Sox8 and Sox9 act redundantly for ovarian-to-testicular fate reprogramming in the absence of R-spondin1 in mouse sex reversals
Abstract
In mammals, testicular differentiation is initiated by transcription factors SRY and SOX9 in XY gonads, and ovarian differentiation involves R-spondin1 (RSPO1) mediated activation of WNT/β-catenin signaling in XX gonads. Accordingly, the absence of RSPO1/Rspo1 in XX humans and mice leads to testicular differentiation and female-to-male sex reversal in a manner that does not requireSry or Sox9 in mice. Here we show that an alternate testis-differentiating factor exists and that this factor is Sox8. Specifically, genetic ablation of Sox8 and Sox9 prevents ovarian-to-testicular reprogramming observed in XX Rspo1 loss-of-function mice. Consequently, Rspo1 Sox8 Sox9 triple mutant gonads developed as atrophied ovaries. Thus, SOX8 alone can compensate for the loss of SOX9 for Sertoli cell differentiation during female-to-male sex reversal.
eLife digest
In humans, mice and other mammals, genetic sex is determined by the combination of sex chromosomes that each individual inherits. Individuals with two X chromosomes (XX) are said to be chromosomally female, while individuals with one X and one Y chromosome (XY) are chromosomally males.
One of the major differences between XX and XY individuals is that they have different types of gonads (the organs that make egg cells or sperm). In mice, for example, before males are born, a gene called Sox9 triggers a cascade of events that result in the gonads developing into testes. In females, on the other hand, another gene called Rspo1 stimulates the gonads to develop into ovaries.
Loss of Sox9 in XY embryos, or Rspo1 in XX embryos, leads to mice developing physical characteristics that do not match their genetic sex, a phenomenon known as sex reversal. For example, in XX female mice lacking Rspo1, cells in the gonads reprogram into testis cells known as Sertoli cells just before birth and form male structures known as testis cords. The gonads of female mice missing both Sox9 and Rspo1 (referred to as “double mutants”) also develop Sertoli cells and testis cords, suggesting another gene may compensate for the loss of Sox9.
Previous studies suggest that a gene known as Sox8, which is closely related to Sox9, may be able to drive sex reversal in female mice. However, it was not clear whether Sox8 is able to stimulate testis to form in female mice in the absence of Sox9.
To address this question, Richardson et al. studied mutant female mice lacking Rspo1, Sox8 and Sox9, known as “triple mutants”. Just before birth, the gonads in the triple mutant mice showed some characteristics of sex reversal but lacked the Sertoli cells found in the double mutant mice. After the mice were born, the gonads of the triple mutant mice developed as rudimentary ovaries without testis cords, unlike the more testis-like gonads found in the double mutant mice.
The findings of Richardson et al. show that Sox8 is able to trigger sex reversal in female mice in the absence of Rspo1 and Sox9. Differences in sexual development in humans affect the appearance of individuals and often cause infertility. Identifying Sox8 and other similar genes in mice may one day help to diagnose people with such conditions and lead to the development of new therapies.
Introduction
During primary sex determination in mammals, a common precursor organ, the bipotential gonad, develops as a testis or ovary. In humans and mice, testicular development begins when SRY and SOX9 are expressed in the bipotential XY gonad. These transcription factors promote supporting cell progenitors to differentiate as Sertoli cells and form sex cords (Gonen et al., 2018; Chaboissier et al., 2004; Barrionuevo et al., 2006), and this triggers a cascade of signaling events that are required for the differentiation of other cell populations in the testis (Koopman et al., 1991; Vidal et al., 2001). In XX embryos, the bipotential gonad differentiates as an ovary through a process that requires RSPO1-mediated activation of canonical WNT/β-catenin (CTNNB1) signaling in somatic cells (Parma et al., 2006; Chassot et al., 2008). Ovarian fate also involves activation of FOXL2, a transcription factor that is required in post-natal granulosa cells (Schmidt et al., 2004; Ottolenghi et al., 2005; Uhlenhaut et al., 2009), which organize as follicles during embryogenesis in humans and after birth in mice (McGee and Hsueh, 2000; Mork et al., 2012). For complete differentiation of testes or ovaries, an active repression of the opposite fate is necessary (Kim et al., 2006). Inappropriate regulation within the molecular pathways governing sex determination can lead to partial or complete sex reversal phenotypes and infertility (Wilhelm et al., 2009).
Studies in humans and mice have shown that the pathway initiated by SRY/SOX9 or RSPO1/WNT/β-catenin signaling are indispensable for sex specific differentiation of the gonads. For example, in XY humans, SRY or SOX9 loss-of-function mutations prevent testis development (Berta et al., 1990; Houston et al., 1983). In mice, XY gonads developing without SRY or SOX9 lack Sertoli cells and seminiferous tubules and differentiate as ovaries that contain follicles (Lovell-Badge and Robertson, 1990; Chaboissier et al., 2004; Barrionuevo et al., 2006; Lavery et al., 2011; Kato et al., 2013), indicating Sry/Sox9 requirement. In XX humans and mice, SRY/Sry or SOX9/Sox9 gain-of-function mutations promote Sertoli cell differentiation and testicular development (Sinclair et al., 1990; Koopman et al., 1991; Bishop et al., 2000; Vidal et al., 2001; Huang et al., 1999), indicating that SRY/SOX9 function is also sufficient for male gonad differentiation.
With respect to the ovarian pathway, homozygous loss-of-function mutations for RSPO1/Rspo1 trigger partial female-to-male sex reversal in XX humans and mice (Parma et al., 2006; Chassot et al., 2008). In XX Rspo1 or Wnt4 mutant mice, Sertoli cells arise from a population of embryonic granulosa cells (pre-granulosa cells) that precociously exit their quiescent state, differentiate as mature granulosa cells, and reprogram as Sertoli cells (Chassot et al., 2008; Maatouk et al., 2013). The resulting gonad is an ovotestis containing seminiferous tubule-like structures with Sertoli cells and ovarian follicles with granulosa cells, indicating that SRY is dispensable for testicular differentiation. In addition, stabilization of WNT/CTNNB1 signaling in XY gonads leads to male-to-female sex reversal (Maatouk et al., 2008; Harris et al., 2018). Thus, RSPO1/WNT/CTNNB1 signaling is required for ovarian differentiation and female development in humans and mice.
Given the prominent role of SOX9 for testicular development (Chaboissier et al., 2004; Barrionuevo et al., 2009), it was hypothesized that SOX9 is responsible for Sertoli cell differentiation in XX gonads developing without RSPO1/Rspo1. This hypothesis was tested by co-inactivation of Rspo1 or Ctnnb1 and Sox9 in Rspo1-/-; Sox9fl/fl; Sf1:creTg/+(Lavery et al., 2012) and in Ctnnb1fl/fl; Sox9fl/fl; Sf1:creTg/+double mutant mice (Nicol and Yao, 2015). Unexpectedly, XY and XX Rspo1 or Ctnnb1 mutant gonads lacking Sox9 exhibited Sertoli cells organized as testis cords (Nicol and Yao, 2015; Lavery et al., 2012). Specifically, gonads in XX Rspo1-/-; Sox9fl/fl; Sf1:creTg/+ double mutant mice developed as ovotestes as in XX Rspo1-/- single mutants, and XY Rspo1-/-; Sox9fl/fl; Sf1:creTg/+ mutant mice developed hypoplastic testes capable of supporting the initial stages of spermatogenesis. These outcomes indicate that at least one alternate factor can promote testicular differentiation in Rspo1 mutant mice also lacking Sox9 in XY mice, and lacking both Sry and Sox9 in XX animals. This or these factors remained to be identified.
Among the candidate genes that could promote testicular differentiation in the absence of Sry and Sox9 are the other members of the SoxE group of transcription factors that includes Sox9, Sox8 and Sox10 (Lavery et al., 2012; Nicol and Yao, 2015). However, Sox10 expression in testes depends on Sox8 and Sox9 (Georg et al., 2012), and Sox10 loss-of-function mice are fertile (Britsch et al., 2001; Peirano and Wegner, 2000), suggesting that Sox10 would not be the best candidate gene. For Sox8, loss-of-function analyses in XY gonads show testicular development, indicating that Sox8 is not required for Sertoli cell differentiation during embryonic development (Sock et al., 2001). However, a Sox8-null background enhanced the penetrance of the testis-to-ovary sex reversal phenotype in mice with reduced Sox9 expression (Chaboissier et al., 2004), suggesting that Sox8 supports the function of Sox9.
Furthermore, in XY Sox9fl/fl; Sf1:creTg/+ single mutant mice and in XY Sox8-/-; Sox9fl/fl; Amh:creTg/+ and Sox8-/-; Sox9fl/fl; Wt1-CreERT2/+ double mutant mice where Sox9 is inactivated after sex determination, the single and double mutant mice initially form testis cords containing Sertoli cells. However, these cells then lose their identity and begin to express granulosa cell markers like FOXL2 (Barrionuevo et al., 2009; Barrionuevo et al., 2016; Georg et al., 2012). In addition, following tamoxifen induction of Cre recombinase and subsequent deletion of Sox9, Sertoli cells in Sox8-/-; Sox9fl/fl; Wt1-CreERT2/+ testes become apoptotic leading to a complete degeneration of the seminiferous tubules. This indicated that a concerted effort by Sox8 and Sox9 is required in XY gonads for the maintenance of Sertoli cells after sex determination. Beyond mice, in humans, SOX8 contributes to testis differentiation or homeostasis, given the 46,XY gonadal dysgenesis phenotype associated with mutations/rearrangements at the SOX8 locus (Portnoi et al., 2018).
Although Sox8 expression is dispensable for Sertoli cell differentiation in XY gonads, it may have a key role for testicular differentiation in XX sex reversal gonads or in cases of Sox9-independent testicular differentiation in XY gonads. This led us to hypothesize that Sox8 can compensate for loss of Sox9 and is the alternate factor capable of: (i) triggering sex reversal in XX Rspo1 knockout gonads lacking Sry and Sox9, and (ii) promoting testicular development in XY Rspo1 knockout gonads lacking Sox9.
To test this hypothesis, we have generated triple Rspo1, Sox8, and Sox9 loss-of-function mutant mice models. We show here that Sox8 and Sox9 are individually dispensable for testicular development in XY and XX mice lacking Rspo1, indicating the presence of redundant testicular pathways. In the absence of both Sox factors, Sertoli cell differentiation is precluded and XY and XX Rspo1-/-; Sox8-/-; Sox9fl/fl; Sf1:creTg/+ triple mutants develop atrophied ovaries. Together, our data show that Sox8 or Sox9 is required to induce testicular development in XY and XX mice lacking Rspo1.
Results
Rspo1, Sox8, and Sox9 are expressed independently
We first performed expression analyses for Rspo1 (Figure 1A a-h), Sox8 (Figure 1 Ba-l), and Sox9 (Figure 1 Ca-l), in control and mutant gonads. We chose to study embryonic day 17.5 (E17.5) fetal gonads, when testis cords form in Rspo1 sex reversal mice, and juvenile postnatal day 10 (P10) gonads, when gonadal fate is likely to be set (Lavery et al., 2012). In XY gonads, Rspo1 is mostly localized to the coelomic epithelium at E17.5 and to the tunica albuginea at P10 (Figure 1 Aa, c). In fetal ovaries, Rspo1 is expressed in somatic cells at E17.5 and down-regulated after birth, as shown in post-natal P10 ovaries (Figure 1 Af,h). In XY and XX mice lacking Sox8 and Sox9 (i.e., Sox8-/-; Sox9fl/fl; Sf1:creTg/+, referred to as Sox8KO Sox9cKO double mutants), high Rspo1 expression levels were observed in embryonic gonads and down-regulated after birth, indicating ovarian differentiation (Figure 1 Ab,d,e,g), as previously described (Chaboissier et al., 2004). Together, these data confirmed that although Rspo1 is expressed in both XY and XX gonads, robust Rspo1 expression in cells throughout the gonad is a feature of ovarian development in fetuses.
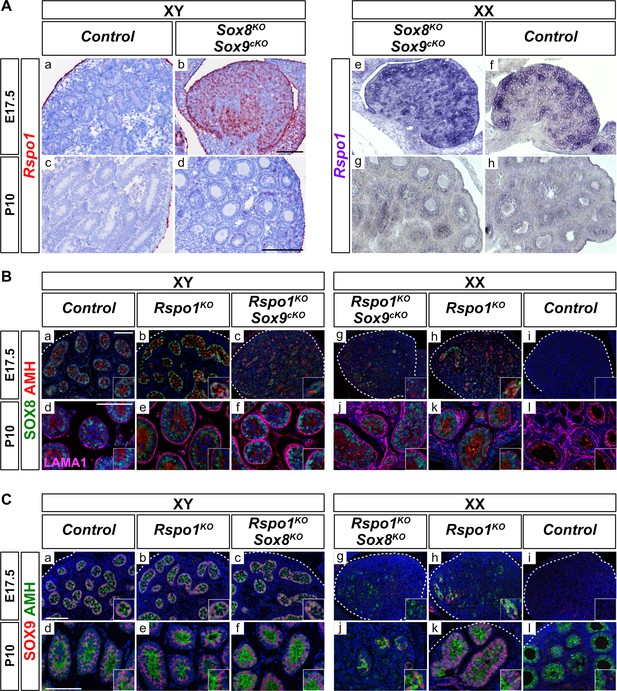
Expression of Rspo1, Sox8, and Sox9 in E17.5 and P10 gonads.
Expression of Rspo1, as revealed by in situ hybridizations (A), and of SOX8 and SOX9, as revealed by immunostaining (B, C) on gonadal sections from embryonic day 17.5 (E17.5) and 10 days post-natal (P10) mice. In XY wild-type testes, Rspo1 is mainly expressed in the coelomic epithelium (Aa) and tunica albuginea (Ac). In XX wildtype ovaries, Rspo1 is expressed throughout the gonad at E17.5 (Af), and down-regulated in post-natal animals, as shown at P10 (Ah). In XY and XX Sox8-/-; Sox9flox/flox; Sf1:creTg/+ (Sox8KO Sox9cKO) mutant mice, the Rspo1 expression profile (Ab,d,e,g) is similar to wildtype ovaries (Af,h), indicating an ovarian fate. For SOX8 expression, in XY control testes (Ba,d) and in XY Rspo1KO gonads developing as testes (Bb,e), SOX8 is expressed in testis cords at E17.5 and P10. Co-immunolabeling with AMH confirmed the identity of Sertoli cells. In XX mice, though AMH is expressed in post-natal control ovaries, these cells were SOX8-negative, indicating that they are granulosa cells (Bj). However, SOX8 and AMH positive testis cords were found in XX Rspo1KO female-to-male sex reversal gonads (Bh,k). SOX8 is also expressed in the absence of Sox9 in XY and XX Rspo1-/-; Sox9flox/flox; Sf1:creTg/+ (Rspo1KO Sox9cKO) gonads at E17.5 and P10 (Bc,g,f,j). At P10, LAMA1 staining demarcates testis cords (Bd-f, j–k) and follicles (Bl). SOX9 expression was found in XY control testes (Ca,d), and in XY Rspo1KO gonads developing as testes (Cb,e). Co-immunolabeling with AMH confirmed the identity of Sertoli cells. As shown, SOX9 and AMH positive testis cords are found in XX Rspo1KO sex reversal gonads at E17.5 and P10 (Ch,k). In addition, SOX9 is also expressed in absence of Sox8 in XY Rspo1-/-; Sox8-/- (Rspo1KO Sox8KO) gonads developing as testes at E17.5 and P10 (Cc,f), and in XX Rspo1KO Sox8KO gonads developing as ovotestes at P10 (Cd). In XX control mice, SOX9 and AMH expression is absent in fetal ovaries (Ci). In post-natal female animals, SOX9 is expressed by theca cells, which are AMH-negative (Cj). All scale bars 100 μm.
In XY control, XY Rspo1-/- (referred to as Rspo1KO), and XY Rspo1-/-; Sox9fl/fl; Sf1:creTg/+ (referred to as Rspo1KO Sox9cKO) mice, immunostaining revealed SOX8 expression in Sertoli cells organized as testis cords at E17.5 and seminiferous tubules at P10, in agreement with previous reports (Figure 1 Ba,d,b,e,g,j; Schepers et al., 2003; Lavery et al., 2012). In XX mice, though SOX8 is not expressed in control ovaries (Figure 1 Bi,l), expression was observed in XX Rspo1KO and XX Rspo1KO Sox9cKO sex reversal gonads (Figure 1 Bh,k,g,j). Co-immunolabeling with AMH confirmed the identity of Sertoli cells (Figure 1 Ba-f, g-h, j-l) and LAMA1 staining at P10 demarcated both testis cords (Figure 1 Bd-f, j-k) and follicles (Figure 1l), which do not express SOX8. In summary, these data corroborated that Sox8 is expressed in gonads lacking Rspo1, and that its expression can be independent of Sox9 (Lavery et al., 2012).
Next, immunostaining revealed SOX9-positive testis cords in XY Rspo1KO testes (Figure 1 Cb,e), XX Rspo1KO ovotestes (Figure 1 Ch, k), as in control testes (Figure 1 Ca, d), as previously described (Chassot et al., 2008). Co-immunolabeling with AMH confirmed the identity of Sertoli cells, since AMH-positive granulosa cells do not express Sox9, and given that ovarian steroidogenic theca cells expressing Sox9 are AMH-negative (Figure 1Cl). In addition, deletion of Sox8 did not alter the expression of Sox9 in XY or XX Rspo1KO gonads (i.e., in Rspo1KO Sox8KO gonads) (Figure 1Cc,f,g,j). Altogether, our results show that Sox8 and Sox9 are expressed in the absence of each other in Rspo1 mutant gonads when testis cords are present or when partial sex reversal occurs.
Ablation of Rspo1 and Sox8 does not impair testis differentiation
Next, we asked how inactivation of both Rspo1 and Sox8 would impact gonad development in XY and XX Rspo1KO Sox8KO double mutants by comparison with controls (Figure 2a–y and Figure 2—figure supplement 1a–h). In XY Rspo1KO Sox8KO mice, the anogenital distance in adult P40 animals was comparable to XY control males (Figure 2a,b). In contrast, XX control females exhibited a short anogenital distance (Figure 2m). Internally, XY Rspo1KO Sox8KO mice developed epididymides, vasa deferensia, seminal vesicles and prostate, as in control males (Figure 2—figure supplement 1a,b). Histological analyses by PAS staining revealed seminiferous tubules with no obvious defects in P10 and P40 XY Rspo1KO Sox8KO animals (Figure 2—figure supplement 1c,d and Figure 2e,f), and these mice were fertile. Testicular development in XY Rspo1KO Sox8KO mice was confirmed by immunostaining experiments on embryonic (E17.5) and post-natal (P10, and P40) gonads that contained SOX9 and DMRT1 positive Sertoli cells forming testicular sex cords and seminiferous tubules (Figure 1Cc,f, Figure 2g–j, and Figure 2—figure supplement 1e–h). DMRT1 expression was also observed in germ cells, which are TRA98-positive (Figure 2i,j and Figure 2—figure supplement 1g,h; Matson et al., 2010). Thus, loss of both Rspo1 and Sox8 does not impair testis differentiation.
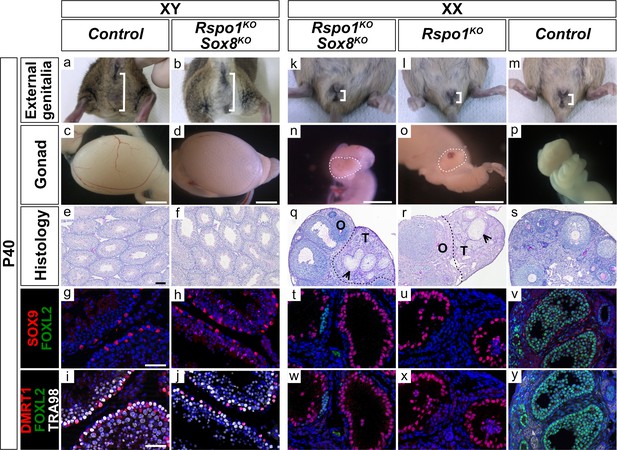
External genitalia and gonad development in adult XY and XX Rspo1KO Sox8KO double mutant mice.
External genitalia from adult P40 mice (a–b, k–m), macroscopic view of gonads (c–d, n–p) (Scale bars 1.5 mm), histology as revealed by PAS staining on gonadal sections (e–f, q–s) (Scale bars 100 µm), and immunostaining of SOX9 (Sertoli cell marker, in red) (g–h, t–v), FOXL2 (granulosa cell marker, in green) (g–j, t–y), DMRT1 (Sertoli and germ cell marker, in red) (i–j, w–y), TRA98 (germ cell marker, in white) (i–j, w–y) and DAPI (nuclear marker, in blue) (g–j, t–y) on gonadal sections (Scale bars 50 µm). Inactivation of both Rspo1 and Sox8 in XY Rspo1KO Sox8KO double mutant mice did not cause a sex reversal (a–j). XY Rspo1KO Sox8KO gonads developed as testes with seminiferous tubules (f) containing SOX9 and DMRT1 positive Sertoli cells (h–j), as in control testes (e, g, i). As shown, XX control ovaries developed follicles (s) containing FOXL2-positive granulosa cells (v, y). Adult ovotestes in XX Rspo1KO Sox8KO mice (n, q) were indistinguishable from XX Rspo1KO mice (o, r). These gonads contained an ovarian ‘O’ compartment with follicles and a testicular ‘T’ compartment with seminiferous tubule-like structures, as indicated by arrowheads (q, r). The seminiferous tubule-like structures in XX Rspo1KO Sox8KO and XX Rspo1KO ovotestes contained SOX9 and DMRT1 positive Sertoli cells (t–u, w–x), as in control testes (g, i), but lacked TRA98-positive germ cells (w, x).
For XX Rspo1KO Sox8KO mice, the question is whether the double mutant gonads developed as ovaries or as ovotestes, as in XX Rspo1KO single mutant (Figure 2k–y and Figure 2—figure supplement 1i–t) and as in XX Rspo1KO Sox9cKO double mutant mice (Lavery et al., 2012). Externally, as in XX control mice, both XX Rspo1KO and XX Rspo1KO Sox8KO mice developed a short anogenital distance, as shown in adult P40 animals (Figure 2k–m). Internally, although XX Rspo1KO Sox8KO mice exhibited rare testis cords during embryonic development (Figure 1Cg), seminiferous tubules devoid of germ cells were apparent at P10, suggesting a delay in ovo-testicular development in double mutant gonads (Figure 2—figure supplement 1l,m). Indeed, by P40, both XX Rspo1KO and XX Rspo1KO Sox8KO mice were essentially indistinguishable with respect to gonad morphology (Figure 2n,o), reproductive tract development (Figure 2—figure supplement 1i,j), ovo-testicular organization (Figure 2q,r), and the presence of SOX9 and DMRT1 positive Sertoli cells in the testicular area (Figure 2t,u,w,x). Altogether, studies performed in Rspo1KO Sox8KO mice demonstrate that like Sox9 (Lavery et al., 2012), Sox8 is dispensable for testicular development in XY and XX Rspo1KO gonads. Moreover, our data suggests that SOX9 likely compensates for the loss of Sox8 in Rspo1KO Sox8KO double mutants.
Ovarian precocious differentiation occurs in XX and XY Rspo1KO Sox8KO Sox9cKO fetuses
Our genetic mouse models allowed us to investigate gonadal fate in XY and XX Rspo1KO mice lacking both Sox8 and Sox9 (i.e., in XY and XX Rspo1KO Sox8KO Sox9cKO triple mutant mice). We first studied gonads in E17.5 fetuses (Figure 3a–j'' and Figure 3—figure supplement 1a–j'), which is when differentiated granulosa cells reprogram as Sertoli cells in XX Rspo1KO gonads (Maatouk et al., 2013). As shown, XX control gonads contained granulosa cells expressing FOXL2, but not Sertoli cells expressing SOX9 or DMRT1 (Figure 3f and Figure 3—figure supplement 1a), indicating ovarian development. The granulosa cells remained quiescent, as evidenced by expression of the mitotic arrest marker CDKN1B (also known as P27) throughout the E17.5 gonad, and the absence of AMH expression indicated that these cells were fetal or pre-granulosa cells (Figure 3a; Maatouk et al., 2013).
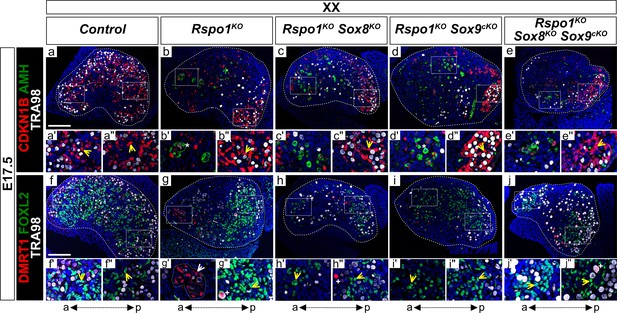
Precocious granulosa cell differentiation in XX Rspo1KO Sox8KO Sox9cKO triple mutant fetuses at E17.5.
Immunofluorescence of CDKN1B (P27) (mitotic arrest marker, in red) (a–e''), AMH (Sertoli marker and mature granulosa cell marker, in green) (a–e''), DMRT1 (Sertoli and germ cell marker, in red) (f–j''), FOXL2 (granulosa cell marker, in green) (f–j''), TRA98 (germ cell marker, in white) (a–j''), and DAPI (nuclear marker, in blue) (a–j'') on gonadal sections from E17.5 fetuses (main panels scale bar 100 µm). The anterior ‘a’ and posterior ‘p’ axis is shown below each column. For main panels (a–j), highlighted anterior and posterior areas are shown in the respective single and double primed letter panels. Yellow arrowheads indicate granulosa cells expressing CDKN1B or FOXL2, asterisks indicate cells expressing AMH, white arrowheads indicate Sertoli cells expressing DMRT1, and plus symbols indicate germ cells expressing DMRT1 and TRA98. Gonads in XX control fetuses developed as ovaries, as shown by FOXL2 and CDKN1B expression in pre-granulosa cells throughout the gonad (a', a'', f', f'', yellow arrowheads). These fetal ovaries lacked mature granulosa cells expressing AMH (a). In contrast, XX Rspo1-/- (Rspo1KO), XX Rspo1-/-; Sox8-/- (Rspo1KO Sox8KO), XX Rspo1-/-; Sox9flox/flox; Sf1:creTg/+ (Rspo1KO Sox9cKO), and XX Rspo1KO Sox8KO Sox9cKO gonads exhibited down-regulation of CDKN1B (b–e) and ectopic AMH expression in the anterior area (b’-e’, asterisks), indicating Sertoli cells or mature granulosa cells. However, while XX Rspo1KO gonads contained Sertoli cells expressing DMRT1 (g', white arrowhead), these cells were rare in XX double and triple mutants (h–j) (for XX triple mutants, 1 out of 8 gonads studied from n = 4 fetuses). Note that some DMRT1-positive cells are germ cells expressing TRA98 (g'', h'', j'', plus symbols). Thus, while granulosa cells differentiate precociously in XX Rspo1KO gonads lacking Sox8 and/or Sox9 at E17.5, these cells have not yet reprogrammed as Sertoli cells in XX Rspo1KO Sox8KO and XX Rspo1KO Sox9cKO mice. In XX triple mutant fetuses, granulosa cell reprogramming as Sertoli cells may be delayed, or blocked.
In contrast, CDKN1B is down-regulated in the anterior area of XX Rspo1KO Sox8KO Sox9cKO triple mutant gonads (n = 4 triple mutant fetuses, Figure 3e), as in XX Rspo1KO single, as well as in XX Rspo1KO Sox8KO and XX Rspo1KO Sox9cKO double mutants (Figure 3b,c,d; Maatouk et al., 2013). In addition, these mutants contained cells expressing AMH (Figure 3b'–e' asterisks), indicating precocious granulosa cell differentiation, as previously described (Maatouk et al., 2013). However, while SOX9 and DMRT1 positive, TRA98-negative Sertoli cells were readily detectable in the anterior area of the XX Rspo1KO gonads (Figure 3—figure supplement 1b' and Figure 3g', white arrowheads), these cells were noticeably absent or rare in XX Rspo1KO Sox8KO Sox9cKO triple mutant fetuses (1 out of 8 XX triple mutant gonads studied from n = 4 fetuses) (Figure 3—figure supplement 1e and Figure 3j). This was also the case in XX Rspo1KO Sox8KO and XX Rspo1KO Sox9cKO double mutants (Figure 3—figure supplement 1c,d and Figure 3h,i). Together with these observations, quantification of immunostained cells expressing DMRT1, FOXL2, and CDKN1B per gonadal section area demarcated by DAPI (Figure 3—figure supplement 2a–f) confirmed the lack of Sertoli cells and presence of granulosa cells in XX double and triple mutant gonads at E17.5 (Figure 3—figure supplement 2a,c,e).
In addition to the presence of mature granulosa cells, gonads in the XX single, double, and triple mutant fetuses also exhibited NR5A1- and HSD3β-positive cells (Figure 3—figure supplement 1g–j), which were absent in XX control ovaries (Figure 3—figure supplement 1f; Chassot et al., 2008; Lavery et al., 2012). Thus, these data indicated that ablation of Sox8 and/or Sox9 in XX fetuses lacking Rspo1 does not prevent the appearance of steroidogenic cells and precocious granulosa differentiation, two characteristics of XX Rspo1KO gonads (Maatouk et al., 2013; Chassot et al., 2008).
We then examined the phenotype of Rspo1KO Sox8KO Sox9cKO gonads in E17.5 XY fetuses (Figure 4a–h''' and Figure 4—figure supplement 1a–h'). As shown, XY Rspo1KO Sox8KO double mutant gonads contained SOX9 and DMRT1 positive Sertoli cells forming testis cords, as in control fetal testes (Figure 4e,f and Figure 4—figure supplement 1a,b). Also, XY Rspo1KO Sox9cKO gonads exhibited DMRT1-positive testis cords (Figure 4g,g'), which were more pronounced than testis cords in XX Rspo1KO and XX Rspo1KO Sox9cKO gonads at this stage (Figure 3g,g',i,i'; Lavery et al., 2012). Thus, in XY fetuses lacking Rspo1, inactivation of one Sox gene is dispensable for Sertoli cells. However, in fetuses lacking both Sox8 and Sox9 in XY triple mutant gonads, Sertoli cells expressing DMRT1 were not readily obvious (6 of 6 XY triple mutant gonads studied from n = 3 fetuses) (Figure 4h and Figure 4—figure supplement 1d). Instead, as in XY Rspo1KO Sox9cKO gonads at this stage, XY triple mutant gonads exhibited FOXL2-positive pre-granulosa cells (Figure 4h,h',h''', yellow arrowheads), and AMH expression suggested that some mature granulosa cells were present (Figure 4d', asterisk). Quantification of cells expressing DMRT1, FOXL2, and CDKN1B confirmed these observations (Figure 3—figure supplement 2b,d,f). Like XX triple mutants, XY triple mutants also contained steroidogenic cells expressing NR5A1 and HSD3β (Figure 4—figure supplement 1h).
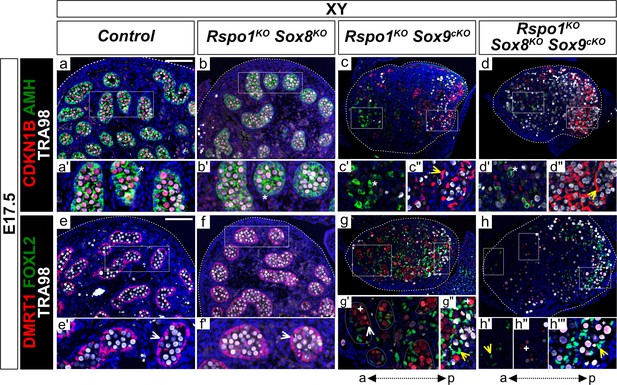
Lack of testis cords in XY Rspo1KO Sox8KO Sox9cKO triple mutant fetuses at E17.5.
Immunofluorescence of CDKN1B (P27) (mitotic arrest marker, in red) (a–d''), AMH (Sertoli marker and mature granulosa cell marker, in green) (a–d''), DMRT1 (Sertoli and germ cell marker, in red) (e–h'''), FOXL2 (granulosa cell marker, in green) (e–h'''), TRA98 (germ cell marker, in white) (a–h'''), and DAPI (nuclear marker, in blue) (a–h) on gonadal sections from E17.5 fetuses (main panel scale bar 100 µm). For gonads in panels (c–d), the anterior ‘a’ and posterior ‘p’ axis is shown below each column. Below each main panels (a–h), highlighted areas are shown in respective primed letter panels. Yellow arrowheads indicate granulosa cells expressing CDKN1B or FOXL2, asterisks indicate cells expressing AMH, white arrowheads indicate Sertoli cells expressing DMRT1, and plus symbols indicate germ cells expressing DMRT1 and TRA98. Gonads in XY Rspo1-/-; Sox8-/- (Rspo1KO Sox8KO) fetuses exhibited AMH and DMRT1 positive Sertoli cells organized as testis cords (b, f) and lacked FOXL2-positive granulosa cells (f), as in control testes (a, e). Cells expressing AMH were found in XY Rspo1-/-; Sox9flox/flox; Sf1:creTg/+ (Rspo1KO Sox9cKO) and XY Rspo1KO Sox8KO Sox9cKO gonads (c’ and d’, asterisks), indicating Sertoli cells or mature granulosa cells. Indeed, both exhibited granulosa cells expressing CDKN1B and FOXL2 (c', d', g', h', h''', yellow arrowheads). However, while XY Rspo1KO Sox9cKO gonads exhibited DRMT1-positive, TRA98-negative Sertoli cells (g', white arrowhead), these cells were scarce in XY triple mutant gonads (h) (6 of 6 XY triple mutant gonads studied from n = 3 fetuses). Note that some DMRT1 expressing cells in XY Rspo1KO Sox9cKO and XY triple mutant gonads are germ cells expressing TRA98 (g', g'', h'', h''', plus symbols). Thus, although XY Rspo1KO Sox9cKO and XY triple mutant gonads contain mature granulosa cells at E17.5, these cells do not reprogram as Sertoli cells in XY triple mutant fetuses.
Altogether, fetal XY and XX Rspo1KO Sox8KO Sox9cKO gonads resembled gonads from XX Rspo1KO Sox8KO, as well as XY and XX Rspo1KO Sox9cKO fetuses, with respect to the presence of steroidogenic cells and mature granulosa cells. However, fetal triple mutant gonads lacked Sertoli cells that were present in fetal (Figure 4f,g) or post-natal (Figure 1Bf,j, Cf,j) double mutant mice. Thus, while pre-granulosa cells in triple mutants differentiated precociously, their reprogramming as Sertoli cells forming testis cords at E17.5 appears to be blocked, or delayed.
Lack of Sertoli cell differentiation in XX and XY Rspo1KO Sox8KO Sox9cKO fetuses
In order to further address the development of triple mutant gonads, we extended our analyses to juvenile (P10) and adult (P40) mice (Figure 5a–d'; Figure 5—figure supplement 1a–x; Figure 5—figure supplement 2a–j and Figure 5—figure supplement 3a–o'''). Both XY and XX Rspo1KO Sox8KO Sox9cKO triple mutant mice developed externally as female with a short anogenital distance, as in XX control mice (Figure 5c,p,r). Internally, both XY and XX triple mutants displayed hermaphroditism of the reproductive tracts, as shown by concomitant presence of vasa deferensia and uteri (Figure 5—figure supplement 1c,m). This was also observed in XY and XX Rspo1KO Sox9cKO mice (Figure 5—figure supplement 1b,n), as well as in XX Rspo1KO and in XX Rspo1KO Sox8KO mice (Figure 2—figure supplement 1i,j). Histological analyses revealed that XY and XX triple mutant gonads developed as ovaries containing primary follicles at P10 (Figure 5—figure supplement 1f,p), which matured up to the antral follicle stage at P40, though some exhibited irregular granulosa cell organization (Figure 5i,v, blue arrowheads). The triple mutant gonads occasionally contained immature or atrophied follicles (Figure 5—figure supplement 2a,b). Both XY and XX Rspo1KO Sox8KO Sox9cKO gonads lacked testicular sex cords (Figure 5i,v, Figure 5—figure supplement 1f,p, and Figure 5—figure supplement 2a,b), which were found in XY and XX Rspo1KO mice lacking Sox8 (Figure 2f,q and Figure 2—figure supplement 1d,l) or Sox9 (Figure 5h,w and Figure 5—figure supplement 1e,q). Immunostaining experiments on P10 and P40 triple mutant gonads confirmed the presence of follicles with granulosa cells expressing FOXL2 and the absence of testis cords with Sertoli cells expressing DMRT1 (Figure 5l,o,y,b’ and Figure 5—figure supplement 1i,l,s,v).
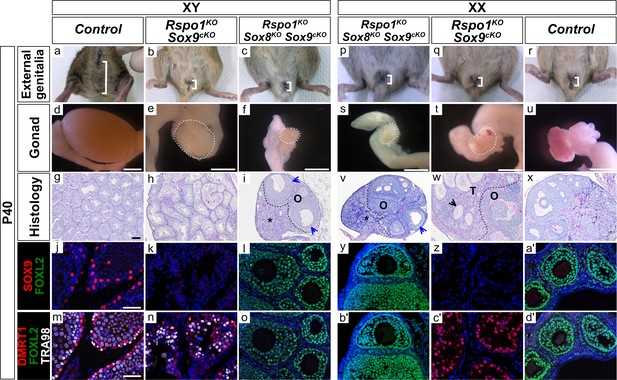
Absence of seminiferous tubules in XY and XX Rspo1KO Sox8KO Sox9cKO triple mutant adult mice.
External genitalia from adult P40 mice (a–c, p–r), macroscopic view of gonads (d–f, s–u) (Scale bars 1.5 mm), histology as revealed by PAS staining on gonadal sections (g–i, v–x) (Scale bars 100 µm), and immunostaining of SOX9 (Sertoli cell marker, in red) (j–l, y–a'), FOXL2 (granulosa cell marker, in green) (j–o, y–d'), DMRT1 (Sertoli and germ cell marker, in red) (m–o, b'–d'), TRA98 (germ cell marker, in white) (m–o, b'–d'), and DAPI (nuclear marker, in blue) (j–o, y–d') on gonadal sections (Scale bars 50 µm). As shown, though adult XY Rspo1-/-; Sox9fl/fl; Sf1:creTg/+ (Rspo1KO Sox9cKO) double mutant gonads developed a short anogenital distance (b), internally these mice developed hypoplastic testes (compare e with d). XX Rspo1KO Sox9cKO gonads developed as ovotestes (t), as in XX Rspo1KO single mutants and XX Rspo1KO Sox8KO double mutants (see Figure 2r, q). Although Sox9fl/fl is inactivated by Sf1:creTg/+ in Rspo1KO Sox9cKO mice (k, z), XY double mutant gonads exhibited seminiferous tubules (h) containing DMRT1-positive Sertoli cells which are TRA98-negative (n), as in control testes (g, m). XX double mutant gonads contained an ovarian compartment ‘O’ with follicles and a testicular ‘T’ compartment with seminiferous tubule-like structures, as indicated by black arrowheads (w). The seminiferous tubule-like structures contained DMRT1-positive Sertoli cells (c'), as in control testes (m), but lacked TRA98-positive germ cells (c'). Both the XY and XX Rspo1KO Sox8KO Sox9cKO triple mutant mice develop externally as female with a short anogenital distance (c, p) as in the double mutants and control female (b, q–r). Despite this, the triple mutants gonads (f, s) developed as atrophied ovaries (i, v), which were smaller than control ovaries (u, x). XY and XX triple mutant gonads exhibited an ovarian ‘O’ compartment and a distinct interstitial compartment, as indicated by asterisks (i, v). The gonads contained follicles up to the antral stage, though some exhibited irregular granulosa cell organization, as indicated by blue arrowheads (i, v). Notably, XY and XX triple mutants lacked testicular sex cords (i, v) that were present in XY and XX Rspo1KO Sox9cKO gonads (h, w). Immunostaining on XY and XX Rspo1KO Sox8KO Sox9cKO gonads confirmed the absence of SOX9 and DMRT1 positive Sertoli cells and the presence of ovarian follicles with granulosa cells expressing FOXL2 (l, o, y, b'), as in control ovaries (a', d'). For these analyses, n = 3 XY and n = 3 XX triple mutant mice were examined.
In 3 of 10 XY and 6 of 16 XX post-natal gonads studied, a cluster of cells expressing DMRT1 were found, but further analyses revealed that these cells did not express the mature Sertoli cell marker GATA1 (Beau et al., 2000; Figure 5—figure supplement 3f,f',f'',l,l',l'',o,o',o''). Instead, these cells expressed the embryonic supporting cell marker GATA4 (Tevosian et al., 2002), which suggests rudimentary testis cord formation (Figure 5—figure supplement 3c,c',c'',i,i',i'', asterisks). We also noticed some cells expressing DMRT1 and FOXL2, though these cells were rare (Figure 5—figure supplement 3l'',l''', arrowheads). In fact, immunostaining for FOXL2 confirmed that the vast majority of the supporting cells in triple mutants were granulosa cells, which did not undergone reprogramming into Sertoli cells (Figure 5l,o,y,b' and Figure 5—figure supplement 1i,l,s,v).
While observing atrophied follicles in adult XY and XX Rspo1KO Sox8KO Sox9cKO triple mutant mice, a distinct interstitial compartment was also apparent (Figure 5i,v, asterisks and Figure 5—figure supplement 2a,b). The identity of this compartment was confirmed by immunostaining for NR5A1 and HSD3β (Figure 5—figure supplement 2g,h). In triple mutant gonads, the interstitial cells were arranged individually or in small clusters when compared with XX control ovaries and XX Rspo1KO ovotestes. In addition, XY and XX triple mutant interstitial cells mildly atrophied, appeared collapsed/dysplastic, and lacked interstitial sinusoids (Figure 5—figure supplement 2e,f). No evidence of neoplasia was present in XY and XX triple mutant and in XX Rspo1KO gonads.
In summary, gonads in XY and XX triple mutants developed as atrophied ovaries. Altogether, our data clearly demonstrate that Sox8 or Sox9 is required and sufficient for testicular differentiation in XY and XX Rspo1KO Sox9cKO or Rspo1KO Sox8KO double mutants, respectively.
Discussion
Our results emphasize the essential role of SOX genes in testis differentiation as we show that Sox genes are required for Sertoli cell differentiation in XX ovotestis. The critical domain of SOX proteins is the DNA binding domain, the HMG (High-Mobility Group)-domain that binds in a sequence-specific manner (Mertin et al., 1999). Remarkably, an HMG-box gene is associated with male sex-specific region in the brown algae Ectocarpus (Ahmed et al., 2014). The sexual cycle of this species consists of an alternation between a diploid sporophyte (with both the U and the V chromosomes), which after meiosis produces either a female haploid gametophyte (with the U chromosome) or male gametophyte (with the V chromosome). The sex-specific region of the Ectocarpus V-chromosome contains an HMG-domain gene, suggesting a conserved function of the HMG-domain containing genes in maleness throughout evolution. In mice, when the HMG box of SRY is replaced with that of SOX3 or SOX9, these composite Sox transgenes induce Sox9 expression and Sertoli cell differentiation (Bergstrom et al., 2000). Also, transgenic expression of Sox3 or Sox10 in XX gonads results in Sox9 expression and testicular differentiation (Sutton et al., 2011; Polanco et al., 2010). These examples demonstrated functional conservation among Sox genes or HMG-box domains and also suggests that male fate centers on transactivation of Sox9.
However, testicular differentiation was reported in XY and XX Rspo1/Ctnnb1 Sox9 double mutant mice (Nicol and Yao, 2015; Lavery et al., 2012), suggesting that another Sox gene can substitute for the absence of Sox9 in this context. Given that Sox8 is up-regulated in the double mutant gonads (Nicol and Yao, 2015; Lavery et al., 2012), we hypothesized that Sox8 and Sox9 can act redundantly for testicular development in mice lacking Rspo1. Here, we demonstrated this by showing that in XY and XX Rspo1KO mice: (i) Sox8 and Sox9 are expressed independently; (ii) Sox8 or Sox9 is sufficient for Sertoli cell differentiation in Rspo1KO Sox9cKO and Rspo1KO Sox8KO mice, respectively; and (iii) Sox8 or Sox9 are required for testicular differentiation, as evidenced by the development of atrophied ovaries in Rspo1KO Sox8KO Sox9cKO triple mutant mice. Together our data show that Sox8 is able to substitute for Sox9 to induce Sertoli cell differentiation in XX sex reversal.
The gonad fate in wildtype, Sox and Rspo1 mutant mice is summarized in Figure 6. In wildtype mice, SOX9 promotes testicular differentiation in XY gonads and RSPO1 promotes ovarian differentiation in XX gonads (Figure 6a). This is also the case in mice lacking Sox8, since it is dispensable for testis and ovarian development (Figure 6a; Sock et al., 2001). As shown, there is an antagonistic relationship between the testis and ovarian pathways, such that the activation of one pathway also leads to the repression of the other to ensure one gonadal fate (Figure 6a). In XY Sox9cKO mice, the testis pathway is not activated, and the ovarian pathway is not repressed, leading to ovarian differentiation (Figure 6b). In XX Sox9cKO mice, loss of SOX9 does not impair ovarian development (Figure 6b). In XY Rspo1KO Sox8KO or XY Rspo1KO Sox9cKO mice, gonads develop as testes or hypo-plastic testes, since one SOX factor is sufficient for Sertoli cell differentiation and seminiferous tubule formation (Figure 6c,d). This is also exemplified by ovo-testicular development in XX Rspo1KO Sox8KO and XX Rspo1KO Sox9cKO mice (Figure 6c,d), where Sertoli cells arise from reprogramming of pre-granulosa cells that have precociously differentiated (Maatouk et al., 2013). We found that inactivation of both SOX factors in mice lacking RSPO1 prevents testicular development in XY and XX animals. In XY and XX Rspo1KO Sox8KO Sox9cKO triple mutant embryos, though pre-granulosa cells differentiate precociously, the absence of both SOX factors impedes granulosa-to-Sertoli reprogramming in embryos and gonads develop as atrophied ovaries (Figure 6e). This atrophied ovary outcome suggests that FOXL2 and other ovarian factors cannot fully compensate for the loss of RSPO1 (Figure 6e).
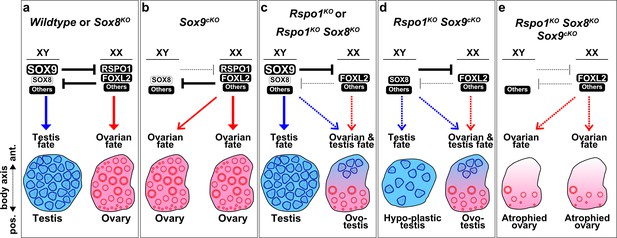
Gonad fate in wildtype, Rspo1, and Sox mutant mice.
In wildtype mice, SOX9, SOX8, and other factors promote testicular differentiation in XY mice, and RSPO1, FOXL2, and other factors promote ovarian differentiation in XX mice, as indicated by arrows (a). Antagonism exists between the testis and ovarian pathway, as indicated by ‘T’ bars (a). SOX9 and RSPO1 are essential for testicular and ovarian differentiation respectively, since XY Sox9cKO mice develop ovaries (b) and XX Rspo1KO mice develop partial sex-reversal ovotestes (c). However, we previously demonstrated that SOX9 is dispensable for testicular development in XX Rspo1KO mice, by studying XX Rspo1KO Sox9cKO mice (d). Also, gonads in XY Rspo1KO Sox9cKO mice develop as hypoplastic testes (d), indicating that RSPO1 is required for ovarian differentiation in XY Sox9cKO mice. In studying a Sox8KO mutation in XY and XX mice or mice lacking Rspo1, it was evident that SOX8 is dispensable for testicular, ovarian, or ovotesticular development (a, c). In this study however, we demonstrated that SOX8 is required for hypoplastic testicular or ovotesticular differentiation in XY and XX Rspo1KO Sox9cKO mice (d) by studying triple mutants (e). Gonads in both XY and XX Rspo1KO Sox8KO Sox9cKO mice lacked testis cords and developed as atrophied ovaries (e). Thus, SOX8 or SOX9 is sufficient and both SOX are required for testicular differentiation in gonads lacking RSPO1.
Interestingly, in XX Rspo1KO single mutant gonads and in XX Rspo1KO gonads lacking Sox8 and/or Sox9 in double and triple mutants, pre-granulosa cells differentiate as mature granulosa cells expressing AMH in an anterior-to-posterior wave or gradient ([Maatouk et al., 2013] and present data). Such a gradient was also found in XX gonads with an Sry transgene – supporting cells transiently express SOX9, after which this ability is lost in an anterior-to-posterior wave (Harikae et al., 2013). This suggests that somatic cell differentiation in ovaries proceeds in a spatiotemporal, anterior-to-posterior, manner. As shown here, apparently, this wave of somatic cell differentiation is conserved in XX sex reversal associated with Rspo1 mutations.
How Sox8 operates in pathophysiological cases of testicular differentiation is not yet known. In wildtype mice, Sox8 expression in XY gonads has been described as coinciding with Sox9 at E11.5 (Jameson et al., 2012; Stévant et al., 2018; Schepers et al., 2003) or occurring after robust expression of Sox9 at E12.5 (Schepers et al., 2003). Together, these observations suggest that SRY might activate Sox8, as predicted (Li et al., 2014), and that Sox8 expression is reinforced by Sox9. However, the activation of Sox8 by SOX9 is likely indirect given that SOX9 does not bind the Sox8 locus in mice. Interestingly, SOX9 binding to Sox8 has been shown in cattle (Rahmoun et al., 2017). Also, the expression of Sox8 and Sox9 are independent in sex cords in XY mouse gonads ((Barrionuevo et al., 2009), our present results). Thus, it is more plausible that SRY activates Sox8 expression in XY Rspo1/Ctnnb1 Sox9 double mutant mice. In fact, in these mice, Sry expression is extended beyond E12.5 (Lavery et al., 2012; Nicol and Yao, 2015), a time when Sry is normally down-regulated in mice (Hacker et al., 1995).
Whereas Sox8 expression can result from SRY activation in XY embryos, it is not obvious how Sox8 is upregulated in the absence of Sry in XX Rspo1/Ctnnb1 Sox9 double mutants. The pro-ovarian factors Rspo1 or Ctnnb1 are required on one hand, to prevent precocious maturation of granulosa cells, which are capable of transdifferentiation into Sertoli cells. On the other hand, these factors repress ectopic steroidogenesis in XX gonads as evidenced by the presence of steroidogenic cells in XX Rspo1 or Rspo1KO Sox9cKO embryonic gonads (Chassot et al., 2008; Lavery et al., 2012).
When E13.5 ovaries are transplanted to kidneys of XY mice, circulating androgens promote partial trans-differentiation towards a testis fate through a mechanism involving up-regulation of Sox8 before Sox9 (Miura et al., 2019). Ablation of Sox8 in the transplanted ovaries did not prevent sex reversal and this outcome is likely attributed to the presence of Sox9. Furthermore, before up-regulation of Sox8, supporting cells in the fetal ovary transplant express Amh, a phenotype that is strikingly similar to sex reversal in XX Rspo1KO gonads (Maatouk et al., 2013). However, inactivation of Amh in the transplanted ovaries was also dispensable for sex reversal, suggesting that TGF-β signaling driven by other TGF-β factors like Activin or unknown factors may promote Sox gene expression in sex reversal conditions.
It is noteworthy that WNT/CTNNB1 signaling regulates the level of ActivinB as evidenced by its up-regulation in XX Wnt4KO or Ctnnb1cKO gonads (Yao et al., 2006; Liu et al., 2010). Moreover ablation of InhibinA/B, two antagonist members of Activin, promotes sex cord development in XX gonads (Matzuk et al., 1992). Thus, TGF-β signaling is likely involved in XX sex reversal when the WNT/CTNNB1 pathway is compromised. The factors including Activin/Inhibin to control Sox gene expression in XX transplanted ovaries and in XX mice lacking Rspo1/Ctnnb1 remain to be identified.
The identification of Sox8 as a key factor in pathophysiological testicular development is somewhat of a paradox, given evidence indicating that aside from Sry and Sox9, no other Sox gene tested so far play key roles in Sertoli cell differentiation in XY wildtype gonads (She and Yang, 2017). In mice, Sox8 is dispensable for Sertoli cell differentiation (Sock et al., 2001), but an inefficient or late deletion of Sox9 leads to XY sex reversal only if there is additional deletion of Sox8 (Lavery et al., 2011; Chaboissier et al., 2004; Barrionuevo et al., 2009). This suggests that Sox8 reinforces Sox9 during testis differentiation. In addition, Sox8 is required for Sertoli cell maintenance along with Sox9, since Sertoli cells in XY Sox8 Sox9 double loss-of-function gonads undergo apoptosis (Barrionuevo et al., 2016). Thus, although Sox8 is an important factor for testis differentiation and maintenance, the rapid and high level of expression of Sox9 induced by SRY, minimizes the role of Sox8 in XY differentiating testes.
During XX sex reversal, early and high induction of Sox8/9 does not occur, given the absence of Sry. Hence, ovarian differentiation is initiated, but absence of pro-ovarian gene such as Rspo1 leads to a succession of events from ectopic steroidogenesis to accelerated maturation of granulosa cells that ultimately promote the expression of Sox8 and Sox9. In XX Rspo1KO gonads, both factors are similarly important and can compensate for the absence of the other to induce transdifferentiation of granulosa cells to Sertoli cells during late embryogenesis. Thus, although Sox8 is dispensable for testicular differentiation in wildtype mice, our current study demonstrates that Sox8 is essential for testicular differentiation in sex reversal conditions.
Functional redundancy between SOX8 and SOX9 does not seem to operate in humans. For example, XY sex reversal can result from inactivating mutations of one SOX9 allele, indicating haploinsufficiency (Wagner et al., 1994; Foster et al., 1994). Also, SOX8 mutations were associated with a range of phenotypes including complete gonadal dysgenesis (streak gonads with immature female genitalia) and hypoplastic testes in three 46, XY patients (Portnoi et al., 2018). Thus, it appears that the impact of a single gene mutation can vary, according to the nature of the mutation and genetic background of the individual. Nevertheless, the human cases of XY sex reversal show that SOX8 is emerging to be an important regulator of testicular gonadal development and by extension, overall male development.
Materials and methods
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Strain, strain (Mus musculus) | Rspo1-/- | (Chassot et al., 2008) | henceforth Rspo1KO | |
| Strain, strain (Mus musculus) | Sox8-/- | (Sock et al., 2001) | henceforth Sox8KO | |
| Strain, strain (Mus musculus) | Sox9fl/fl; Sf1:creTg/+ | (Lavery et al., 2011) | henceforth Sox9cKO | |
| Strain, strain (Mus musculus) | Rspo1-/-; Sox8-/- | This paper | henceforth Rspo1KO Sox8KO | |
| Strain, strain (Mus musculus) | Rspo1-/-; Sox9fl/fl; Sf1:creTg/+ | (Lavery et al., 2012) | henceforth Rspo1KO Sox9cKO | |
| Strain, strain (Mus musculus) | Rspo1-/-; Sox8-/-; Sox9fl/fl; Sf1:creTg/+ | This paper | henceforth Rspo1KOSox8KO Sox9cKO | |
| Antibody | anti-AMH/MIS (C-20) (Goat polyclonal) | Santa Cruz RRID:AB-649207 | Cat# sc-6886 | IF(1:100) |
| Antibody | anti-DMRT1 (Rabbit polyclonal) | Sigma RRID:AB_10600868 | Cat# HPA027850 | IF(1:100) |
| Antibody | anti-FOXL2 (Goat polyclonal) | Novus RRID:AB_2106188 | Cat# NB100-1277 | IF(1:200) |
| Antibody | anti-GATA1 (N6) (Rat monoclonal) | Santa Cruz RRID:AB_627663 | Cat# sc-265 | IF(1:200) |
| Antibody | anti-GATA4 (C20) (Goat polyclonal) | Santa Cruz RRID:AB_2108747 | Cat# sc-1237 | IF(1:200) |
| Antibody | anti-3β-HSD (P18) (Goat polyclonal) | Santa Cruz RRID:AB_2279878 | Cat# sc-30820 | IF(1:200) |
| Antibody | anti-P27/CDKN1B (Kip1) (Rabbit polyclonal) | Santa Cruz RRID:AB_632129 | Cat# sc-528 | IF(1:200) |
| Antibody | anti-Laminin LAMA1 (Rabbit polyclonal) | Sigma RRID:AB_477163 | Cat# L9393 | IF(1:150) |
| Antibody | anti-NR5A1/SF-1 (Rabbit polyclonal) | Gift from Ken Morohashi | IF(1:1000) | |
| Antibody | anti-SOX8 (Guineapig, polyclonal) | Gift from Elisabeth Sock (Stolt et al., 2005) | IF(1:1000) | |
| Antibody | anti-SOX9 (Rabbit polyclonal) | Sigma RRID:AB_1080067 | Cat# HPA001758 | IF(1:200) |
| Antibody | anti-TRA98 (Rat monoclonal) | Abcam RRID:AB_1659152 | Cat# ab82527 | IF(1:200) |
| Recombinant DNA reagent | Rspo1 riboprobe | (Parma et al., 2006) | ||
| Recombinant RNA reagent | Rspo1 (RNAscope riboprobe) | Advanced Cell Diagnostics | ||
| Software, algorithm | Affinity Photo | Serif Europe Ltd., Nottingham United Kingdom | https://affinity.serif.com/en-us/photo/ | |
| Software, algorithm | Affinity Designer | Serif Europe Ltd., Nottingham United Kingdom | https://affinity.serif.com/en-us/ | |
| Software, algorithm | Graphpad Prism | Graphpad Software, Inc, La Jolla, CA | http://www.graphpad.com/ | |
| Other | DAPI stain | Vector Laboratory | H-1500 | (1µg/ml) |
Mouse strains and genotyping
Request a detailed protocolThe experiments described here were carried out in compliance with the relevant institutional and French animal welfare laws, guidelines, and policies. These procedures were approved by the French ethics committee (Comité Institutionnel d’Ethique Pour l’Animal de Laboratoire; number NCE/2011–12). All mouse lines were kept on a mixed 129Sv/C57BL6/J background. Rspo1-/- (Chassot et al., 2008), Sox8-/- (Sock et al., 2001), Sox9fl/fl (Akiyama et al., 2002), and Sf1:creTg/+ (Bingham et al., 2006) mice were obtained previously, and the generation of Sox9fl/fl; Sf1:creTg/+ (Lavery et al., 2011) and Rspo1-/-; Sox9fl/fl; Sf1:creTg/+ (Lavery et al., 2012) mice was described previously. For Rspo1KO Sox8KO mice: Rspo1-/- males were mated with Sox8-/- females to obtain Rspo1+/-; Sox8+/- males and females. Matings between these littermates allowed us to obtain Rspo1-/-; Sox8-/- double mutant mice, referred to as Rspo1KO Sox8KO mice, and control animals. For Rspo1KO Sox8KO Sox9cKO mice: first, Rspo1-/-; Sox8-/- males were mated with Sox8-/-; Sox9fl/fl; Sf1:creTg/+ females to generate Rspo1+/-; Sox8-/-; Sox9fl/+ males and Rspo1+/-; Sox8-/-; Sox9fl/+; Sf1:creTg/+ females. Matings between these littermates then produced Rspo1-/-; Sox8-/-; Sox9fl/fl males and Rspo1+/-; Sox8-/-; Sox9fl/fl; Sf1:creTg/+ females. Finally, matings between these littermates then allowed us to obtain Rspo1-/-; Sox8-/-; Sox9fl/fl; Sf1:creTg/+ triple mutant mice, referred to as Rspo1KO Sox8KO Sox9cKO mice, and control animals. Embryos were collected from timed evening matings that was confirmed by the presence of a vaginal plug the following morning. This marked embryonic day 0.5 (E0.5). The day of delivery was defined as post-natal day 0 (P0). Genotyping was performed as described in Chaboissier et al. (2004); Chassot et al. (2008); Bingham et al. (2006) by using DNA extracted from tail tip or ear biopsies of mice. The presence of the Y chromosome was determined, as described previously (Hogan et al., 1994).
In situ hybridization
Request a detailed protocolGonad samples were fixed with 4% paraformaldehyde overnight, processed for paraffin embedding, and then sectioned at 5–7 μm thick. The in situ hybridizations for Figure 1e–h were carried out essentially as described by Lavery et al. (2012). For analyses in Figure 1a–d, RNAscope technology was used (Wang et al., 2012). The Rspo1 probe was purchased from the manufacturer (Advanced Cell Diagnostics) and the protocol was performed according to the manufacturer’s instructions using the Fast Red dye, which can be visualized using light or fluorescence microscopy. The in situ hybridization experiments were performed on gonads from at least three mice for each genotype.
Immunological analyses
View detailed protocolGonad samples were fixed with 4% paraformaldehyde overnight, processed for paraffin embedding, and sectioned at 5 μm thick. The following dilutions of primary antibodies were used: AMH/MIS (c-20, sc-6886, Santa Cruz), 1:200; DMRT1 (HPA027850, Sigma), 1:100; FOXL2 (NB100-1277, Novus), 1:200; GATA1 (N6, sc-265, Santa Cruz), 1:200; GATA4 (C20, sc-1237, Santa Cruz), 1:200; 3βHSD (P18, sc-30820, Santa Cruz), 1:200; P27 (Kip1, sc-528, Santa Cruz), 1:200; LAMA1 (L9393, Sigma), 1:150; SF1 (kindly provided by Ken Morohashi), 1:1000; SOX8 (kindly provided by Elisabeth Sock [Stolt et al., 2005]), 1:1000; SOX9 (HPA001758, Sigma), 1:200; and TRA98 (ab82527, Abcam), 1:200. Counterstain with DAPI was used to detect nuclei. Immunofluorescence of secondary antibodies were detected with an Axio ImagerZ1 microscope (Zeiss) coupled to an Axiocam mrm camera (Zeiss) or a LSM 780 NLO inverted Axio Observer.Z1 confocal microscope (Carl Zeiss Microscopy GmbH, Jena,Germany) using a Plan Apo 10X dry NA 0.45 objective. Images were processed with Axiovision LE and Serif Affinity Photo software. Immunostaining experiments were performed on gonads from at least three mice for each genotype.
Cell quantification
Request a detailed protocolImmunostaining analyses were performed, as described above. For analyses at E17.5, immunostaining were performed on 2 to 17 sections spaced 20–30 μm apart in each gonad. Then, for each section, the ratio of cells positive for DMRT1, FOXL2, or CDKN1B to total gonad area, as visualized by DAPI staining, were manually tabulated. Next, the individual ratios and mean for each genotype were plotted in a histogram using Graphpad software. Finally, the data was analysed by one-way ANOVA and Tukey-Kramer post tests. For p-values<0.05,<0.01,<0.001, and <0.0001, asterisks (*, **, ***, ****) represent significant differences compared with XY control cell numbers, respectively and ampersands (&, &&, &&&, &&&&) represent significant differences compared with XX control cell numbers, respectively.
Histological analyses
Request a detailed protocolGonad samples were fixed with Bouin’s solution overnight, processed for paraffin embedding, sectioned at 5 μm thick, and then stained according to standard procedures for periodic acid Schiff (PAS) or hematoxylin and eosin (H and E) staining. Images were taken with an Axiocam mrm camera (Zeiss) and processed with Serif Affinity Photo software. Histology staining was performed on gonads from at least three mice for each genotype.
Data availability
All data generated or analysed during this study are included in the manuscript and supporting files.
References
-
A haploid system of sex determination in the Brown alga Ectocarpus spCurrent Biology 24:1945–1957.https://doi.org/10.1016/j.cub.2014.07.042
-
Homozygous inactivation of Sox9 causes complete XY sex reversal in miceBiology of Reproduction 74:195–201.https://doi.org/10.1095/biolreprod.105.045930
-
GATA-1 is a potential repressor of anti-Müllerian hormone expression during the establishment of puberty in the mouseMolecular Reproduction and Development 56:124–138.https://doi.org/10.1002/(SICI)1098-2795(200006)56:2<124::AID-MRD2>3.0.CO;2-J
-
Activation of beta-catenin signaling by Rspo1 controls differentiation of the mammalian ovaryHuman Molecular Genetics 17:1264–1277.https://doi.org/10.1093/hmg/ddn016
-
Heterogeneity in sexual bipotentiality and plasticity of granulosa cells in developing mouse ovariesJournal of Cell Science 126:2834–2844.https://doi.org/10.1242/jcs.122663
-
BookManipulating the Mouse EmbryoCold Spring Harbor: Cold Spring Harbor Laboratory Press.
-
Autosomal XX sex reversal caused by duplication of SOX9American Journal of Medical Genetics 87:349–353.https://doi.org/10.1002/(SICI)1096-8628(19991203)87:4<349::AID-AJMG13>3.0.CO;2-N
-
Production of Sry knockout mouse using TALEN via oocyte injectionScientific Reports 3:3136.https://doi.org/10.1038/srep03136
-
XY female mice resulting from a heritable mutation in the primary testis-determining gene, tdyDevelopment 109:635–646.
-
Stabilization of beta-catenin in XY gonads causes male-to-female sex-reversalHuman Molecular Genetics 17:2949–2955.https://doi.org/10.1093/hmg/ddn193
-
Initial and cyclic recruitment of ovarian folliclesEndocrine Reviews 21:200–214.https://doi.org/10.1210/er.21.2.200
-
The DNA-binding specificity of SOX9 and other SOX proteinsNucleic Acids Research 27:1359–1364.https://doi.org/10.1093/nar/27.5.1359
-
Foxl2 is required for commitment to ovary differentiationHuman Molecular Genetics 14:2053–2062.https://doi.org/10.1093/hmg/ddi210
-
R-spondin1 is essential in sex determination, skin differentiation and malignancyNature Genetics 38:1304–1309.https://doi.org/10.1038/ng1907
-
Mutations involving the SRY-related gene SOX8 are associated with a spectrum of human reproductive anomaliesHuman Molecular Genetics 27:1228–1240.https://doi.org/10.1093/hmg/ddy037
-
SOX8 is expressed during testis differentiation in mice and synergizes with SF1 to activate the amh promoter in vitroJournal of Biological Chemistry 278:28101–28108.https://doi.org/10.1074/jbc.M304067200
-
Sry and SoxE genes: how they participate in mammalian sex determination and gonadal development?Seminars in Cell & Developmental Biology 63:13–22.https://doi.org/10.1016/j.semcdb.2016.07.032
-
Idiopathic weight reduction in mice deficient in the high-mobility-group transcription factor Sox8Molecular and Cellular Biology 21:6951–6959.https://doi.org/10.1128/MCB.21.20.6951-6959.2001
-
Identification of SOX3 as an XX male sex reversal gene in mice and humansJournal of Clinical Investigation 121:328–341.https://doi.org/10.1172/JCI42580
-
Gonadal differentiation, sex determination and normal sry expression in mice require direct interaction between transcription partners GATA4 and FOG2Development 129:4627–4634.
-
Sox9 induces testis development in XX transgenic miceNature Genetics 28:216–217.https://doi.org/10.1038/90046
-
RNAscope: a novel in situ RNA analysis platform for formalin-fixed, paraffin-embedded tissuesThe Journal of Molecular Diagnostics : JMD 14:22–29.https://doi.org/10.1016/j.jmoldx.2011.08.002
-
Antagonism of the testis- and ovary-determining pathways during ovotestis development in miceMechanisms of Development 126:324–336.https://doi.org/10.1016/j.mod.2009.02.006
Article and author information
Author details
Funding
Agence Nationale de la Recherche (ANR-11-LABX-0028-01)
- Nainoa Richardson
Agence Nationale de la Recherche (Graduate student fellowship)
- Nainoa Richardson
Agence Nationale de la Recherche (ANR-19-CE14-0022-SexDiff)
- Marie-Christine Chaboissier
The funders had no role in study design, data collection and interpretation, or the decision to submit the work for publication.
Acknowledgements
We thank Ken Morohashi and Elisabeth Sock for the SF-1/NR5A1 and SOX8 antibodies, respectively. We also thank Samah Rekima and the Experimental Histopathology Platform for assistance with samples. The microscopy was done at MICA facility and the help of Simon Lachambre is acknowledged. Thanks to the Schedl team for helpful discussions, and Anne-Amandine Chassot and Aitana Perea-Gomez for critical reading of the manuscript. This work was supported by the Agence Nationale de la Recherche (ANR-11-LABX-0028–01; ANR-19-CE14-0022-SexDiff).
Ethics
Animal experimentation: This study was performed in strict accordance with the relevant institutional and European animal welfare laws, guidelines, and policies. These procedures were approved by the French ethics committee (Comité Institutionnel d'Ethique Pour l'Animal de Laboratoire; number NCE/2011-12).
Copyright
© 2020, Richardson et al.
This article is distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use and redistribution provided that the original author and source are credited.
Metrics
-
- 3,615
- views
-
- 297
- downloads
-
- 27
- citations
Views, downloads and citations are aggregated across all versions of this paper published by eLife.
Citations by DOI
-
- 27
- citations for umbrella DOI https://doi.org/10.7554/eLife.53972






