The clonal structure and dynamics of the human T cell response to an organic chemical hapten
Figures
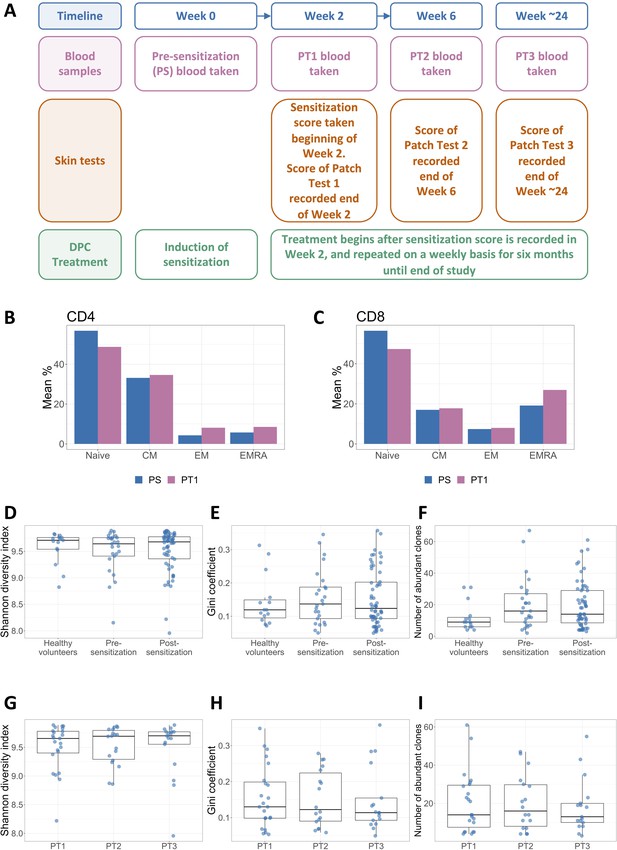
Repeated exposure to diphenylcyclopropenone (DPC) does not alter the global structure of the peripheral blood T cell receptor (TCR) repertoire.
(A) The study outline showing when bloods were drawn, skin tests were performed, and DPC treatment was applied during the ~24 weeks of participation in the study. The four time points at which sensitization scores were recorded and blood samples were taken are referred to throughout the paper as PS (pre-sensitization, week 0), PT1 (Patch Test 1, week 2), PT2 (Patch Test 2, week 6) and PT3 (Patch Test 3, around week 24). In all cases bloods were drawn prior to application of the patch test. (B) and (C) The PS and PT1 blood samples of 10 patients were analyzed using flow cytometry. The mean percentage of total naive, central memory (CM), effector memory (EM), and effector memory RA (EMRA) expressing cells in the (B) CD4 and (C) CD8 compartments are shown. Paired t-tests with Benjamini–Hochberg correction for multiple testing were performed to check for significant differences between the pre- and post-sensitization cell number distributions for each subpopulation. All p-values were considerably higher than the 0.05 significance threshold. (D) The Shannon diversity index of the healthy volunteers (n = 15 samples from five individuals), pre-sensitization (n = 25), and post-sensitization (n = 58; from all three time points) TCR repertoire samples. All samples were randomly subsampled to the minimum sample size (21,838 beta TCRs), and the Shannon diversity index of the subsample was then calculated. Each sample is represented by a dot. The box plots show the median, and lower and upper quartiles of each group. Differences in the distribution of the three groups were tested using a Kruskal–Wallis rank sum test and were non-significant (p=0.87). (E) The Gini inequality coefficient of the healthy volunteers, pre-sensitization and post-sensitization TCR repertoire samples, subsampled as in (D). Differences in the distribution of the three groups were tested using a Kruskal–Wallis rank sum test and were non-significant (p=0.89). (F) The number of TCRs that appear with a frequency of 1/1000 or higher in each sample (termed ‘abundant TCRs’), for the healthy volunteers, pre-sensitization and post-sensitization samples, subsampled as in (D) and (E). A Kruskal–Wallis rank sum test revealed no statistical difference between the groups (p=0.14). (G)– (I) Sensitized samples were separated according to time point: PT1 (n = 23), PT2 (n = 18), and PT3 (n = 17). The Shannon diversity index (G), the Gini coefficient (H), and the number of abundant clones (I) of these subsamples were then calculated. Kruskal–Wallis rank sum tests were used to compare between the three groups in each case. All tests showed no statistically significant difference, with p-values p=0.97, p=0.96, and p=0.90 respectively.
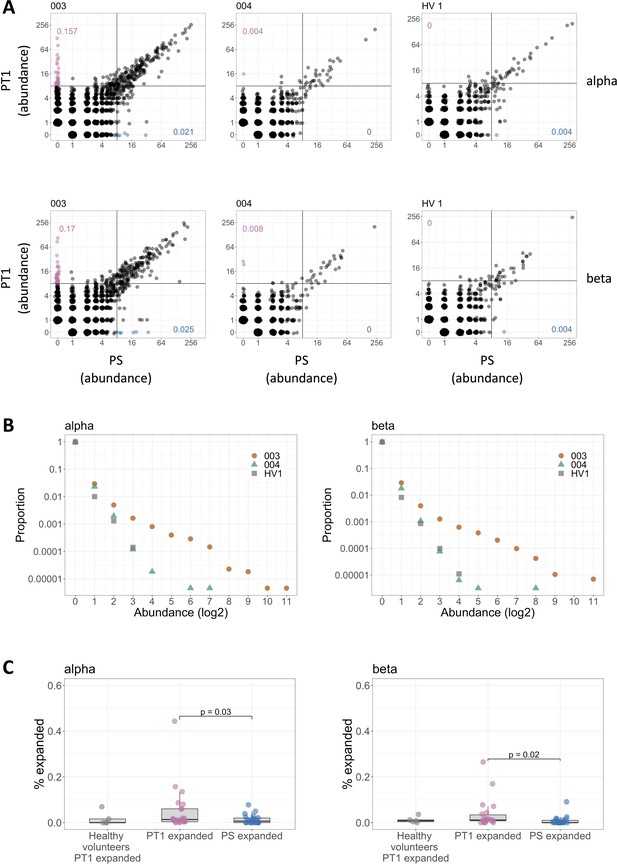
Sensitization with diphenylcyclopropenone (DPC) induces a transient expansion in the frequency of a small subset of the T cell receptor (TCR) repertoire.
(A) The abundance distribution of TCRs at PS and PT1. All samples were subsampled to the same number of TCRs (28,000). Each unique TCR is represented by a dot, and the axes represent the number of times it is observed in the PS (x-axis) and PT1 (y-axis) sample of the same individual. The left and center panels represent two representative individuals. The right panels show one representative healthy volunteer at 0 and 2 weeks (equivalent timings to PS and PT1). The plots for all individuals are shown in Supplementary file 3. The pink dots identify a population of TCRs absent in the PS sample and expanded (abundance ≥8) in the PT1 sample. The blue dots identify a population of TCRs absent in the PT1 sample and expanded (≥8) in the PS sample. The numbers indicate the percentage of PT1 expanded TCRs (pink) and PS expanded TCRs (blue). (B) The abundance distribution profile of TCRs present at PT1 and absent at PS corresponding to the left panel in (A) (orange), middle panel (green), and right panel (gray). The y-axis shows the proportion of the TCRs which are found at the abundance indicated by the x-axis. (C) The distribution of the percentages of PT1 expanded (≥8) TCRs in healthy volunteers (n = 5) and in sensitized individuals (n = 22), and the distribution of the percentages of PS expanded (≥8) TCRs in sensitized individuals (n = 22), all subsampled as in (A). Bars show Wilcoxon signed–rank comparisons.
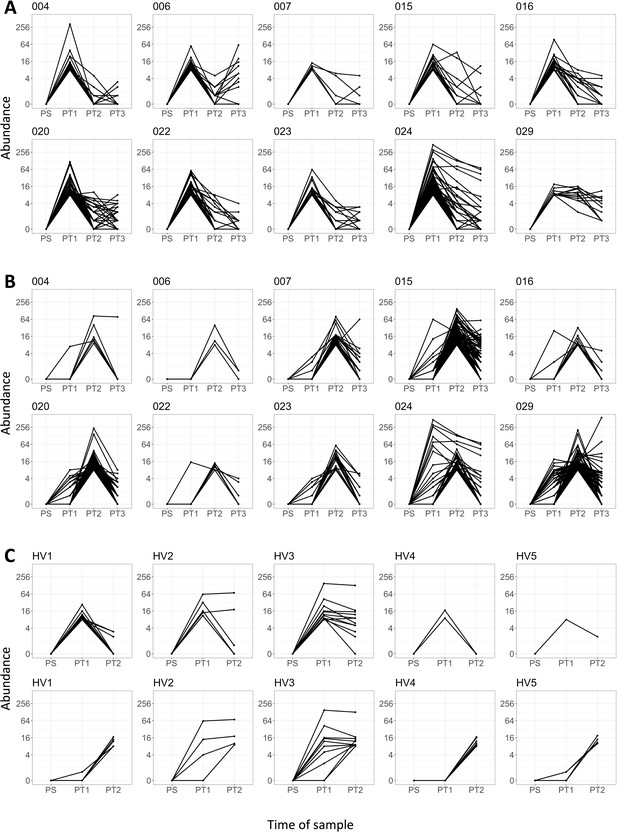
Dynamic changes in T cell receptor (TCR) frequency following sensitization.
(A) The abundances of the PT1 expanded (threshold ≥8) beta TCRs at the four time points: PS, PT1, PT2, and PT3. Each panel is a different patient (n = 10). (B) The abundances of the PT2 expanded (threshold ≥8) beta TCRs at the four time points: PS, PT1, PT2, and PT3. Each panel is a different patient (n = 10). (C) Equivalent time points (0 weeks, 2 weeks, and 6 weeks) for five healthy volunteers. Top row is PT1 expanded beta TCRs; bottom row is PT2 expanded beta TCRs.
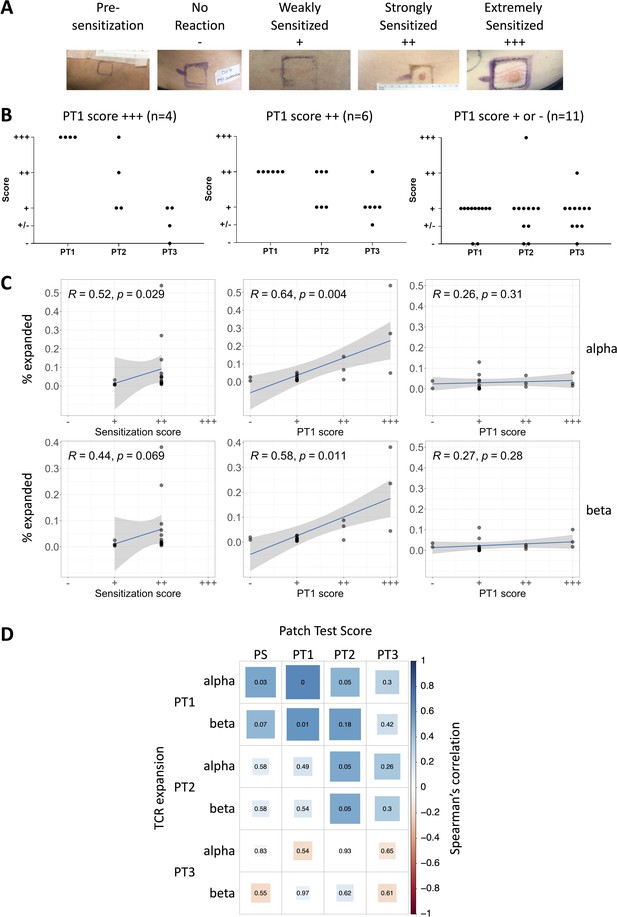
T cell receptor (TCR) expansion after exposure to diphenylcyclopropenone (DPC) correlates with the magnitude of allergic contact dermatitis.
(A) Photographs showing examples of the varying levels of skin reaction observed in response to DPC; the reactions were classified according to standards set by the International Contact Dermatitis Research Group (no reaction [−], +, ++, +++). (B) The changes in patch test scores during treatment in patients with PT1 score of +++ (n = 4, left panel), ++ (n = 6, middle panel), and + or − (n = 11, right panel). (C) Left panels: The number of PT1 expanded (≥8) clones are plotted against the sensitization score. Center panels: The number of PT1 expanded (≥8) clones are plotted against the PT1 patch test score. Right panels: The number of PS expanded (≥8) clones are plotted against the patch test score at PT1. The blue line indicates the best fit linear model, with the model’s 95% confidence intervals in gray. The inset text shows the Spearman’s correlation coefficient, rho. (D) Correlation matrix representing the Spearman correlation between the number of PT expanded TCRs and the reaction recorded at the sensitization site or the patch test scores at each time point. The color and size of each square correspond to Spearman’s rho, and non-adjusted p-values are shown.
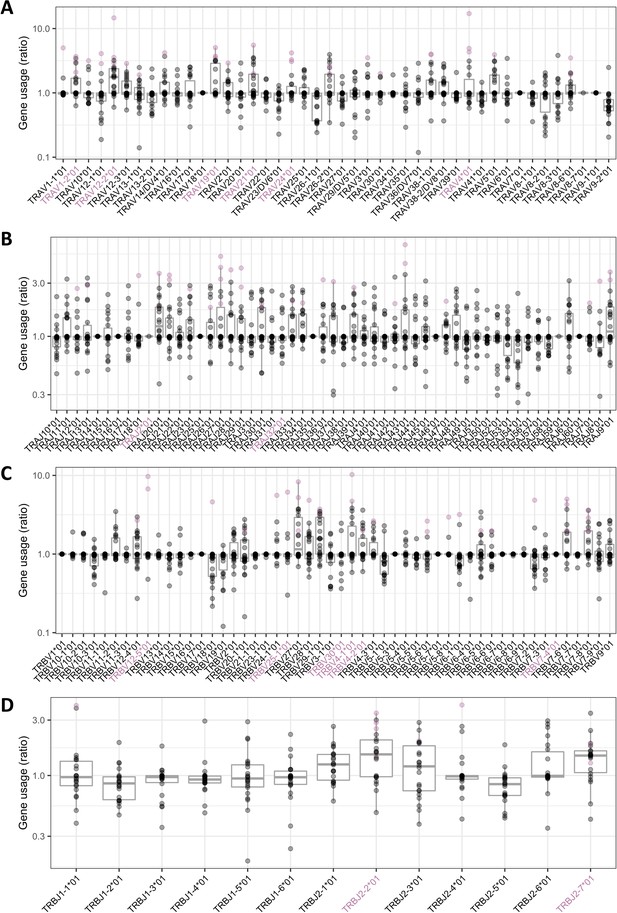
The expanded T cell receptors (TCRs) show characteristics of antigen-driven responses in their V and J gene usage.
Relative frequency of V and J alpha and beta gene usage in the set of PT1 expanded TCRs (≥8) from 22 individuals, compared to the frequency in their respective pre-sensitization repertoires. Each dot is a patient. Genes significantly over-represented in the expanded set (see text for statistical test) are colored pink. Under-represented genes are not included in this figure, since the small number of expanded TCRs in some patients resulted in a large but less meaningful set of under-represented genes due to sampling. Genes colored pink on the x-axis are those that were significantly over-represented in the combined expanded set (1858 unique alpha TCRs and 2019 unique beta TCRs).
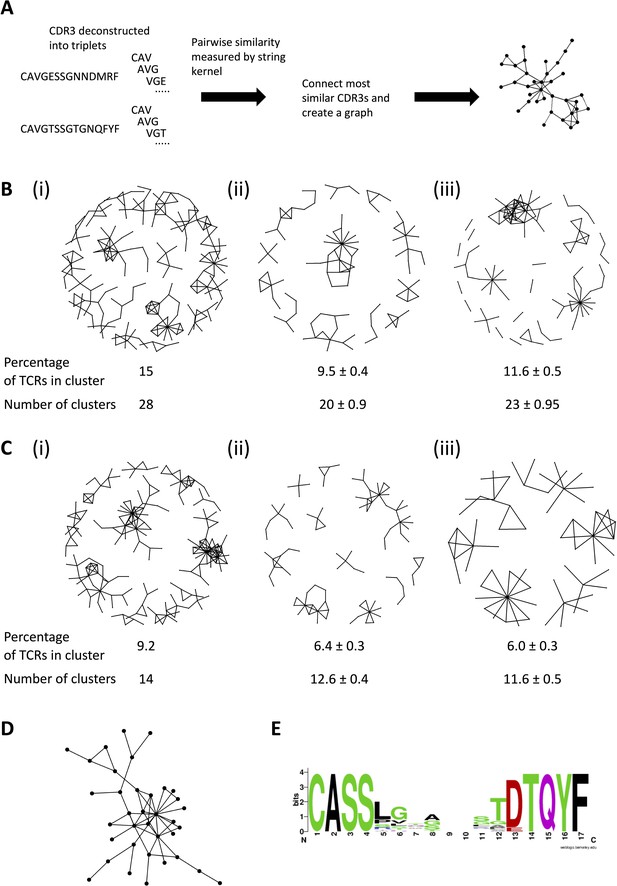
The expanded T cell receptors (TCRs) show clustering characteristics of antigen-driven responses.
(A) Diagram illustrating the similarity graph construction process. Individual CDR3s are deconstructed into overlapping series of contiguous amino acid triplets, and the pairwise similarity between two CDR3s is calculated as the normalized string kernel. Two CDR3s which have a pairwise similarity of >0.75 are connected by an edge. (B) Clusters formed from the CDR3 sequences of (i) the PT1 expanded alpha TCRs (≥ 8), (ii) an equal-sized (1858) set of alpha TCRs sampled randomly from the combined pre-sensitization repertoires of the same patients, and (iii) a size-matched set randomly sampled from the combined healthy volunteer alpha TCRs. The numbers under each network diagram show the percentage of the 1858 TCRs which are incorporated in a cluster, and the number of clusters. The numbers for the pre-sensitization and healthy volunteers show the average ± standard error of the mean for each parameter. (C) As (), but for beta sequences (2019 expanded TCRs, or the same number of control TCRs). (D) The largest (35 unique CDR3s) cluster of PT1 expanded TCR beta sequences, from panel (C) (i). (E) An alignment of CDR3 sequences from the cluster shown in panel (D). The alignment is illustrated as a sequence logo (https://weblogo.berkeley.edu/logo.cgi). The full alignment is shown in Supplementary file 5.
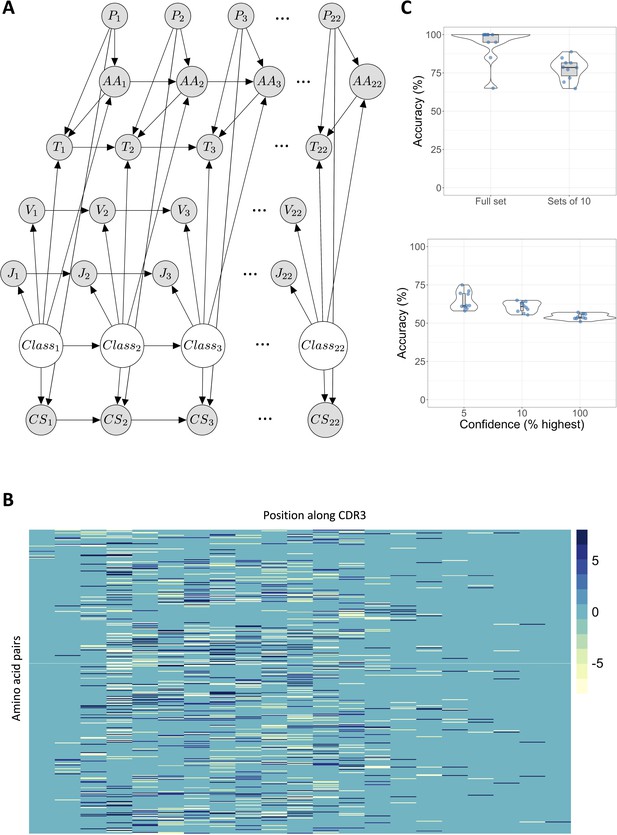
A dynamic Bayesian network (DBN) can predict sensitization based on T cell receptor (TCR) sequence.
(A) The directed acyclic graph depicts the DBN structure, unrolled over 22 time slices (positions). Directed edges capture probabilistic dependencies. Pi indicates position i along the CDR3 sequence; AAi the amino acid in position i; Ti is the triplet score for the triplet of amino acids in positions ; Vi and Ji are the V and J genes (constant for each sequence); CSi the class score at position i, and Classi determines whether the sequence comes from the diphenylcyclopropenone (DPC)-related expanded set, or the control set. (B) The probability of observing pairs of neighboring amino acids differs between DPC-related sequences and control sequences from the pre-sensitization repertoire of the same individuals. Each column in the heatmap corresponds to a position along the CDR3 sequence, each row to an ordered pair of amino acids (total 400). If row i in the heatmap corresponds to amino acid pair , then position in the matrix is the ratio of the probability of observing amino acid Y in position j+1 in the expanded sequences against the probability of observing Y in the same position in an equal-sized random sample of control sequences, given amino acid X has been observed in position j. (C) The upper panel shows the mean classification accuracy for classifying the full DPC/control test sets, and sets of 10 DPC/control sequences. The lower panel shows mean classification accuracy for individual sequences. 5% confidence refers to the top 5% of sequences with greatest log likelihood difference between the models, and similarly for 10%. 100% is the accuracy when classifying every sequence. Each dot depicts the mean of one of the train-test sets from 10-fold cross validation over 10 generations of the model. Each training set consisted of 3618 expanded sequences and control sequences in equal proportions, and the test sets 402 sequences in equal proportions (201 expanded and 201 control).
-
Figure 7—source data 1
Table of expanded T cell receptors (TCRs).
The list of TCRs which is used as the DPC set to generate the DBN which is illustrated in Figure 7. Each row contains a unique combination of CDR3 sequence, V gene, and J gene annotation.
- https://cdn.elifesciences.org/articles/54747/elife-54747-fig7-data1-v2.csv
-
Figure 7—source data 2
Table of control T cell receptors (TCRs).
The list of TCRs which is used as the control set to generate the DBN which is illustrated in Figure 7. Each row contains a unique combination of CDR3 sequence, V gene, and J gene annotation.
- https://cdn.elifesciences.org/articles/54747/elife-54747-fig7-data2-v2.csv
Tables
Demographics of the study population.
| ID | Age | Sex1 | Alopecia2 | TCRseq sets3 | Flow cytometry4 | Patch test scores5 |
|---|---|---|---|---|---|---|
| 1 | 50–59 | F | AA (>50%) | 0 | X | |
| 2 | 10–19 | M | AU | 0 | X | |
| 3 | 20–29 | F | AT | 3 | X | |
| 4 | 50–59 | F | AT | 4 | X | X |
| 5 | 50–59 | M | AA (>50%) | 3 | X | X |
| 6 | 40–49 | M | AA (>50) | 4 | X | X |
| 7 | 40–49 | M | AU | 4 | X | X |
| 8 | 30–39 | F | AA (>50%) | 3 | X | |
| 9 | 30–39 | F | AT | 0 | X | X |
| 10 | 20–29 | F | AT | 1 | X | |
| 11 | 30–39 | M | AU | 1 | ||
| 12 | 20–29 | F | AA (>50%) | 0 | X | X |
| 13 | 30–39 | F | AT | 1 | X | |
| 14 | 30–39 | F | AA (<50%) | 3 | X | |
| 15 | 40–49 | F | AU | 4 | X | X |
| 16 | 40–49 | F | AA (<50%) | 4 | X | X |
| 17 | 50–59 | F | AT | 3 | X | |
| 18 | 20–29 | F | AT | 1 | ||
| 19 | 50–59 | F | AU | 2 | ||
| 20 | 50–59 | F | AU | 4 | X | |
| 21 | 10–19 | M | AA (>50%) | 3 | X | |
| 22 | 30–39 | F | AA (>50%) | 4 | X | |
| 23 | 50–59 | F | AU | 4 | X | |
| 24 | 40–49 | F | AA (<50%) | 4 | X | |
| 25 | 40–49 | F | AT | 3 | X | |
| 26 | 60–69 | F | AA (<50%) | 2 | ||
| 27 | 30–39 | F | AA (>50%) | 1 | ||
| 28 | 30–39 | M | AA (<50%) | 3 | X | |
| 29 | 50–59 | F | AT | 4 | X | |
| 30 | 60–69 | F | AT | 3 | X | |
| 31 | 30–39 | F | AA (>50%) | 1 | ||
| 32 | 20–29 | F | AA (<50%) | 0 | X | |
| 33 | 40–49 | F | AU | 3 | ||
| 34 | 40–49 | F | AA (>50%) | 3 |
-
1F: female; M: male; 2AA: alopecia areata (<50% or >50% scalp involvement); AT: alopecia totalis; AU: alopecia universalis. 3The number of time points for which TCRseq data was obtained. 4The patients for whom PBMC flow cytometry data were available. 5The patients for whom patch test scores were available.
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Software, algorithm | Decombinator V4 | https://github.com/innate2adaptive/Decombinator | RRID:SCR_006732 | This software suite is under active development; latest versions available at the GitHub site. |
| Other | TCRseq protocol | Uddin et al., 2019b PMID:31727254 | This protocol is in a continuous state of development. The full details of the current stable version including primer sequences, PCR conditions etc. are all in the attached reference. For latest development contact the corresponding author on b.chain@ucl.ac.uk |
Additional files
-
Supplementary file 1
Unique and total TCR numbers for each TCRseq sample.
‘_TRA’ are the TCR alpha chain samples, and ‘_TRB’ the beta chain samples.
- https://cdn.elifesciences.org/articles/54747/elife-54747-supp1-v2.docx
-
Supplementary file 2
Repeated exposure to DPC does not alter the global structure of the peripheral blood TCR repertoire.
(A) The Shannon diversity index of the healthy volunteers (n = 14 samples from five individuals), pre-sensitization (n = 25), and post-sensitization (n = 58; from all three time points) TCR repertoire samples. All samples were randomly subsampled to the minimum sample size (20,172 alpha TCRs), and the Shannon diversity index of the subsample was then calculated. Each sample is represented by a dot. The box plots show the median, and lower and upper quartiles of each group. Differences in the distribution of the three groups were tested using a Kruskal–Wallis rank sum test and were non-significant (p=0.7658). (B) The Gini inequality coefficient of the healthy volunteers, pre-sensitization and post-sensitization alpha chain TCR repertoire samples, subsampled as in (A). Differences in the distribution of the three groups were tested using a Kruskal–Wallis rank sum test and were non-significant (p=0.9062). (C) The number of alpha TCRs that appear with a frequency of 1/1000 or higher in each sample (termed ‘abundant TCRs’), for the healthy volunteers, pre-sensitization and post-sensitization samples, subsampled as in (A) and (B). A Kruskal–Wallis rank sum test revealed no statistical difference between the groups (p=0.9256). (D) – (F) The alpha chains of the sensitized samples were separated according to time point: PT1 (n = 23), PT2 (n = 18), and PT3 (n = 17). The Shannon diversity index (D), the Gini coefficient (E), and the number of abundant clones (F) of these subsamples were calculated. Kruskal–Wallis rank sum tests were used to compare between the three groups in each case. All tests showed no statistically significant difference, with p=0.9608, p=0.9281, and p=0.8681 respectively.
- https://cdn.elifesciences.org/articles/54747/elife-54747-supp2-v2.docx
-
Supplementary file 3
Sensitization with DPC induces a transient expansion in the frequency of a small subset of the TCR repertoire.
(A) The abundance distribution of TCRs at PS and PT1. All samples were subsampled to the same number of TCRs (28,000). Each unique TCR is represented by a dot, and the axes represent the number of times it is observed in the PS (x-axis) and PT1 (y-axis) sample of the same individual, and equally spaced time points for the healthy volunteers. The pink dots identify a population of TCRs absent in the PS sample and expanded (abundance ≥8) in the PT1 sample. The blue dots identify a population of TCRs absent in the PT1 sample and expanded (≥8) in the PS sample. The numbers indicate the percentage of PT1 expanded TCRs (pink) and PS expanded TCRs (blue). (B) The correlation between the percentage of PT1 expanded alpha chain TCRs (x-axis) and the percentage of PT1 expanded beta chain TCRs (y-axis) for each individual (n = 22), subsampled as in (A). Spearman’s rho = 0.85, p<0.0001. The line x=y is shown.
- https://cdn.elifesciences.org/articles/54747/elife-54747-supp3-v2.docx
-
Supplementary file 4
Dynamic changes in TCR frequency following sensitization.
(A) The abundances of the PT1 expanded (threshold ≥8) alpha TCRs at the four time points: PS, PT1, PT2, and PT3. Each panel is a different patient (n = 10). (B) The abundances of the PT2 expanded (threshold ≥8) alpha TCRs at the four time points: PS, PT1, PT2, and PT3. Each panel is a different patient (n = 10). (C) Equivalent time points (0 weeks, 2 weeks, and 6 weeks) for four healthy volunteers for whom we had all three times points. Top row is PT1 expanded alpha TCRs; bottom row is PT2 expanded alpha TCRs. The fourth volunteer had no PT1 expanded alpha TCRs. The fifth volunteer had no PT1 expanded alpha TCRs and no PT2 alpha sample.
- https://cdn.elifesciences.org/articles/54747/elife-54747-supp4-v2.docx
-
Supplementary file 5
Sequence alignment of the CDR3 sequences from the largest cluster of TCR beta PT1 expanded CDR3s.
The CDR3s of the largest cluster of PT1 expanded CDR3 beta sequences (see Figure 6D and E) were aligned using the MUSCLE alignment algorithm in Aliview (https://ormbunkar.se/aliview/).
- https://cdn.elifesciences.org/articles/54747/elife-54747-supp5-v2.docx
-
Transparent reporting form
- https://cdn.elifesciences.org/articles/54747/elife-54747-transrepform-v2.docx





