triton X-100 and SDS-PAGE, and subsequently probed for alpha-synuclein (αSyn) via western blot, or (E) left unfractionated and assessed by western blot for phospho-Serine129 αSyn (pS129). (F–H) Quantification of western blots by integrated density. Representative images from of mono-colonized ASO mice of the (I) midbrain stained for proteinase K-resistant αSyn (green), (J) substantia nigra stained for tyrosine hydroxylase (TH, green) and pS129 (red), and (K) frontal cortex stained for pS129 (red) and quantified in (L). (M) Representative images of the myenteric plexus within proximal large intestine of mono-colonized ASO animals stained for PGP9.5 (white) and pS129αSyn (green,), and counts of PGP9.5+ cells bodies per ganglia. (N) Proximal large intestine homogenates of mono-colonized ASO animals were assessed by SDS-PAGE and analyzed by western blot for pS129αSyn. Quantification of insoluble αSyn fibrils in the (O) duodenum and (P) proximal large intestine by dot blot assay. (Q) Total striatal dopamine quantified by ELISA. (R) TH expression in the midbrain by qPCR. (S) Quantification of TH+ cells in representative sections of the SN. n = 3 (A–H, L), n = 4 (O, P), n = 7–9 (Q), n = 3–4 (R), n = 4–5 (S). Full-length immunoblots available in associated Source Data file. Points represent individuals, bars represent the mean and standard error. Data analyzed by one-way ANOVA with Tukey post-hoc test for A-C, Q, or two-tailed t-test for F-H, L, O, P, R, S. *p≤0.05; **p≤0.01; ***p≤0.001; ****p≤0.0001. Scale bars = 100 μm.
-
Figure 1—figure supplement 2—source data 1
Source data and statistical analysis.
- https://cdn.elifesciences.org/articles/53111/elife-53111-fig1-figsupp2-data1-v1.xlsx
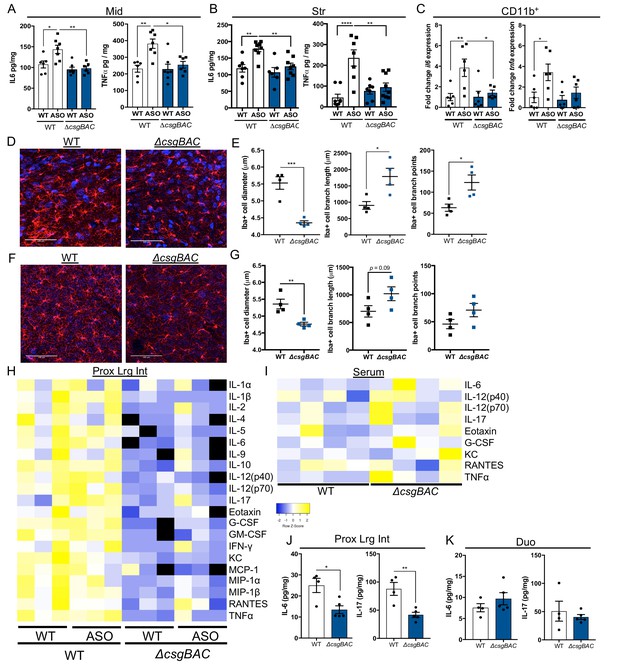
Mono-colonization with curli-sufficient bacteria induces altered inflammatory responses in the brain and intestine.
Germ-free (GF) wild-type (WT) or Thy1-αSyn (ASO) animals were mono-colonized with either wild-type, curli-sufficient E. coli (WT) or curli-deficient E. coli (ΔcsgBAC). Quantification of IL-6 and TNFα by ELISA from homogenates derived from the (A) midbrain or (B) striatum. (C) CD11b+ cells enriched from total brains and gene expression of il6 and tnfa quantified by qRT-PCR, relative to gapdh. Representative thin sections of (D) substantia nigra and (F) striatum derived from ASO mice stained for Iba1 (microglia). 3D cellular reconstructions of microglia generated and morphological characteristics quantified of (E) substantia nigra- and (G) striatum—resident microglia. Multiplex ELISA performed for cytokines and chemokines derived from (H) homogenates derived from proximal large intestine tissue of indicated animals or (I) serum of ASO animals. Quantification of IL-6 and IL-17 by ELISA from tissue homogenates of the (J) proximal large intestine or (K) duodenum derived from ASO animals. n = 6–7 (A–C), n = 4 (D–G) (averaged from 20 to 40 cells for diameters, or 5–7 cells for branching), n = 3 (H), n = 4–5 (J, K). Points represent individuals, bars represent the mean and standard error. Data analyzed by one-way ANOVA with Tukey post-hoc test for A–C), or two-tailed t-test for E, J) and K). *p≤0.05; **p≤0.01; ***p≤0.001; ****p≤0.0001. Scale bars = 100 μm.
-
Figure 1—figure supplement 3—source data 1
Source data and statistical analysis.
- https://cdn.elifesciences.org/articles/53111/elife-53111-fig1-figsupp3-data1-v1.xlsx
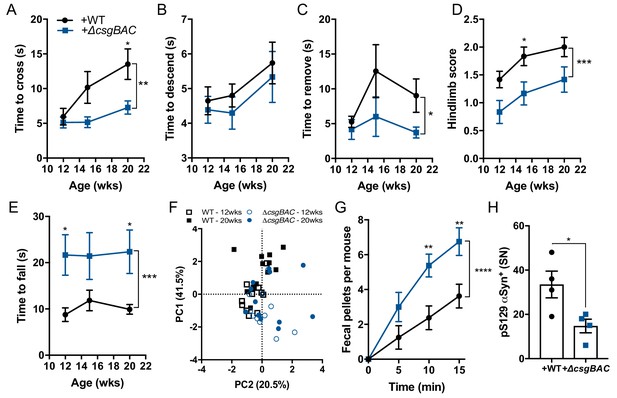
Curli biosynthesis within a complex microbiome contributes to motor and GI deficits Germ-free Thy1-αSyn (ASO) mice were colonized with fecal microbes derived from healthy human at 5–6 weeks of age, and concurrently supplemented with either wild-type E. coli MC4100 (+WT) or a curli-deficient strain (+ΔcsgBAC).
Motor function was tested longitudinally at 12, 15, and 20 weeks of age in the (A) beam traversal, (B) pole descent, (C) adhesive removal, (D) hindlimb clasping score, (E) wirehang tests. (F) Principal component analysis of compiled motor scores from tests in (A–E). (G) Fecal output over a 15-min period observed at week 21 of age. Quantification of (H) pS129αSyn by immunofluorescence microscopy in the substantia nigra. n = 12 (A–F), n = 6 (G), n = 4 (H). Data points represent individuals, bars represent the mean and standard error. Time courses analyzed by two-way ANOVA, with Sidak post-hoc test for between group comparisons indicated above individual time points, and brackets indicating significance between colonization status. Data in (H) analyzed two-tailed t-test. *p≤0.05; **p≤0.01; ***p≤0.001; ****p≤0.0001. Motor data are compiled from two independent cohorts.
-
Figure 2—source data 1
Source data and statistical analysis.
- https://cdn.elifesciences.org/articles/53111/elife-53111-fig2-data1-v1.xlsx
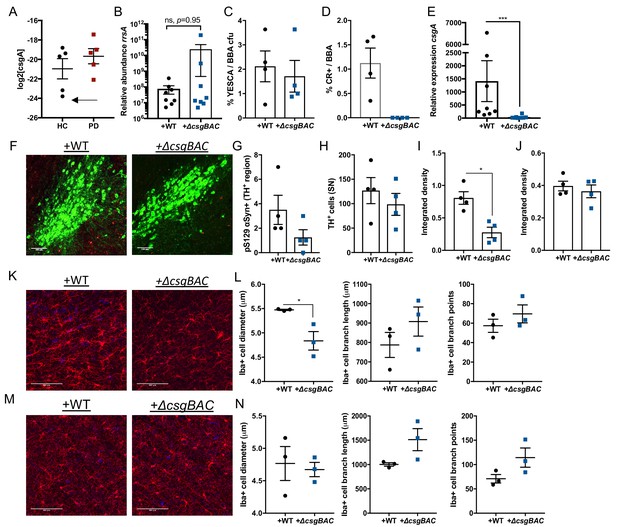
Amyloid-producing bacteria in humanized animals modulate microglia responses.
(A) Human samples from previous cohort (ENA Accession: PRJEB17694) were analyzed by PICRUSt to infer abundance of csgA encoded within each population, arrow indicates sample utilized for transplantation. Fecal pellets of Thy1-αSyn (ASO) mice receiving healthy-human derived fecal microbes enriched with either wild-type, curli-sufficient E. coli (WT) or curli-deficient E. coli (ΔcsgBAC) were analyzed by (B) qPCR for rrsA abundance relative to 16 s rRNA present in fecal bacterial DNA and ratios of (C) YESCA/blood agar aerobic colony forming units and (D) congo red-positive/blood agar colony forming units calculated. (E) qPCR analysis for csgA expression relative to rrsA in fecal bacterial RNA. (F) Representative images of the substantia nigra in humanized ASO mice stained for TH (green) and pS129 (red) and quantified in (G, H). Quantification of insoluble αSyn fibrils in the (I) midbrain and (J) striatum by dot blot assay. Representative thin sections of (K) substantia nigra and (M) striatum derived from ASO mice stained for Iba1 (microglia; red). 3D cellular reconstructions of microglia generated and morphological characteristics quantified of (L) substantia nigra- and (N) striatum—resident microglia. n = 5 (A), n = 8 (B, E), n = 4 (C–J), n = 3 (K–N) (averaged from 20 to 40 cells for diameters, or 5–7 cells for branching) Points represent individuals, bars represent the mean and standard error. Data in (B, E, I) analyzed by two-tailed Mann-Whitney test or t-test in L. *p≤0.05; ***p≤0.001. Scale bars = 100 μm.
-
Figure 2—figure supplement 1—source data 1
Source data and statistical analysis.
- https://cdn.elifesciences.org/articles/53111/elife-53111-fig2-figsupp1-data1-v1.xlsx
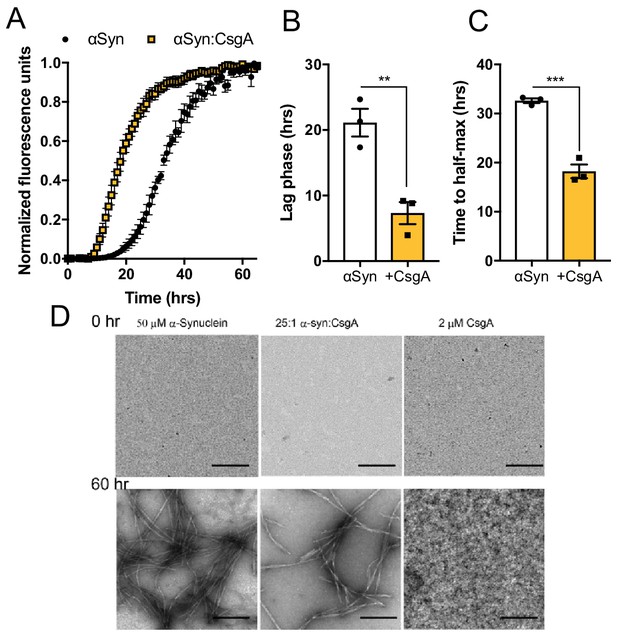
The bacterial amyloid protein, CsgA, accelerates αSyn fibrilization In vitro biophysical analysis with purified αSyn and CsgA proteins.
(A) Aggregation as measured by Thioflavin T fluorescence over time during αSyn amyloid formation alone or in the presence of CsgA monomers (25:1 molar ratio). Time to reach (B) exponential fibrilization/lag phase and (C) half-maximum from reactions in (A). (D) Representative transmission electron micrographs of αSyn or CsgA alone, or in combination, at 0 and 60 hr post-aggregation. n = 3 (A–C). Bars represent the mean and standard error. Data are analyzed by two-tailed, t-test. **p≤0.01; ***p≤0.001.
-
Figure 3—source data 1
Source data and statistical analysis.
- https://cdn.elifesciences.org/articles/53111/elife-53111-fig3-data1-v1.xlsx
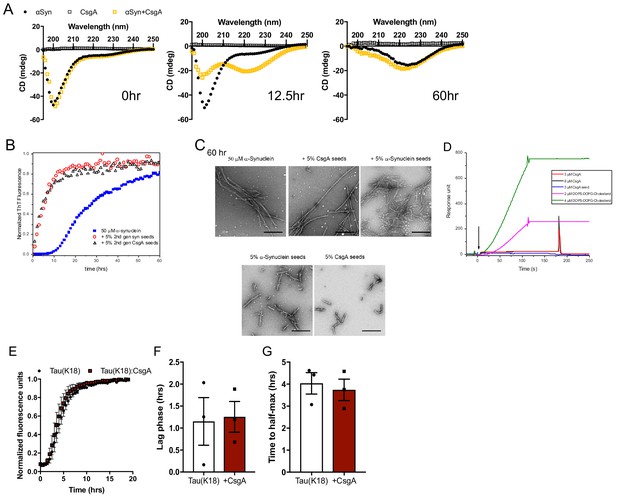
CsgA seeds αSyn aggregation and propagation in vitro.
(A) Circular dichroism spectroscopic analysis of αSyn fibrilization alone or in the presence of CsgA at 0, 12.5, and 60 hr post-aggregation. (B) Thioflavin T fluorescence during αSyn amyloid formation alone or in the presence of 5% seeds previously generated by addition of CsgA monomer to αSyn (as in Figure 2a) or αSyn alone. (C) Transmission electron micrograph of fibril structures generated by the addition of above seeds and of seeds themselves. (D) Surface plasmon resonance measurements of surface immobilized αSyn with additions of either CsgA monomer or seeds, or DOPS-DOPG cholesterol liposomes as positive control. (E) Amyloid aggregation as measured by Thioflavin T fluorescence with Tau alone or in the presence of CsgA monomers (25:1 molar ratio, red). Time to reach (F) exponential fibrilization/lag phase and (G) half-maximum from reactions in (E).
-
Figure 3—figure supplement 1—source data 1
Source data and statistical analysis.
- https://cdn.elifesciences.org/articles/53111/elife-53111-fig3-figsupp1-data1-v1.xlsx
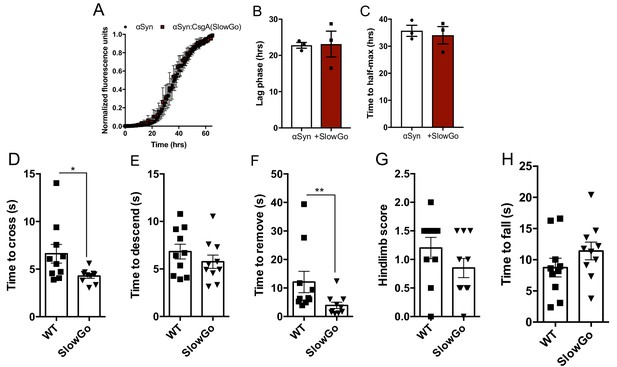
CsgA seeds αSyn aggregation in vitro and in vivo through native amyloidogenic properties.
(A) Amyloid aggregation as measured by Thioflavin T fluorescence with αSyn alone or in the presence of non-amyloidogenic CsgA monomers (SlowGo, 25:1 molar ratio, red). Time to reach (B) exponential fibrilization/lag phase and (C) half-maximum from reactions in (A). (D–H) Germ-free Thy1-αSyn mice (ASO) were mono-colonized with E. coli producing either wild-type CsgA (WT) or SlowGo (SlowGo), and motor function assessed at 12-13 weeks of age in (D) beam traversal, (E) pole descent, (F) adhesive removal, (G) hindlimb reflexes, and (H) wirehang. Points represent individuals, bars represent the mean and standard error, n = 10–11 (D–H). Data in (D–H) analyzed by two-tailed Mann-Whitney test. *p≤0.05; **p≤0.01.
-
Figure 3—figure supplement 2—source data 1
Source data and statistical analysis.
- https://cdn.elifesciences.org/articles/53111/elife-53111-fig3-figsupp2-data1-v1.xlsx
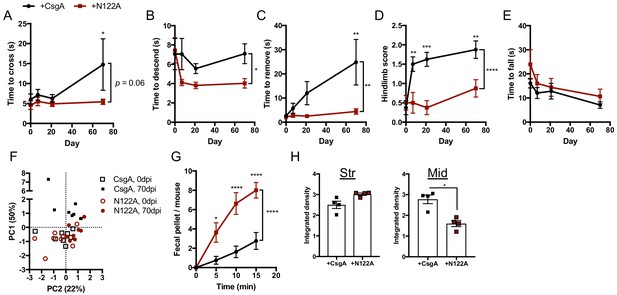
Intestinal injection of amyloidogenic curli promotes progressive motor dysfunction.
Conventionally-raised Thy1-αSyn (ASO) mice were injected intestinally with 30 μg of synthetic CsgA hexamer (CsgA; N-QYGGNN-C) or non-amyloidgenic peptide (N122A; N-QYGGNA-C). Motor function assessed overtime at 0, 7, 21, and 70 days post-injection in the (A) beam traversal, (B) pole descent, (C) adhesive removal, (D) hindlimb clasping score, and (E) wirehang tests. (F) Principal component analysis of compiled motor scores of ASO mice. (G) Fecal output observed at day 70 in ASO mice. (H) Quantification of insoluble αSyn fibrils in the striatum and ventral midbrain by dot blot assay. n = 8 (A–G), n = 4 (H). Points represent individuals, bars represent the mean and standard error. Time courses analyzed by two-way ANOVA, with Sidak post-hoc test for between group comparisons indicated above individual time points, and brackets indicating significance between treatments. Data in (H) analyzed by two-tailed Mann-Whitney test. *p≤0.05; **p≤0.01; ***p≤0.001; ****p≤0.0001.
-
Figure 3—figure supplement 3—source data 1
Source data and statistical analysis.
- https://cdn.elifesciences.org/articles/53111/elife-53111-fig3-figsupp3-data1-v1.xlsx
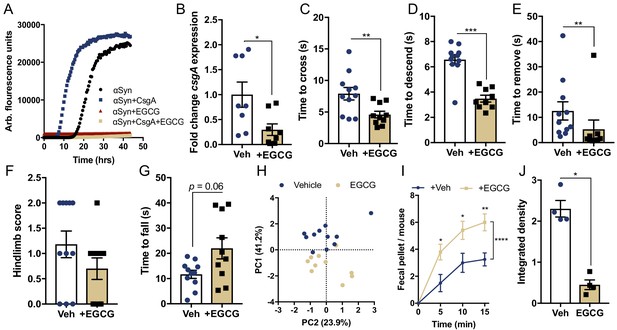
Curli-driven pathophysiology in mice requires functional amyloid formation.
(A), Representative in vitro αSyn aggregation measured by Thioflavin T fluorescence during αSyn amyloid formation alone or in the presence of CsgA (25:1 molar ratio), with and without EGCG (50 μM) treatment. (B–H) Germ-free Thy1-αSyn mice (ASO) were mono-colonized with WT E. coli at 5–6 weeks of age, and given water alone (Vehicle: Veh) or treated with EGCG ad lib in drinking water (+EGCG). (B) RNA was extracted from fecal pellets and csgA expression quantified by qRT-PCR, relative to rrsA. Motor function was assessed at 15–16 weeks of age by quantifying (C) beam traversal time, (D) pole descent time, (E) nasal adhesive removal time, (F) hindlimb clasping score, (G) wirehang tests. (H) Principal component analysis of compiled motor scores from tests in (C–G). (I) Fecal output over a 15 min period. (J) Quantification of insoluble αSyn fibrils in the ventral midbrain by dot blot assay. n = 8 (B); n = 10–11 (C–G); n = 4–5 (I, J). Points represent individuals, bars represent the mean and standard error. Data analyzed by two-tailed Mann-Whitney for B–G, (J) and two-way ANOVA with Sidek’s post-hoc test for I with between group comparisons indicated above individual time points, and brackets indicating significance between treatment status. *p≤0.05; **p≤0.01; ***p≤0.001; ****p≤0.0001. Motor data compiled from two independent cohorts.
-
Figure 4—source data 1
Source data and statistical analysis.
- https://cdn.elifesciences.org/articles/53111/elife-53111-fig4-data1-v1.xlsx

Inhibition of functional amyloid formation dampens progressive pathophysiology.
(A) Wild-type E. coli was inoculated into brain-heart infusion broth, containing indicated concentrations of EGCG. Cultures were incubated, with orbital shaking and growth was measured by optical density at 600 nm over time. (B) Crystal violet staining of wild-type E. coli biofilm at 4 days in static culture, with indicated concentrations of epigallocatechin gallate (EGCG); assessed by optical density (OD). (C–R) Germ-free ASO mice were mono-colonized with wild-type E. coli and treated with water alone (Vehicle, Veh) or given EGCG ad lib in drinking water (+EGCG). (C) Fecal pellets were cultured on YESCA agar to determine total viable E. coli. Motor function was assessed at 10, 12, and 15 weeks of age by quantifying (D) beam traversal time, (E) pole descent time, (F) nasal adhesive removal time, (G) hindlimb reflex score, (H) wirehang tests. (I) Principal component analysis of compiled motor scores. (J) Quantification of insoluble αSyn fibrils in the striatum by dot blot assay. (K) Representative images of the substantia nigra in humanized ASO mice stained for TH (green) and pS129 (red) and quantified in (L–N). Representative thin sections of (O) substantia nigra and (Q) striatum were stained for Iba1 (microglia; red) and morphological characteristics of (P) substantia nigra- and (r) striatum-resident microglia quantified. n = 3 (A, O–R) (averaged from 20 to 40 cells for diameters, or 5–7 cells for branching); n = 6–7 (B); n = 4–5 (C, J), n = 10–11 (D–I); n = 3–4 (K–N). Points represent individuals, bars represent the mean and standard error. Time courses analyzed by two-way ANOVA, with Sidak post-hoc test for between group comparisons indicated above individual time points, and brackets indicating significance between treatments. Data in (J) analyzed by two-tailed Mann-Whitney test and (L, P, R) analyzed by two-tailed t-test. *p≤0.05; **p≤0.01; ****p≤0.0001.
-
Figure 4—figure supplement 1—source data 1
Source data and statistical analysis.
- https://cdn.elifesciences.org/articles/53111/elife-53111-fig4-figsupp1-data1-v1.xlsx
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Strain, strain background (Mus musculus) | Thy1- alpha-synuclein (Line 61), BDF1 background | Rockenstein et al., 2002 | ASO (alpha-synuclein over-expressing) | With permission, UCSD |
| Strain, strain background (Escherichia coli) | Str. K12, MC4100 | Zhou et al., 2013 | WT (wild-type) | |
| Strain, strain background (Escherichia coli) | Str. K12, MC4100:ΔcsgBAC | Zhou et al., 2013 | ΔcsgBAC | |
| Strain (Escherichia coli) | Str. K12, MC4100: CsgA:Q49A/N54A/Q139A/N144A | Wang and Chapman, 2008 | SlowGo | |
| Antibody | Anti-alpha synuclein, mouse monoclonal | BD | Cat#: 610787 | (1:1000) |
| Antibody | Anti-pS129 alpha-synuclein, rabbit monoclonal | AbCam | Cat#: Ab51253 | (1:1000) |
| Antibody | Anti-tyrosine hydroxylase, mouse monoclonal | Millipore | Cat#: MAB318 | (1:1000) |
| Antibody | Anti-Iba1, rabbit | Wako | Cat#: 019–19741 | (1:1000) |
| Antibody | Anti-aggregated alpha-synuclein, rabbit polyclonal | AbCam | Cat#: MJFR-14-6-4-2 | (1:1000) |
| Antibody | Anti-mouse IgG-546, goat polyclonal | Life Technologies | Cat#: A-11003 | (1:1000) |
| Antibody | Anti-rabbit IgG-546, goat polyclonal | Life Technologies | Cat#: A-11010 | (1:1000) |
| Antibody | Anti-mouse IgG-488, goat polyclonal | Life Technologies | Cat#: A-11001 | (1:1000) |
| Antibody | Anti-rabbit IgG-HRP, goat polyclonal | Cell Signaling | Cat#: 7074 | (1:1000) |
| Peptide, recombinant protein | CsgA (N’-QYGGNN-C’) | Bio-Synthesis, Inc; Tükel et al., 2009 | CsgA | |
| Peptide, recombinant protein | N122A (N’QYGGNA-C’) | Bio-Synthesis, Inc; Tükel et al., 2009 | N122A | |
| Sequence-based reagent | 16S rRNA: 5’-TCCTACGGGAGGCAGCAGT-3’ and 5’-GGACTACCAGGGTATCTAATCCTGTT-3’ | IDT | qPCR primer | PrimerBank |
| Sequence-based reagent | rrsA: 5’-AGTGATAAACTGGAGGAGGTG-3‘ and 5’-GGACTACGACGCACTTTATGAG-3’ | IDT | qPCR primer | PrimerBank |
| Sequence-based reagent | csgA: 5’-ATGACGGTTAAACAGTTCGG-3’ and 5’-AGGAGTTAGATGCAGTCTGG-3’ | IDT | qPCR primer | PrimerBank |
| Sequence-based reagent | gapdh: 5’-CATGGCCTTCCGTGTTCCTA-3’ and 5’-CCTGCTTCACCACCTTCTTGAT-3’ | IDT | qPCR primer | PrimerBank |
| Sequence-based reagent | il6: 5’-TAGTCCTTCCTACCCCAATTTCC-3’ and 5’-TTGGTCCTTAGCCACTCCTTC-3’, | IDT | qPCR primer | PrimerBank |
| Sequence-based reagent | TH: 5’-CCAAGGTTCATTGGACGGC-3’ and 5’-CTCTCCTCGAATACCACAGCC-3’ | IDT | qPCR primer | PrimerBank |
| Sequence-based reagent | tnfa; 5’-CCCTCACACTCAGATCATCTTCT-3’ and 5’-GCTACGACAGTGGGCTACAG-3’ | IDT | qPCR primer | PrimerBank |





